NH 2
advertisement

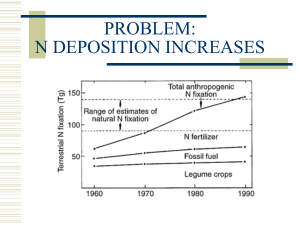
N2 Cosmic abundance of the elements 12 H log abondance 6 (atomes / 10 Si) 10 He 8 CO Ne Si S N 6 Fe Ca Ni 4 2 Li 0 B Sc Pt Be Tc -2 0 10 20 30 40 Pb Th U Pm 50 Mass number 60 70 80 90 100 14.0 N -3,0,3,5 Nitrogen species nitrate NO3– N+V stable oxide of N, highly soluble as an anion nitrite NO2– N+III intermediate between NO3– and NH4+ nitrous oxide N2O N+I from lightning and internal combustion engines nitrogen N2 N0 hydroxylamine NH2OH N–I intermediate species during oxidation of NH4+ ammonia NH3 N–III un-ionized ammonia gas ammonium NH4+ N–III ionized ammonia (dominates below pH 9.23) urea CO(NH2)2 N–III amino N R–NH2 elemental nitrogen gas N–III common fertilizer organic nitrogen as amine, measured as TKN Nitrogen redox NO2–/N2 1 15 NO3–/N2 O2/H2O 0.8 10 0.6 – NO3 /NO2 – Fe(OH)3/Fe2+ 5 0.2 NO3–/NH4 0 0 SO42–/HS– -0.2 CO2/CH4 -5 H2O/H2 -0.4 -10 -0.6 5 6 7 pH 8 9 Eh (V) pe 0.4 Essential nutrient Elemental composition of algae C106H263O110N16P(S) Protein :NN: Fixing nitrogen for photosynthesis C106H263O110N16P(S) Natural N2 fixation by Rhizobia and the nitrogenase enzyme (Fe and Fe-Mo proteins) N2 + 8H+ + 8e− + 16ATP → 2NH3 + H2 + 16ADP + 16 P adenosine triphosphate (ATP) adenosine diphosphate (ATP) And organic N scavenged from biodegradation R–NH2 + 2H + NH3 NH3 + O2 NO3– It’s all about getting NH3 and NO3–. Human intervention into the nitrogen cycle NH3 Urea from animal waste, urine CO(NH2)2 + H2O 2NH3 + CO2 Guano from Chile and the Pacific islands – NO3 Nitroglycerine 4 C3H5(ONO2)3 12 CO2(g) + 10 H2O(g) + 6 N2(g) + O2(g) Saltpetre KNO3 from India Haber-Bosch, 1908-10 ½N2(g) + 3/2H2(g) ⇌ 2NH3(g) ΔG = –16.5 kJ/mol CH4(g) + 2H2O(g) → CO2 (g) + 4H2(g) N2 and H2 are reacted over a ferric iron catalyst with Al2O3 & K2O at 250 atm and 450-500°C. Essential nitrogen reactions Degradation of organic N: –NH2 + H+ NH3 Organically-bound nitrogen is a component of all proteins and plant biomass. Aerobic and anaerobic degradation of such carbon compounds releases this reduced nitrogen in the form of ammonia. Where this occurs in unsaturated materials such as soils or manure, the ammonia can volatilize, or dissolve into water. Essential nitrogen reactions Degradation of organic N: –NH2 + H+ NH3 Organically-bound nitrogen is a component of all proteins and plant biomass. Aerobic and anaerobic degradation of such carbon compounds releases this reduced nitrogen in the form of ammonia. Where this occurs in unsaturated materials such as soils or manure, the ammonia can volatilize, or dissolve into water. Decomposition of urea: CO(NH2)2 + H2O 2NH3 + CO2 Urea is a common form of organic nitrogen that is produced naturally in animals and industrially. It is often applied as fertilizer in granulated form, and breaks down by a bacterially mediated reaction (urease enzyme) to release ammonia for plants. Ammonia transformations Ionization of ammonia: NH3 + H+ NH4+ KT = 10–9.23 • high solubility of ammonia in water at neutral pH • at high pH NH3 represents a large fraction of the total ammonia • NH3 = NH4+ at pH 9.23. Ammonia transformations Ionization of ammonia: NH3 + H+ NH4+ KT = 10–9.23 • high solubility of ammonia in water at neutral pH • at high pH NH3 represents a large fraction of the total ammonia • NH3 = NH4+ at pH 9.23. Volatilization of ammonia: NH3(aq) NH3(g) KH = 101.76 • high Henry’s Law constant for ammonia • un-ionized NH3 = 246 mg/L at 25˚C for a NH3 partial pressure of one atmosphere • volatilization from manure, soils and surface waters • loss from groundwater below the water table is minimal Ammonia transformations Ionization of ammonia: NH3 + H+ NH4+ KT = 10–9.23 • high solubility of ammonia in water at neutral pH • at high pH NH3 represents a large fraction of the total ammonia • NH3 = NH4+ at pH 9.23. Volatilization of ammonia: NH3(aq) NH3(g) KH = 101.76 • high Henry’s Law constant for ammonia • un-ionized NH3 = 246 mg/L at 25˚C for a NH3 partial pressure of one atmosphere • volatilization from manure, soils and surface waters • loss from groundwater below the water table is minimal Sorption of ammonium: Na–clay + NH4+ NH4–clay + Na+ • cation exchange of ammonium onto clay minerals in soils and aquifers • erosion of NH4-bearing soils is a major sources of contamination in surface waters • selectivity coefficient for ammonium varies with the clays and competing cations • transport of NH4+ in groundwater is retarded. Ammonia oxidation Aerobic nitrification of ammonium: NH4+ + 2O2 NO3– + H2O + 2H+ DG°r = –266.5 kJ/mol • NH4+ can be oxidized to NO3–by reaction with elemental oxygen (O2) • significant energy yield is favorable for bacteria • two step reaction of oxidation to nitrite by a Nitrosomonas, Nitrobacter and Nitrosospira, and oxidation of nitrite to nitrate by Nitrobacter Pseudomonas. • reaction is restricted to aerobic environments – manure piles, soils and surface waters. Ammonia oxidation Aerobic nitrification of ammonium: NH4+ + 2O2 NO3– + H2O + 2H+ DG°r = –266.5 kJ/mol • NH4+ can be oxidized to NO3–by reaction with elemental oxygen (O2) • significant energy yield is favorable for bacteria • two step reaction of oxidation to nitrite by a Nitrosomonas, Nitrobacter and Nitrosospira, and oxidation of nitrite to nitrate by Nitrobacter Pseudomonas. • reaction is restricted to aerobic environments – manure piles, soils and surface waters. Anaerobic nitrification of ammonium – anammox: 3 NO3– + 5 NH4+ ® 4 N2 + 9 H2O + 2H+ DG°r = –282.30 kJ/mol-NH4+ = –470.50 kJ mol-NO3– • Recently discovered (1995) less well-known reaction • thermodynamically very favorable for bacteria • anaerobic environments with both ammonium and nitrate species are present, such as in waste-water streams, anoxic marine waters and soils. • NH4+ as an electron donor, with NO3–, and NO2–, as an electron acceptors, producing N2. • only known biologically-mediated reaction for conversion of NH4+ to N2. Nitrate reduction back to N2 Denitrification: 5CH2O + 4NO3– + 4H+ 2N2 + 5CO2 + 7H2O DG°r = –252.47 kJ/mol • anaerobic reaction - O2 – free conditions required • low-pe electron donor such as carbon or sulphide • nitrate is an electron acceptor with nearly the same energy yield as O2 • Pseudomonas denitrificans reduces NO3– to N2 using fixed carbon (biomass) • Denitrification can also be mediated by chemotrophs such as Thiobacillus denitrificans, which uses sulfide (H2S or pyrite) as a substrate. • N2 from denitrification becomes overpressured in water as dissolved nitrogen gas • anaerobic waters with low nitrate concentrations (NO3– limited), denitrification to N2 gas may not be complete, resulting in the production of N2O gas. Nitrate Cycle UREA HYDROLISIS AND VOLATILIZATION OF AMMONIA NH3 (gas) CO (NH2) NH3 NH4+ NO315NH 3 + 14NH4+ (aq) 14NH3 (gas) + 15NH4+ (aq) Isotope fractionation factor = 1.034 DENITRIFICATION 4NO3 + 5CH2O + 4H 2N2 + 5CO2 + H2O - + 14NO3 + 5FeS2 +14H 7N2 + 10SO4 + 5Fe + H2O - + - +2 e15NNO3-N2 ~ 15 to 20 permil e18ONO3-H2O ~ 8 permil Isotope Data in Nitrate of Different Origins Ref: Wassenaar, L. 1995. Applied Geochem. 10:391-405 Groundwater flow system Nitrate distribution in mg/L as NO3- From Aravena, R., Evans, M.L., and Cherry, J.A. 1993. Ground Water, 31: 180-186 Septic system plume based on Na concentration Nitrate Concentration in mg/L as NO3- 15N data (‰) in Nitrate Ref:Aravena, R and Robertson, W. 1998. Ground Water, 36: 975-982 Nitrate distribution in mg/L as N Oxygen (o) and DOC () concentration profiles 15N (o) and nitrate () concentration profiles Isotope enrichment trend showing denitrification Chemical and Isotope Depth Profiles WHY RIPARIAN ZONES ARE IMPORTANT • Nitrate is a major groundwater pollutant in agricultural landscapes • Riparian zones act as a buffer zones to attenuate nitrate associated to contaminated groundwater discharging in rivers and lakes Conceptual Groundwater Flow Regimes in Riparian Zones Geological Cross Section of Study Area Ref: Cey E.E., Rudolph, D.L., Aravena, R., and Parkin, G et al., 1999. Journal of Contaminant Hydrology, 37: 45-67. Buffer strip Stream Manure Spreading Instrumentation transect perpendicular to the stream Nitrate vs 15N Data 15N vs 18O data Nitrate vs DOC