Grade 11 - Predicting Products of Chemical
advertisement

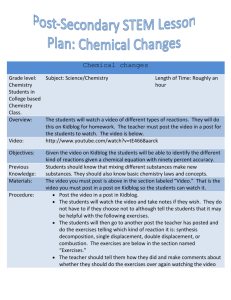
Micro-Teaching Lesson Plan i) ii) iii) iv) v) Specific Expectations for Academic grade 11 Chemistry: Chemical Reactions Write balanced chemical equations to represent synthesis, decomposition, single displacement, double displacement, and combustion reactions using the IUPAC nomenclature system Materials needed: Demo: piece of magnesium metal, Bunsen burner, flint, safety goggles, tongs Class activity: handout with chemical reaction problems Time sequence: Approximately 1 minute to go over learning goal and agenda Approximately 5 minutes for Demo and related discussion Approximately 4 minutes to review reaction patterns, polyatomic ions, and balancing equations Approximately 5 minutes to teach predicting products of chemical reactions Approximately 5 minutes for take-up of class activity Introduction Today’s lesson will be on predicting the products of chemical reactions The learning goal for today’s lesson is to predict the products of synthesis, decomposition, single displacement, and double displacement, and combustion reactions and write balanced chemical equations for the reaction Here is the agenda for today: 1) Demo 2) Review of reaction patterns, polyatomic ions, and balancing equations 3) Lesson on predicting products of chemical reactions 4) Activity take-up We will start the class with the demo: magnesium combustion Key questions: Demo: 1. What type of reaction do you expect to see when something is burned? (expected answer: combustion) 2. What are the starting reagents? (expected answer: magnesium metal, oxygen) 3. What might the product be? (expected answer: MgO) Review: 1. What are the four types of reactions you learned last year? 2. When writing out a complete reaction, what must we do to preserve mass/molecule composition on both the reagent and product sides? (expected answer: balance) 3. Are the elements in a polyatomic ion separable? Lesson: 1. Can you predict the product of this reaction based on the patterns we discussed? vi) vii) 2. What could be the first step in helping you predict the product of a reaction? (expected answer: finding the pattern) 3. What type of reaction could a combustion reaction be? (expected answer: synthesis – related to the demo) Concluding Idea/Summary Keep in mind: make sure equations are balanced, consider that the elements in polyatomic ions are all one entity If a particular reaction appears difficult, start off by identifying the pattern Expectations: be able to predict products and write balanced chemical equations to represent synthesis, decomposition, single displacement, double displacement, and combustion reactions for quizzes, tests, assignments Chalkboard outline Board 1 Title: Predicting Products of Chemical Reactions Learning Goal: To predict products of synthesis, decomposition, single displacement, double displacement and combustion reactions and write balanced chemical equations for the reaction Agenda Demo: burning magnesium Review reaction patterns, polyatomic ions, balancing equations Lesson: Predicting products of chemical reactions Class activity and take-up Board 2 4 Reaction patterns: 1) 2) 3) 4) A + B -> AB AB -> A + B AB + C -> AC + B AB + CD -> AC + BD Synthesis Decomposition Single Displacement Double Displacement Polyatomic ions Review – Inseparable! PO43-, CO32-, SO42-, NO3Board 3 Predicting products for each reaction pattern + balancing 1) 2Na + Cl2 -> 2NaCl Synthesis 2) 2HgO -> 2Hg + O2 Decomposition 3) 2Fe + 3H2SO4 -> 3H2 + Fe2(SO4)3 Single displacement 4) 2NaOH + 2CuCl2 -> 2NaCl + Cu(OH)2 Double displacement 5) 2Mg + O2 -> 2MgO Combustion Board 4 Take-up of class activity * indicate polyatomic ion* * point out hydroxide * *Demo*