Physics 8: The Electromagnetic Spectrum
advertisement

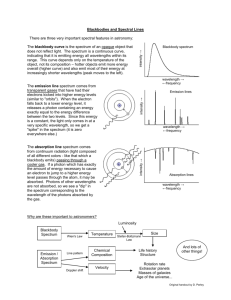
The Electromagnetic Spectrum Introduction Electromagnetic waves are produced when charged particles are accelerated or change their energy state. The light you see is a form of electromagnetic radiation, typically produced when electrons lose energy. The “lost” energy is emitted in the form of electromagnetic radiation. Radio waves, TV waves, microwaves, X-rays, gamma rays, infrared rays, and ultraviolet rays are also examples of electromagnetic radiation. Visible light is only a small part of the electromagnetic spectrum. In general, electromagnetic waves that are more energetic than visible light (to the right of visible light in the figure above) are damaging to humans. There are exceptions; microwaves can interact with water molecules in your tissues to cook you from inside out. In today’s lab, you will use your eyes as your “radiation detector,” so let’s focus on the visible spectrum. ROY G BIV! “White” is actually what you perceive when “all” colors are present (or—in the case of a monitor—red, green, and blue pixels are all fully illuminated). “Black” is what you perceive when no light is present (or—in the case of a monitor—red, green, and blue pixels are not illuminated at all) To study the spectrum of a light source, we need a device that shows all the colors present. You are familiar with prisms, which separate white light into “rainbow” colors. For today’s lab, you will use diffraction gratings, which perform the same task. On the left, you see “magenta” light being “split” into blue and red. On the right, you see the spectrum of light from the gas inside the glowing “white” tube. I will provide you with a sheet of diffraction grating, a “spectroscope” which uses a small piece of diffraction grating, and several light sources. Some of the sources produce light by passing a high-voltage current through a tube filled with a gas. The current “excites” electrons in the gas atoms by giving them extra energy. The electrons quickly get “tired” of being “excited,” and emit light in the process of returning to their normal energy state. Because electrons in atoms exist in only well-defined energy states, only light corresponding to the energy difference between those well-defined states is emitted. If all that sounds too complicated, just think of the spectrum as being the “fingerprint” of an atom. Each atom, molecule, chemical, compound, etc., has its own unique fingerprint. Has anybody been to the sun? How do we know what is in the sun? One more thing. A continuous spectrum is produced by an object emitting all colors. An emission spectrum is produced by a glowing gas. An absorption spectrum is produced when white light passes through a cold gas. If the gas is the same, emission and absorption spectra are the “inverse” of each other, as pictured above. As an example, below is the spectrum of xenon gas (rather complex!). Your lab today involves observing and describing a variety of spectra. I will provide at least one unknown. To complete today’s lab, complete the observation sheet, identify the unknowns, and hand in the observation sheet with your name on it. You may work in groups of up to three, so three names may appear on your observation sheet. Some web sites to visit: http://cse.cosm.sc.edu/hses/StarEvol/pages/morelec.htm http://www.colorado.edu/physics/2000/waves_particles/ http://hyperphysics.phy-astr.gsu.edu/hbase/phyopt/grating.html http://amazing-space.stsci.edu/resources/explorations/light/ems-frames.html http://hyperphysics.phy-astr.gsu.edu/hbase/ems3.html#c2 The Electromagnetic Spectrum Lab Activities Part 1. Diffraction grating. I will provide each of you with a sheet of diffraction grating material. I will suggest several activities that will help you observe and understand its properties. Part 2. Observations. Observe each of the light sources with your spectroscope. Sketch the spectrum you observe and state whether the spectrum appears to be an absorption spectrum, an emission spectrum, or a continuous spectrum. See the notes below. Warning: if you experience visual problems or a headache while doing this lab, immediately stop and discuss the problem with your lab instructor. When you get to the laser, do not look directly at the laser. Source Sketch of Spectrum Emission/Abs/Cont? Mercury _____________________________ ________________ Sodium _____________________________ ________________ Hydrogen _____________________________ ________________ Helium _____________________________ ________________ Krypton _____________________________ ________________ Argon _____________________________ ________________ Neon _____________________________ ________________ Solar Spectrum1 _____________________________ ________________ Fluorescent Tube2 _____________________________ ________________ Incandescent Bulb _____________________________ ________________ Computer Monitor3 _____________________________ ________________ HeNe Laser4 _____________________________ ________________ Unknown _____________________________ Identify: _______________ Unknown _____________________________ Identify: _______________ Notes. 1. Attempt to view the solar spectrum. You will probably observe a continuous spectrum because the thousands of absorption lines in the sun are too narrow to see with your spectroscope. If there are clouds, you will not be able to observe the solar spectrum with your spectroscopes. Do not look directly at the sun! Look at a bright reflection. 2. Observe one of the fluorescent tubes either in the laboratory or in the hall. 3. Bring up a screen that is mostly white (e.g., blank document in Microsoft Word), and observe the white with your spectroscope. Keep in mind that monitors are usually RGB monitors. 4. Lasers are especially useful because their light has a single wavelength and is coherent (all of the light waves are in phase). Point the laser at the wall and observe the dot on the wall through your spectroscope. Never look directly at the laser!