Answers to rates of reaction Practice Test
advertisement

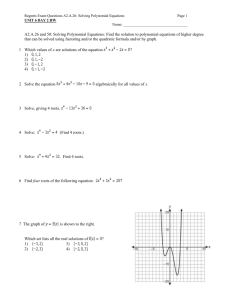
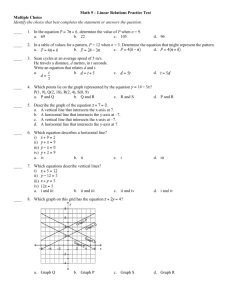
Rates of reactions Answer Section MULTIPLE CHOICE 1. ANS: D DIF: average REF: K/U LOC: EC 2.01 2. ANS: C DIF: average REF: K/U LOC: EC 2.01, EC 1.03 3. ANS: A DIF: average REF: K/U LOC: EC 2.01 4. ANS: C DIF: average REF: K/U LOC: EC V.01, EC 2.06 5. ANS: D DIF: average REF: K/U LOC: EC V.01, EC 2.06 6. ANS: E DIF: average REF: K/U LOC: EC V.01, EC 2.06 7. ANS: B DIF: easy REF: K/U LOC: EC V.01, EC 1.04 8. ANS: E DIF: average REF: I LOC: EC 2.06, EC V.01 9. ANS: A DIF: easy REF: K/U LOC: EC 2.06 10. ANS: E DIF: easy REF: K/U LOC: EC 1.06 11. ANS: D DIF: average REF: K/U LOC: EC 1.04 12. ANS: A DIF: easy REF: K/U, I LOC: EC 1.06 13. ANS: A DIF: average REF: K/U, I LOC: EC 1.06 14. ANS: D DIF: easy REF: K/U LOC: EC 1.04 15. ANS: B DIF: easy REF: K/U LOC: EC 1.04 16. ANS: A DIF: average REF: K/U LOC: EC 1.04 17. ANS: D DIF: average REF: K/U LOC: EC 2.06 18. ANS: D DIF: easy REF: K/U LOC: EC 1.05 19. ANS: A DIF: easy REF: K/U LOC: EC 1.05 20. ANS: B DIF: easy REF: K/U LOC: EC 1.05 21. ANS: C DIF: average REF: I, K/U LOC: EC 1.04 SHORT ANSWER 22. ANS: An elementary reaction involves a single molecular event, such as a simple collision between atoms, molecules, or ions. It may be one step in a series of steps that make up a reaction mechanism. DIF: 23. ANS: easy REF: K/U LOC: EC 1.06 The mechanism of a reaction is the series of steps that make up the overall reaction. Each step involves an elementary reaction: a single molecular event, such as a simple collision between particles. DIF: easy REF: K/U LOC: EC 1.06 24. ANS: A bimolecular elementary reaction is an elementary reaction in which two particles collide and react. DIF: easy REF: K/U LOC: EC 1.06 25. ANS: A catalyst does not affect the enthalpy change of a chemical reaction. A catalyst increases the rate of a chemical reaction by decreasing the activation energy required, but it does not affect the difference in enthalpy between products and reactants. DIF: easy REF: K/U, C LOC: EC 1.04 26. ANS: This is a first-order reaction. (The sum of the exponents is 1 DIF: easy REF: K/U easy REF: K/U 1.) 2 3.) LOC: EC 1.03 27. ANS: This is a third-order reaction. (The sum of the exponents is 1 DIF: 0 LOC: EC 1.03 28. ANS: The rate of dissolution of the lump of sugar can be increased by - crushing the lump of sugar in order to increase the surface area of sugar exposed to the tea - stirring the tea in order to increase the amount of contact between undissolved sugar and tea that has a low concentration of dissolved sugar molecules DIF: average REF: K/U, C LOC: EC 1.04 29. ANS: Particles in the gaseous state move much faster than particles in the solid or liquid state. They have a higher kinetic energy. This allows a greater percent of all collisions to be successful. Because gas reactants exist as elementary molecules or atoms, the bonds that hold the molecules or atoms together in the liquid or solid state do not have to be broken. Therefore, lower activation energy is required. The rapid movement of individual molecules or atoms provides maximum surface area for interaction. All these factors combine to make gaseous reactions extremely fast. DIF: difficult REF: K/U, C LOC: EC 1.04 30. ANS: Ratio of moles NO to moles NO2 3:1 \Rate of loss of NO 3 Rate of production of NO2 3 [0.035 mol/(L s)] 0.105 mol/(L s) The rate of loss of NO is DIF: average 0.105 mol/(L s). REF: I 31. ANS: Since this is a first order reaction, LOC: EC V.01, EC 2.01 Reaction rate k [X]1 (0.25 s 1)(0.05 mol/L) 1.25 10 2 mol/(L s) -2 10 mol/(L s). The rate of the reaction is 1.25 DIF: average REF: I LOC: EC 1.03 32. ANS: The following two requirements must be met for a reaction to occur: - A collision must occur between reactant particles that have the correct orientation. - Reactants must have sufficient collision energy to be able to overcome the activation energy barrier. DIF: easy REF: K/U LOC: EC 1.04 33. ANS: An activated complex is a chemical species that exists at the transition state, or change-over point, of a reaction. The activated complex is neither reactant nor product. It has partial bonds between aligned reactants and is highly unstable. DIF: average REF: K/U LOC: EC 1.04 34. ANS: a) mass of calcium carbonate, pH, volume of carbon dioxide gas if pressure constant, change in pressure if volume constant, change in electrical conductivity b) change in pressure if constant volume, change in volume if constant pressure c) pH, mass of zinc, volume of hydrogen at constant pressure or pressure due to hydrogen at constant volume, change in electrical conductivity DIF: average REF: I LOC: EC 2.06 35. ANS: a) pH, volume of CO2(g) at constant pressure or pressure due to CO2(g) at constant volume, change in electrical conductivity b) change in colour due to the iodine in the water c) change in volume at constant pressure or change in pressure at constant volume due to change in the number of moles, colour change due to N2O4(g) DIF: average REF: I LOC: EC 2.06 36. ANS: The equation has only CH3CHO(g) as a reactant, and the molar coefficient is one. Therefore, this is a first-order reaction. For a first-order reaction, 0.693 k 0.693 (8.6 8.06 s The half-life is 8.06 s. DIF: difficult 10 REF: I 2 s 1) LOC: EC 1.03 37. ANS: The equation has only N2O5(g) as a reactant, and the molar coefficient is one. Therefore, this is a first-order reaction. For a first-order reaction, 0.693 k 0.693 (6.0 1.16 103 s 19.3 min The half-life is 19.3 min. DIF: difficult 10 4 s 1) REF: I LOC: EC 1.03 38. ANS: A homogeneous catalyst exists in the same phase as the reactants. An example is the use of ZnCl2(aq) as the catalyst in the reaction between aqueous solutions of (CH3)2CHOH and HCl. A heterogeneous catalyst exists in a phase that is different from the phase of the reaction it catalyses. An example is the use of platinum or palladium to catalyse the hydrogenation of alkenes. DIF: easy REF: K/U LOC: EC 1.04 PROBLEM 39. ANS: a) b) c) The average rate is determined by drawing the secant to join the beginning of the required time interval to the end of this time interval. The average rate is equal to the slope of the secant. d) The instantaneous rate is determined by drawing the tangent to the curve at the time for which the instantaneous rate is required. The instantaneous rate is equal to the slope of the tangent. DIF: average REF: K/U LOC: EC 2.06 40. ANS: a) The reaction is endothermic. The enthalpy of the products is higher than the enthalpy of the reactants. This means that energy is absorbed as the reaction proceeds. b) A c) C d) B DIF: easy REF: K/U LOC: EC 1.05 average REF: K/U LOC: EC 1.05 easy REF: K/U LOC: EC 1.05 41. ANS: DIF: 42. ANS: DIF: 43. ANS: a) b) Slope of the tangent at 20 min Slope of the tangent at 180 min DIF: average REF: I (0.31 mol/L 0.21 mol/L) 0.001 mol/L/min (0.20 mol/L 0.14 mol/L) 0.0005 mol/L/min 92 min 130 min LOC: EC 2.06 44. ANS: a) Draw a tangent to the curve at A. Then calculate the slope of the tangent to the curve. b) The rate of reaction at B is less than the rate of reaction at A. As the reaction proceeds, the concentration of H2O2 decreases because it is used up in the reaction. This decrease in concentration results in fewer successful collisions and hence a reduced reaction rate. c) DIF: average REF: C, K/U LOC: EC 2.06 45. ANS: a) b) A 3B E G c) Hr H1 H2 H3 Hr 42 21 42 21 kJ/mol d) The rate-determining step is the first step because it has the highest activation energy barrier. DIF: 46. ANS: average REF: K/U, I LOC: EC 1.05 a) B b) A: A lighted match is needed to ignite the candle, hence a high activation energy barrier is needed. Since the reaction is self-sustaining, however, it must be exothermic. DIF: average REF: K/U LOC: EC 1.05 47. ANS: a) A, B, C b) A: The rusting of iron takes place without the application of energy from outside the system, and yet the rusting of iron is a fairly slow reaction. This is consistent with an activation energy barrier that is not too small (which would be a fast reaction) and a barrier that is not too high (which would be a very slow or non-spontaneous reaction). Thus, diagram A represents this reaction. DIF: average REF: K/U LOC: EC 1.05, EC 3.04 48. ANS: a) C: It has the lowest activation energy barrier, therefore a greater percent of the particles have sufficient energy for a successful collision. b) B: Two solids have the highest activation energy barrier because more bonds have to be broken to form products. DIF: average REF: K/U LOC: EC 1.05 49. ANS: a) B: It has the lowest activation energy barrier therefore a greater percent of the particles have sufficient energy for a successful collision. b) C: The ionization of sulfuric acid takes place very quickly, with the release of a large amount of energy. Diagram C has a low activation energy barrier, with a subsequent large negative enthalpy change. Thus, diagram C best matches the ionization of sulfuric acid. DIF: average REF: K/U LOC: EC 1.05 50. ANS: Students might include a diagram to show that breaking a solid into smaller pieces provides a greater surface area. Students might also show, on the same diagram, that more particles of the solvent (or other reactants) collide more frequently with the increased surface area. The increase in surface area results in an increase in the number of collisions. According to collision theory, this should result in an increase in reaction rate. DIF: 51. ANS: average REF: K/U, C LOC: EC 1.04 As the Maxwell-Boltzmann distribution diagram shows, the number of particles with energy that is equal to or greater than the activation energy is increased. This increases the number of successful collisions, thereby increasing the reaction rate. DIF: average REF: K/U, C LOC: EC 1.04 52. ANS: As shown in the diagram, an increase in the number of particles of one or more of the reactants increases the probability of collision. The increase in the number of collisions per unit of time results in an increase in reaction rate. DIF: average REF: K/U, C LOC: EC 1.04 53. ANS: A catalyst provides an alternate reaction pathway, or reaction mechanism, that has a lower activation energy barrier. Lowering the activation energy barrier results in an increase in the number of particles with sufficient energy to have a successful collision. This results in an increase in the reaction rate. DIF: average REF: K/U, C LOC: EC 1.04 54. ANS: a) Since doubling the concentration of hydroxide ions causes the reaction rate to double, this reaction is first order with respect to hydroxide ions. b) Doubling the concentration of both reactants causes the reaction rate to quadruple, and doubling the hydroxide ion concentration causes the reaction rate to double. Thus, doubling the concentration of bromoethane would also cause the reaction rate to double. Therefore, this reaction is first order with respect to the bromoethane. c) The overall reaction order is 2(1 . DIF: 55. ANS: a) Rate b) Rate difficult REF: I, C LOC: EC 1.03 k[A][B] k[A][B] The rate constant is 3.33 10 1 s 1(mol/L) 1 for this reaction. c) The rate constant is temperature specific because any change in temperature results in a change in the reaction rate even though there has not been a change in reactant concentrations. DIF: average REF: I LOC: EC 1.03 56. ANS: a) The overall reaction order is first order. (The sum of the exponents is 1 0 1.) b) The reaction is first order with respect to (CH3)3CBr. c) The reaction order is not dependent on the concentration of water. d) Since the reaction order is not dependent on the concentration of water, doubling the concentration of water has no effect on the reaction rate. e) If the concentration of (CH3)3CBr is halved, the reaction rate is also halved, since the reaction rate is first order with respect to the concentration of (CH3)3CBr. DIF: average REF: K/U, I, C LOC: EC 1.03 57. ANS: a) Rate of formation of H2 Initial rate for decomposition of PH3 PH3 2.0 10 4 mol/(L s) 3.0 10 4 mol/(L s) Mole ratio of H2 to b) Rate of formation of P4 DIF: 58. ANS: Step 1: Step 2: DIF: average Initial rate for decomposition of PH3 PH3 2.0 10 4 mol/(L s) 3.3 10 5 mol/(L s) REF: I LOC: EC 1.03 O3(g) O2(g) O(g) O3(g) O(g) 2O2(g) 2O3(g) 3O2(g) average REF: I LOC: EC 1.06 59. ANS: Balanced equation: 2NO(g) O2(g) 2NO2(g) Step 1: NO(g) O2(g) NO3(g) Step 2: NO3(g) NO(g) 2NO2(g) 2NO(g) O2(g) 2NO2(g) DIF: 60. ANS: Step 1: Step 2: DIF: average REF: I LOC: EC 1.06 SO2(g) O2(g) SO3(g) O(g) SO2(g) O(g) SO3(g) 2SO2(g) O2(g) 2SO3(g) difficult REF: I LOC: EC 1.06 Mole ratio of P4 to 61. ANS: a) b) and c) Reaction rate Slope of tangent to curve at 100 s 2.5 10 4 mol/(L•s) The reaction rate at 100 s is 2.5 10 4 mol/(L•s). DIF: average REF: I LOC: EC 2.06 62. ANS: Compare trials 1 and 2: Doubling the concentration of H2(g) causes the reaction rate to double. Therefore, the reaction rate is first order with respect to the concentration of H2(g). Compare trials 1 and 3: Doubling the concentration of NO(g) causes the reaction rate to quadruple. Therefore, the reaction rate is second order with respect to NO(g). Reaction rate k[H2(g)]1[NO(g)]2 DIF: difficult REF: I LOC: EC 1.03 63. ANS: Compare trials 1 and 2: Doubling the concentration of SO2Cl2(g) causes the reaction rate to double. Therefore, the reaction rate is first order with respect to the concentration of SO2Cl2(g). Comparing trials 1 and 3 verifies this conclusion. The tripling of the concentration of SO2Cl2(g) causes the reaction rate to triple. Reaction rate k[SO2Cl2 (g)]1 DIF: average REF: I LOC: EC 1.03 64. ANS: Compare trials 1 and 2: Doubling the concentration of cyclopropane causes the reaction rate to double. Therefore, the reaction rate is first order with respect to the concentration of cyclopropane. Comparing trials 1 and 3 verifies this conclusion. Tripling the concentration of cyclopropane causes the reaction rate to triple. Reaction rate k[cyclopropane]1 DIF: average REF: I, MC LOC: EC 1.03 65. ANS: Compare trials 1 and 2: Doubling the concentration of OCl causes the reaction rate to double. Therefore, the reaction rate is first order with respect to the concentration of OCl . Compare trials 1 and 3: Doubling the concentration of I also causes the reaction rate to double. Therefore, the reaction rate is first order with respect to I . Reaction rate k[OCl ]1[I ]1 DIF: difficult REF: I LOC: EC 1.03 66. ANS: Reaction rates play an important role in our everyday lives, from the decay of food to the way we start a fire. Foods stored at room temperature decay quite quickly. Milk sours in a day, while fruit goes rotten after a few days. The rates of both these reactions can be slowed down by a decrease in temperature. Hence, we store food in a refrigerator or (for longer periods of time) in a freezer, in order to keep it fresh longer. We start a fire using shavings and small twigs, because increased surface area results in an increased reaction rate, which allows us to start the fire much more easily. A catalyst, such as palladium, is put in the fire heads of portable heaters, which are used in confined spaces, such as tents. The catalyst allows the reaction to take place more readily, thus reducing the need for an open flame to provide the energy to keep the reaction self-sustaining. Students may provide a variety of examples, based on their personal experiences and the examples discussed in class. DIF: average REF: C, MC LOC: EC 3.04, EC 1.04 67. ANS: Catalysts are important in industries because they make some reactions, which might otherwise be too expensive, efficient and financially possible. Catalysts also help to increase the rate at which products can be produced, and subsequently sold, in order to be profitable. For example, platinum is used in the hydrogenation of unsaturated fats and oils in the food industry. Oil is converted to a solid when the process of hydrogenation reduces the number of unsaturated hydrocarbons (oil to margarine). Platinum or palladium is used in the industrial process of hydrogenating ethene. Students may provide other examples to illustrate their answers. DIF: average REF: C, MC LOC: EC 3.03, EC 1.04 68. ANS: a) Enzymes are enormous protein molecules that catalyse biological processes. Enzymes are important because they make possible many functions in the human body, such as digestion and metabolism. b) One example of an enzyme that is important in the human body is lactase, which catalyses the breakdown of lactose, a sugar in milk. People who are lactose-intolerant are usually missing lactase and have to take supplements that contain lactase. Another example is amylase, an enzyme in saliva. Amylase is important in the breakdown of carbohydrates into simple sugars in digestion. DIF: average REF: C, MC LOC: EC 3.04 69. ANS: The rate of a chemical reaction involving a solid reactant is affected by the amount of surface area that is exposed to the other reactant(s). Grain dust has a much larger surface area than grain. Thus, if a reaction is initiated, it will proceed at a very fast rate, because a large number of collisions can quickly occur between the grain dust and oxygen. DIF: easy REF: C, MC LOC: EC 1.04 70. ANS: Fires can be started more easily using small twigs, because the increase in surface area results in an increased reaction rate. DIF: easy REF: C, MC LOC: EC 1.04 71. ANS: Food that is stored at room temperature decays quite quickly. Milk sours in a day, and fruit goes rotten after a few days. The reaction rates of both these processes can be slowed down by a decrease in temperature. Hence, we store food in a refrigerator or (for longer periods of time) in a freezer, in order to keep it fresh longer. Although a decrease in temperature decreases the reaction rate, it does not stop the reaction. Thus, food that is left in a refrigerator will eventually spoil. DIF: average REF: C, MC LOC: EC 3.04, EC 1.04