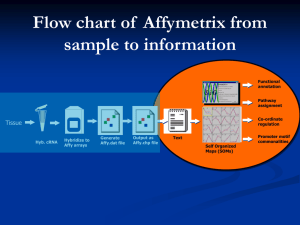
Statistical analysis of
expression data:
Normalization, differential
expression and multiple testing
Jelle Goeman
Outline
Normalization
Expression variation
Modeling the log Fold change
Complex designs
Shrinkage and empirical Bayes (limma)
Multiple testing (False Discovery Rate)
Measuring expression
Platforms
Microarrays
RNAseq
Common:
Need for normalization
Batch effects
Why normalization
Some experimental factors cannot be
completely controlled
Amount of material
Amount of degradation
Print tip differences
Quality of hybridization
Effects are systematic
Cause variation between samples and
between batches
What is normalization?
Normalization =
An attempt to get rid of unwanted systematic
variation by statistical means
Note 1: this will never completely succeed
Note 2: this may do more harm than good
Much better, but often impossible
Better control of the experimental conditions
How do normalization methods work?
General approach
1. Assume: data from an ideal experiment would
have characteristic A
E.g. mean expression is equal for each sample
Note: this is an assumption!
2. If the data do not have characteristic A, change
the data such that the data now do have
characteristic A
E.g. Multiply each sample’s expression by a factor
Example: quantile normalization
Assume:
“Most probes are not differentially expressed”
“As many probes are up and downregulated”
Reasonable consequence:
The distribution of the expression values is identical for
each sample
Normalization:
Make the distribution of expression values identical for
each sample
Quantile normalization in practice
Choose a target distribution
Typically the average of the measured distributions
All samples will get this distribution after normalization
Quantile normalization:
Replace the ith largest expression value in each sample by the
ith largest value in the target distribution
Consequence:
Distribution of expressions the same between samples
Expressions for specific genes may differ
Less radical forms of normalization
Make the means per sample the same
Make the medians the same
Make the variances the same
Loess curve smoothing
Same idea, but less change to the data
Overnormalizing
Normalizing can remove or reduce true
biological differences
Example: global increase in expression
Normalization can create differences that are not
there
Example: almost global increase in expression
Usually: normalization reduces unwanted
variation
Batch effects
Differences between batches are even stronger
than between samples in the same batch
Note: batch effects at several stages
Normalization is not sufficient to remove batcheffects
Methods available (comBat) but not perfect
Best: avoid batch effects if possible
Confounding by batch
Take care of batch-effects in experimental
design
Problem: confounding of effect of interest
by batch effects
Example: Golub data
Solution: balance or randomize
Expression variation
Differential expression
Two experimental conditions
Treated versus untreated
Two distinct phenotypes
Tumor versus normal tissue
Which genes can reliably be called differentially
expressed?
Also: continuous phenotypes
Which gene expressions are correlated with phenotype?
Variation in gene expression
Technical variation
Variation due to measurement technique
Variability of measured expression from experiment to
experiment on the same subject
Biological variation
Variation between subjects/samples
Variability of “true” expression between different
subjects
Total variation
Sum of technical and biological variation
Reliable assessment
Two samples always have different expression
Maybe even a high fold change
Due to random biological and technical variation
Reliable assessment of differential expression:
Show: fold change found cannot be explained by
random variation
Assessment of differential expression
Two interrelated aspects:
Fold change:
How large is the expression difference found?
P-value:
How sure are we that a true difference exists?
LIMMA:
Linear models for gene expression
Modeling variation
How does gene expression depend on
experimental conditions?
Can often be well modeled with linear models
Limma:
linear models for microarray analysis
Gordon Smyth, W. and E. Hall Institute, Australia
Multiplicative scale effects
Assumption: effects on gene expression work in
a multiplicative way (“fold change”)
Example: treatment increases gene expression
of gene MMP8 by a factor 2
“2-fold increase”
Treatment decreases gene expression of gene
MMP8 by a factor 2
“2-fold decrease”
Multiplicative scale errors
Assumption: variation on gene expression
works in a multiplicative way
A 2-fold increase by chance is just as
likely as a 2-fold decrease by chance
When true expression is 4, measuring 8 is
as likely as measuring 2
Working on the log scale
When effects are multiplicative, log-transform!
Usual in microarray analysis: log to base 2
Remember: log(ab) = log(a)+log(b)
2 fold increase = +1 to log expression
2 fold decrease = -1 to log expression
Log scale makes multiplicative effects symmetric
½ and 2 are not symmetric around 1 (= no change)
-1 and +1 are symmetric around 0 (= no change)
A simple linear model
Example: treated and untreated samples
Model separately for each gene
Log Expression of gene 1: E1
E1 = a + b * Treatment + error
a: intercept = average untreated logexpression
b: slope = treatment effect
Modeling all genes simultaneously
E1 = a1 + b1 * Treatment + error
E2 = a2 + b2 * Treatment + error
…
E20,000 = a20,000 + b20,000 * Treatment +
error
Same model, but
Separate intercept and slope for each gene
And separate sd sigma1, sigma2, … of error
Estimates and standard errors
Gene 1: Estimates for a1, b1 and sigma1
Estimate of treatment effect of gene 1
b1 is the estimated log fold change
standard error s.e.(b1) depends on sigma1
Regular t-test for H0: b1=0:
T = b1/s.e.(b1)
Can be used to calculate p-values.
Just like regular regression, only 20,000 times
Back to original scale
Log scale regression coefficient b1
Average log fold change
Back to a fold change: 2^b1
b1= 1 becomes fold change 2
b1 = -1 becomes fold change 1/2
Confounders
Other effects may influence gene
expression
Example: batch effects
Example: sex or age of patients
In a linear model we can adjust for such
confounders
Flexibility of the linear model
Earlier: E1 = a1 + b1 * Treatment + error
Generalize:
E1 = a1 + b1 * X + c1 * Y + d1 + Z + error
Add as many variables as you need.
Variance shrinkage
Empirical Bayes
So far: each gene on its own
20,000 unrelated models
Limma: exchange information between
genes
“Borrowing strength”
By empirical Bayes arguments
Estimating variance
For each gene a variance is estimated
Small sample size: variance estimate is
unreliable
Too small for some genes
Too large for others
Variance estimated too small: false positives
Variance estimated too large: low power
Large and small estimated variance
Gene with low variance estimate
Likely to have low true variance
But also: likely to have underestimated variance
Gene with high variance estimate
Likely to have high true variance
But also: likely to have overestimated variance
Limma’s idea:
Use information from other genes to assess whether
variance is over/underestimated
True and estimated variance
Variance model
Limma has a gene variance model
All gene’s variances are drawn at random
from an inverse gamma distribution
Based on this model:
Large variances are shrunk downwards
Small variances are shrunk upwards
Effect of variance shrinkage
Genes with large fold change and large
variance
More power
More likely to be significant
Genes with small fold change and small
variance
Less power
Less likely to be significant
Limma and sample size
Shrinkage of limma only effective for small
sample size (< 10 samples/group)
Added information of other genes
becomes negligeable if sample size gets
large
Large samples: Doing limma is the same
as doing regression per gene
Differential expression in RNAseq
RNAseq data: counts
Gene id
Y1
Y2
Y3
Y4
Y5
Y6
Y7
Y8
Y9
Y10
ENSG0000
0110514
69
178
101
58
101
31
165
108
70
1
ENSG0000
0086015
115
52
86
88
146
84
59
85
86
0
ENSG0000
0115808
285
190
467
295
345
532
369
473
423
5
ENSG0000
0169740
502
184
363
195
403
262
225
332
136
3
ENSG0000
0215869
0
7
0
0
0
0
0
2
0
0
ENSG0000
0261609
20
31
76
20
25
158
23
18
23
1
ENSG0000
0169744
488
529
470
505
1137
373
1392
3517
192
1
ENSG0000
0215864
1
0
0
0
0
0
0
0
0
0
Modelling count data
Distinguish three types of variation
Biological variation
Technical variation
Count variation
Count variation is important for lowexpressed genes
Generally biological variation most
important
Overdispersion
Modelling count data: two stages
1. Model how gene expression varies from
sample to sample
2. Model how the observed count varies by
repeated sequencing of the same sample
Stage 2 is specific for RNAseq
Two approaches
Approach 1: Model the count variation and the
between-sample variation
edgeR
Deseq
Approach 2: Normalize the count data and
model only the biological variation
Voom + limma
Approach 3: Model count variation only
Popular but very wrong!
Multiple testing
20,000 p-values
Fitting 20,000 linear models
Some variance shrinkage
Result:
20,000 fold changes
20,000 p-values
Which ones are truly differentially
expressed?
Multiple testing
Doing 20,000 tests: risk false positive
20,000 times
If 5% of null hypotheses is significant,
expect 1,000 significant by pure chance
How to make sure you can really trust the
results?
Bonferroni
Classical way of doing multiple testing
Call K the number of tests performed
Bonferroni: significant = p-value < 0.05/K
“Adjusted p-value”
Multiply all p-values by K, compare with 0.05
Advantages of Bonferroni
Familywise error control
=Probability of making any type I error < 0.05
With 95% chance, list of differentially
expressed genes has no errors
Very strict
Easy to do
Disadvantages of Bonferroni
Very strict
“No” false positives
Many false negatives
It is not a big problem to have a few false
positives
Do validation experiments later
False discovery rate
(Benjamini and Hochberg)
FDR = expected proportion of false
discoveries among all discoveries
Control of FDR at 0.05 means in the long
run experiments average about 5% type I
errors among the reported genes
Percentage: longer lists of genes are
allowed to have more errors
Benjamini and Hochberg by hand
1. Order the p-values small to large
Example: 0.0031, 0.0034, 0.02, 0.10, 0.65
2. Multiply the k-th p-value by m/k, where m is the number
of p-values, so
0.0031 * 5/1, 0.0034 * 5/2, 0.02 * 5/3, 0.10 * 5/4, 0.65 * 5/5
which becomes
0.0155, 0.0085, 0.033, 0.125, 0.65
3. If the p-values are no longer in increasing order, replace
each p-value by the smallest p-value that is later in the
list. In the example, we replace 0.0155 by 0.0085. The
final Benjamini-Hochberg adjusted p-values become
0.0085, 0.0085, 0.033, 0.125, 0.65
FDR warnings
FDR is susceptible to cheating
How to cheat with FDR?
Add many tests of known false null
hypotheses…
Result: reject more of the other null
hypotheses
Example limma results
Conclusion
Testing for differentially expressed genes
Repeated application of a linear model
Include all factors in the model that may
influence gene expression
Limma: additional step “borrowing strength”
Don’t forget to correct for multiple testing!