aic14903-sup-0001-suppinfo
advertisement

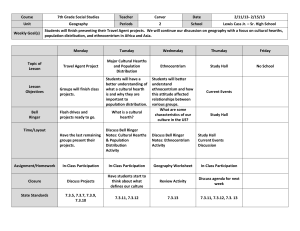
Appendix A – Heat transfer coefficients Nomenclature Roman letters A Area (m2) cp specific heat capacity (J/molK) DH characteristic diameter (m) g gravimetric constant (=9.81 m/s2) kg thermal conductivity (W/mK) L characteristic dimension (m) P perimeter (m) v average velocity (m/s) Greek letters α kinetic viscosity β volumetric expansion coefficient (1/T(K)) ε emissivity, radiative exchange ratio ρ density (kg/m3) ν thermal diffusivity µ dynamic viscosity (Pa s) Heat transfer between the solid bed and the gas phase The heat transfer coefficient hcgs is calculated using the Nusselt’s number presented in Equation A.1. 𝑁𝑢 = ℎ𝐿 𝑘𝑔 In this case the characteristic dimension is the length of the heat transfer surface shown in Figure A.1. ( A.1 ) L Figure A.1. The solid bed surface from up showing the characteristic length L. For the hearths 1 to 6 the conditions for the Nusselt number calculation are assumed to be forced convection, turbulent (Re > 1*105) and internal flow. For this case the Nusselt number is given by Equation A.2, as suggested in1 4 𝑁𝑢 = 0.23 ∗ 𝑅𝑒 5 ∗ 𝑃𝑟 0.35 ( A.2 ) The Reynold number in Equation A.2 is calculated as 𝑅𝑒 = 𝜌𝑣𝐷ℎ 𝜇 ( A.3 ) The Prandtl number describing the ratio between the momentum diffusivity and thermal diffusivity of the fluid is calculated as 𝑃𝑟 = 𝑐𝑝 𝜇 𝑘 ( A.4 ) The conditions on the hearths 7 and 8 are assumed to be natural convection on the top of a hot surface. Now, the solid bed is assumed to be in a higher temperature than the gas phase, otherwise there would not be a cooling effect. According to Incropera et al.1, the Nusselt number for this particular case can be evaluated as 𝑁𝑢 = 0.54𝑅𝑎1/4 ( A.5 ) The Rayleigh number is written as 𝑅𝑎 = 𝑔𝛽(𝑇𝑠 − 𝑇𝑔 )𝐿3 𝜈𝛼 ( A.6 ) The characteristic length L is defined as 𝐿= 𝐴 𝑃 The gas-solid heat exchange is calculated separately for each four or five solid volumes depending on the hearth. ( A.7 ) Heat transfer between the solid bed and the walls The emissivity between the solid bed and the walls εsw is calculated as in2: 𝜀𝑠𝑤 = 𝜀𝑠 𝜀𝑤 (1 − 𝜀𝑔 ) 1−𝑈 ( A.8 ) where εs, εw and εg present emissivities of the solid bed, walls and gas phase, respectively. The variable U is defined as in Equation A.9. 𝑈 = (1 − 𝜀𝑤 )(1 − 𝜀𝑔 )[𝑣(1 − 𝜀𝑔 )(1 − 𝜀𝑠 ) + (1 − 𝑣)] ( A.9 ) where the variable v presents the ratio between the heat exchange area of the solid bed and the gas phase A sg and the heat exchange area between the walls and the gas phase Agw: 𝑣= 𝐴𝑠𝑔 𝐴𝑔𝑤 ( A.10 ) The emissivity of the walls is assumed to be 0.90 as with a refractory material being an average radiator. Heat transfer between the inner walls and the gas phase The heat transfer coefficient hcgw is calculated using the same equations as for the heat exchange between the gas phase and the solid bed for the hearths 1 to 6. For the hearths 7 and 8, the heat exchange surface is assumed to be a vertical plate with natural convection due to the stagnant gas phase. The Nusselt number for the hearths 7 and 8 is calculated as 1 𝑁𝑢 = 0.68 + 0.67𝑅𝑎4 (1 + ( 4 9 9 16 0.492 ) ) 𝑃𝑟 ( A.11 ) The Raleigh number (Ra) is calculated with Equation A.6 where the characteristic length is now the height of the wall. The Prandtl number (Pr) is calculated with Equation A.4. Conduction inside the walls To solve the boundary surface temperatures of the wall layers, the heat fluxes between the wall and the gas phase and between the wall and the solid bed are calculated first. Next, the outer wall temperature is iterated so that the heat flux inside the wall and the heat flux to the ambient air are equal. The furnace wall consists of four layers defined according to the wall materials described in Table A.1. As the heat flux through the different layers of the wall was assumed constant, the heat flux inside the wall can be calculated using Equation A.12 suggested in3 𝑄𝑤 = 2𝜋𝐿(𝑇𝑖𝑛𝑛𝑒𝑟 𝑤𝑎𝑙𝑙 − 𝑇𝑜𝑢𝑡𝑒𝑟 𝑤𝑎𝑙𝑙 ) 𝑟 𝑟 1 𝑟2 1 1 𝑟 1 ln ( ) + ln ( 3 ) + ln ( 4 ) + ln ( 5 ) 𝜆1 𝑟1 𝜆2 𝑟2 𝜆3 𝑟3 𝜆4 𝑟4 ( A.12 ) where L is the height of the wall section, λi is the thermal conductivity of a wall layer and, rj is the radius of the surface from the centre of the hearth. After updating the inner wall surface temperature, the temperatures of the other surface layers can be calculated using Equation A.13: 𝜕𝑇𝑤𝑎𝑙𝑙 𝑠𝑒𝑐𝑡𝑖𝑜𝑛 𝑟𝑖+𝑖 1 = −𝑄𝑤 ∗ ln ( ) 𝜕𝑡 𝑟𝑖 2𝜋𝜆𝑖 ℎ𝑤𝑎𝑙𝑙 ( A.13 ) where subscript i is the number of the wall layer (number 1 presents the most inner layer and the inner surface) and h presents the height of the wall section. The heat exchange properties of the wall materials are given in Table A.1. Table A.1. Wall material properties. Thermal Width Material Heat capacity Density (J/kg K) (kg/m3) conductivity (mm) (W/m K) Refractory brick DSF 594 152 1.6 1000 2480 Fire brick HTJ5 114 0.32 1000 769 Backfill Caldecast XL 1066 75 0.16 1000 2350 Mild steel (0.5% carbon)7 25 54 465 7833 Heat transfer between the outer wall and the ambient air The heat transfer between the outer wall surface and ambient air is assumed to be natural convection on a vertical hot plate. The heat exchange coefficient is calculated using the Rayleigh number, Equation A.6. Now, 𝑅𝑎𝑤𝑎 > 109 and equation for the Nusselt number becomes1 1 3 𝑁𝑢𝑤𝑎 = 0.10 ∗ 𝑅𝑎𝑤𝑎 ( A.14 ) Heat transfer between the gas and the central shaft and the rabble arms For the heat exchange between the gas phase and the central shaft at the hearths 1 to 6, the following assumptions are made: the shaft is in external gas flow, the shaft is considered as a flat plate parallel to the flow and the gas flow is turbulent. Now, the Nusselt number for the heat transfer coefficient can be calculated using Equation A.15. 4 1 ( A.15 ) 𝑁𝑢𝑔𝑠 = 0.0296 ∗ 𝑅𝑒 5 ∗ 𝑃𝑟 3 The Reynold number is an average of the Reynold number in the annulus between the central shaft and the hearth and of the Reynold number in the gas phase close to the shaft. For the hearths 7 and 8, the heat exchange coefficient is approximated to be the same as the coefficient between the walls and the gas phase since, in both cases, the heat exchange conditions are the same: natural convection on a vertical plate. For the heat exchange between the rabble arms and the gas phase, the Nusselt number is calculated with the same equation A.14 on the hearths 1 to 6. In the hearths 7 and 8, the Nusselt number is calculated using Equation A.16 given in1, which applies to a long horizontal cylinder for a wide Rayleigh number range Ra<10 12 1 𝑁𝑢 = 0.60 + 0.387𝑅𝑎 6 9 0.559 16 (1 + ( ) ) 𝑃𝑟 8 27 ( A.16 ) Heat transfer between cooling air and the central shaft and the rabble arms Nusselt number for the heat transfer between cooling air and the central shaft and the rabble arms is calculated using Equation A.17, which applies for a flow inside a tube in turbulent conditions. 1 𝑁𝑢 = 0.023 ∗ 𝑅𝑒 0.8 ∗ 𝑃𝑟 3 ( 𝜇̅ 0.14 ) 𝜇𝑤 ( A.17 ) For the rabble arms the characteristic length Dh is given by Equation A.18: 𝐷ℎ = 4 ∗ 𝐶𝑟𝑜𝑠𝑠 𝑠𝑒𝑐𝑡𝑖𝑜𝑛𝑎𝑙 𝑎𝑟𝑒𝑎 𝑜𝑓 𝑡ℎ𝑒 𝑎𝑟𝑚 𝑃𝑒𝑟𝑖𝑚𝑒𝑡𝑒𝑟 𝑜𝑓 𝑡ℎ𝑒 𝑎𝑟𝑚 ( A.18 ) Appendix B – The emissivity of aluminium oxide and the silicone oxide The comparison of the curve presented in equation (28) and the literature values of the emissivity is presented in Figure B.1. Figure B.1. The literature values of emissivity of aluminium oxide and silicon oxide and the curve defined by Equation Error! Reference source not found.. Appendix C. Furnace response under gas and air flows changes on the Hearth 6 The gas and air flows to Hearth 6 were introduced a 6.6 kg/h (5 percent) step increase to investigate the response of the system presented in Figure C.1 and Figure C.2. The mullite formation reaction begins immediately to intensify on Hearth 7 as seen in Figure C.2. As the gas temperature on Hearth 6 rises the product formation intensifies at 870 minutes, especially in the third volume on the same hearth as shown in Figure C.1. Since, the amount of gas fed to Hearth 6 is approximately half of what is fed to Hearth 4, the observed changes in the reactions are not as intensive as in the previous case. The exhaust gas temperature and the temperatures of Hearths 2, 3, and 5 increases instantly after the step change and afterwards they settle to a higher steady state than the initial temperature was. The Hearth 4 temperature decreases slightly as a result of the increased gas flow from previous hearth, but afterwards increases steadily to a higher temperature. The gas temperatures on Hearths 6 to 8 increase over the time and settle to new steady state values. The final single input step test was a 6.6 kg/h (5 percent) decrease in the gas and air flows to Hearth 6. The model response is given in Figure C.3 andFigure C.4. As a result of decreased heating the spinel phase formation moves to Hearth 7 at the time of 1540 minutes as seen in Figure C.4. This also noticeably increases the Hearth 7 temperature and decreases the Hearth 6 temperature as seen in Figure C.3. As the gas and air flow to Hearth 6 drops, the temperatures of the exhaust gas and Hearths 2, 3 and 5 decrease first with a steep step and then more steadily before reaching a new steady state. In contrast, the temperature of Hearth 4 increases first slightly and then decreases steadily for the whole recorded time period. Figure C.1. The response of the gas phase temperature next to the wall while increasing the gas and air flows to Hearth 6. Figure C.2. The response of the product content in Hearths 6 and 7 while increasing the gas and air flows to Hearth 6. Figure C.3. The response of the gas phase temperature next to the wall while decreasing the gas and air flows to Hearth 6. Figure C.4. The response of the product content in Hearths 6 and 7 while decreasing the gas and air flows to Hearth 6. Literature cited 1. Incropera F, DeWitt D, Bergman T, Lavine A. Fundamentals of Heat and Mass Transfer (6th ed). Hoboken: Wiley, 2007. 2. Ginsberg T, Modigell M. Dynamic modelling of a rotary kiln for calcination of titanium dioxide white pigment. Comput. Chem. Eng. 2011; 35:2437-2446. 3. Engineers Edge, LLC. Conduction-cylindrical coordinates heat transfer. Available at: http://www.engineersedge.com/heat_transfer/conduction_cylidrical_coor.htm Accessed August 29, 2014. 4. DSF Refractories & Minerals Ltd. DSF 59 - The Product description sheets, 2009. 5. BNZ Materials, Inc. Insulating fire brick. Product data sheet, 2009. 6. Calderys. Technical data sheet - Calde Cast XL 106 C/G, 2009. 7. Engineers Edge, LLC. Thermal Properties of Metals, Conductivity, Thermal Expansion, Specific Heat. Available at: http://www.engineersedge.com/properties_of_metals.htm Accessed December 23, 2013.