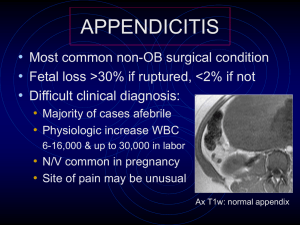
std testing, treatment and referral of pregnant women
advertisement

STD TESTING, TREATMENT AND REFERRAL OF PREGNANT WOMEN INTRODUCTION Intrauterine or perinatally transmitted STDs can have severely debilitating effects on pregnant women, their partners, and their fetuses. All pregnant women and their sex partners should be asked about STDs, counseled about the possibility of perinatal infections, and ensured access to treatment, if needed. The health department has a primary emphasis on getting pregnant women to be seen rapidly by an obstetrical practitioner so that appropriate testing, counseling and treatment can begin promptly. However, the availability of certain services by our STD clinic may allow treatment to begin before they are able to see their obstetrician, family practitioner or midwife. This standing order facilitates initiation of testing, counseling and treatment prior to the woman’s first visit to their obstetrical provider. INDICATIONS Testing, counseling and treatment of pregnant females may be performed in the STD Clinic under the following circumstances: 1. 2. 3. 4. Pregnant females referred by local, state or federal disease intervention specialist. Pregnant females referred by outside providers. Pregnant contacts of known positives. Pregnant females whom are symptomatic. DIAGNOSTIC TESTING 1. 2. 3. 4. 5. Neisseria gonorrhoeae and Chlamydia trachomatis: The health department’s STD clinic does not routinely have available a urine test for GC and Chlamydia. Using a cervical swab test for GC and Chlamydia might disrupt the pregnancy; therefore cervical testing is deferred until the patient reaches her obstetrical provider. If urinary testing for GC and Chlamydia becomes available for our clinic, it may be used. All pregnant women should be offered voluntary HIV testing. Care should be taken to offer this as Standard for all pregnant women regardless of past medical history, heritage or income. A serologic test for syphilis should be performed on all pregnant women. A serologic test for Hepatitis B surface antigen (HBsAg) should be performed on all pregnant women. A test for Hepatitis C antibodies (anti-HCV) should be performed on all pregnant women whom are not known to already have been infected by Hepatitis C virus. Note: Invasive cervical procedures and wet mounts are not to be performed on pregnant women within the STD clinic. TREATMENT AND REFERRAL Treat according to the health department’s standing orders including those in the STD Manual. For pregnant females who are highly suspicious of PID, urethritis, or have laboratory evidence of an STD, an effort should be made to reach their obstetrical provider or primary care provider by D:\219571701.docPage 1 of 3 phone so that phone orders may be obtained to initiate treatment promptly. If the patient’s provider cannot be reached in a timely fashion, contact the medical director to consider initiation of therapy. These females require close follow-up by the physician. All positives for Gonorrhea, Chlamydia, HIV or Syphilis must be interviewed by a DIS. Patients without an obstetrician midwife or obstetrically practicing family practitioners will be assisted in locating an obstetrical provider so prompt follow-up may occur. All pregnant women will be referred to an obstetrical provider for continuation of care. * * * * * Establish health care coverage and make appropriate referrals for application to Medicaid or Michcare. Ask pregnant female who their preferred health care provider would be and make appropriate referral. Provide a list of community resources to pregnant females who do not have an obstetrician, midwife or obstetrically practicing family physician. Obtain Release of Information as appropriate and provide test results that would be pertinent to health care provider. Note: Release of HIV results must be specified on form (blank release no acceptable). When possible call and schedule initial OB visit for client while they are in the STD clinic. OTHER INSTRUCTIONS 1. 2. 3. 4. 5. 6. Advise clients and their sex partners to avoid sex for one week after receiving treatment for an STD infection. Advise clients regarding possible side effects of anti-microbial therapies. Advise client to use condom after one week of abstaining from sexual intercourse and treatment with anti-microbial therapy. Advise client to share disease history with their primary care provider and obstetrical provider. Educate client regarding the risk to the unborn child regarding STDs. Collect identifying information regarding all sexual contacts of pregnant female within the last 6 months and establish a plan for contacting all sexual partners. FOLLOW-UP Follow-up as necessary to insure proper contact tracing, treatment, and continuity of care by the patient’s obstetrical provider. The results of testing obtained by the STD Clinic should be sent to the patient’s obstetrical provider for follow-up, once appropriate consent has been obtained. OTHER STD-RELATED CONCERNS HBsAg-positive women should be reported to the local and/or state health department to ensure that they are entered into a case-management system and that appropriate prophylaxis is provided for their infants. In addition, household and sexual contacts of HBsAg-positive women should be vaccinated. No treatment is available for anti-HCV-positive pregnant women. However, all women found to be anti-HCV-positive should receive appropriate counseling. No vaccine is available to prevent HCV transmission. D:\219571701.docPage 2 of 3 In the absence of lesions during the third trimester, routine serial cultures for HSV are not indicated for women who have a history of recurrent genital herpes. Prophylactic cesarean section is not indicated for women who do not have active genital lesions at the time of delivery. The presence of genital warts is not an indication for cesarean section. Not enough evidence exists to recommend routine screening for Trichomonas vaginalis in asymptomatic pregnant women. REFERENCES 1. Centers for Disease Control and Prevention, Sexually transmitted diseases treatment guidelines 2006. MMWR 2006;51(No. RR-6):6-8. Rev: 9/27/06 STD/Manual/PregnantWomenAndSTDs D:\219571701.docPage 3 of 3