Name ______ Per - Liberty Union High School District
advertisement

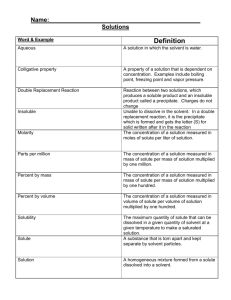
SG 07 Solutions Name ____KEY________ Per. _________ We can divide most everyday matter that we encounter into two categories: pure substances and mixtures. Substances are either elements or ___compounds___ (neutrinos/ compounds/ noble gases), which are when 2 or more elements have bonded chemically. Mixtures are when 2 or more substances have physically blended together without a chemical change occurring. When a mixture is composed entirely of particles that are the size of individuals atoms, ions, or molecules it is called a __homogeneous__ (homogeneous/ heterogeneous) mixture. When a mixture is composed of particles that are larger, meaning clumps of atoms ions, or molecules, it is called a __heterogeneous__ (homogeneous/ heterogeneous) mixture. Homogeneous mixtures include solutions, alloys, and mixtures of gases. Heterogeneous mixtures include __colloids__ (suspensions/ colloids), which are somewhat stable and have medium-sized particles, and __suspensions__ (suspensions/ colloids), which are more unstable and have larger-sized particles. When 2 substances form a homogeneous mixture and the substance doing the dissolving is a liquid, together they form a ___solution___ (solvent/ solute/ solution). The dissolving substance, which there is usually more of, is called the ___solvent___ (solvent/ solute/ solution). The substance being dissolved, which there is usually less of, is called the ___solute___ (solvent/ solute/ solution). Concentration is a measure of how much of a substance is dissolved in another substance. A substance’s solubility is the ___maximum___ (minimum/ maximum) amount of that substance that can be dissolved in a solvent. Most solid solutes ___increase____ (increase/ decrease) their solubility in water with increasing temperature. Most gaseous solutes ___decrease___ (increase/ decrease) their solubility in water with increasing temperature and increasing pressure. A solute that can dissolve in a solvent is described as being ____soluble____ (soluble/ insoluble). A solute that cannot dissolve in a solvent is described as being ___insoluble___ (soluble/ insoluble). The dissolving process can be speeded up by stirring or using a solute with a ___large___ (large/ small) surface area, for example using a powder instead of large chunks of the solute. If a solution has reached its maximum concentration, aka solubility, it is __saturated__ (unsaturated/ saturated/ supersaturated). If a solution has not yet reached its solubility it is __unsaturated__ (unsaturated/ saturated/ supersaturated). If a solution has somehow exceeded its solubility it is _supersaturated_ (unsaturated/ saturated/ supersaturated). The __precipitate__ (precipitate / solvent) is the solid that appears at the bottom of a container because too much has dissolved or a chemical reaction has occurred. The basic rule for predicting solubility is “like dissolves ____like____ (dislike/ like/ different)”. What it means is that polar substances are more likely to dissolve in ____polar____ (nonpolar/ polar) solvents and nonpolar substances are more likely to dissolve in __nonpolar__ (nonpolar/ polar) solvents. Fats, oils, lipids, and gasoline are all examples of __nonpolar__ (nonpolar/ polar) substances. Water, vinegar, and alcohol are all examples of ___polar___ (nonpolar/ polar) substances. In biology, vitamins A and D are examples of __nonpolar__ (nonpolar/ polar) substances because they can accumulate in the body’s fatty tissues. Vitamins C and E are examples of __polar__ (nonpolar/ polar) substances because they are soluble in water and any excess will be flushed out of the body with the urine. Applications for solutions chemistry include using salt to melt ice on roads because the salt ___raises___ (raises/ lowers) the melting point of water and preventing radiator fluid from boiling out by ___lowering___ (raising/ lowing) the boiling point of water. These are called colligative properties. Acids and ionic compounds are sometimes called ___electrolytes___ (colloids/ electrolytes) because when dissolved in water they conduct electricity. Separations are utilized in chemistry to isolate and measure various substances in a mixture. _Centrifugation_ (Distillation/ Centrifugation/ Chromatography/ Filtration) spins a mixture at a very high speed and causes the heavier material to settle on the bottom of a tube. ___Filtration___ (Distillation/ Centrifugation/ Chromatography/ Filtration) collects solid precipitates or suspension solids on paper while dissolved solutes pass through a funnel with the solvent. __Distillation__ (Distillation/ Centrifugation/ Chromatography/ Filtration) takes advantage of substances’ differing intermolecular attractions. _Chromatography_ (Distillation/ Centrifugation/ Chromatography/ Filtration) takes advantage of liquid substances’ differing boiling points by having the substance with the lowest boiling point boil off first and then condensed and collected. Circle all of the following units that can be used for measuring concentration. grams (g) % composition milliliters (mL) g solute/ L solvent meters (m) liters (L) parts per billion (ppb) kilopascals (kPa) moles (mol) Kelvin (K) degrees Celsius (˚C) g solute/100 g H2O mol/L kilograms (kg) parts per million (ppm) M1 × V1 = M2 × V2 Molarity (M) = Molarity (M) # moles solute 1 L solution 1) On a molecular level, describe the process of grains of solid table salt, sodium chloride (NaCl), dissolving into water. As a solid, the sodium chloride is stuck in a crystal with alternating positive and negative charges. When it hits the water, the water (with its slightly positive and negative ends) pulls away oppositely charged ions one at a time as the molecules move randomly. 2) On a molecular level, describe how the ions from an electrolyte facilitate electricity, electrons travelling, through normally nonconductive water. The negative electrons are able to hop from positive ion to positive ion, which are dissolved in the water, as the move from the negative electrode to the positive electrode. 3) If 21 moles of KI are dissolved in a 7 L solution, then what is the molarity of the solution? 21 mol KI 7 L solution = 3 M KI solution 4) How many moles of NaCl are dissolved in 400 mL of a 0.4 M NaCl solution? 0.4 mol NaCl x 400 mL = 0.400 L = 1 L solution 0.4 L solution 0.16 = 1 x x = 0.16 mol NaCl Use the below graph for questions 5 – 7. 5) 10 grams of KClO3 are dissolved in 50 g of water at 50 ˚C. Will the solution be unsaturated, saturated, or supersaturated? 1,000 = 50 x 10 g KClO3 x = 50 50 50 g H2O 100 g H2O = 20 g KClO3 / 100 g H2O 6) 15 grams of NH4Cl are dissolved in 20 g of water at 70 ˚C. Will the solution be unsaturated, saturated, or supersaturated? 1,500 = 20 x 15 g NH4Cl x = 20 20 20 g H2O 100 g H2O = 75 g NH4Cl / 100 g 7) 2 grams of KClO3 are dissolved inH10 2O g of water at 80 ˚C. Will the solution be unsaturated, saturated, or supersaturated? 200 = 10 x 2 g KClO3 x = 10 10 10 g H2O 100 g H2O = 20 g KClO3 / 100 g H2O 8) 3 liters of a 6.0 M solution of potassium iodide (KI) is diluted with water until the new solution has a volume of 36 liters. What is the new molarity of the potassium iodide solution? 18 = 36 M2 M1 × V1 = M2 × V2 M2 = 0.5 M KI solution 36 36 (6.0) × (3) = M2 × (36) 9) 4 liters of a 6.0 M solution of sodium chloride (NaCl) is diluted with water until the new solution has a molarity of 0.12 M. What is the new volume of the sodium chloride solution? 24 = 0.12 V2 M1 × V1 = M2 × V2 V2 = 200 L 0.12 0.12 (6.0) × (4) = (0.12) × V2