EMS ppt review
advertisement

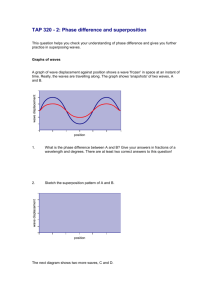
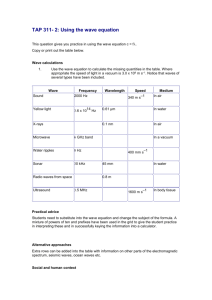
EMS ppt review 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. Electron number equals what number? Light emissions from different elements are ____. Light emissions show no nuclear decay, so from where is energy being emitted? Describe light and waves. What are the key characteristics of waves? Diagram a wave, label the characteristics and how they are measured. Make a chart or table of wave characteristics. Go to www.phet.colorado.edu/sims/wave-on-a-string What are the units for speed? What is the unit for frequency? (YOU MAY USE little f as the symbol) What is symbol for wavelength, name and symbol? Speed of wave= How many Hz in a kHz? What is the scientific notation for kHz? What is the scientific notation for 24.5 kHz? How many Hz is a megahertz? Sci notation? Speed of light = __ in m/s and in km/s? Record both values above in sci notation. What is the mathematical relationship of wavelength to frequency? If frequency is high, wavelength is E = hf or E= hv what does the equation mean? Planck’s constant= numerals and units! What is the wavelength of a photon that carries 2.56 eV energy? (1 eV=1.602 x 10-19 J) List the EM waves in order from longest to shortest wavelengths. List the EM waves in order from lowest to highest energies. List the EM waves in order from greatest to lowest frequencies. What is the difference between the bright line and the visible spectrum? Gas heated gives off Refracting light does what? What is the emission spectra? What are spectral lines supposed to represent? A spectroscope is an object that