Supplementary Figure Legends (doc 110K)
advertisement

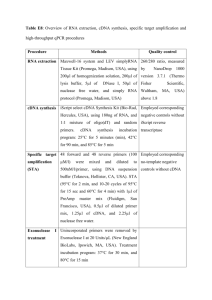
SUPPLEMENTARY FIGURE LEGENDS FIGURE S1: E2F family proteins are NEDDylated in cells. A–F. HEK293T cells were transfected with GFP along with a plasmid encoding either His6-tagged ubiquitin (His-Ub), NEDD8 (His-NEDD8), or the corresponding empty vector, along with a plasmid for each protein as indicated. Total cell lysates (bottom panels) as well as His6-tagged proteins purified there from (top panels) were subjected to immunoblot analysis with antibodies to the indicated proteins (bottom panels) or to the test protein (top panels). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and GFP were examined as loading and transfection controls, respectively. 3×FLAG- pRB and HA-HAUSP was detected with antibodies to FLAG and HA, respectively. Asterisks represent nonspecifically precipitated proteins. G. HEK293T cells were transfected with a plasmid encoding 3×FLAG-tagged forms of ubiquitin or NEDD8, after which cell lysates were prepared and subjected to immunoblot analysis with antibodies to FLAG. H. HEK293T cells were transfected with a plasmid encoding His-NEDD8, or the corresponding empty vector. 18 h after transfection, cells were treated with 1 μM MLN4924 and incubated for 6 h. Cells were analyzed as described in A-F. MLN4924 was purchased from Active Biochemicals Co. FIGURE S2: E2F family proteins are deNEDDylated by SENP8. A–D. HEK293T cells were transfected with plasmids encoding GFP and His6-NEDD8, along with a plasmid encoding each protein as indicated, and either a plasmid encoding HA-tagged SENP8 or the corresponding empty plasmid. Cell lysates and His6-tagged proteins purified from there were then subjected to immunoblot analysis with antibodies to the indicated proteins. Asterisks indicate nonspecifically precipitated proteins. E. HEK293T cells were transfected with a plasmid encoding 3×FLAG-tagged forms of ubiquitin or NEDD8 and with a plasmid for HA-SENP8 as indicated. Cell lysates were subjected to immunoblot analysis with antibodies to FLAG.. FIGURE S3: NEDDylation of E2F1 is attenuated in response to DNA damage in a SENP8-dependent manner A. H1299 cells were transfected first with SENP8 or control siRNAs (10 nM) for 24 h and then with plasmids for His6-NEDD8, for GFP, and for E2F1 for 24 h. Cell lysates as well as His6-tagged proteins purified from there were subjected to immunoblot analysis with antibodies to E2F1. The asterisk indicates a nonspecifically precipitated protein. B. U2OS cells were transfected first with SENP8 or control siRNAs (5 nM) for 24 h and then with plasmids for His6-NEDD8, for GFP, and for E2F1 for 24 h. The cells were then incubated in the absence or presence of 2 μM doxorubicin for 12 h, after which cell lysates as well as His6-tagged proteins purified from there were subjected to immunoblot analysis with 1 antibodies to E2F1. The asterisk indicates a nonspecifically precipitated protein. FIGURE S4: SENP8 is dispensable for E2F1’s ability to promote cell cycle. SAOS-2 cells stably expressing ER-E2F1 were transfected with SENP8 or control siRNAs (5 nM) for 48 h and then incubated in the absence or presence of 500 nM 4-OHT for 12 h. Cells were then collected and analyzed for cell cycle profile by flow cytometry. 2 SUPPLEMENTARY MATERIALS Plasmids pT7-7-His6-Ubc12 and pT7-7-HA-His6-NEDD8GG were kindly provided by K. Iwai; pT7-T-UbcH5b and pGEX6P1-Ub by K. Tanaka; pRc/CMV-HA-E2F1, -E2F2, -E2F3, and -E2F4 as well as the E2F2-luciferase reporter construct by A. Zubiaga; pcDNA3.1-3×FLAG-RB by M. Kitagawa; pCI-FLAG-PCAF by K. Ishiguro; pcDNA3-HA-YY1 by M. Miyagishi; pcDNA3-HA-p300 by Y. Mori; and pCG-N-HA-Jab1 by J. Kato. The pCAGEN-His6 vector was constructed by ligating the hybridized oligonucleotides 5′-AATTATGGGGGGTTCTCATCATCATCATCATCATGAATTCC-3′ and 5′-TCGAGGAATTCATGATGATGATGATGATGAGAACCCCCCAT-3′ into the EcoRI-XhoI sites of pCAGEN. NEDD8 cDNA was amplified from HeLa cell cDNA and cloned in pCAGEN-His6 or in frame with the 3×FLAG tag in pCAGEN. SENP8 cDNA was amplified from HeLa cell cDNA and cloned in frame with the HA or 3×FLAG tags in pCAGEN. HAUSP cDNA was amplified from HeLa cell cDNA and cloned in frame with the HA tag in pCAGEN. DP1 cDNA was amplified from HeLa cell cDNA and cloned in frame with the FLAG tag in pcDNA3.1 (Invitrogen). A DNA fragment encoding chimpanzee ubiquitin was isolated from pGEX6P1-Ub and subcloned into the EcoRI-XhoI sites of pCAGEN-His6 or in frame with the 3×FLAG tag in pCAGEN. MCPH1 cDNA was amplified from HeLa cell cDNA and cloned in frame with the 3×FLAG tag in pCAGEN. Slc7a1 cDNA was amplified from mouse embryonic fibroblast cDNA and cloned in frame with the HA tag in pcDNA 3.1. A DNA fragment encoding human E2F1 was isolated from pRc/CMV-HA-E2F1 and subcloned into the BamHI-NotI sites of pcDNA3 (Invitrogen) or of pCMV/SV2-GAL4DBD (kindly provided by H. Sasaki). Mutagenesis was performed by PCR according to standard protocols. A DNA fragment corresponding to amino acids 1 to 368 of E2F1 was amplified by PCR from pcDNA3-E2F1 and subcloned together with DNA encoding the TAD of VP16 into the BamHI-EcoRI sites of pCS2. The p73 gene promoter (nucleotides –4091 to +438 relative to the transcription start site) was amplified by PCR from the human genome BAC clone RP5-1092A11 (BACPAC Resources) and cloned into the MluI-NheI sites of pGL3-basic (Promega). 3 Antibodies Antibody FLAG (M2) M2 Affinity Gels E2F1 (C-20) E2F2 (C-20) E2F3 (C-18) E2F4 (A-20) DP1 (K-20) GFP (598) HAUSP Cleaved PARP (#9541) SENP8 p53 (Ab-8) HA (3F10) GAPDH (6C5) pRB (G3-245) Caspase-3 (19) p73 (5B429) Cyclin E (HE12) NEDD8 (Y297) Company Sigma Sigma Santa Cruz Biotechnology Santa Cruz Biotechnology Santa Cruz Biotechnology Santa Cruz Biotechnology Santa Cruz Biotechnology MBL Bethyl Cell Signaling Enzo Life Science Calbiochem Roche Chemicon BD Pharmingen BD Pharmingen Imgenex Santa Cruz Biotechnology Epitomics Primer sequences for real-time PCR Gene Forward PPIA TTGCTGACTGTGGACAACTC p73 CAGACAGCACCTACTTCG NOXA CGAAGATTACCGCTGGC SENP8 TGATTCCCATAGCAGGAGC CCNE1 AGCCTTGGGACAATAATGC CDC25A CCTACTGATGGCAAGCG E2F2 CCTGACCTCAAGTGATCC AXIN2 TGGACCAAGTCCTTACACTC Reverse ACAAAGATTCTAGGATACTGCGA GACGTCCATGCTGGAATC GCAACAACAACAATGCACT GGCCAGTTTGTCTCCTT GCACGTTGAGTTTGGGTAA GGCGATCTCTCTCTCTCACATA GTCTACCTGGTCCCTAAAGAAA GCAAACCAGAAGTCTAAGGTATC SUPPLEMENTARY METHOD Quantification of Proteins The bands resulting from immunoblotting were quantified with the use of ImageQuant LAS and ImageQuant TL software (GE Healthcare) according to the manufacturer’s protocol. 4