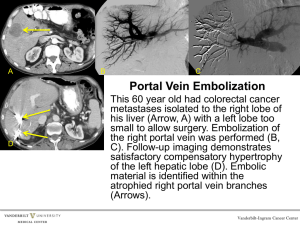
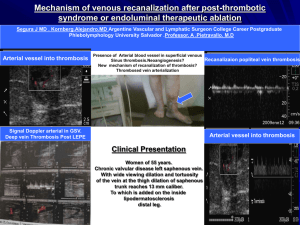
Supplementary table 1 | Common risk factors for portal vein
advertisement

Supplementary table 1 | Common risk factors for portal vein thrombosis in patients with liver cirrhosis Risk factors for PVT Results of univariate analysis Sources of evidence Multivariate analysis performed? Results of multivariate analysis Interpretation of results Type of analysis Study design Patients ( n) Relevant papers Characteristics of portal flow Decreased portal flow velocity (cut-off: 15 cm/s) P <0.001 Yes OR = 44.9 (95% CI 5.3–382.7, P <0.001) Significant in both univariate and multivariate analyses Chi-square test, logistic regression analysis Prospective cohort study 73 Zocco et al. (2009)S1 Lower mean portal vein flow velocity P <0.001 Yes OR = 4.5 (95% CI 3.7–8.2, P <0.001) Significant in both univariate and multivariate analyses Chi-square test, logistic regression analysis Retrospective study 56 Pellicelli et al. (2011)S2 Portal trunk vessel diameter NS Uncertain N/A NS in univariate analysis Portal trunk flow velocity NS Uncertain N/A NS in univariate analysis Student’s t-test Retrospective study 150 Maruyama et al. (2013)S3 Lower portal trunk flow volume P = 0.016 Uncertain N/A Significant in univariate analysis Yes N/A Yes N/A Cox regression analysis Retrospective study 150 Maruyama et al. (2013)S3 Yes HR = 3.992 (95% CI 3.697–4.415, P <0.0001) Significant in univariate, but not in multivariate analysis Significant in univariate, but not in multivariate analysis Significant in both univariate and multivariate analyses P = 0.78 No N/A NS in univariate analysis Chi-square test Retrospective study 63 Amitrano et al. (2000)S4 P <0.05 No N/A Significant in univariate analysis Chi-square test Retrospective study 64 Erkan et al. (2005)S5 NS No N/A NS in univariate analysis Chi-square test Retrospective study 219 Mangia et al. (2005)S6 Characteristics of largest collateral vessel Larger vessel diameter Higher flow velocity (cut-off: 10.2 cm/s) Higher flow volume (cutoff: 398 ml/min) HR = 1.422 (95% CI 1.210–1.732, P = 0.0103) HR = 2.876 (95% CI 2.691–3.170, P <0.0001) HR = 3.894 (95% CI 3.189–4.798, P <0.0001) Characteristics of coagulation abnormalities Factor V Leiden mutation 1 Prothrombin G20210A mutation MTHFR C667T mutation OR = 1.52 (95% CI 0.64–3.60, NS) No N/A NS Meta-analysis Meta-analysis 5 studies Dentali et al. (2008)S7 P = 0.002 No N/A Significant in univariate analysis Chi-square test Retrospective study 63 Amitrano et al. (2000)S4 P <0.01 No N/A Significant in univariate analysis Chi-square test Retrospective study 64 Erkan et al. (2005)S5 NS No N/A NS in univariate analysis Chi-square test Retrospective study 219 Mangia et al. (2005)S6 OR = 3.68 (95% CI 1.58–8.57) No N/A Significant in univariate analysis Meta-analysis Meta-analysis 3 studies Dentali et al. (2008)S7 P <0.001 No N/A Significant in univariate analysis Chi-square test Retrospective study 63 Amitrano et al. (2000)S4 NS No N/A NS Chi-square test Retrospective study 64 Erkan et al. (2005)S5 NS (homozygous) No N/A NS in univariate analysis Chi-square test Retrospective study 219 Mangia et al. (2005)S6 No N/A Significant Chi-square test Retrospective study 76 Gabr et al. (2010)S8 Yes OR = 4.1 (95% CI 3.2–7.3, P <0.001) Significant in both univariate and multivariate analyses Chi-square test, logistic regression analysis Retrospective study 56 Pellicelli et al. (2011)S2 No N/A Significant in univariate analysis Chi-square test Retrospective study 73 Violi et al. (1994)S9 P = 0.037 (homozygous) P <0.001 (homozygous) Positive anticardiolipin antibodies Positive lupus anticoagulant P = 0.015 P = 0.0008 No N/A Significant in univariate analysis 2 Hypofibrinolysis TAFI: P = 0.001; activated TAFI: P = 0.037; PAI-1: P = 0.004 No N/A Significant in univariate analysis PAI-1: P = 0.947; t-PA: P = 0.205 No N/A NS in univariate analysis P <0.001 Yes OR = 15.57 (95% CI 4.09–59.14, P <0.01) Significant in univariate and multivariate analysis P <0.01 No N/A Significant in univariate analysis NS No N/A NS No NS Retrospective study 66 Rossetto et al. (2013)S10 Retrospective study 116 Zhang et al. (2010)S11 Retrospective study 116 Zhang et al. (2010)S11 Mann-Whitney U-test Retrospective study 188 Zhang et al. (2013)S12 NS in univariate analysis Student’s t--test Retrospective study 63 Amitrano et al. (2000)S4 N/A NS in univariate analysis Student’s t--test Retrospective study 64 Erkan et al. (2005)S5 No N/A NS in univariate analysis Student’s t--test Retrospective study 76 Gabr et al. (2010)S8 AT: P = 0.119; PS: P = 0.028; PC: P = 0.007 Yes PS: NS; Increased PC: OR = 0.48 (95% CI 0.24–0.95, P = 0.036) PC and PS were significant in univariate analysis, but only PC was significant in multivariate analysis Student’s t-test & logistic regression analysis Retrospective study 116 Zhang et al. (2010)S11 AT: P = 0.24; PS: P = 0.006; PC: P = 0.16 No N/A Only PS was significant in univariate analysis Meta-analysis Meta-analysis 9 studies Qi et al. (2013)S13 AT: P = 0.53; PS: P = 0.125; PC: P = 0.058 No N/A NS in univariate analysis Student’s t--test Retrospective study 60 Chen et al. (2013)S14 Increased levels of D-dimer Decreased levels of AT, PC and PS Nonparametric Mann-Whitney U test 3 Mann-Whitney U-test & student’s t-test Mann-Whitney U-test & student’s t-test Abbreviations: AT, antithrombin; HR, hazard ratio; N/A, not available; NS, not significant; OR, odds ratio; PC, protein C; PS, protein S; PVT, portal vein thrombosis; PAI-1, plasminogen activator inhibitor; TAFI, thrombin activatable fibrinolysis inhibitor; t-PA, tissue-type plasminogen activator. S1. Zocco, M. A. et al. Thrombotic risk factors in patients with liver cirrhosis: correlation with MELD scoring system and portal vein thrombosis development. J. Hepatol 51, 682–689 (2009). S2. Pellicelli, A. M. et al. Clinical and genetic factors associated to development of portal vein thrombosis in cirrhotic patients without hepatocellular carcinoma. J. Hepatol. 54, S77 (2011). S3. Maruyama, H., Okugawa, H., Takahashi, M. & Yokosuka, O. De novo portal vein thrombosis in virus-related cirrhosis: predictive factors and long-term outcomes. Am. J. Gastroenterol. 108, 568–574 (2013). S4. Amitrano, L. et al. Inherited coagulation disorders in cirrhotic patients with portal vein thrombosis. Hepatology 31, 345–348 (2000). S5. Erkan, O. et al. Thrombophilic gene mutations in cirrhotic patients with portal vein thrombosis. Eur. J. Gastroenterol. Hepatol. 17, 339–343 (2005). S6. Mangia, A. et al. Causes of portal venous thrombosis in cirrhotic patients: the role of genetic and acquired factors. Eur. J. Gastroenterol. Hepatol. 17, 745–751 (2005). S7. Dentali, F., Galli, M., Gianni, M. & Ageno, W. Inherited thrombophilic abnormalities and risk of portal vein thrombosis. A meta-analysis. Thromb. Haemost 99, 675–682 (2008). S8. Gabr, M. A., Bessa, S. S. & El-Zamarani, E. A. Portal vein thrombosis in Egyptian patients with liver cirrhosis: Role of methylenetetrahydrofolate reductase C677T gene mutation. Hepatol. Res. 40, 486–493 (2010). S9. Violi, F. et al. Relation between lupus anticoagulant and splanchnic venous thrombosis in cirrhosis of the liver. BMJ 309, 239–240 (1994). S10. Rossetto, V. et al. Does decreased fibrinolysis have a role to play in the development of non-neoplastic portal vein thrombosis in patients with hepatic cirrhosis? Intern. Emerg. Med. http://dx.doi.org/10.1007/s11739-013-0929-7 S11. Zhang, D., Hao, J. & Yang, N. Protein C and D-dimer are related to portal vein thrombosis in patients with liver cirrhosis. J. Gastroenterol. Hepatol 25, 116–121 (2010). S12. Zhang, D. L., Hao, J. Y. & Yang, N. Value of D-dimer and protein S for diagnosis of portal vein thrombosis in patients with liver cirrhosis. J. Int. Med. Res. 41, 664–672 (2013). S13. Qi, X., Chen, H. & Han, G. Effect of antithrombin, protein C and protein S on portal vein thrombosis in liver cirrhosis: a meta-analysis. Am. J. Med. Sci 346, 38–44 (2013). S14. Chen, H. et al. Coagulation imbalance may not contribute to the development of portal vein thrombosis in patients with cirrhosis. Thromb. Res 131, 173–177 (2013). 4 Supplementary table 2 | Effect of portal vein thrombosis on the prognosis of cirrhotic patients with bleeding Effect of PVT on prognosis Results of univariate analysis Multivariate analysis performed? Results of multivariate analysis Interpretation of results Sources of evidence Type of analysis Study design Target population Patients ( n) Relevant papers 5-day failure (failure to control bleeding, rebleeding, or death within 5 days of admission) P <0.05 Yes OR = 3.19 (95% CI 1.53–6.67, P = 0.002)* OR = 3.06 (95% CI 1.39-6.68, P = 0.005)# P = 0.047 Yes OR = 2.942 (95% CI 0.884–9.790, P = 0.079) Significant in univariate, but not in multivariate analysis Logistic regressio n analysis Prospective study LC with EVB 185 Amitrano et al. (2012)S16 OR = 7.54 (95% CI 3.25–17.50, P <0.01) No N/A Significant in univariate analysis Logistic regressio n analysis Retrospective study LC with acute EVB 342 Xu et al. (2011)S17 HR = 2.62 (95% CI 1.18–5.79, P = 0.018) Yes HR = 2.734 (95% CI 1.228–6.088, P = 0.014) Significant in both univariate and multivariate analyses Cox regressio n analysis Cohort study LC with active EVB 101 Chen et al. (2012)S18 P = 0.141 No N/A NS in univariate analysis Fisher’s exact test Retrospective study LC with EVB 97 Lee et al. (2009)S19 P = 0.543 No N/A NS in univariate analysis Prospective study LC with recent EVB 383 Amitrano et al. (2012)S20 P = 0.382 No N/A NS in univariate analysis Prospective study LC with recent EVB 83 Amitrano et al. (2012)S20 HR = 3.344 (95% CI 1.613–6.897, P = 0.001) Yes HR = 4.049 (95% CI 1.908–8.621, P <0.001) Significant in both univariate and multivariate analyses Randomized controlled trial LC with acute GVB 95 Hung et al. (2012)S21 Increased 5-day failure Significant in both univariate and multivariate analyses Logistic regressio n analysis Multi-center, prospective cohort study LC with hematemesis and/or melena 291* 207# D’Amico et al. (2003)S15 Bleeding Increased 2-week rebleeding Increased 6-week rebleeding Increased 1-year rebleeding Increased rebleeding during final follow-up 5 Chisquare test Chisquare test Cox regressio n analysis NS Yes OR = 0.17 (95% CI 0.02–1.69, NS) NS in both univariate and multivariate analyses Logistic regressio n analysis Retrospective study LC with acute GVB 83 Wu et al. (2002)S22 HR = 3.19 (95% CI 1.59–6.41, P = 0.001) Yes N/A Significant in univariate, but not in multivariate analysis Cox regressio n analysis Cohort study LC with active EVB 101 Chen et al. (2012)S18 Retrospective study LC with EVB 97 Lee et al. (2010)S23 Prospectively built cohort LC with acute EVB 164 Augustin et al. (2009)S24 Prospective study LC with recent EVB 383 Amitrano et al. (2012)S20 Prospective study LC with recent EVB 383 Amitrano et al. (2012)S20 Mortality Increased 6-week mortality Increased mortality during 1-year follow-up Increased mortality during final follow-up P = 0.102 No N/A NS in univariate analysis Univariate (Cox regressio n analysis) P = 0.01 No (PVT was not included in multivariate analysis) N/A Significant in univariate analysis Logistic regressio n analysis P = 0.382 No N/A NS in univariate analysis P = 0.040 No N/A Significant in univariate analysis HR = 6.024 (95% CI: 2.770–13.158, P <0.001) Yes HR = 3.390 (95% CI 1.499–7.692, P = 0.003) Significant in both univariate and multivariate analyses Cox regressio n analysis Randomized controlled trial LC with acute GVB 95 Hung et al. (2012)S21 HR = 12.6 (95% CI 5.93–26.72, P <0.01) Yes HR = 6.99 (95% CI 2.42–20.16, P <0.01) Significant in both univariate and multivariate analyses Cox regressio n analysis Retrospective study LC with acute GVB 83 Wu et al. (2002)S22 Notes: * data in patients independent of source of bleeding; # Chisquare test Chisquare test data in patients with bleeding from varices. Abbreviations: EVB, esophageal variceal bleeding; GV, gastric variceal bleeding; HR, hazard ratio; LC, liver cirrhosis; N/A, not available; NS, not significant; OR, odds ratio; PVT, portal vein thrombosis. S15. D’Amico, G. & De Franchis, R. Upper digestive bleeding in cirrhosis. Post-therapeutic outcome and prognostic indicators. Hepatology 38, 599–612 (2003). S16. Amitrano, L. et al. The effectiveness of current acute variceal bleed treatments in unselected cirrhotic patients: refining short-term prognosis and risk factors. Am. J. 6 Gastroenterol 107, 1872–1878 (2012). S17. Xu, L., Ji, F., Xu, Q. W. & Zhang, M. Q. Risk factors for predicting early variceal rebleeding after endoscopic variceal ligation. World J. Gastroenterol. 17, 3347–3352 (2011). S18. Chen, P. H. et al. Delayed endoscopy increases re-bleeding and mortality in patients with hematemesis and active esophageal variceal bleeding: a cohort study. J. Hepatol. 57, 1207–1213 (2012). S19. Lee, S. W., Lee, T. Y. & Chang, C. S. Independent factors associated with recurrent bleeding in cirrhotic patients with esophageal variceal hemorrhage. Dig. Dis. Sci. 54, 1128–1134 (2009). S20. Amitrano, L. et al. Splanchnic vein thrombosis and variceal rebleeding in patients with cirrhosis. Eur. J. Gastroenterol. Hepatol. 24, 1381–1385 (2012). S21. Hung., H. H. et al. Efficacy of non-selective beta-blockers as adjunct to endoscopic prophylactic treatment for gastric variceal bleeding: a randomized controlled trial. J. Hepatol. 56, 1025–1032 (2012). S22. Wu, C. Y., Yeh, H. Z. & Chen, G. H. Pharmacologic efficacy in gastric variceal rebleeding and survival: including multivariate analysis. J. Clin. Gastroenterol. 35, 127–132 (2002). S23. Lee, S. W. et al. Independent factors associated with early outcome in Chinese cirrhotic patients after cessation of initial esophageal variceal hemorrhage. J. Clin. Gastroenterol. 44, e123–e127 (2010). S24. Augustin, S. et al. Predicting early mortality after acute variceal hemorrhage based on classification and regression tree analysis. Clin. Gastroenterol. Hepatol. 7, 1347–1354 (2009). 7 Supplementary Table 3 | Incidence and type of TIPS procedure-related complications Study Enrollment period Target population Pati ents ( n) Gaba et al. 2013, USAS25 June 2010— January 2012 LT candidates with LC and partial PVT 4 Luca et al. 2011, ItalyS26 January 2003— February 2010 Han et al. 2011, ChinaS27 December 2001— September 2008 Perarnau et al. 2010, FranceS28 1990— 2004 Van Ha et al. 2006, USAS29 December 1995— December 2003 LC with PVT LC with PVT LC with PVT LC with PVT 70 57 Characteristics of PVT Success rate of TIPS Incidence of procedurerelated complications Type of procedurerelated complications HE Shunt dysfunction Degree: partial PVT (n = 4) 4/4 (100%) 0/4 (0%) N/A Rate of HE: 1/4 (25%) N/A Migration of the stent in the MPV (n = 1) Rate of HE at 12 and 24 months: 27% and 32% Rate of dysfunction at 12 and 24 months: 38% and 85% (bare stent); 21% and 29% (covered stent) 1- and 2-year cumulative rate: 25% and 27% 1- and 2-year cumulative rate: 21% and 32% (bare stent) Acute and chronic HE: 4/27 (14.8%); 2/27 (7.4%) Long-term stenosis: 8/27 (29.7%) Rate of early HE: 1/13 (7.7%) Shunt revision: 1/13 (7.7%) Degree: stenosis<50% (n = 31); >50% (n = 39); CTPV (n = 0) Extension: MPV (n = 67); SMV (n = 55); SV (n = 18); one vessel (n = 18); 2 vessels (n = 34); 3 vessels (n = 18) Degree: stenosis>50% (n = 35); complete occlusion (n = 14); fibrotic cord (n = 8); CTPV (n = 30) Extension: MPV (n = 57); SMV (n = 43); SV (n = 45) 70/70 (100%) 43/57 (75%) 34 Degree: complete PVT (n = 34); CTPV (n = 19) 27/34 (79%) 15 Degree: partially or acute PVT (n = 11); chronic PVT with CTPV (n = 4) Extension: MPV (n = 15); SMV (n = 2) 13/15 (87%) 8 1/70 (1.4%) 4/57 (7%) 6/34 (17.6%) 1/15 (6.7%) Hepatic capsule perforation (n = 2); caudate-lobe hematoma (n = 1); the bile duct was punctured (n = 1) Early thrombosis (n = 3); haemobilia (n = 1); digestive bleeding (n = 1); death from intravascular disseminated coagulopathy (n = 1) An access site hematoma in the neck due to inadvertent carotid puncture (n = 1) Bauer et al. 2006, USAS30 1999— 2005 LT candidates with end-stage liver disease and PVT Blum et al. 1995, GermanyS31 January 1990—March 1994 LC with noncavernomatous PVT 9 Degree: stenosis >50% (n = 7); <50% (n = 2); CTPV (n = 4) Extension: PV alone (n = 1); PV+SMV (n = 4); PV+SV (n = 2); PV+SMV+SV (n = 2); 9/9 (100%) 7 Degree: complete PVT (n = 7); CTPV (n = 0) 7/7 (100%) 0/9 (0%) N/A N/A Shunt occlusion: 1/9 (11.1%) 0/7 (0%) No major TIPS-related complications such as intraperitioneal bleeding or PE N/A Shunt insufficiency: 1/7 (14%) Abbreviations: CTPV, cavernous transformation of the portal vein; HE, hepatic encephalopathy; LC, liver cirrhosis; LT, liver transplantation; MPV, main portal vein trunk; N/A, not available; PE, pulmonary embolism; PV, portal vein; PVT, portal vein thrombosis; SMV, superior mesenteric vein; SV, splenic vein. S25. Gaba, R. C. & Parvinian, A. Transjugular intrahepatic portosystemic shunt for maintenance of portal venous patency in liver transplant candidates. J. Clin. Imaging Sci. 3, 28 (2013). S26. Luca, A. et al. Short- and long-term effects of the transjugular intrahepatic portosystemic shunt on portal vein thrombosis in patients with cirrhosis. Gut..60,846-52 (2011). S27. Han, G. et al. Transjugular intrahepatic portosystemic shunt for portal vein thrombosis with symptomatic portal hypertension in liver cirrhosis. J. Hepatol. 54, 78–88 (2011). S28. Perarnau, J. M., Baju, A., D’Alteroche, L., Viguier, J. & Ayoub, J. Feasibility and long-term evolution of TIPS in cirrhotic patients with portal thrombosis. Eur. J. Gastroenterol. Hepatol. 22, 1093–1098 (2010). S29. Van Ha, T. G. et al. Transjugular intrahepatic portosystemic shunt placement in patients with cirrhosis and concomitant portal vein thrombosis. Cardiovasc. Intervent. Radiol. 29, 785–790 (2006). S30. Bauer, J. et al. The role of TIPS for portal vein patency in liver transplant patients with portal vein thrombosis. Liver Transpl. 12, 1544–1551 (2006). S31. Blum, U. et al. Noncavernomatous portal vein thrombosis in hepatic cirrhosis: treatment with transjugular intrahepatic portosystemic shunt and local thrombolysis. Radiology 195, 153–157 (1995). 9