Forced oscillation techniques for the assessment of
advertisement

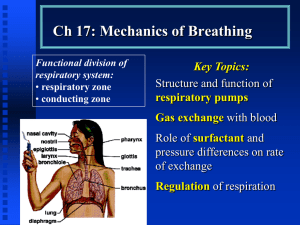
FORCED OSCILLATION TECHNIQUE (FOT) FOR THE ASSESSMENT OF BREATHING MECHANICS IN ANIMALS H.-J. Smith, Erich Jaeger GmbH, Wuerzburg, Germany 1. Introduction Lung function measurements in animals should be effort independent, able to provide objective information and discriminate between the major components of the respiratory tract, taking into account the particular lung physiology of the measured species. Considering the huge differences in size and weight between small animals like rodents [1, 6] and large animals, for example horses [2, 3], the same measuring principle should be applicable with only minor modifications in the mechanical components of the system, to fit it to the respiratory parameters of the examined species and the needs of parameter determination. Furthermore, it is required that the test is non-invasive and can be easily performed in a short time. The oscillometric methodology not only fulfils the above mentioned points, it also offers additional properties which make the technique exceptionally well suited for the measurement of the breathing mechanics in animals and the assessment of their bronchial hyperreactivity. The determination of respiratory impedance by the forced oscillation technique was originally described by Du Bois et al. [4] in 1956. In 1993, a workshop on mechanical respiratory impedance sponsored by the Commission of the European Communities took place in Zeist (NL). As a result, major recommendations concerning technology and clinical application of oscillometry were established and published [5]. Although this workshop was mainly focused on human medicine, these general recommendations can also be transferred to respiratory measurements in animals. The traditional technique to determine total pulmonary resistance RL and dynamic compliance Cdyn by means of simultaneous recording of airflow and oesophageal pressure correlates with oscillometry but is invasive. The assessment of airflow limitation is most commonly achieved by the spirometric technique, which is non-specific and less sensitive. 2. Forced oscillation technique A characteristic feature of oscillometry is, that it does not derive pulmonary impedance from the respiration signals, but from the pressure-flow-relationship of artificial test signals which are produced by an external generator. These artificial signals are superimposed on the respiratory tidal breathing waveform within the measuring system. In this way they enter the respiratory tract and allow determination of its properties. The advantage of artificial test signals is the incomparable higher frequency contents with a relatively high consistency as far as frequency range and amplitude are concerned. The distribution of all of these frequencies is called spectrum. The knowledge of the physical behaviour of every frequency is pre-condition for detailed differentiation of the respiratory system. 2.1. Principle The principle of input impedance measurements is shown in Fig. 1. The subject whose respiratory impedance Zrs is to be determined, breathes ambient air via a pneumotachograph, Y-adapter and terminating resistor. The terminating resistor controls system pressure within the measuring head. Its low value guarantees an insignificant effect on breathing. However, the resistance is high enough to make sure, that a certain amount of the oscillometric pressure, which is produced by the external generator, is forced into the respiratory tract. Two separated transducers register flow V’ and the corresponding pressure P. The respective signals of total pressure and total flow consisting of spontaneous breathing and the superimposed test signal are recorded. The principle of transfer impedance measurements [6, 7], which is mostly used in small animals, differs from input impedance. In this technique the flow is still recorded at the airway opening, whilst oscillometric pressure is applied to the chest. In order to determine the respiratory impedance from the test signal, it has to be separated from the breathing by means of frequency discrimination. For the oscillometric evaluation program, the range between 0.1 Hz and 50 Hz is of particular importance. The frequency range of spontaneous breathing is considerably less and therefore can be easily eliminated from the frequencies of the test signal. 2.2. Technical aspects To achieve high quality of all acquired data, the amplitude and phase of flowand pressure transducer must be matched. Both transducers should have linear response and an extremely good common mode rejection ratio of more than 60 dB [5, 8]. This is especially important for the flow transducer. Wave tube techniques [9] can improve the matching of the transducers, because the flow sensor is replaced by a reference impedance and a second pressure transducer of the same type. However, the dimensions of the wave tube and its limited linearity properties have limited the usefulness of this technique. 2.3. Test signals The various oscillometric techniques use different test signals. In the past sinusoidal signals were more often applied, standard of today is the use of multifrequent signals like pseudo random noise or impulses [10, 11, 22]. The generation of these test signals ranges from electrically controlled membrane deflections of loudspeakers to electrically or mechanically controlled pumps, to sophisticated jet techniques which produce a pulse-shaped pressure flow excitation. 3. Important oscillometric parameters In multi-frequent applications such as Pseudo Random Noise Technique or Impulse Oscillometry, the characteristics of the respiratory system are displayed in form of an impedance spectrum Zrs. Depending on the measured species, it is necessary to identify which of the low frequencies can be effected by spontaneous breathing and which higher frequencies have low sensitivity in the diagnostic evaluation. The respiratory impedance Zrs, which mathematically can be described by its two components, resistance Rrs and reactance Xrs, can be considered as an objective and differentiated image of lung mechanics. Resistance and reactance reflect quite specific and completely different properties of the lung. Pulmonary resistance Rrs covers all frictional components which consume breathing energy within the respiratory tract. The breathing mechanical equivalent of the resistance is the flow resistance in the airways, dependent on airway calibre and structure of the surface of the airway walls. In comparison, reactance Xrs describes the ability of the lung to store energy, which is needed for passive expiration. The reactance itself consists of two components which also differ in their properties. The negative part of reactance Xrs reflects capacitive behaviour (C) and corresponds not only to the thoracopulmonary elasticity but also to changes in the available lung volumes in the distal compartments of the lung, including the visco-elastic properties of the lung tissue. The positive part of reactance Xrs has inertial characteristics (I) describing the moving air column in the bronchial tree. Additionally, the point of intersection of the reactance/frequency plot with the zero axis, the resonant frequency Fres, is useful for clinical interpretation of the results of the measurement. The Fast Fourier Transformation (FFT) is used to separate complex multifrequent test signals into their different sinusoidal components (spectrum) and assumes linearity of the measured system. The linearity of the respiratory system can be documented by the calculation of the coherence function for pressure and flow. Criterion for linearity is a coherence which is the square of the modulus of the cross power spectrum, divided by the product of the pressure and flow auto spectrum at any particular frequency higher than 0.95 [12]. 4. The clinical contents of measured impedance parameters and their relationship to abnormal lung function 4.1. Interpretation of pulmonary resistance Rrs and reactance Xrs For clinical interpretation of lung function measurements, it is necessary to establish a link between measured impedance parameters and their assessment of lung physiology. Weibel [13] described the physiology of the lung with the help of a trumpet model. To get an impression of the dimensions of this trumpet one should remember that the surface of all alveoli covers an area which is 10,000 times larger than the cross sectional area of the trachea. Although the calibre of the single airway is decreasing in the respective airway generation, the cross sectional area of all airways in this generation increases because of the quadratic increase of the number of airways. Simultaneously, with the increase of cross sectional area, the airflow decreases. Whereas the central airways limit the flow because of their narrow calibre and therefore generate airway resistance, in distal lung compartments, which comprise approximately 60% of the total lung volume, there is no flow limitation and therefore no resistance at all. Respiratory impedance is well suited to establish a relationship to this special lung physiology. The resistive part is located more or less in the central and upper airways. In contrast, the peripheral chamber is nearly independent of resistance, expressed by the low frequent capacitive reactance. Increasing resistance Rrs, but also decrease of reactance Xrs (loss of capacitance), describe abnormalities of the respiratory system. The absolute value of these parameters is equivalent to the degree of disability. 4.2. The influence of a face mask Conscious animals are adapted to the measuring apparatus via a face mask. As the face mask influences the outcome of the measurement, it has to be considered in the interpretation of the results. The face mask itself generates a shunt compliance which appears in parallel to the respiratory impedance Zrs. Furthermore, the nasal cavity of the animal adds a resistance which is in series to the lung. With increasing size of total resistance (nose + lung), the effect of mask compliance becomes more important, especially in higher frequencies and nasal resistance can dominate across pulmonary resistance. As a consequence, low frequent reactance becomes more sensitive in the determination of pulmonary properties compared to resistance. 5. Mathematical modeling Conventional lung function diagnostics is mainly based on the time trend of different signals like flow (V'), volume (V) or pressure (P), which are derived from the respiratory tract while the subject is breathing. The behaviour of these signals is easy to follow and to interpret. Quite different is the outcome of oscillometry with mainly spectral parameters. The clinical interpretation therefore needs a completely different approach. To overcome this problem and to be able to use the advantages of oscillometry, mathematical models were developed in order to transfer spectral data into structural data describing different structures of the lung. This is a modern form of interpretation of the measurement and a method of significant data reduction. Although different models are available [14, 15, 16, 17, 18, 19], none of them are able to fulfil the most important clinical requirement: to provide an extensive interpretation of any measured respiratory system. The definition of the model structure as well as the algorithms for numerical parameter calculation are problematic. A high number of elements in the mathematical model simplifies interpretation and fits to the complex structure of the respiratory tract, but makes parameter calculation unstable. Simple models allow reliable calculations but are still difficult to interpret. However, some authors use models for further partitioning of pulmonary impedance into tissue and airway components [19, 20]. Other authors imply models to recalculate the influence of the face mask on the acquired impedance [21]. 6. Conclusions Abnormal lung function means limited fitness of animals. Oscillometry offers sufficient sensitivity to detect the changes in the respiratory system noninvasively. The knowledge of the specific characteristics of resistance and reactance, their relationship to lung physiology and the physical properties of various frequencies, which are used in the multifrequent versions of the oscillometric technique, offer the possibility to differentiate proximal and distal airway resistance and to determine the capacitance of lung and thorax. This is possible even if we take into account the distinguishing characteristics of measurements in animals, as lung measurement across the nose and nose breathing. References 1. Zwart A, Hessel EM. Oscillatory mechanics of the mouse respiratory system. Eur Respir Rev 1994; 4 (19): 182-186. 2. Young SS, Tesarowski D. Respiratory mechanics of horses measured by conventional and forced oscillation techniques. J App Physiol 1994; 76(6): 24672472. 3. Young SS, Tesarowski D, Viel L. Frequency dependence of forced oscillatory respiratory mechanics in horses with heaves. J App Physiol 1997; 82(3): 983987. 4. Dubois AB, Brody AW, Lewis DH, Burgess JR BF. Oscillation mechanics of lungs and chest in man. J App Physiol 1956; 19: 653-658. 5. Van de Woestijne KP, Desager KN, Duiverman EJ, Marchal F. Recommendations for the measurement of respiratory input impedance by means of forced oscillation method. Eur Respir Rev 1994; 4 (19): 235-237. 6. Oostveen E, Zwart A. Effects of Pentobarbital and halothane anaesthesia on the respiratory transfer impedance of rats. Eur Resp Rev 1994; 4 (19): 172-177. 7. Marchal F, Colin G, Chalon B, Duvivier C, Crance JP, Peslin R. Transfer respiratory impedance and thoracic gas volume to estimate airway and tissue impedance in infants. Eur Respir Rev 1994; 4: 159-164. 8. Duvivier G, Rotger M, Felicio da Silva J, Peslin R, Navajas D. Static and dynamic performances of variable reluctance and piecoresistive pressure transducers for forced oscillation measurements. Eur Respir Rev 1991; 1; 3: 146-150. 9. Brusasco V, Cipooloni C, Barabino P, Basano L, Ottonello P. Evaluation of a modified device for measuring respiratory input impedance without using a flowmeter. Eur Respir Rev 1991; 1; 3: 163-166. 10. Reinhold P, MacLeod, D, Lekeux P. Comparative evaluation of impulse oscillometry and monofrequency forced oscillation technique in clinically healthy calves undergoing bronchochallenges. Research in Veterinary Science 1996; 61: 206-213. 11. Vogel J, Smidt U. Impulse Oscillometry. Pmi Verlagsgruppe GmbH, Frankfurt am Main1994 12. Grant LJ, Scott RS. The use of multiple coherence functions in evaluating the reliability of respiratory impedance data. Eur Respir Rev 1994; 19: 121–125. 13. Weibel ER, Taylor CR. Design of the mammalian respiratory system. Respiration Physiology 1981; 44:1-164. 14. Rotger M, Farré R, Navajas D. Importance of the frequency range for the reliability of Hildebrandt’s model parameters for lung tissue in humans. Eur Respir Rev 1994; 4 (19): 187-190. 15. Zwart A, Arts JHE, Kuper CF. Wave propagation: a new parameter in the description of mechanical airway impedance. Eur Respir Rev 1994; 4 (19): 203209. 16. Bates JHT. Understanding lung tissue mechanics in terms of mathematical models. Monaldi Arch Chest Dis 1993. 48 (2): 134-139. 17. Tomalak W, Peslin R, Duvivier C, Gallina C. Optimal frequency range to analyze respiratory transfer impedance with six-element model. J Appl Physiol 1993; 75 (6): 2656-2664. 18. Lorino AM, Zerah F, Mariette C, Harf A, Lorino H. Respiratory resistive impedance in obstructive patients: linear regression analysis vs viscoelastic modelling. Eur Respir J 1997; 10: 150-155. 19. Hantos Z, Adamicza A, Govaerts E, Daroczy B. Mechanical impedances of lungs and chest wall in the cat. J Appl Physiol 1992; 73: 427-433. 20. Hantos Z, Daroczy B, Suki B, Nagy S, Fredberg JJ. Input impedance and peripheral inhomogeneity of dog lungs. J Appl Physiol 1992; 72: 168-178. 21. Reinhold P, Smith HJ, Close R, Genicot B, Lekeux P. Validation of impulse oscillometry in Friesian and Blue Belgian calves with respect to changes in extrathoracic upper airway resistance. Research in Veterinary Science 1998; 65: 93-102. 22. Daroczy B, Hantos Z. Generation of optimum pseudorandom signals for respiratory impedance measurements. Int J Biomed Comput 1990; 25: 21-31.