Mike Jones WRIT 340 Illumin Article 12 April 2013 From Scotch
advertisement

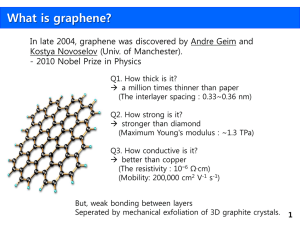
Mike Jones WRIT 340 Illumin Article 12 April 2013 From Scotch Tape to Electric Cars: Graphene, the Wonder Material Although its properties have been hypothesized for years, graphene has only recently been isolated in forms large enough to enable experiments to be conducted. These experiments have revealed that graphene, comprised of carbon atoms arranged in repeated hexagonal rings, possesses some of the amazing properties that had been anticipated. These properties, including strength and conductivity, may enable graphene to be implemented in a wide array of fields, hopefully with much success. Multimedia Suggestions: Source [2]. It is a video that explores Ric Kaner’s research to create a graphene supercapacitor, and can be found on vimeo[dot]com. Mike Jones is a sophomore studying Chemical Engineering with a Biochemical emphasis. He is a tutor at the Viterbi Academic Resource Center and is involved with his fraternity. Introduction Every day, students all across the world pick up their pencils and drag these sticks of carbon graphite across their papers, creating drawings, essays, and the occasional love note. With the naked eye, we merely see the creation of metallic gray lines, but what is happening on the molecular scale? In fact, when the graphite is pressed and dragged against the surface of the paper, small sheets of carbon atoms flake off and adhere to the paper. One single sheet of these carbon flakes is called graphene, whose extraordinary properties just might revolutionize the world of electronic devices. History When we think of pure carbon, three materials traditionally come to mind: diamond, coal, and graphite. These materials are called allotropes, or different structural arrangements of the same atom, and are made possible by carbon’s desire to form four bonds. Diamond contains carbon in a very rigid 3-dimensional crystal lattice, coal contains carbon in random, or amorphous, arrangements, and graphite contains carbon atoms linked together in repeating hexagonal rings that form sheets stacked on top of one another, as seen in figure 1. To put the size of each layer in perspective, there are 3 million layers for every millimeter of thickness of graphite [1]. Fig 1. A model showing layers of graphite split apart to reveal the hexagonally linked carbon atoms. Web Elements For many years, it had been theorized that a single layer of graphite would have very interesting electrical properties, but isolating a sheet large enough to conduct tests proved difficult. Eventually, Andre Geim and Konstantine Novoselov, two scientists from Manchester University, applied a small chunk of graphite to a piece of Scotch tape and found that, after sticking and unsticking the tape to and from the graphite chunk a few times, they could isolate relatively large single layers of graphite. They deposited these layers, an example of which is depicted in figure 2, onto oxidized silicon, ran some tests, and the world of graphene research was born. Geim and Konstantine won the 2010 Nobel Prize in Physics both for the significance of their own experiments as well as for igniting a wave of graphene research across the globe. Fig 2. A model of a large sheet of graphene, emphasizing its near 2dimensionality. Jannik Meyer/Science vol 324, 15 May 2009 Properties This wave revealed just how unique graphene truly is. Graphene is essentially a 2-dimensional material, as the thickness of a carbon atom is absolutely minuscule compared to the area of even a very small sheet. This means that graphene has a very high surface area, 2630 m2/gram [2], and is nearly transparent, absorbing only 2.3% of any visible spectrum light shining at it [1]. However, it is also extremely strong, given the very stable nature of the repeating carbon hexagon rings. It boasts a 2dimensional breaking strength of 42 Newtons per meter, which is 100 times greater than that of a hypothetical steel sheet similar in dimension to graphene [1]. Figure 3 puts this strength into perspective. Fig 3. A hypothetical 1m2 graphene hammock would be able to support a 4kg cat, yet weigh about as much as one of the cat’s whiskers and be nearly invisible [8]. Graphene is also a very good conductor, despite it being a nonmetal. Linear electrical conductance is the reciprocal of linear electrical resistance, which is measured in Ωcm, or ohm*centimeters, and linear thermal conductance is measured in Wm-1K-1, or watts per meter per Kelvin. Graphene conducts electricity at 0.96x10^6 Ω-1 cm-1,which is approximately 50% better than copper [1], and conducts heat at 5000 Wm-1K-1, which outperforms any other known material [3]. Applications Graphene’s special properties – conductivity, surface area, transparency, and strength – have launched it to the forefront of many application-based research initiatives. One field in which graphene could have a large impact is that of biomedicine, which has taken specific interest in graphene oxide, a form of graphene that contains oxygen atoms and other oxygen-containing groups. For biomedical purposes, graphene oxide performs as a framework onto which different “accessory” compounds can be attached in order to carry out various tasks, which include delivering compounds to certain cells of the body. When folic acid and sulfonic acid are attached to graphene oxide, it delivers doxorubicin and camptothecin in order to target and kill human breast cancer cells. When a compound called polyethenimine is attached, graphene oxide can condense and transport DNA and RNA, possibly treating diseases involving genetic defects like cystic fibrosis or Parkinson’s disease. Graphene oxide can even function as a nanoscale biosensor to detect various molecules in the body, and can provide a “scaffolding” on which new tissues can be grown [2]. These are just a few of the roles for graphene that biomedical scientists are exploring. However, whatever splash graphene may make in the biomedical realm, it is a proverbial drop in the bucket compared to the impact it could have on the future of electronic devices. Currently, circuits, motors, screens, or anything else in an electronic device that needs energy is powered by either a battery or a capacitor. A battery, such as the lithium-ion type in a cellular or smartphone, uses an electrochemical reaction to create electric power [4], whereas a capacitor, such as those in radio tuning circuits, delivers energy by storing electrons between two oppositely charged plates and then releasing them [5]. Currently, batteries have a high capacity for energy, but long charge times, and capacitors have very quick charge times, but very low energy capacities. With its remarkable electrical properties, graphene seeks to combine the best of both into what is called a supercapacitor—a capacitor that has the energy capacity of a battery, but the charge time of a capacitor. The graphene supercapacitor is still in the research phase, and many variations are being tested, including one that uses hybridized ribbons of vanadium oxide and graphene. This Rice University-developed material can reach full charge in a mere twenty seconds, and after 1,000 charge and discharge cycles, can still be charged to 90% of its original capacity [6]. UCLA chemistry professor Ric Kaner has worked with pure graphene, layering it with simple plastic and electrolytes to create a capacitor that can store the same amount of energy as a traditional battery, but can go through a discharge-charge cycle one hundred to one thousand times faster [7]. Impact on the Future Imagine that when the warning appears on a laptop indicating 5% battery life remaining, the owner could plug the laptop into an outlet for a mere five minutes and the laptop’s graphene supercapacitor would be fully charged. This would undoubtedly increase the convenience of many people’s lives. However, implementing the same technology into the transportation sector, however, could be revolutionary. Electric family cars, taxis, buses, and possibly even electric semi-trucks could fully recharge during a one hour lunch break instead of the current commitment of eight to nine hours overnight [8]. It is by no means an immediate reality, but graphene has the potential to serve as the foundation for the transportation, electronic device, pharmaceutical, and materials science sectors, as seen in figure 4. And perhaps, graphene could revolutionize fields we have not yet considered. Limitations Unfortunately, this graphene-embedded future may not be realized for quite some time. As with any engineered chemical product, graphene is bounded by three primary parameters: cost, production scale, and quality of the product. And, unfortunately, there is no production method currently available that can fulfill all three parameters, which is impeding the widespread implementation of graphene. In addition to the various fields of application, figure 4 also portrays the pros and cons of various graphene production methods. CKMNT Fig 4: The current methods of producing graphene sheets and their respective pros, cons, and applications. Conclusion There is no debate over just how extraordinary graphene is. It is the first 2-dimensional material to be heavily studied, boasting transparency, strength, and conductivity, and despite the limitations that may exist currently, the applications of graphene in biomedicine, electronics, and other fields are promising enough to push forward with any and all research and industrial initiatives. It wouldn’t be the first material for which scientists and engineers needed to overcome significant obstacles in its development, and like many others graphene could be well worth the effort. [1] “The Nobel Prize in Physics 2010: Background Information.” Nobel Prize. [On-line]. [29 Mar 2013] [2] H Shen, et al. (2012). Biomedical applications of graphene. Theranostics. pp.283–94. [3] “The Nobel Prize in Physics 2010: Information for the Public.” Nobel Prize. Internet. [29 Mar 2013]. [4] Brain, Marshall, Charles W. Bryant, and Clint Pumphrey. "How Batteries Work." HowStuffWorks. Internet. [1 Apr. 2013]. [5] Brain, Marshall, and Charles W. Bryant. "How Capacitors Work." HowStuffWorks. Internet. [1 Apr. 2013]. [6] Mike Williams. "Hybrid Ribbons a Gift for Powerful Batteries." Rice University News. [Internet]. [2 Apr. 2013]. [7] Jennifer Marcus. “Researchers Develop Graphene Supercapacitor Holding Promise for Portable Electronics." UCLA Engineering. Internet. [1 Apr. 2013]. [8] “Fuel Economy.” Internet: http://www.fueleconomy.gov [02 Apr. 2013]. [9] Brian G. Davis, Director, “The Super Supercapacitor”. [Video]. Focus Forward Films. [21 Oct. 2012].