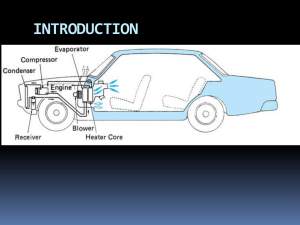
Refrigeration
advertisement

Refrigeration Plants – combine carbon dioxide from atmosphere, moisture and traces of chemicals from the soil to form all constituents they need. Energy from sunlight and green parts of plants can effect this synthesis. • Animals cannot do this They have to get food from plants or other animals. From their food they absorb what they require in their digestive system and reject the rest. Many micro-organisms live on the same food as men. Our battle is to get the food before the micro-organisms. • In this battle the micro-organisms are well placed – size so small they are ever present in the atmosphere. Any food exposed to atmosphere at ordinary temperature – appropriate organisms will start to grow. They consume the food like animal – absorbing what they like and rejecting the remainder. Rejected remainder is often toxic – leading to food poisoning. • Micro-organisms can be divided in three groups – moulds, yeast and bacteria. • Moulds – start microscopic – can develop into clearly visible growths. • Yeast – reproduce into microscopic individuals – presence known by the alcohol their digestive process rejects – leading to fermentation. • Bacteria - also multiply as microscopic individuals- their waste products often giving rise to evil smells of putrefaction as well as being toxic. • Each type of micro-organisms has its own environment of temperature, atmosphere (gaseous composition and humidity) and food for optimum growth. Water is required by everyone. Hence drying can also lead to food preservation. • Control of micro-organisms by subjecting them to low temp. is basis of refrigerated carriage of foodstuffs. • There are three main temperature group of microorganisms – 1. 36.70 C for optimal growth. This group includes bacteria. They practically cease growth at 00 C. 2. Optimal growth at 210 C, which are capable of rapid growth at 00 C and which will grow on unfrozen substances below this temperature. This group includes yeast and moulds and bacteria. 3. Micro-organisms, mainly yeasts and moulds, which will grow on frozen substances down to about -70 C. • Below –80 C no growth or reproduction of microorganisms occurs. • For each living process there is an optimum temp. at which the process occurs at the maximum rate. As temp. falls below the optimum, the rate of living falls off until at low enough temperatures life ceases. As temperature rises above the optimum, different chemical processes take over from the normal living reactions until at high enough temperature life again ceases . Optimum temperature varies for each life process, but all lie within the range of –80 C to 1000 C, outside which life is virtually unknown. Dead animal products • With dead animal produce, the primary purpose of refrigeration is to delay or prevent the developments of microorganisms (moulds, yeast and bacteria) which live on the produce. Cold storage also retards the slow chemical changes, such as the oxidation of fats, which whilst not rendering the produce unfit for human consumption do adversely affect flavours. • Dead animal products are carried as frozen cargo below –80 C. None of the microorganisms that cause decay and putrefaction grow and multiply below this temperature, although some may survive in dormant state to resume growth if temperature is raised subsequently. Further temperature reduction is required to slow chemical changes that impair flavour. For beef and lamb –100 C is adequate. For pork –12 to –150 C. Poultry and fish –180 C is desirable. Fruits and vegetables • The living process continues after picking of fruits and vegetables. After picking at immature stage the ripening process continues until it is fully ripe and finally overripe. Cool storage delays the ripening process. Second benefit is delaying of onset of mouldiness as they are likely to be attacked by moulds. Hence by refrigeration, we keep it alive and let it ripen slowly. The water content of fruits and vegetables is 80-90%. If brought below freezing point, changes in physical structure will take place and will be killed. They are to be carried in chilled condition above their freezing point. • Common between fruit and animal produce –provide conditions unsuitable for growth of organisms which live on the produce. Chemical reaction proceed more slowly at low temperature. 50C drop in temperature halves the rate of reaction. Units of Refrigeration Capacity • Tonnage – One Ton of Refrigeration implies latent heat of one tonne of ice or the heat required to convert one tonne of water to ice. Equivalent to 12,000 BTUs. One BTU is the heat required to raise one pound of water by one degree Fahrenheit. • One calorie is the heat required to raise one gram of water by one degree Celsius. • Joule is the mechanical equivalent of heat, work done when one a force of one Newton moves through one metre. • One Joule = 4.2 calories • Calculate the conversion factor from BTUs to Kilocalories Refrigerant • Refrigeration depends upon a substance called the refrigerant, which can readily be converted from liquid into a vapour (evaporation) and also from a vapour into a liquid (condensation) within a narrow range of pressures. Refrigerants are those fluids, which are used as working fluids, for example in vapour compression refrigeration systems. These fluids provide refrigeration by undergoing a phase change process in the evaporator. • A refrigerant gives up heat by condensing at high temperatures and pressures and absorbs heat by evaporating at low temperatures and pressures. REFRIGERANTS Halocarbon based Ethane based CFCs Non-Halocarbon based Methane based HCFCs HFCs Primary Secondary Pure Azeotropic Blended Near Azeotropic Zeotropic • Azeotropic Refrigerants: Mixture of two or more refrigerants with similar boiling points. Where the change of phase takes place at a specific single temperature.ex: R-500;R-502; R-503; R-507. They can be charged as a liquid or vapour. • Zeotropic Refrigerants: Mixture of two or more refrigerants with dissimilar boiling points. Where the change of phase takes place over a temperature range of over 5.56 degrees C (10 deg F).ex: R404a; 407c. They should be charged as a liquid. • Near Azeotropic Refrigerants: : Mixture of two or more refrigerants with dissimilar boiling points, but the change of phase takes place over a temperature range of less than 5.56 degrees C (10 deg F). R410a. They should be charged as a liquid. • TEMPERATURE GLIDE is the range of temperature over which change of phase occurs. Important because fractionating of refrigerant occurs especially when there is a leak. Also complicates the charging process. Generation of Refrigerants • First Generation 1830-1930s ex: CO2; NH3; HCs;SO2 etc. usefulness of volatile compounds • Second Generation: 1931-1990s ex: CFCs; HCFCs; safety & durability • Third Generation; 1990-2010s ex: HCFCs and HFCs; Ozone Layer protection • Fourth Generation; 2010 onwards ex: Pure & Blended HCs; Global warming; high efficiency Refrigerant selection criteria • Selection of refrigerant for a particular application is based on the following requirements: 1. Thermodynamic and thermo-physical properties 2. Environmental and safety properties, and 3. Economics Thermodynamic and thermophysical properties • The requirements are: 1. Suction pressure: At a given evaporator temperature, the saturation pressure should be above atmospheric for prevention of air or moisture ingress into the system and ease of leak detection. Higher suction pressure is better as it leads to smaller compressor displacement 2. Discharge pressure: At a given condenser temperature, the discharge pressure should be as small as possible to allow light-weight construction of compressor, condenser etc. 3. Pressure ratio: Should be as small as possible for high volumetric efficiency and low power consumption 4. Latent heat of vaporization: Should be as large as possible so that the required mass flow rate per unit cooling capacity will be small 5. Isentropic index of compression: Should be as small as possible so that the temperature rise during compression will be small 6. Liquid specific heat: Should be small so that degree of subcooling will be large leading to smaller amount of flash gas at evaporator inlet 7. Vapour specific heat: Should be large so that the degree of superheating will be small 8. Thermal conductivity: Thermal conductivity in both liquid as well as vapour phase should be high for higher heat transfer coefficients 9. Viscosity: Viscosity should be small in both liquid and vapour phases for smaller frictional pressure drops 10. The freezing point of the refrigerant should be lower than the lowest operating temperature of the cycle to prevent blockage of refrigerant pipelines. 11. High critical temperature Environmental properties • 1. 2. 3. 4. 5. The important environmental properties are: Ozone Depletion Potential (ODP) Global Warming Potential (GWP) Total Equivalent Warming Index (TEWI) Atmospheric Lifetime Chlorine Leading Potential Safety and Other Properties 1. 2. 3. 4. 5. 6. 7. 8. TLV (Threshold Limit Value) Toxicity Flammability Chemical stability Compatibility with common materials Miscibility with lubricating oils Dielectric strength Ease of leak detection Ozone Depletion Potential (ODP): According to the Montreal protocol, the ODP of refrigerants should be zero, i.e., they should be non-ozone depleting substances. Refrigerants having non-zero ODP have either already been phased-out (e.g. R 11, R 12) or will be phased-out in near-future (e.g. R22). Since ODP depends mainly on the presence of chlorine or bromine in the molecules, refrigerants having either chlorine (i.e., CFCs and HCFCs) or bromine cannot be used under the new regulations Global Warming Potential (GWP) Refrigerants should have as low a GWP value as possible to minimize the problem of global warming. Refrigerants with zero ODP but a high value of GWP (e.g. R134a) are likely to be regulated in future. Total Equivalent Warming Index (TEWI) • The factor TEWI considers both direct (due to release into atmosphere) and indirect (through energy consumption) contributions of refrigerants to global warming. Naturally, refrigerants with as a low a value of TEWI are preferable from global warming point of view. Atmospheric Lifetime • HFC125, the major component of HFC blend refrigerants, has an atmospheric life of 29 years, while the atmospheric life of HFC32 is only five years. Toxicity Ideally, refrigerants used in a refrigeration system should be non-toxic. However, all fluids other than air can be called as toxic as they will cause suffocation when their concentration is large enough. Thus toxicity is a relative term, which becomes meaningful only when the degree of concentration and time of exposure required to produce harmful effects are specified. Toxicity • Some fluids are toxic even in small concentrations. Some fluids are mildly toxic, i.e., they are dangerous only when the concentration is large and duration of exposure is long. Some refrigerants such as CFCs and HCFCs are nontoxic when mixed with air in normal condition. However, when they come in contact with an open flame or an electrical heating element, they decompose forming highly toxic elements (e.g. phosgene-COCl2). Toxicity • 1. 2. 3. 4. 5. In general the degree of hazard depends on: Amount of refrigerant used vs total space Type of occupancy Presence of open flames Odor of refrigerant, and Maintenance condition • Thus from toxicity point-of-view, the usefulness of a particular refrigerant depends on the specific application. Flammability • The refrigerants should preferably be nonflammable and non-explosive. For flammable refrigerants special precautions should be taken to avoid accidents. Chemical stability & Compatibility • The refrigerants should be chemically stable as long as they are inside the refrigeration system. • Compatibility with common materials of construction (both metals and non-metals) Miscibility with lubricating oils • Oil separators have to be used if the refrigerant is not miscible with lubricating oil (e.g. ammonia). Refrigerants that are completely miscible with oils are easier to handle (e.g. R12). However, for refrigerants with limited solubility (e.g. R 22) special precautions should be taken while designing the system to ensure oil return to the compressor Dielectric strength • This is an important property for systems using hermetic compressors. For these systems the refrigerants should have as high a dielectric strength as possible Ease of leak detection • In the event of leakage of refrigerant from the system, it should be easy to detect the leaks. Economic properties • The refrigerant used should preferably be inexpensive and easily available. Halocarbon Refrigerants • Methane (CH4)Based will have a two-digit number ex:R22 First figure shows no. of Hydrogen atoms plus 1 Second figure shows no. of fluorine Atoms R22=CHClF2 Chlorofluoromethane. The total number of Hydrogen and replacement atoms should be 4; R11; R12; R13; R23 etc. • Ethane (C2H6) Based will have a three-digit number ex: R113. First figure shows no. of Carbon atoms minus 1; Second figure shows no. of Hydrogen atoms plus 1; Third figure shows no. of fluorine Atoms R113 =CCl2FCClF2 Tricholorofluoroethane; R114; R115 etc. • R134a=C2H2F4 Tetrafluoro ethane The letter a signifies isomer ( having same chemical composition but different atomic arrangement) • Refrigerants starting with 4 and 5 indicate blended refrigerants • The most important members of the group have been – • Chlorofluorocarbons (CFC) • Hydrochlorofluorocarbon (HCFC) • Hydrofluorocarbon(HFC) Chlorofluorocarbons (CFC) • CCl2F2 - Dichlorodifluoromethane (Freon 12 or R12) • CCl3F - Trichlorofluoromethane (Freon 11 or R11) • C2Cl2F4 - Dichlorotetrafluoroethane (Freon 114 or R114) • C2Cl3F3 - Trichlorotrifluoroethane (Freon 113 or R113) Hydrochlorofluorocarbon (HCFC) • CHClF2 – monochlorodifluoromethane (Freon 22 or R22) • R123 – C2HCl2F3 • R124 - C2HClF4 Hydrofluorocarbon(HFC) • • • • • C2H2F4 - R134a C2H4F2 - R152a C2HF5 - R125 CH2F2 - R32 C2H3F3 - R143a Inorganic refrigerants • These are designated by number 7 followed by the molecular weight of the refrigerant (rounded-off). Mixtures • Azeotropic mixtures (containing two gases with same boiling point ) are designated by 500 series, where as zeotropic (containing two or more gases not having same boiling point ) refrigerants (e.g. nonazeotropic mixtures) are designated by 400 series. Pure Hydrocarbon Refrigerants Depletion of stratospheric ozone layer • The depletion of stratospheric ozone layer was attributed to chlorine and bromine containing chemicals such as CFCs, HCFCs. If released to atmosphere, they are broken down by photolysis to release chlorine atoms, which catalytically destroy ozone, the stratospheric gas which acts as a filter to ultra violet (UV) light from the sun. Effect of UV light • Scientists predict that increased UV light on earth as a result of ozone depletion will, amongst other possible consequences, cause skin cancer, interfere with immune systems and harm aquatic systems and crops. Strong pressure was exerted to phase out CFCs and HCFCs, resulted in the Montreal Protocol being adopted in 1987. Regulation 12(2) of Annex VI of MARPOL 73/78 • New installations which contain ozonedepleting substances shall be prohibited on all ships, except that new installations containing hydrochloroflourocarbons (HCFCs) are permitted till 1 January 2020. Alternate refrigerants • They can be classified into two broad groups: 1. Non-ODS, synthetic refrigerants based on Hydro-Fluoro-Carbons (HFCs) and their blends 2. Natural refrigerants including ammonia, carbon dioxide, hydrocarbons and their blends Refrigeration oils 1. 2. 3. 4. 5. 6. 7. Lubricating oils for refrigeration compressors are selected for their suitability with the different refrigerant, compressor type and the plant’s operating temperatures. Refrigeration oils should possess the following properties: Good chemical stability Good thermal stability Low viscosity Low wax content Low pour point Low Floc Point Moisture free Good chemical and thermal stability • Good chemical stability: There should be little or no chemical reaction with the refrigerant or materials normally found in the system. • Good thermal stability: They should not form hard carbon deposits at hot spots in the compressor (such as valves or discharge ports). Low viscosity As the oil particles are carried along with the refrigerant through out the system, the oil is subjected to extreme temperatures - low temperature in the evaporator and high in the cylinder head. Therefore it should be in a position to flow freely at both low and high side temperature. This is the ability of an oil to maintain good lubrication properties at high temperatures and good fluidity at low temperatures, i.e. to provide a good lubricating film at all times. Low wax content & low pour point • • Low wax content. Plants are operating at low evaporating temperatures, as separation of wax particles from the refrigerant-oil mixture may cause problems by blocking expansion and regulating valves. Low pour point. Ability of the oil to remain in a fluid state at the plant’s lowest evaporating temperature. Low Floc Point The refrigerant oil may contain certain amount of paraffin which will precipitate at low temperature. The floc point is the temperature at which wax will start to precipitate from a mixture of 90% of refrigerant and 10% of oil by volume. If an oil of high floc point is used, wax will separate at expansion valve and restrict flow of refrigerant. So a low floc point is necessary for an oil to be used in the refrigeration system. Moisture free Any moisture added with oil may cause corrosion, and in the case of CFC and HCFC refrigerants would form as ice in a choked expansion or regulating valve. • When adding oil to a compressor, or doing an oil change, it is therefore important that only the type specified in the manufacturer’s operating manual is used. The oil must be clean and have no moisture content. Oil should always be stored in tightly sealed containers, in a warm place, to ensure it does not absorb moisture from the atmosphere. It is important that the procedures given in the compressor operating manual for changing and topping- up the oil are strictly followed. Refrigeration • Refrigeration is a process of cooling by the transfer of heat. Heat is a form of energy and is indestructible so if heat is removed from a space or substance to cool it to a temperature below that of its surroundings, the heat removed must be discarded to some substance at a higher temperature where it is of no consequence. Since heat will not flow freely from a body at a low temperature to another at a higher temperature, it is necessary to expend mechanical work, heat, or electrical energy from an external source to achieve it. Refrigeration thus depends on thermodynamics, heat transfer and fluid flow for its practical achievement. • The refrigerant, if first stored as a liquid under pressure, then allowed to flow at reduced pressure through an evaporator coil in the closed system, will withdraw heat from its surroundings during the evaporation stage. The heat so absorbed is removed from the refrigerated area when the vapour returns to that portion of the refrigeration equipment designed to cool down and compress it again to the liquid state for reuse. • The two main refrigeration systems in commercial use are the absorption system and the vapour compression system. Most marine refrigerating plants are of the vapour compression type. Vapour compression cycle • The vapour compression cycle takes place in a closed system, comprising a compressor, a condenser, a liquid receiver, an evaporator, and a flow control or expansion valve, interconnected by discharge, liquid and suction lines • The liquid refrigerant e.g. R22 is stored at high pressure in the liquid receiver. The liquid flows from the liquid receiver through the liquid line to the expansion valve, which regulates the rate of flow to the evaporator to suit the rate of evaporation. As it passes through the expansion valve, the pressure of the liquid is reduced to the evaporating pressure, so that the saturation temperature of the refrigerant entering the evaporator is below that required in the refrigerated space. Note that as it passes through the expansion valve, a portion of the liquid evaporates instantly (flash gas) in order to reduce the temperature of the remaining liquid to the evaporating temperature. • The liquid-vapour refrigerant mixture then flows through the evaporator, where it extracts heat from the refrigerated space, and changes to a dry saturated vapour at approximately the same temperature and pressure as that at which it left the expansion valve. • The evaporating pressure is maintained constant by the action of the compressor, which removes vapour from the evaporator at the same rate as that at which it is formed. In practice, the expansion valve regulating the refrigerant flow is designed to ensure that the vapour leaving the evaporator is slightly superheated, thus ensuring that only dry vapour is handled by the compressor. • In the compressor, the temperature and pressure of the vapour are raised by compression. The compressed vapour flows through the ‘hot gas’ discharge line from the compressor into the condenser, using water or air as the cooling medium. The vapour in the condenser first gives up its superheat as it is cooled from the discharge temperature to the saturation temperature corresponding to the condensing pressure, and then gives up its latent heat as it condenses back to a liquid. The liquid then flows from the bottom of the condenser into the liquid receiver, thereby completing the cycle. When its temperature is below the condensing temperature, it is said to be subcooled. Molliers Chart • Diagrammatic Representation of properties of Refrigerant. Useful for designing the Refrigeration system • Gives enthalpy of refrigerant at various pressures and physical states (liquid, vapour, mixture) • Also called Pressure-Enthalpy Chart. • Enthalpy is the total heat content in a substance BTUs or KJs • Specific Enthalpy is Enthalpy per unit mass BTU/lb or KJ/kg Constant Entropy LInes Constant Temperature Lines Pressure-enthalpy (P-H) or Mollier diagram • • • • • • • • Line A to B represents the change from high to low pressure, or expansion process Line B to B’ represents the amount of liquid ‘flashed-off’ in the expansion valve cooling the remaining liquid. Line B to C represents the evaporation process at constant saturation temperature and pressure in the evaporator. At point C the refrigerant is a dry saturated vapour. Line C to C’ represents the superheat absorbed by the dry saturated vapour Line C’ to D represents the compression process. Line D to E represents the superheat given up by the vapour in the condenser. At point E the refrigerant is a dry saturated vapour. Line E to F represents the condensation process at constant saturation temperature and pressure. At point F the refrigerant is a saturated liquid. Line F to A represents the sub cooling of the condensed liquid Refrigerating effect • The amount of heat absorbed by each unit mass of refrigerant as it flows through an evaporator is known as the refrigerating effect, and is equal to the difference between the enthalpy of the vapour leaving the evaporator and the enthalpy of the liquid at the flow control. • Thus, for the system shown in Fig 3, refrigerating effect, Refrigerating capacity • The rate at which a system will absorb heat from the refrigerated space or substance is known as the refrigerating capacity, and is expressed as, refrigerating capacity, • where m = mass flow of refrigerant through the evaporator (kg/s). • To achieve a specified refrigerating capacity of 15OkW, say, the required mass flow rate is Compressor capacity • The capacity of a compressor must be such that it removes the vapour from the evaporator at the same rate as that at which it is formed. To maintain a specified operating condition, a compressor must have a swept volume equal to the volume of vapour formed in the evaporator per unit time (m3/h). • To maintain constant operating conditions and produce the required refrigeration duty would require a compressor with a swept volume: • V = m x v m3 • where v = specific volume of the vapour at the compressor suction inlet, m3/kg, and v at -25°C and 1.32 bar • = 0.18m3/kg. • i.e. V = 0.94 x 0.18 x 3600 = 609 m3/h. Heat of compression • The energy input from the compressor motor to raise the pressure of the vapour to the required condensing temperature is known as the heat of compression, and is equal to the difference between the enthalpy of the vapour at the compressor outlet and inlet. • Heat of compression, Condenser duty • The rate of heat transfer from the refrigerant in the condenser to the cooling medium is known as the condenser duty, and is expressed as, • = 0.94 (470 – 230.3) = 225.3 kW Coefficient of performance • The ratio of refrigerating effect to the heat of compression is known as the coefficient of performance (COP). • Thus for the system shown in Fig 3, Properties of Marine Refrigerants Ammonia R11 ( R 717) R12 R22 R502 - 33.3 23.6 -29.6 -40.6 -45.6 Absolute evaporator pressure 2.3 at -15°C (Bar) 0.2 1.6 3.0 3.5 Absolute condensing pressure at 30°C (Bar) 11.6 0.9 7.4 12.0 13.0 Latent heat at -15°C (kJ/kg) 1314 194 159 217 166 Coefficient of performance at -150C evaporator, 30°C condensing 4.77 5.03 4.71 4.67 4.37 Evaporating temperature at atmospheric pressure (0C) Automatic freon system on board ship • Each cold compartment has a thermostatic expansion valve, which acts as the regulator through which the correct amount of refrigerant is passed. • The sketch is for a three compartment system but shows only the detail of one. Each room has a solenoid, thermostatic expansion valve and evaporator. Air blown through the evaporator coils acts as the secondary refrigerant. Regular defrosting by means of electric heating elements keeps the evaporator free from ice. The automatic defrost time switch de-energizes the solenoids to shut down the system and diverts electrical current to the heaters, for a set period. “Pump-down cycle.” • The refrigerated space temperature is monitored by a thermostat which acts to open and close the solenoid valve in the liquid line. The routine starting and stopping of the compressor is controlled by the low pressure cutout switch in the compressor suction line. As the space temperature is reduced to the set point of the thermostat, the thermostat contacts open, deenergizing the solenoid valve and stopping the flow of refrigerant to the evaporator. Continued compressor operation reduces the suction pressure to the set point of the low pressure switch, stopping the compressor. • Due to the stopping of refrigeration, the space temperature will slowly rise. When the temperature reaches the thermostat set point, the contacts close, energizing the solenoid valve, thus permitting liquid refrigerant to enter the evaporator. The refrigerant vaporizes, raising the suction pressure to the cut-in point of the low pressure switch, thus starting the compressor. There is also an HP (high pressure) cut-out with a hand re-set which operates to shut down the compressor in the event of excessively high discharge pressure. System components 1. 2. 3. 4. 5. 6. 7. Pressure gauges Compressors Oil separators Condensers Liquid receiver Liquid indicator Evaporators 8. Expansion valves 9. High pressure cut-out 10. Low pressure cutin/cut-out 11. Oil pressure cut-out 12. Room temperature control 13. Solenoid valves 14. Drier Pressure gauges • The pressure gauge on the compressor suction and discharge shows the gas pressure and also has marked on it the relative condensing temperature. It helps to check correct pressuretemperature relationships for refrigerants. Pressure gauge in the lub oil line is normally of differential type. Compressors • Refrigeration compressors are usually reciprocating type for marine refrigeration. It may be of the rotary screw displacement type or of the scroll type. Reciprocating compressors • Reciprocating compressors for systems cooling domestic store rooms are usually of the vertical in-line type. The larger reciprocating compressors have their cylinders arranged in either V or W formation with 4,6, 8, 12 or even 16 cylinders. Compressor speeds have been increased considerably over the years from 500 rev/min to the high speed of 1500 to 2000 rev/min. V-belt drive • Compressors are usually driven by Vbelts. Most are driven at less speed than the motor speed. Pulleys must be in perfect alignment and the pulley shafts (compressor and motor) must be parallel to each other. • Each crank of the crankshaft for the compressor shown carries three bottom ends. The aluminium alloy pistons operate in cast iron liners, which are honed internally. Piston rings may be of plain cast iron but special rings having phosphor-bronze inserts are sometimes fitted. These assist when running in. Connecting rods are H section steel forgings with white metal lined steel top end bushes. The crankcase and cylinders comprise a one-piece iron casting. Main bearings are white metal lined steel shells. • Gas from the evaporator passes through a strainer housed in the suction connection of the machine. This is lined with felt to trap scale and impurities scoured from the system by the refrigerant particularly during the running-in period. Freons tend to clean the circuit but the impurities will cause problems unless removed by strainers. Any oil returning with the refrigerant drains to the crankcase through holes in the diaphragm plate. Suction and Discharge Valve • There is a valve plate under cylinder head with plate type of suction and discharge valve located in it. Large diameter and very small lifts of plates offer the least resistance to the flow of refrigerant gas. Heavy springs on the discharge valve cage permit a greater valve lift to protect compressor in case of severe liquid refrigerant or oil pumping. Lubrication • Oil is supplied to the bearings and crankshaft seal by means of a gear pump driven from the crankshaft. Oil pressure is about 2 bar above crankcase pressure and the differential oil pressure gauge is necessary to compare oil pressure with that of the gas in the crankcase. There is a relief valve in the oil system set to about 2.5 bar above crankcase pressure. Protection against oil failure is provided by a differential oil pressure switch. Crankcase heaters • A certain amount of refrigerant will always be dissolved in the lubricating oil. However, large amounts of refrigerant in the oil are undesirable. Excessive dilution can result in inadequate lubrication. In addition, during compressor start-up, the lowering of the crankcase pressure will cause oil foaming due to the vaporization of the refrigerant. In severe cases, this can disrupt lubrication and can cause carryover of the liquid refrigerant and oil into the cylinder. Since marine systems typically operate on the pump-down cycle, the low crankcase pressure at shutdown limits refrigerant absorption by the oil. Crankcase heaters which come on automatically during the compressor off cycle are used to keep the oil warm and reduce refrigerant absorption. Shaft seal • A mechanical seal is fitted around the crankshaft at the drive end of the crankcase. This prevents leakage of oil and refrigerant from the crankcase. All seals use two rubbing surfaces. One surface turns with the crankshaft and is sealed to the shaft with an O-ring of synthetic material. The other surface is stationary and mounted on the housing with gasket. The rubbing surfaces can be hardened steel and bronze or ceramic and carbon. The mechanical seal is lubricated from the compressor system. Unloader mechanism • The most common method of varying the capacity of multi-cylinder compressors is to vary the number of active cylinders by holding the suction valves open. The capacity control system unloads cylinders (i.e., cuts cylinders out of operation) in response to decreases in refrigeration load based on suction pressure. • A bellows device, actuated by suction pressure can serve to cut out one or more cylinders. Under high loads (high suction pressures) none of the suction valves are held open, and all the cylinders are in operation. As the load decreases (and the suction pressure falls off), the cylinders are cut out in sequence. If the suction pressure continues to fall off, the compressor will stop on the low pressure switch. Compressor lubricating oil is used to operate the valve lifting mechanism. Since oil pressure is required to load the cylinder, the compressor will start with all controlled cylinders unloaded, thus reducing the starting load on the compressor motor. Oil separators • Oil separators may be fitted in hot gas discharge lines from the compressor. It is a closed vessel fitted with a series of baffles or a knitted wire mesh through which the oil-laden vapour passes. The reduction in velocity of the vapour as it enters the larger area of the separator allows the oil particles, which have greater momentum, to impinge on the baffles. The oil then drains by gravity to the bottom of the vessel where a float valve controls flow to the compressor crankcase. Manual Valves • Manual valves are installed at various locations in the refrigeration system to facilitate system operation, to isolate components for maintenance and for other purposes. Most of the valves used in refrigeration systems are of the packed valve type. They are of the backseating type. When in the open position, the valve is backseated to minimize the possibility of leakage. Condensers • Most marine refrigeration condensers are of the watercooled, multipass, shell-and-tube type. Seawater is circulated through the tubes, and the hot gas from the compressor discharge is admitted to the shell and condenses on the outer surfaces of the tubes. The condenser is typically constructed of a steel shell, copper-nickel tubes and tube plates, and bronze waterheads. Gas inlet, liquid outlet, purge, and water regulating valve control connections are provided. Liquid Receiver • The receiver collects the liquid refrigerant draining from the condenser. It consists of a steel shell with steel dished heads welded on each end. Sight glasses or a liquid level indicator is installed to permit determination of the amount of liquid refrigerant in the receiver. The receiver will typically have sufficient capacity to hold the entire system refrigerant charge and will retain a small liquid level during full load operation. High levels indicate overcharge and low levels indicate undercharge. • Sometimes bottom part of condenser serves as the liquid receiver Liquid Indicators (Sight Glasses) • Liquid indicators or sight glasses are commonly installed in the liquid line to indicate a proper refrigerant charge. Bubbles appearing in the liquid stream are an indication of a shortage of refrigerant. Some indicators also include a moisture indicator. A portion of the indicator will change color based on the relative moisture content of the refrigerant. Refrigerant drier • Shell is filled with a desiccant such as activated alumina or silica gel which absorbs moisture and also acts as a filter. Even small amounts of moisture can cause problems such as frozen thermostatic expansion valves, so it is important to remove sufficient moisture to prevent the release of water in the low pressure portions of the system. Evaporators • Marine evaporators are usually forced convection evaporators. They are in common use in ship’s provision refrigeration and air conditioning systems. The units consist of a cooling coil with finned tubes, a motor-driven fan, and drain pan, all enclosed in a sheet metal casing. Units designed for sub-zero temperature applications are usually fitted with an electric resistance defrost system. Expansion device • The basic functions of an expansion device used in refrigeration systems are to: 1. Reduce pressure from condenser pressure to evaporator pressure, and 2. Regulate the refrigerant flow from the high-pressure liquid line into the evaporator at a rate equal to the evaporation rate in the evaporator • Under ideal conditions, the mass flow rate of refrigerant in the system should be proportional to the cooling load. It is desirable that liquid refrigerant should not enter the compressor. In such a case, the mass flow rate has to be controlled in such a manner that only superheated vapour leaves the evaporator. Thermostatic Expansion Valve • While there are a number of devices available to control the flow of refrigerant to the evaporator, such as the capillary tube, the float valve, and the constant-pressure expansion valve, the thermostatic expansion valve is the device most commonly found in marine systems. The thermostatic expansion valve responds to the evaporator temperature and pressure and maintains a constant superheat at the outlet of the evaporator. As refrigerant is fed to the evaporator, the liquid boils off into a vapour. By the time the refrigerant gas reaches the end of the evaporator, it is superheated. Feeding more refrigerant to the evaporator will lower the superheat temperature, while feeding less refrigerant will raise the superheat temperature. • This consists of a feeler bulb that is attached to the evaporator exit tube so that it senses the temperature at the exit of evaporator. The feeler bulb is connected to the top of the bellows by a capillary tube. The feeler bulb and the narrow tube contain some fluid that is called power fluid. system. To ensure the correct operation of the valve, the bulb must be securely clamped to the evaporator outlet line. • Pb - Bulb pressure on the upper side of the diaphragm, tending to open the valve, where Pb is the saturation pressure of the refrigerant in the bulb, corresponding to the temperature of the gas at the evaporator outlet. • Po - Evaporator pressure on the lower side of the diaphragm, tending to close the valve, where Po is the saturation pressure of the refrigerant at the evaporator inlet, and Δp is the pressure drop between the evaporator inlet and outlet. • Ps - Pressure exerted by the regulating spring, tending to close the valve. The spring tension, set by the regulating spindle, controls the degree of superheat; a typical superheat value is 4°C to 6°C. • At any constant operating condition, these forces are balanced and Pb = Po + Ps • If the superheat starts to rise, the bulb pressure increases, Pb > Po +Ps and the valve is moved in the opening direction, admitting more liquid and restoring the constant operating condition. • If the superheat falls, Pb < Po + Ps and the valve is moved to the closing position, reducing the supply of liquid. • In practice, to achieve the desired degree of superheat at the evaporator outlet, dry expansion evaporators require up to 20 per cent of their cooling surfaces to be available to superheat the gas, the precise area varying with demand. TEV with external equalizing connection • There is an appreciable pressure drop in the large evaporators. Additional control is introduced by incorporating a pressure equalising connection. This connection eliminates further increase in the superheat temperature to compensate for the reduction in pressure, and so allows an increase in the effective area of the evaporator. Back pressure valve in vegetable room • If the evaporator in vegetable room is kept at common pressure of meat room and fish room, then ice formation will take place in this evaporator also. This ice will be formed from the moisture of fruits and vegetables stored in the room and they will become desiccated. • The bellows pressure area and the valve area are equal. The adjustment spring and the evaporator pressure change can operate the valve motion. The valve has a gauge opening to check pressure of the evaporator. For R22 system, this pressure may be 4 bar gauge. Solenoid valve • The solenoid valve is thermostat controlled valve which provides automatic opening of and closing of liquid line to the evaporator. When the coil (3) is energised, the pilot orifice (4) is opened and the diaphragm (1) moves into open position (vice versa when coil is deenergised). Thermostats • Thermostats are temperature-controlled electric switches. It is used to control the temperature in a refrigerated space by ‘opening and closing’ a solenoid valve in the liquid line. • Three types of element are used to sense and relay temperature changes to the electrical contacts. 1. A fluid-filled bulb connected through a capillary to a bellows. 2. A thermistor 3. A bi-metal element High Pressure Cut-out • There are a number of faults which cause high discharge pressure, including loss of condenser cooling, air in the system and overcharge. • In the event of overpressure on the condenser side of the compressor the high pressure cut-out will cause the compressor to shut down. The device is re-set by hand. • The bellows in the cut-out is connected by a small bore pipe between the compressor discharge and the condenser. The bellows tends to be expanded by the pressure and this movement is opposed by the spring. The adjustment screw is used to set the spring pressure. During normal system operation, the switch arm is held up by the switch arm catch and holds the electrical contact in place. Excessive pressure expands the bellows and moves the switch arm catch around its pivot. The upper end slips to the right of the step and releases the switch arm so breaking the electrical contact and causing the compressor to cutout. The machine cannot be restarted until the trouble has been remedied and the switch re-set by hand. Low pressure controller • The low pressure control stops the compressor when low suction pressure indicates closure of all cold compartment solenoids. When the pressure in the compressor suction rises again due to one or more solenoids opening, the low pressure control restarts the compressor. • The controller is operated through a bellow which monitors pressure in the compressor suction. A pressure differential between cut out and cut in settings is necessary to avoid hunting. The push pin operates the switch through a contact which is flipped open or closed through a coiled spring plate. With the contacts open the spring is coiled as shown. Outward movement of the pin compresses the spring and this then flips the contact to close the compressor starting circuit. Oil pressure safety cut-out • This is used to protect against too low oil pressure in forced lubrication systems. It is a differential control, using two connections. One side responds to the suction pressure of compressor and the other responds to the oil pressure. If the differential oil pressure fails, or falls below a minimum value, the control stops the compressor after a certain time delay. Carriage of refrigerated cargo • Refrigerated cargo can be carried in 1. Specialised “Reefer Ships” 2. Refrigerated containers. Reefer Ships • Reefer ships are effectively large refrigerators, heavily insulated with modern glass fibre or similarly efficient insulation , shuttered with bright metal that prevents taint and is easy to clean. They are ships that tend to be divided into many more spaces than conventional dry cargo ships, with several ’tweendecks spaces, so that different commodities can be separated and carried, if required, at different temperatures. Cleanliness and the maintenance of optimum temperatures are the pre-requisites. • The main features of a modern, 450,000 ft3 reefer vessel are as follows. • Four holds with 4 or 5 cargo decks, each with the same clear head of 2.2m to minimize lost load space when storing standard pallets (1.2m long x 1.0 m wide x 2.1 m high maximum). These decks are arranged usually in eight air tight temperature zones, with the air coolers placed along the bulk-heads serving one or sometimes two decks. Variable speed fans are placed above the coolers forcing air through, under the grating then vertically from bottom to top through the cargo and back to the coolers. • Four fast cranes able to handle a 40 ft container laden, for example, with frozen meat. • Space for eighty or more integral containers on the weather deck, and space for fork lift trucks and pallet cages. Air circulation and refreshing • Between 90 and 120 air changes of the net volume per hour is usually provided in holds. Such volume flow guarantees good and uniform cooling rate of palletized and bulk cargo and allows for imperfect storage. This air rate will be reduced on completion of cooling down and when frozen cargo is carried. • Air refreshing rates to remove carbon dioxide, ethylene and other volatiles, can be two or three air changes per hour. Brine battery and air • In this system, brine instead of primary refrigerant is circulated through the batteries. Air is cooled in a brine cooling system and cold air ducted to the cargo spaces. • Brine is relatively easy to regulate. The system shown is arranged with three separate refrigeration and brine circuits with connections from both brine systems to the air cooler batteries (or grids). Brine is inexpensive, being made with calcium chloride and fresh water to a gravity of about 1.25. Sodium dichromate or lime may be added to maintain the brine in an alkaline condition. Calcium Chloride Brine CALCIUM CHLORIDE / BRINE Specific Gravity Hydrometer Reading Freezing Point of Solution (Twaddell) °C °F 1·20 40 -21 -6 1·21 42 -23 -9 5 1·22 44 -25 -13 1·23 46 -27 -17 1·24 48 -30 -21.5 1·25 50 -32 -26 1·26 52 -35 -31 1·27 54 -38 -37 1·28 50 -42 -44 1·29 58 -51 -60 Showing third pair of headers served by a brine heater and third pump, so that any battery can be individually defrosted by circulating the warm brine. Also introduced is a brine "injection cross connection from the delivery of pump No. 1 to the suction of' pump No. 2. Brine injection is used so that evaporator No. 1 can assist evaporator No. 2 when No. 1 is set to deliver brine at a lower temperature than No. 2. A further refinement of this injection is the by-pass arranged across the inlet and outlet of evaporator No. 2 so that the cooling, of the brine circulating in No. 2 system can be achieved entirely by injection if desired. Additionally, there also has to be a brine make-up tank, in which solid calcium chloride is dissolved, for topping up the system. An overflow connection from the header tank, a safety pressure relief line from the brine heater, and a sighting connection to which the return from any space can be diverted, are all arranged to terminate over this make up tank. Decline of reefer ships • The conventional reefer fleet shows a negative growth rate since 1994, while the container fleet expanded at an even higher rate than anticipated. There are approx. 1250 reefer ships larger than 10,000 ft3 in operation today, with an average size of 267,000 ft3. • The largest reefer capacity in a single ship is now found, not on a "traditional" reefer, but aboard a large container ship. Refrigerated containers • Two basic types of refrigerated containers were developed for use in the international reefer trade. 1. The insulated box connected to the ship’s central plant and a cold air circulation system; sometimes known as an isotherm container, or porthole container. 2. The insulated box incorporating its own ‘plug-in’ refrigeration unit within the standard module; usually known as an integral container. • The containers most frequently found in practice are 20ft (6.097m) or 40ft (12.19m) long, 8 ft (2.4m) wide and either 8ft or 8.5ft (2.56m) high. Porthole container • Systems designed for the cooling of refrigerated containers employ trunkings arranged so that containers stowed in stacks between built-in guide rails, can be connected to the suction and delivery air ducts of the ship’s refrigeration plant by bellows pieces operated pneumatically. The air is cooled either by brine or direct expansion batteries and the containers are arranged so that one cooler can maintain a stack of containers at a given temperature. The temperature of the return air duct for each container is monitored. Provision of a cooler and trunking system for maintaining container temperatures must also be provided at container terminals. Refrigeration machinery • Refrigeration systems in porthole container ships are generally similar to those used in conventional reefers, i.e. the primary refrigerant-usually R22-is circulated through compact shell and tube evaporators with refrigerant flow being controlled by conventional thermostatic expansion valves. • The brine, when cooled to the appropriate temperature, is circulated through finned tube air-to-brine heat exchangers, with some form of bypass valve to maintain the air at the prescribed temperature. • It is advisable to keep a mean temperature difference between brine and air of 3°C to 40C, so that relative humidity within containers is maintained at a high level of approximately 90%. • Defrosting of coolers is usually achieved by circulating hot brine, the time taken being about 30 minutes. Intermodal Refrigerated Containers • The number of units in operation in 2005 is approximately 750,000, equivalent to 1,270,000 TEU. TEU “Twenty foot Equivalent Unit” refers to a unit of volume corresponding to a twenty foot ISO container, which used to be the standard container. But today 40 foot containers of the “HighCube” type dominate the market for new reefer containers. Their size surpasses two TEU and therefore the increase in container traffic exceeds the increase in the number of units at the present time. • There are no more than 18,000 containers of the porthole type in operation now and no new porthole containers are built. • About 200 new container ships and 160,000 TEU new refrigerated containers were built annually during the last few years. Integral refrigerated container • The integral container will have an independent refrigeration unit which enables the container operator to carry cargoes in the temperature range -25°C to +2O0C. These units are mostly electrically driven and are plugged in to appropriate power points on shore or onboard ship. Nowadays, a number of units are compact enough to allow for a removable diesel alternator set to be fitted when the container is travelling on the road, or sited in areas where a suitable 3-phase power supply is not available. Ships for the carriage of integral containers • The holds of these ships are normally uninsulated. In order to maintain the hold temperature below ambient, a very powerful ventilation system is fitted to forward and aft bulkheads with air intakes positioned in the vicinity of each container integral unit. Fresh air may be supplied to the holds at tank top level or through openings in the vicinity of hatches. As soon as the containers are loaded and positioned up to 6 high in guides, the refrigerating units are plugged to the electrical power supply and those containers having water cooled condensers are connected to fresh or sea water cooling systems permanently fitted onboard. • The fresh water is circulated in a closed circuit and cooled by sea water in heat exchangers, its outlet temperature being 4 to 5°C higher than the sea water outlet temperature from the heat exchanger. The fans of air cooled condensers will not be running. Some ships can carry over 600 integral containers under the deck. Refrigerated container on deck • Individual containers with their own refrigeration plant are connected to the 440 or 220 V a.c. sockets provided on deck. These containers may be arranged for ships’ systems with either 440 or 220 V by provision of a direct connection for a 22OV supply to the self-contained refrigerator and a 440V connection through a step down transformer. Air cooler fans • Fans may be either centrifugal or of the propeller type; the air circulation systems being based on a pressure requirement of about 50 mm W.G. (water gauge). All of the electrical energy of the fan motors is dissipated in the form of heat and has to be removed by the refrigerating plant. Fan output should be variable so that it can be reduced as heat load diminishes. There was no problem with d.c. motors but with a.c. either the motors are two speed, or each Controlled atmosphere • Fruits and vegetables are actually alive, their cells are metabolising, consuming oxygen and carbohydrate and producing water vapour, carbon dioxide and heat. Also small amounts of ethylene gas (C2H2) are produced. Therefore it is not enough to just refrigerate the cargo, a supply of fresh air must also be provided. Controlled atmosphere • If insufficient fresh air is supplied, several things can happen: 1. Too much carbon dioxide can produce tissue damage, in apples this condition is known as brown heart 2. Not enough oxygen can result in anaerobic respiration, in this case the metabolic pathway is incomplete and the final product becomes alcohol resulting in alcoholic fruit 3. Too much water vapour can encourage the development of moulds, rots and fungi. However, not enough is just as bad as it promotes desiccation of fruit and vegetables 4. Ethylene gas is ripening hormone, very small quantities building up can promote premature ripening in fruit or degreening in green vegetables Controlled atmosphere • Rate of breathing or respiration can be lowered and thus ripening slowed down either by lowering the temperature or by reducing the amount of oxygen and increasing the amount of carbon dioxide in the surrounding atmosphere. • Controlled atmosphere is an inert gas system used to extend the storage life of seasonal perishable products and has been used for many fruits and vegetables; primarily apples and pears in the past, and now mainly for bananas. Controlled atmosphere • To successfully store fruit for long periods, the natural ripening of the produce has to be delayed without affecting the eating quality. This is achieved by reducing the temperature of the fruit to the lowest level possible without causing damage through freezing or low temperature breakdown. To further delay ripening, the oxygen supply in the space is reduced to levels below that of the natural atmosphere. This level is below the level required to support human life. Controlled atmosphere • The precise levels of temperature, oxygen and carbon dioxide required to maximize storage life and to minimize storage disorders are extremely variable, depending on type of produce, growing conditions and maturity. Optimum storage conditions can vary from farm to farm and from season to season. • On reefer vessels, oxygen (02) and carbon dioxide (CO2) levels and relative humidity (RH) in controlled atmosphere zones (cargo chambers) can be independently controlled within close tolerances, irrespective of type, temperature and volume of cargo carried and the length of the voyage. • A typical modern controlled atmosphere marine system would be expected to have flexibility to control gas levels within the following ranges: • 02 : < 1 – 8 % • CO2 : 0-15% • RH : 40-90% • The required oxygen and carbon dioxide levels can be achieved in a number of different ways. Oxygen level • The oxygen level can be decreased by: • a ) injecting pure nitrogen as a gas or liquid from bottles or storage tanks; • b) burning propane in an open flame burner, or a burner with a catalyst; • c) generating gas (nitrogen with a low oxygen level) on board from compressed dry and clean air, using high pressure membranes, etc. Carbon dioxide level • • • • • • • The carbon dioxide level can be increased by: a ) injecting carbon dioxide gas; b) fruit respiration; The carbon dioxide level can be decreased by: a ) fresh air or gas injection; b) hydrated lime; c) water scrubbing. Relative humidity • • • • Relative humidity can be increased by: a) injecting water mist; b) steam; c) evaporating water, etc. Human body temperature • The human body is warm-blooded and that it must maintain a body temperature within very close limits. It uses food as a fuel, converting it into energy. Some of this energy may leave the body in the form of external work done; the balance represents heat production within the body and is available for the maintenance of body temperature. As the body must produce heat continuously, it must also lose it at a rate that provides control of body temperature. • The human body maintains a basic minimum rate of heat production at about 75 watts during sleep and about 120 watts when awake but sedentary. As bodily activity increases, the rate of oxidation of food, with its attendant release of energy, must increase. The level of heat production for light work will be about 190 watts, the extreme value for heavy work, about 700 watts. • It is possible to recognize the main ways in which the body will lose heat and to relate them in a simple equation as follows: • H=S+E+R+C • where H = rate of internal heat production S = rate of heat storage in the body E = rate of loss by evaporation R = rate of radiant energy exchange with surroundings C = rate of loss by convection • Most of the heat, however, must be rejected from the body surfaces through convection losses to the surrounding air, by radiation exchanges with surrounding surfaces, and by the evaporation of perspiration from the skin when required. The body involuntarily makes adjustments that influence these processes by increasing or decreasing the rate of heat loss as required. It can attempt to induce evaporation by pouring out perspiration on the skin when it is too warm. • Whether or not these measures are effective depends on the temperature, moisture content and motion of the air, and the temperature of the surrounding surfaces. The amount of clothing becomes a major factor also since it is interposed between the skin and the influence of the surroundings and becomes involved in the convective, evaporative, and radiative losses for those areas of the body that are covered. • Increasing the air temperature tends to reduce convective and radiative losses and to increase evaporative losses under sweating conditions. If air temperature rises above skin or clothing surface temperature there will be a heat gain rather than a heat loss by convection, and this must be offset by increased losses in other ways. Perspiration and respiration • Perspiration and respiration components of the evaporative loss are dependent upon the rate at which water is actually evaporated. This depends in turn upon the degree of saturation of the air, which may be measured in terms of relative humidity. Thus, there can be no evaporative loss with air saturated at 37°C, regardless of the rate of perspiration unless the body temperature rises above normal. On the other hand, under conditions that lead to comfort, with only light activity the main evaporative loss is that from the lungs, with little or no contribution from perspiration, at least until the upper limits of comfort are reached. Air motion • Air motion is another factor that can have a marked effect. Increased air speed over the body and clothing surfaces can increase convective losses and, when there is perspiration, the evaporative losses as well. Thus, under conditions of high temperature and high humidity, discomfort can often be greatly reduced by increasing the air flow. It is of more than passing interest to note that under these conditions even high air speeds can be pleasant. With cooler conditions, however, even small localized air circulation may give rise to complaints of chilliness. Effective Temperature • It is the temperature of still, saturated air which would produce the same feeling of warmth. Effect of Relative humidity • A marked influence of relative humidity was found. The value in summer at which 97 per cent of the subjects were comfortable was 21.50C Effective Temperature, which corresponds to conditions of 27.50C degrees at 10 per cent, 24.50C at 50 per cent and 21.50C at 100 per cent relative humidity. Correspondingly, the value for winter conditions at which the greatest proportion of subjects was comfortable was about 200C, which can be obtained with 260C at 10 per cent and 25.50C at 50 per cent relative humidity. Air purification • Outside air must be introduced to all living spaces, although the amount of fresh air necessary to sustain life is very small indeed. They are governed by factors such as body odours and smoking, which may require a fresh air supply of 12 litre/s per person or more. Air conditioning • 1. 2. 3. 4. Air conditioning is the process of treating air so as to control simultaneously its temperature, humidity, cleanliness and distribution to meet the requirements of the conditioned space. Action involved: Temperature control Humidity control Air filtering, cleaning and purification Air movement and circulation • Winter conditioning relates to increasing temperature and humidity whilst summer conditioning relates to decreasing temperature and humidity. Basically the practical difference is dependent upon whether the air fluid is passed over a hot grid (steam) or cold grid (brine or direct expansion refrigerant). Per Cent Relative Humidity • Is the mass of water vapour per m3 of air compared to the mass of water vapour per m3 of saturated air at the same temperature. This also equals the ratio of the partial pressure of the actual air compared to the partial pressure of the air if it was saturated at the same temperature i.e. Dalton’s Laws of Partial Pressures • The pressure exerted by a mixtures of gases and vapours is the sum of the pressure which each would exert if it occupied the same space alone, assuming no interaction of constituents. • Barometer pressure = partial pressure of N2 +p.p. O2 +p.p. of H2O • from Dalton’s Laws, viz: 1. Pressure exerted by and the quantity of, the vapour required to saturate a given space (i.e. exist as saturated steam) at any given temperature, are the same whether that space is filled by a gas or is a vacuum. Dew Point • When a mixture of dry air and water vapour has a saturation temperature corresponding to the partial pressure of the water vapour it is said to be saturated. Any further reduction of temperature (at constant pressure) will result in some vapour condensing. This temperature is called the dew point. Air at dew point contains all the moisture it can hold at that temperature, as the amount of water vapour varies in air then the partial pressure varies, so the dew point varies. • It can be seen that cooling at constant pressure brings the low pressure superheated vapour to the dew point after which condensation occurs. It can also be noted that cooling at constant temperature increases the partial pressure until the saturation point is reached thus relative humidity can be found . Dry and Wet bulb temperature • Dry bulb temperature - Air temperature indicated by a thermometer in a dry condition. • Wet bulb temperature – If a moist wick is placed over a thermometer bulb, the evaporation of the moisture from the wick will lower the temperature reading. This temperature is known as ‘Wet bulb temperature’. Psychrometric Chart • Psychrometry is the study of the properties of mixtures of air and water vapour. This subject is important to airconditioning because the systems handle air-water vapor mixtures, not dry air. Some air-conditioning processes involve the removal of water from the air-water vapor mixture (dehumidification) while some involve the addition of water (humidification). A convenient way to represent the properties of air-water vapor mixtures is the psychrometric chart. On the chart, such properties as dry bulb temperature, wet bulb temperature, dew point, relative humidity, humidity ratio, specific volume, and enthalpy are presented in graphical form. Understanding the psychrometric chart Reading the psychrometric chart • In order to locate any condition of air on the chart, two independent properties must be determined. The air condition can then be plotted on the chart, and all other properties can be read from the chart. Dry bulb temperatures are read along the horizontal scale at the bottom of the chart, humidity ratio is read along the right-hand vertical scale, and the wet bulb temperature, dew point temperature, and enthalpy are read along the diagonal scale at the upper left. Lines of constant relative humidity and specific volume are labeled in the body of the chart. • From wet and dry bulb readings the various properties of the air-vapour mixture can be estimated. Enthalpy is a function of the wet bulb temperature, and moisture content and vapour pressure are functions of dew point. The chart gives a quick performance check on the air entering and leaving the cooling coil, dew point, temperature, humidity, enthalpy, etc. Comfort Conditions • Comfort under summer conditions is dependent on dry and wet bulb readings i.e. relative humidity as well as air motion. For a given degree of air turbulence (75 mm/s to 127 mm/s), relative humidity between 30% and 70%, average 50%, and thermometer readings 19°C to 250C, average 220C, gives the best degree of summer comfort. Air at low temperature and high humidity can be as comfortable as air at high temperature and low humidity. 6 psychometric zone classification 6 zones worldwide based on temp and humidity conditions • Hot & Dry: Temp: >33C; RH: 40-12% • Hot & Humid: Temp: >33C; RH: 33-60% • Warm & Dry: Temp: 27-33C;RH:4-80% • Warm & Humid: Temp: 27-33C; RH:20-55% • Moderate: Temp: 20-27C; RH:30-70% • Cool: Temp: 15-20C; RH:45-70% Comfort Conditions Comfort Conditions • As the temperature of air is reduced, so too is its capacity for carrying water vapour. Air, with an initial dry bulb temperature of 360C and relative humidity of about 60%, will, when cooled to 27°C dry bulb temperature, have a relative humidity of 100%. The temperature drop reduces the capacity of the air to carry moisture in suspension. Further cooling will cause moisture to be precipitated. Air cooled to a comfortable temperature level of 21°C but having a relative humidity of IOO%, would not be able to take up further moisture and perspiration would not be evaporated. People in an atmosphere at 21°C with IOO% relative humidity would be uncomfortable. • The remedy of dehumidifying the air is achieved by overcooling to precipitate excess moisture, (removed via the drain) so that when air is brought to the correct temperature, its humidity will be at an acceptable level. Thus the air could be overcooled to about 10°C dry bulb temperature so that warming to about 210C, would bring humidity to about 50%. The air is warmed in the trunking or by contact with warmer air in the space. • An adequate drain is required to remove what can be a considerable flow of water from dehumidification of the air. Heating • When the temperature of air is increased, so too is its capacity for carrying water vapour. Air, with a very low initial dry bulb temperature of -5°C and relative humidity of about 5O%, will, when heated to 21°C dry bulb temperature, have a relative humidity of about 10%. The temperature rise increases the capacity of the air to carry moisture in suspension. Air heated to a comfortable temperature level of 210C but having a relative humidity of 10% will readily take up moisture whether from perspiration or from the nasal passages and throat. People in an atmosphere at 21°C but 10% relative humidity, would experience discomfort from dryness in their nose and throat and on the skin. • The remedy is to humidify the air with a hot water or steam spray. This action increases humidity towards 100% relative humidity and also increases the temperature from -5°C and 50% relative humidity to say, + 7°C. Straight heating by the zone heater bringing the air to about 210C, will drop relative humidity to 40%. The humidity will be at an acceptable level but is kept low to minimize condensation on any very cold external bulkheads. Exercise • Outside air at 35°C and 60% relative humidity is to be conditioned by cooling and heating so as to bring the air to within the "comfort zone". Using the Psychrometric Chart plot the required air conditioning process and estimate (a) the amount of moisture removed (b) the heat removed and (c) the amount of heat added Cooling to DBT27 RH100% Over cooling to DBT15 RH100% Heating to DBT25 RH50% Solution • (a) the amount of moisture removed [22-10=11.5g of H20/kg of dry air] • (b) the heat removed [(1)-(2), qcool 88-40= 48kJ/kg-dry-air] • (c) the amount of heat added [(2)-(3), qheat 50-40= 10kJ/kg-dryair]. Air conditioning circuit Marine air conditioning unit Types of air conditioning systems • Air conditioning systems may be divided into two main classes – the central unit type in which the air is distributed to a group of spaces through ducting and the self-contained type, installed in the space it is to serve. • The central unit type is the most widely used, in one or other of a number of alternative systems, characterized by the means provided to meet the varying requirements of each of the spaces being conditioned. The systems in general use are as follows: 1. Zone control system; 2. Double duct system; Zone control system • The accommodation is divided into zones, having different heating requirements. Separate air heaters for each zone are provided at the central unit and the temperature of the air leaving the heater is controlled. Air quantity control in each room served gives individual refinement. • The regulation of temperature by individual air quantity control in this system can give rise to difficulties. For instance, a concerted move to reduce the air volume in a number of cabins would cause increased air pressure in the ducts, with a consequent increase in air flow and possibly in noise level at other outlets. Double duct system • In this system, two separate ducts, one with cool dehumified air and the other with warm humid air. These separate airstreams are mixed just before they reach the space to be conditioned. Through dampers that control and balance air, each different space in the ship can be conditioned as needed. The air control in this system is excellent. A single duct air return is used. Double duct system SAFETY 1. Mechanical hazards 2. Electrical hazards 3. Chemical hazards • Freon cannot be seen or smelt! • Freon is heavier than air so it will settle and remain at the bottom of the compartments. • Freon is extremely harmful if it comes into contact with the eyes. • Freon is suffocating because it displaces air. • If you inhale high concentrations of Freon, it attacks the nerve system. • When Freon comes into contact with hot surfaces and starts to burn, it can give off poisonous gases. • Freons, if released into the air, may cause depletion of the Ozone Layer which contributes to the greenhouse effect. Refrigerants are not to be released into the atmosphere. They must be drawn into the condenser/receiver or into a separate cylinder. • Most refrigerants mix with oil so oil drained from a refrigeration system must be clearly labelled and disposed of separately. • Refrigerants must not be mixed. • If you start feeling faint or dizzy as you enter a compartment - don't think twice - evacuate! If a refrigerant leak occurs • Evacuate compartment immediately. • Sound alarm and get crew in an up-wind position. • If leak is in engine room shut down machinery. • Turn vessel into wind if still possible. • Do not enter compartment without ventilating the compartment. • Ventilate compartment. Remember Freon sinks to the bottom of the compartment and is very hard to remove. Try to force airflow down into the bottom of the compartment to force the Freon upwards. • Refrigerant pipes are lagged and constantly damp. This means that pipe coatings and surface can deteriorate relatively quickly. Check pipes regularly and make sure the coating is maintained. • Inspect Blower Rooms regularly and keep them clean and dry. Often, they are neglected areas. It is a great idea to fit an exhaust fan for the blower room and start the same and wait for a few minutes before entering. • When entering compressor rooms, start the exhaust fan and wait for a few minutes before entering. • Where flexible hoses are used, only use refrigerant tolerant hoses. Try to avoid using flexible hoses wherever possible. • Maintain fittings such as valves and gauges in good order. • Mark pipes to show what type of refrigerant they have in them. • Refrigerants are supplied in metal cylinders which will corrode in the salt environment. Make sure these are left in dry storage (preferably ashore). • When working on compressor crankcase, for draining/changing oil, ensure the crankcase is totally depressurised. Retain a firm grip on the drain plugs and other connections so that they do not fly off uncontrollably. Safe handling of refrigerants. • Ensure that personnel who handle refrigerants are properly trained in their safe use and handling, and have reviewed the MSDS for the refrigerant used. • Wear safety goggles and gloves at all times when handling refrigerants or servicing a refrigeration system. • Wear the proper respiratory protection while working with refrigerants. Check the MSDS for the proper level of protection required. • Proper ventilation or respiratory protection is required for any work on equipment in an enclosed area where a leak is suspected. • Always ventilate or test the atmosphere of an enclosed area before beginning work. Many refrigerants which may be undetectable by human senses are heavier than air and will replace the oxygen in an enclosed area causing loss of consciousness. • Inhaling refrigerants can cause sudden death. Intentional inhalation of refrigerants to produce intoxication can cause the heart to cease functioning properly and may be fatal. • Be certain that the Refrigerant Recovery Cylinder being used is the Refillable Type and has the capacity to contain the refrigerant to be added to its contents. • Refrigerant cylinders should never be filled over 80% of their capacity (liquid expansion may cause the cylinder to burst). • Label the cylinder with the contents using the appropriate colour code • Check the I.C.C. cylinder stamp to ensure the cylinder is safe. Always check the refrigerant number before charging to avoid mixing refrigerants. • Always check for the correct operating pressure of the refrigerant used. Use gauges to monitor the system pressure. • Always charge refrigerant into the low side of the system to avoid damaging the compressor, or causing the system to rupture. • R-717 and R-764 are very irritating to the eyes and lungs. Avoid exposure to these refrigerants. • R-717 is slightly flammable and mixed with the proper proportions of air may form an explosive mixture. • Fluorocarbon refrigerants should be treated as toxic gases. In high concentrations, these vapors have an anesthetic effect, causing stumbling, shortness of breath, irregular or missing pulse, tremors, convulsions, and even death. • Ammonia is a respiratory irritant in small concentrations and is a life threatening hazard at 5,000 parts per million (ppm). • Ammonia is also flammable at a concentration of 150,000-270,000 ppm • Always stand to one side when operating an ammonia valve. Ammonia can burn and damage the eyes, or cause loss of consciousness. Ammonia leaks may be detected by their smell, or with a sulfur candle or sulfur spray vapor. • Refrigerant oil in a hermetic compressor is often very acidic causing severe burns. Avoid skin contact with this oil. • Liquid refrigerant on the skin may freeze the skin surface causing frostbite. If contact with the skin occurs, wash immediately with water, treat any damaged skin area for frostbite, and seek medical treatment. • Never cut or drill into an absorption refrigeration mechanism. The high pressure ammonia solutions are dangerous and may cause blindness if the solution contacts your eyes. • Ensure that all liquid refrigerant is removed and the pressure is at 0 psi before disassembling a system. • Do not smoke, braze, or weld when refrigerant vapors are present. Vapors decompose to phosgene acid vapors and other products when exposed to an open flame or hot surface. • When soldering, brazing, or welding on refrigeration lines, the lines should be continuously purged with low pressure carbon dioxide or nitrogen. • Following work, the lines should be pressure tested with carbon dioxide or nitrogen. • If refrigerant makes contact with the eyes, immediately wash with mineral oil as this absorbs the refrigerant. Then wash your eyes with a prepared boric acid solution. • If the refrigerant is ammonia, wash with water for at least 15 minutes. Seek medical attention as soon as possible. • Purged refrigerants must not be released into the atmosphere. Federal law governs their disposal, and they must be collected and disposed of properly. • Do not allow temperatures where refrigerant cylinders are stored to reach 125 degrees F. Temperatures can easily exceed 125 degrees F in your vehicle during hot weather. • Inspect refrigerant cylinders regularly. Do not use the cylinders if they show signs of rust, distortion, denting, or corrosion. Store cylinders secured and upright in an area where they will not be knocked over or damaged. • Beware of valve spindles and other components which can fly off because of high pressures. Enough fatal accidents have been reported which have occurred because of personnel coming in direct line of loose flying off parts fitted on pressurized equipments. • Special Note: Always check MSDS before handling any refrigerant and follow all safety requirements. Exposure to large concentrations of fluorocarbon refrigerants can be fatal. In high concentrations, these vapors have an anesthetic effect, causing stumbling, shortness of breath, irregular or missing pulse, tremors, convulsions, and even death. Take care and be safe. Precautions when working in Refrigerated Spaces: • Keep refrigerated spaces dry and free of water and condensate accumulation due to choked drains. Moisture inside refrigerated spaces will reduce the efficiency of refrigeration by accumulating on the evaporator coils as frost. It will also make the floor slippery and can cause accidents due to slips, trips and falls. • Take all personal safety precautions not to inhale any refrigerant vapours which could have possibly leaked out into the chamber which can also cause chemical poisoning of the human system. Refrigerant Vapour under high temperature can liberate Phosgene gas, which is highly poisonous. • Always vent refrigerated spaces for sufficient duration before personnel entry. • When working in refrigerated spaces with frozen cargoes, wear protective warm clothing. • When working in refrigerated spaces with chilled cargoes, especially fruits, beware of accumulated pockets of carbon-di-oxide and ethylene which could cause an oxygen deficient atmosphere. This is particularly applicable for controlled, regulated and modified atmospheres. • It is safer always for at least two persons to enter and work in refrigerated spaces at a time. • All personnel should be aware of the push button alarms, nearest escape routes and emergency exits from refrigerated spaces. Mechanical hazards • Personnel should be aware at all times that refrigeration systems contain liquids and vapours under pressure. Suitable precautions must be taken when opening any part of the system to guard against the pressure hazard. • Compressors must be operated within their design parameters. Mechanical hazards • Personnel must not start the compressor until they have taken steps to verify that: 1. All guards on coupling, belts drives, and fans are in place, and other personnel are not in positions that might be hazardous when the plant is in operation; 2. The compressor discharge stop valve is open. Mechanical hazards • Opening up part of the system will necessitate the loss of a certain amount of refrigerant to atmosphere. It is essential that the amount of refrigerant which escapes is kept to a minimum and appropriate steps are taken to prevent hazardous concentrations of refrigerant accumulating. Under certain conditions, liquid refrigerant at low temperature may be present. Contact with this liquid must be avoided. Electrical hazards • Before carrying out maintenance or repair procedures, persons concerned must ensure that equipment is isolated from the electrical supply and tests made to verify that isolation is complete. Whenever possible, precautions must be taken to prevent the circuit being inadvertently energised i.e. withdraw the mains fuses. Chemical hazards • HCFC and HFC refrigerants can present a danger to life by excluding air. Inhalation of very high concentrations of the vapour, even for short periods, must be avoided since this maybe dangerous and can produce unconsciousness or prove suddenly fatal due to oxygen deficiency. The refrigerant vapour is heavier than air, and in static or poorly ventilated situations may be slow to disperse. Anyone suffering from the effects of inhalation of the vapour should move or be moved to the open air. Chemical hazards • Care must be exercised before entering any area where the presence of high vapour concentration is suspected. The vapour will displace air upwards out of cargo chambers, ships’ engine rooms, etc., and tend to collect at deck level and in pits and trenches. • Should accidental escape of the refrigerant occur indoors, adequate fan assisted ventilation must be used to disperse the vapour, preferably by extraction at ground level, before entering the area. When any doubt exists it is recommended that breathing apparatus should be worn. Chemical hazards • HCFC and HFC refrigerants are non-flammable, but refrigerant vapour coming into contact with temperatures of 315°C and above (burning cigarettes, gas burners, electrical heating elements, etc.), will de-compose to form phosgene, hydrogen fluoride and hydrogen chloride. These compounds have extremely harmful physiological effect on human beings. Naked flame and smoking must be prohibited in the presence of refrigerant vapour and refrigerant must be purged from pipes or vessels before carrying out cutting or welding operations. Chemical hazards • Approved methods of leak detection only should be used. If a halide test lamp is used, remember that the heating or combustion effect will produce toxic byproducts which could be dangerous if inhaled. Chemical hazards • Liquid refrigerant in contact with the eyes or skin will cause freezing and injuries similar to a burn. Care must be taken when opening pipes or vessels which may contain liquid. • Thus it is essential when loosening a connection on any part in which refrigerant is confined, goggles to be worn to protect the eyes. Ammonia (R717) • Ammonia is normally considered to be the most dangerous of the primary refrigerants and has inherent material and physiological hazards. A limited range of ammonia/air mixtures (16 27%) ammonia by volume can be ignited by flame and an explosion may result. Ammonia must not be allowed to come into contact with iodine, bromine, chlorine, hypochlorite or mercury. There is an explosion hazard in each case. Ammonia Vapour • Low concentrations may cause only irritation and discomfort. • High concentrations can destroy body tissue. The action is more pronounced on moist tissues: eyes, nose, breathing passages, and moist areas of the skin may be burned by high concentrations. Ammonia Liquid • In this form ammonia can cause severe burning of the skin and eyes. As the eyes are particularly delicate organs, even small amounts of ammonia can be harmful. The full effects of ammonia on the eyes may not be apparent for 8-10 days but, ultimately, blindness may result. Ammonia • Maintenance procedures must not be carried out unless adequate ventilation has been provided to avoid risk of explosion and physiological harm. Naked flames must not be permitted in the area. The pungency of ammonia will usually warn personnel against remaining in locations where dangerously high concentrations of vapour exist. Personnel must not be permitted to work without wearing a gas mask, even for short periods, in a concentration which causes any discomfort to the eyes or affects breathing. Operation of refrigeration plant • The refrigerating capacity of any refrigerating plant is at its maximum when the greatest possible quantity of refrigerant is evaporated in the evaporator to obtain the required room temperature or to obtain the required brine temperature. • The evaporating temperature and hence pressure must be kept as high as possible consistent with the temperatures. For a given design this means ensuring that all heat transfer surfaces are kept clean, so that the temperature difference across them is at its minimum. It is also essential to ensure that evaporator surfaces are supplied with liquid refrigerant at the correct temperature and in the correct condition. • The condensing temperature must be kept as low as possible so as to keep the compressor delivery pressure to a minimum. This again means keeping all heat transfer surfaces clean and ensuring the correct flow of cooling water or air through the condenser. It is also important to keep air out of the systems as, being non-condensable, it will collect in the condenser vapour space so raising the effective compressor delivery pressure artificially. • As per Dalton’s law of partial pressures, the delivery pressure is the sum of that due to the air and that at which the refrigerant is condensing. Air in the condenser will be indicated by an excessively high condenser gauge reading in relation to the temperature of the cooling water or air. Air can be purged out of the top of the condenser, though some refrigerant will also be lost with it. • The compressor must be maintained in the best possible condition. This means that suction and delivery valves must not leak and piston clearance must be kept to a minimum. The correct operation of unloading devices is also important, as the partial operation of these can affect compressor performance. Valve leakage or excessive clearance both result in reexpansion of gas from delivery to suction pressure, and hence in reduced pumping capacity. • The temperatures at inlet and outlet of each side of the heat exchanger, if fitted, must be correct to ensure correct superheat and sub-cooling temperatures. The refrigerating capacity of a plant is directly proportional to the weight of refrigerant evaporated in the evaporator. This in turn is directly related to the mass of vapour pumped by the compressor. The latter is the sum of the flash gas formed at the expansion valve and the vapour evaporated in the evaporator; it is therefore important to keep the flash gas to a minimum. • The mass of refrigerant vapour pumped by a given compressor depends directly on the temperature and hence pressure difference between the evaporator and the condenser, and this must be kept to a minimum. Furthermore, the density of the refrigerant vapour varies directly with the evaporating pressure and hence temperature, and it is therefore important to keep the evaporating temperature as high as possible in relation to the required load conditions. • The swept column of a given compressor at a constant speed is constant, and the volume pumped varies relatively slightly for normal plants, although for a given condensing temperature it drops rapidly at low evaporating temperatures, i.e. the volumetric efficiency of a compressor is directly related to the pressure ratio between the compressor suction and delivery, which in turn corresponds to the temperature difference between the evaporating and condensing temperatures. • A rise in condensing temperature has much less effect on the refrigerating capacity than a corresponding drop in evaporating temperature; both affect the volumetric efficiency, but lowering the evaporating temperature also reduces the density of the gas entering the compressor suction. • It is wrong, therefore, to assume that a lower evaporating temperature will improve the refrigerating capacity. While heat transfer may be improved in the evaporator, this is much more than counterbalanced by the reduced weight of gas pumped by the compressor, due to its reduced density as well as to the reduced compressor volumetric efficiency. MAINTENANCE • 1. 2. 3. 4. The plant will need a minimum of care and maintenance if it is kept: free of moisture; free of impurities; free of freon leaks; free of frost. • Experience has shown that most problems with marine refrigerating plant involve refrigerant shortage caused by leakage. In rooms where frozen or chilled cargo below 5°C is carried, it is necessary to keep evaporators free from frost. Cleaning of filters is also important. • In cases where equipment is opened up, neither air nor moisture must enter the refrigeration system, as either will cause trouble, e.g. in the form of increased condensing pressure. To avoid moisture, filter driers are installed in HCFC/HFC plants. • The drying agent should be changed every time any part of the system is opened. This also applies when charging with oil or refrigerant. Oil should not be filled from vessels that have not been tightly closed. Drop in oil level • A leakage-free refrigerating plant does not consume any oil. The oil which has disappeared from the crankcase or oil separator is always somewhere in the system. Oil level drops quickly at the start • This may be due to refrigerant being dissolved in the oil. At evaporation the oil is drawn with the refrigerant into the system. Fill the system with a small quantity of oil, as the ejected oil will gradually come back. Oil level drops slowly 1. the plant is operated at lower evaporating temperature than usual or the refrigerant charge is too small; 2. refrigerant leakage in the system, by which the level in the evaporator has become too low; 3. condensing temperature is too low, the minimum condensing temperature should be maintained; 4. the cooling demand is too low, so the gas velocity becomes too low in the evaporator and the oil remains in the system. • In those plants with piston compressors which have oil separators, the shut-off valve in the oil return line should always be kept closed for about 1/2 hour after compressor start in order to avoid the carriage of condensate from the oil separator to the crankcase. Brine specific gravity • In those cargo refrigerating plants where brine serves as the heat transfer medium, it is of great importance that the correct brine specific gravity for the required cargo temperature is maintained. If this specific gravity is not checked, functional problems may occur. Calcium Chloride Brine CALCIUM CHLORIDE / BRINE Specific Gravity Hydrometer Reading Freezing Point of Solution (Twaddell) °C °F 1·20 40 -21 -6 1·21 42 -23 -9 5 1·22 44 -25 -13 1·23 46 -27 -17 1·24 48 -30 -21.5 1·25 50 -32 -26 1·26 52 -35 -31 1·27 54 -38 -37 1·28 50 -42 -44 1·29 58 -51 -60 Daily maintenance • 1. 2. 3. 4. 5. The daily maintenance for a R22 installation should be completed as follows: Check that condensing pressure and evaporating pressure are correct. Inspect the compressor unit and check that there are no abnormal noises or vibrations. Check the oil level. Check the tightness of the shaft seal. Oil leakage can be tolerated whereas gas leakage can not. If an oil separator is installed, check that oil is returned to the crankcase, and that the oil return line is warmer than the crankcase. Leak detection • Refrigerating plants must be gas-tight to prevent refrigerant leakage and air entering the low-pressure side when under a vacuum. Systems which have been opened to the atmosphere during repairs, must be pressure tested for mechanical strength and leaks before charging with refrigerant. Pressure tests • Pressure tests should be done with nitrogen. Water or other fluids must not be used as a test medium. The plant’s compressors must not be used to pressurise the plant. The pressure is gradually increased by pressurising with nitrogen gas the pressure is equal to 1.5 times the maximum working pressure of the system. This pressure should be maintained for about 10 minutes. • Each joint must be examined thoroughly for signs of gas bubbles which indicate a leak. After sealing any leaks, pressurise the system again with the test medium and some refrigerant as a trace gas, and repeat the leak test using an electronic leak detector, etc. It is imperative that all leaks are found and sealed before the system is charged with refrigerant, as even the tiniest of leaks can result in the loss of the whole refrigerant charge. Leak detection equipment • Electronic leak detectors • Electronic leak detectors are the most sensitive and accurate method of leak detection. The detector contains an internal pump that draws air into a probe, or tube. If refrigerant gas is present in the sample, the electrodes in the sensing element generate a current, and an output signal is obtained. Halide lamps • Halide lamp is used to locate leaks of HCFC refrigerants. This method is based on the colour of a flame that surrounds a glowing copper element. The flame turns blue-green if the air being consumed contains the refrigerant. • These lamps should only be used in well ventilated spaces. Routine inspections • The high pressure side of the system may easily be tested for leaks with the plant running, but it may be necessary to stop the compressor, and allow the pressure in the low pressure side to rise sufficiently for leak test in the low pressure side. The amount of refrigerant in the system should also be strictly monitored as a drop in level may indicate the presence of a leak. In the event of a major leak, the initial leak test should be made using the soap bubble method, specially in areas where there are signs of an oil leak. Drying by evacuation • 1. 2. 3. 4. This method of removing moisture is based on the fact that the boiling point of water decreases with falling pressure. In the course of evacuation, any water or ice in the plant will evaporate, and is carried away by the vacuum pump. Connect a vacuum pump to the system using a short length of large bore pipe, and open all valves in the system (expansion valves, solenoid valves, etc., may have to be jacked open). Evacuate the system to a pressure of 6mm Hg or less. If possible, carry out the evacuation at ambient temperatures above 10°C. Close the line between the system and the vacuum pump. The pressure in the system may not rise more than 2mm Hg within five minutes. A rise of more than 2mm Hg indicates the presence of water, and/or a leak. Where water is present, the system will be colder than its surroundings. Check for water and/or leaks, carry out any repairs, and repeat the evacuation procedure until the pressure rise is less than 2mm Hg. When this is achieved the system is free of moisture and non-condensable gases and ready for refrigerant charging. Charging procedures • Refrigerating plants should not be overcharged with refrigerant, as this may overload or damage the compressor. • To ensure that the correct amount is added, the refrigerant should be weighed during charging. • The liquid refrigerant is decanted from the refrigerant bottle into the system via a charging valve just after, the liquid receiver. Charging Procedure 1. 2. 3. 4. 5. 6. 7. 8. Weigh refrigerant bottle Connect refrigerant bottle to charging valve with flexible charging line. Crack bottle liquid valve before tightening line to blow out air. Close main liquid line valve and pump down system. Open charging valve and carefully open liquid valve on refrigerant bottle. Liquid refrigerant will flow into the system. Start compressor. Continue charging until required amount of refrigerant has been charged. 9. Check weighing scale reading and observe liquid level in receiver. 10.Close charging valve and open main liquid valve and observe liquid flow through sight glass. Bubbles indicate the need for further charging. 11.If charge is complete, close bottle valve, and disconnect charging line. 12.Store empty refrigerant bottle for reuse. Condensers • Once a year, the tubes should be cleaned with a tube brush in order to remove deposits which would cause a high condensing pressure. The gaskets must be glued to the condenser end plate with good contact. If the partition wall gasket is not properly installed, there is a risk that it will ‘blow’, leading to an excessively high water velocity and to damage to the tube plate. • If a tube has become defective, it does not have to be replaced immediately. Up to 10% in each pass (flow direction) can be plugged. • Corrosion plugs should be renewed if required. Defrosting • Another task which maybe regarded as maintenance is to keep frost deposits on provision and cargo refrigerating plants under control. Frequent defrosting assures that the plant will cause few problems. Investigating trouble • 1. 2. 3. 4. 5. 6. 7. 8. In investigating trouble, there are certain things to which attention should always be paid in the first instance: the temperature of the refrigerated space; evaporating pressure; condensing pressure; suction pipe temperature; discharge temperature; liquid line temperature; compressor running time; noise from compressor, motor, expansion valve, etc. TROUBLE SHOOTING • When problems are incurred in a refrigerating plant, these can be attributed in most cases to a shortage of refrigerant. Bearing this in mind, always commence trouble-shooting by checking the refrigerant charge. • For example, in the case of HCFC plants, too little refrigerant prevents the oil, which always circulates in the system, from being returned as the gas velocity is low, and this leads to various functional troubles. CONTAMINANTS • If the moisture present in a refrigerating system exceeds the amount that the refrigerant can hold in solution it will exist as free water. At temperatures of 0°C or lower, the free water will freeze into ice in the expansion valve or evaporator, restricting the flow of refrigerant. To avoid freeze-ups, the moisture content in low temperature HCFC refrigerant systems must be maintained at a very low level. Oil • In refrigerating systems some oil is always carried over from the compressor into the condenser by the refrigerant gas, from where it is carried by the liquid into the evaporator. The presence of oil in the circulating refrigerant reduces the heat transfer capacity of the various heat exchangers, the problem being greatest in the evaporator, since oil becomes more viscous and tends to congeal at low temperature. To prevent oil related problems, the operation of the oil separator should be checked regularly to ensure oil is being returned to the compressor lubrication system. The amount of oil added to the lubrication system should also be strictly monitored; an excessive amount indicates that oil is being trapped in the evaporator or suction line. Air and non condensable gases • The presence of air and other non-condensable gases is detrimental to the efficient operation of a refrigerating plant, as these gases collect in the condenser, and so increase the condensing pressure. Abnormally high condensing pressures cause overheating of the compressor, excessive discharge temperatures, losses in compressor capacity and efficiency, excessive power consumption and possible overloading of the drive motor. Testing for Non-condensable Gases • Air and non-condensable gases, if present in the system, are pumped through the system and discharged by the compressor into the condenser. • These gases are trapped in the condenser and cause excessive condensing pressures. In order to check the condenser for the presence of air or non-condensable gases, it is essential that gauges and thermometers be accurate and that the system has sufficient charge so that the liquid refrigerant present in the receiver will seal the liquid line connection. Check for non-condensable gases Close liquid line valve and allow system to pump down. • 1. Shut off compressor and close suction line valves. 2. The thermometer in the sea water outlet of the condenser will indicate the actual condensing temperature, when there is no further drop in temperature. 3. Record the condensing pressure. 4. On a refrigerant pressure gauge, look up the saturation temperature that corresponds to the condensing pressure. 5. If the condensing temperature is less than the corresponding saturation temperature of the condensing pressure, it is necessary to purge. Purging Non-condensable Gases 1. Pump down the refrigerant by shutting the liquid valve at the outlet of liquid receiver. 2. Continue cooling the condenser for 10 to 15 minutes. 3. Open purge valve on top of condenser, and slowly release gases. 4. Since it is difficult to tell if excessive refrigerant is being purged with the non-condensables, purge slowly. Compressor Short-Cycles Possible Cause Low sea water temperature. Low refrigerant charge. Reduced evaporator capacity. Action Throttle condenser sea water outlet valve. This will raise the compressor discharge pressure, thereby raise the compressor suction pressure. Check refrigerant charge. Add as required. Check for frosted coils. Compressor Runs Continuously Possible Cause Low refrigerant charge. Compressor valves leaking. Action Check for proper charge. If low, repair any leaks and recharge. Pump down, remove cylinder heads, and check. Worn piston rings Pump down, disassemble, and/or cylinder liner and then check. Legionella bacteria • A type of pneumonia which may be fatal to older people, has been blamed on the presence of a bacteria associated with the air conditioning plant of large buildings. Because the outbreak which heralded the disease, occurred at a convention for American ex-servicemen (The American Legion), the identified cause of the problem, was labelled legionella bacteria and the sickness is referred to as legionnaires disease. • There is a risk that the bacteria could flourish in the air conditioning systems of ships. The organisms breed in stagnant water or in wet deposits of slime or sludge. Possible locations for bacteria colonies, are mentioned as being at the air inlet area and below the cooler (stagnant water), in the filter, in humidifiers of the water spray type and in damaged insulation. • Provision of adequate drainage is recommended to remove stagnant water. • Regular inspections and cleaning as necessary of filters and other parts, using a 50ppm super-chlorinated solution as the sterilizing agent is required. The solution is to be used also on the cooler drain area at not more than three month intervals. Regular sterilization is necessary for water spray type humidifiers (steam humidifiers being preferred).