Application Form
advertisement
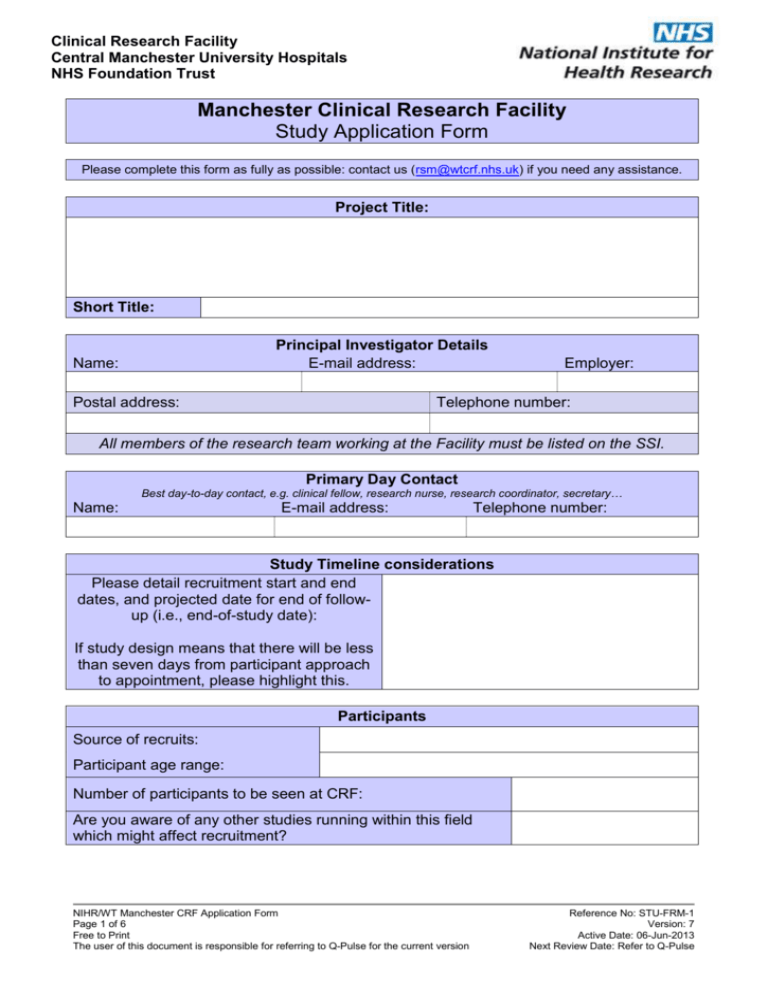
Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust Manchester Clinical Research Facility Study Application Form Please complete this form as fully as possible: contact us (rsm@wtcrf.nhs.uk) if you need any assistance. Project Title: Short Title: Principal Investigator Details E-mail address: Name: Postal address: Employer: Telephone number: All members of the research team working at the Facility must be listed on the SSI. Primary Day Contact Best day-to-day contact, e.g. clinical fellow, research nurse, research coordinator, secretary… Name: E-mail address: Telephone number: Study Timeline considerations Please detail recruitment start and end dates, and projected date for end of followup (i.e., end-of-study date): If study design means that there will be less than seven days from participant approach to appointment, please highlight this. Participants Source of recruits: Participant age range: Number of participants to be seen at CRF: Are you aware of any other studies running within this field which might affect recruitment? NIHR/WT Manchester CRF Application Form Page 1 of 6 Free to Print The user of this document is responsible for referring to Q-Pulse for the current version Reference No: STU-FRM-1 Version: 7 Active Date: 06-Jun-2013 Next Review Date: Refer to Q-Pulse Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust Value Added by Use of MCRF: Specialist clinical support One stop shop for participants Environment Specialist equipment Administration/Appointments support Other Please explain how using the Facilities will improve delivery of the study: Study Type First Area Second Area Second Area First Area Second Area First Area Phase I Please select the most appropriate project type: (If 'Other', please give details) Please select the two most appropriate principal research areas: Blood Infection Oral & Gastrointestinal Cancer Inflammatory & Immune System Other Cardiovascular Injuries & Accidents Renal & Urogenital Congenital Disorders Mental Health Reproductive Health & Childbirth Ear Metabolic & Endocrine Respiratory Eye Musculoskeletal Skin Generic Health Neurological Stroke NIHR/WT Manchester CRF Application Form Page 2 of 6 Free to Print The user of this document is responsible for referring to Q-Pulse for the current version Reference No: STU-FRM-1 Version: 7 Active Date: 06-Jun-2013 Next Review Date: Refer to Q-Pulse Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust Main Funding Organization:* Please select the category of the main funding organization for this study: Please give the name of the main funding organization, and the total amount awarded: Name: Award Amount: Secondary Funding Organization: Please select the category of the second funding organization for this study, if there is n/a one: Please give the name of the second funding organization, and the total amount awarded: Name: Award Amount: Grant Administrating Organization: Please select organization which will University of Manchester administer the research grant: * Please give the grant reference (if known), and details of organization if ‘Other’: Research Governance Sponsor: Please give details of the organization acting as research governance sponsor: Has research governance sponsorship been confirmed? Has the study been adopted by a study network? (If yes, please give details of network, and whether the adoption is in application, or has been confirmed.) Eg: Cancer RN, CCRN, DeNDRoN, Diabetes RN, MCRN (or PNMR), Mental Health RN, Primary Care RN, Stroke RN. Are any research fellowships, or Masters or PhD students, attached to or supported by the study? (If yes, please give details.) NIHR/WT Manchester CRF Application Form Page 3 of 6 Free to Print The user of this document is responsible for referring to Q-Pulse for the current version Reference No: STU-FRM-1 Version: 7 Active Date: 06-Jun-2013 Next Review Date: Refer to Q-Pulse Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust MCRF Resources Overview Please briefly outline your requirements for MCRF support: Clear explanation of resource needs is crucial for assessing applications. Please note that children are usually seen at the CCRF in RMCH, and adults (18 years and over) in the CRF in the MRI. Would you like your study to run in: CRF CCRF Both Will the study require CRF staff to conduct visits outside the Facility? Yes/No (if yes please give details) Will all visits in the protocol require CRF support (staff or space)? Yes/No (if no then please indicate which visits need support) Will any visits be Day case visits (4-8 hours long)? Yes/No (if yes please give details) Will any visits be In-patient visits (8 hours or longer)? Yes/No (if yes please give details) Are other departments in CMFT involved in supporting the study? Yes/No (if yes please give details) Will you be paying travel expenses? (Details of how this will be organized will be discussed at the application meeting.) Yes/No Does the study require any of the following? Nursing Medical Cover (see below) Pharmacy Human Performance 3T MR Imaging Minor Procedures lab Room Equipment (please state what equipment is required) NIHR/WT Manchester CRF Application Form Page 4 of 6 Free to Print The user of this document is responsible for referring to Q-Pulse for the current version Laboratory Facilities Ultrasound Temperature controlled room Reference No: STU-FRM-1 Version: 7 Active Date: 06-Jun-2013 Next Review Date: Refer to Q-Pulse Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust If the pharmacy will be involved, please contact Carolyn Davies, MRI Clinical Trials Pharmacist (CMFT) 0161 276 4623 clinical.trials@cmft.nhs.uk If laboratory support is needed, please contact Aisling Flatley, Senior Biomedical Scientist at the CRF, or Annelise Waite at the CCRF 0161 906 7500 aisling.flatley@wtcrf.nhs.uk or annelise.waite@cmft.nhs.uk You will need to discuss any study with imaging needs before submitting an application: please contact Frances Davies, MRIF Project Manager 0161 275 0006 frances.davies@manchester.ac.uk Please ensure that the necessary contact is made at the same time as applying to the Manchester CRF, and before the study’s application meeting. Medical Cover The CRF may be able to provide, at a cost, a general medic and/or Advanced Nurse Practitioner (AP) to support study activities such as physical examination and medical history. They can also review ECG’s and adverse events if needed, as well as provide immediate support for drug reactions. Any specialist medical support for the study will need to be provided by the PI or his team. Please indicate below who will provide any specialist medical support and be available if needed in the event of any serious adverse events or reactions MCRF AP/MedicError! Reference source not found. Please note: The MCRF cannot provide 24 hour medical cover for studies or out of hours support NIHR/WT Manchester CRF Application Form Page 5 of 6 Free to Print The user of this document is responsible for referring to Q-Pulse for the current version Reference No: STU-FRM-1 Version: 7 Active Date: 06-Jun-2013 Next Review Date: Refer to Q-Pulse Clinical Research Facility Central Manchester University Hospitals NHS Foundation Trust A1: MR Imaging Appendix Please complete this section only if the 3T MR scanner at the MCRF is required. If the study requires the use of other MR or PET equipment, please complete the Joint Imaging Facilities Application form instead of this document. See www.mhs.manchester.ac.uk/mrif/ Is a full scanning protocol available? * If ‘no’, please detail specific protocol: n/a: no MRI If assistance is required in developing the scanning protocol, please contact Sue Stubbs before submitting the application. Scan cost for MR imaging £ per hour Number of participants being scanned: * Number of scans per participant: * Scheduled time per scan: * hours Total scanning time for study: * hours Estimated scanning time/month (based on recruitment hours target): * Does the scanning component of the study have funding? Yes If yes: account code to be charged: If no: is this an application for MRIF Grant for Scan Time? Not Applicable Will the study require MR scan contrast? No Total study costs for contrast agent(s): £ Account code to be charged for contrast agents: NIHR/WT Manchester CRF Application Form Page 6 of 6 Free to Print The user of this document is responsible for referring to Q-Pulse for the current version Reference No: STU-FRM-1 Version: 7 Active Date: 06-Jun-2013 Next Review Date: Refer to Q-Pulse