File
advertisement

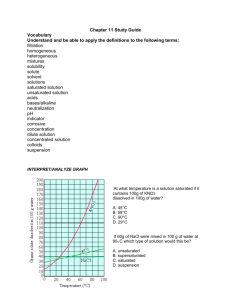
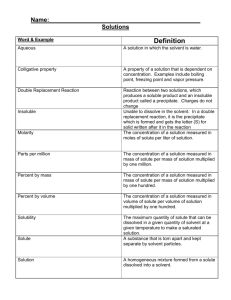
Best of luck Class IX SCIENCE TEST I. Multiple choice questions:- 1 Mark each 1. Which of the following provides an example of a true solution? (A) Blood (B) Milk (C) Starch solution (D) Sugar solution 2. Which of the following can be classified as a pure substance? M.M.50 20 (A) Milk (B) Sea - water (C) Ice (D) Cast iron 3. Which of the following is a compound? (A) air (B) Milk (C) Iodine (D) Water 4. The particle size of solute in true solution is of the order of (A) 10-6 m (B) 10-7 m (C) 10-8 m (D) 10-9 m 5. Which of the following statement is not true about true solution? (A) It can pass through filter paper. (B) It is homogeneous in nature. (C) At constant temperature, particles of solute settle down. (D) From a true solution, the solute can easily be recovered by evaporation or crystallization. 6. The concentration of solution is the mass of the solute in grams, which is present in (A) 10 gm of solvent (B) 10 gm. of solution (C) 100 gm of solvent (D) 100 gm of solution 7. A solution, which at a given temperature dissolves as much solute as it is capable of dissolving, is said to be a(A) Saturated solution (B) semi saturated solution (C) Unsaturated solution (D) super saturated solution 8. Which of the following is not a compound? (A) Marble (B) Washing soda (C) Quick lime (D) Brass 9. The elements which given out harmful radiation are called (A) Normal elements (B) representative elements (C) Radioactive elements (D) none of these 10. Air is regarded as a (A) Compound (B) mixture (C) element (D) electrolyte 11. Which of the following is/are example of suspension? (A) Muddy water (B) Slaked lime (C) Paints (D) All 12. Which of the following statement is not true about suspension ? (A) The particles of suspension can be separated from solvent by the process of filtration. (B) When the suspension is kept undisturbed the particles of suspension settle down. (C) A suspension is homogeneous in nature. (D) Scattering of light take place in suspension. 13. Fog is an example of (A) Foam (B) Emulsion (C) Aerosol (D) Gel 14. Which of the following statement is not true about colloidal solution? (A) These are visible under powerful microscope. (B) Their particles do not settle down with passage of time 1|Page IS MATTER AROUND US PURE (C) Their particles are electrically charged. (D) These are homogeneous in nature. 15. Which of the following is an example of emulsion? (A) Face cream (B) Shaving cream (C) Honey (D) Smoke 16. Soap solution is an example of (A) True solution (B) suspension (C) colloidal solution (D) none of these 17. When a beam of light is passed through a colloidal solution, it gets (A) Reflected (B) absorbed (C) scattered (D) refracted 18. Which of the following is an example of gel? (A) Colored gem (B) Jelly (C) Smoke (D) Shaving cream 19. Which of the following will show Tyndall effect? (A) Starch solution (B) Sodium chloride solution (C) Copper sulphate solution (D) Sugar solution 20. Colloidal particles can be normally seen by (A) naked eye (B) optical microscope (C) electron microscope (D) telescope II. Fill the blanks: 1 Marks each 1. The component present in the solution in greater quantity is ----------. 2. The component present in the solution in lesser quantity is -------------. 3. Water can dissolve in it a great number of substances. So water is considered a 4. The amount of solute dissolved in a fixed amount of solvent is called ----------- of solution 5. If the amount of solute dissolved is greater, it is a -----------solution 10 6. if the amount of solute dissolved is lesser, it is ---------- solution 7. The amount of solute dissolved in 100g of water to make it saturated is the ------------- of the solute in water 8. A solution in which maximum amount of solute is dissolved at a certain temperature is its ----------- solution at that temperature. 9. ----------- is a form of mixture which is neither a solution nor a suspension 10. When a strong beam of light is passed through a colloid, its path becomes visible. This phenomenon is called--------------Q3. Distinguish between physical and chemical change. Any Four Q4. With the help of a slow diagram show the process of obtaining different gases from air. Q5. Arrange the gases present in air in increasing order of their boiling point. Q6. What is meant by concentration of a solution? Which is the most common method of expressing The concentration of a solution? Q7. Explain the method of distillation and fractional distillation with example. Q8. Differentiate between (i) Mixtures and Compound (ii) Homogenous and heterogeneous mixtures 2 3 1 IS MATTER AROUND US PURE 2 | Page 2 3+3 3+3