Lecture Two – Introduction to Chemistry
advertisement

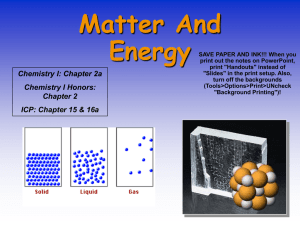
CHEM 5013 Applied Chemical Principles Chapter One Professor Bensley Alfred State College Chapter Objectives Explain the usefulness of the macroscopic, microscopic, and symbolic perspectives in understanding chemical systems. Explain the differences between physical and chemical properties of matter and provide examples of each. Define the following types of matter: substance, element, compound, mixture. The Study of Chemistry The study of chemistry involves three levels of understanding or three perspectives. 1. _ 2. _ 3. _ The Macroscopic Perspective Matter: Matter is observed through two types of changes: 1. 2. _ _ The Macroscopic Perspective Physical properties: Examples: Chemical properties: Examples: Physical Property vs. Chemical Property Example: Potassium is a soft, silvery-colored metal that melts at 64°C. It reacts vigorously with water, with oxygen, and with chlorine. Identify all of the physical properties given in this description. Identify all of the chemical properties given. The Macroscopic Perspective 1. 2. 3. Three phases of matter: The Microscopic or Particulate Perspective Substances: Examples: Elements: Examples: Elements Elements: sulfur, arsenic, iodine, magnesium, bismuth, mercury. The Microscopic or Particulate Perspective Compounds: Examples: Mixtures: Examples: Mixtures Two types of mixtures: 1. Examples: 2. Examples: Which of the following are pure substances and which are mixtures? For each, list all of the different phases present. a) bromine liquid and its vapor b) paint, containing a liquid solution and a dispersed solid pigment c) partially molten iron d) baking powder containing sodium hydrogen carbonate and potassium hydrogen tartrate e) A glass of ice water The Microscopic or Particulate Perspective For solids, the molecules or atoms maintain a regular ordered structure, so a sample maintains its size and shape. The Microscopic or Particulate Perspective For liquids, the molecules or atoms remain close to one another, but the ordered array breaks down. This allows the liquid to flow and take the shape of its container. The Microscopic or Particulate Perspective For gases, the molecules or atoms are very widely separated, and move independently of one another. This allows the gas to fill the available volume of the container. The Microscopic or Particulate Perspective The Microscopic or Particulate Perspective The Symbolic Representation What are the symbols used to represent each of these substances?