sampling
advertisement

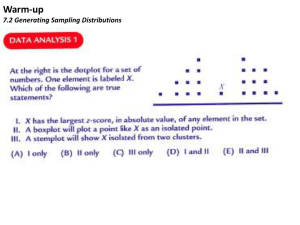
Mercury Collection and Analysis in Ambient and Effluent Waters using EPA Method 1631 Overview of Sampling and Analysis William Telliard U.S. EPA Office of Science and Technology Office of Water Mercury workshop- sampling.ppt Contamination Control Philosophy Ensure any object or substance that contacts the samples is nonmetallic and free from any material that may contain metals of concern Mercury must be eliminated or reduced to a level that will not compromise the measurement Sample bottles Sampling equipment Reagents Laboratory environment Laboratory glassware and equipment Office of Water Mercury workshop- sampling.ppt Equipment Cleaning Preclean All equipment and containers must be non-metallic Clean all sampling equipment and containers using detergent, mineral acids, and reagent water as described in Method 1669 Verify cleanliness Generate acceptable equipment blanks Demonstrates equipment and containers are free from contamination Storage Fill cleaned sample containers with 0.1% (v/v) ultrapure HCl and individually double-bag Bag or wrap all sampling equipment for storage and shipment Office of Water Mercury workshop- sampling.ppt Sample Collection Using Method 1669 “Clean Hands/Dirty Hands” A two-person sampling team is required One person is designated “Clean Hands” and performs all operations involving direct contact with the sample and bottles The other person is designated “Dirty Hands” and is responsible for operation of machinery and activities not involving direct contact with the sample Sampling personnel must wear clean, non-talc gloves, and may need to wear other clothing, such as disposable coveralls or nylon windsuits, caps, and shoulder-length glove liners, to prevent sample contamination Office of Water Mercury workshop- sampling.ppt Sample Collection Using Method 1669 The presence of potential sources of contamination is of extreme importance in site selection To minimize contamination— Collect samples facing upstream and upwind Avoid collecting samples during precipitation events – – During a 9 POTW Great Lakes study, the Hg field blank collected on the only rainy day had a Hg concentration 3 times higher than any other field blank in the study Collect samples as far as possible from metal supports, bridges, wires, and heavily traveled roads Office of Water Mercury workshop- sampling.ppt Sample Collection Using Method 1669 (cont.) Method includes procedures for collection of samples for determination of total recoverable metals and dissolved metals Sample Filtration For dissolved metals, samples are filtered through a 0.45-um capsule filter at the field site A continuous-flow sampling system with an in-line filter can simplify sample collection and filtration Sample Preservation Conducted in the field or in the laboratory Field preservation must be performed in a glove bag or a designated clean area Office of Water Mercury workshop- sampling.ppt Quality Assurance/Quality Control Equipment Blanks Field Blanks Bottle blanks Sampler Check Blanks Must be collected to assess the likelihood of contamination in the samples. Field blanks are collected before samples Field Duplicates Collected to assess precision of the sampling and analytical processes Office of Water Mercury workshop- sampling.ppt Blanks and Definitions Equipment Blank Bottle Blank - generated by filling a sample bottle with reagent water acidified to pH < 2, allowing the bottle to stand for 24 hours, and analyzing the water Sampler Check Blank - generated at the lab by processing reagent water through the sampling equipment using the same procedures that will be used in the field, and collecting and analyzing the water Field Blank - generated by filling a large carboy with reagent water in the laboratory, transporting the container to the field, processing the reagent water through the entire sampling equipment system, and analyzing the sample Office of Water Mercury workshop- sampling.ppt Summary Rigorous sample handling procedures are necessary to ensure samples are not contaminated Stringent QC is necessary to ensure the validity of the analytical results Not all of the procedures described in Method 1669 may be necessary for effluent sampling Level of protection required depends on sampling environment, monitoring levels of interest, and metals of interest The low levels of Hg (<36 ng/L) found in a 9 POTW Great Lakes Study suggests these clean techniques may be required for some metals Office of Water Mercury workshop- sampling.ppt Laboratory Analysis using Method 1631 For determination of mercury in filtered and unfiltered water by oxidation, purge and trap, desorption, and cold-vapor atomic fluorescence spectrometry (CVAFS) Method detection limit is 0.2 ng/L and the range is 0.5 - 100 ng/L The ease of contamination samples cannot be overemphasized Method includes suggestions for improvements in facilities and analytical techniques that minimize contamination and maximize the ability of the laboratory to make reliable trace metals determinations Performance-based Accompanied by Method 1669 Office of Water Mercury workshop- sampling.ppt Laboratory Analysis using Method 1631 (cont.) A 100-2000 mL sample is collected directly into a specially cleaned, pretested, fluoropolymer bottle using appropriate sample handling techniques For dissolved mercury, the sample is filtered through a 0.45 uM capsule filter The sample is preserved by adding 5mL/L of pretested HCl or 5 mL/L BrCl solution. BrCl solution is added to oxidize all Hg compounds to Hg(II) The sample is pre-reduced with NH2OH-HCl to destroy the free halogens Office of Water Mercury workshop- sampling.ppt Laboratory Analysis using Method 1631 (cont.) The sample is then reduced with SnCl to convert Hg(II) to volatile Hg(0). The Hg(0) is separated from solution by purging with nitrogen onto a gold-coated sand trap The trapped Hg is thermally desorbed from the gold trap into an inert gas stream that carries the released Hg(0) into the cell of a CVAFS for detection Quality is assured through calibration and testing of the oxidation, purging, and detection systems. Office of Water Mercury workshop- sampling.ppt Figure 1. Cold Vapor Atomic Fluorescence Spectrometer (CVAFS) System (Method 1631) Office of Water Mercury workshop- sampling.ppt Figure 2. Automated Mercury Fluorescence System (Method 245.7) Office of Water Mercury workshop- sampling.ppt Clean Spaces Guidance Draft guidance on the establishment of trace metal clean rooms in existing facilities was completed in April 1995, with minor revisions in January 1996. The guidance includes: A general design of a trace metals clean laboratory; Delineation of the hierarchy of cleanliness needed within the laboratory, including change rooms; Description of HEPA-filtered air and clean water supplies, which are essential in a trace metals laboratory; and Detailed descriptions of materials and modifications used to construct a trace metals clean room in an existing laboratory. Office of Water Mercury workshop- sampling.ppt Clean Spaces Guidance (cont.) Basic requirements metal free work surfaces and hoods positive pressure with HEPA-filtered air clean water Achieved with commercially available materials Installed into existing facilities Paint the walls with metal-free paint (epoxy- or latex-based) to which has been added a small amount of sulfur powder to react with mercury that could diffuse out of the underlying surfaces All metal fixtures and appliances should be replaced with non-metal counterparts Office of Water Mercury workshop- sampling.ppt Analysis Tips Clean all work surfaces in which samples will be processed Reagent water should be monitored for Hg Samples known to contain high levels of mercury (greater than 100 ng/L) should be diluted prior to bringing them into the clean room or area Sample processing and analysis should occur as far as possible from sources of airborne contamination Effluent from the CVAFS should pass through either a column of activated charcoal or a trap containing gold or sulfur to amalgamate or react mercury vapors Office of Water Mercury workshop- sampling.ppt