Mineralogy Poster
advertisement

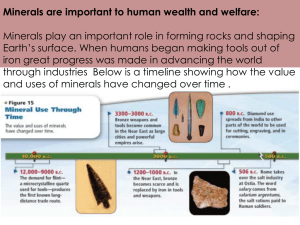
Fluorite, Calcium Fluoride, CaF2 Jamison Brizendine, Economic Importance: GEOS 350, Mineralogy Fluorite has many industrial and practicable uses. Fluorite is used as a component in iron and aluminum smelting, special fluxes in welding rods, toothpastes, ceramics. and optics. One of the most widely used organic fluoride compounds, the refrigerant Freon 12®, is no longer produced in the United States. The chlorine in the compound is thought to damage the protective ozone layer that shields the earth from ultraviolet radiation. Department of Geosciences Brizeja@earlham.edu Fluorite in a Nutshell: Color: Green, purple, blue, orange, red, yellow, and clear. Hardness of 4, scratches Calcite, but can be scratched by Apatite. Fluorite is the ideal mineral for it’s hardness. Upper Left: Minerva #1 Mine, Cave-In-Rock, Illinois, www.mineralauctions.com Luster: Vitreous Up: Okarusu Mine, Otjiwarongo, Namibia: www.mineral-auctions.com Upper Right: Boltburn Mine, Weardale, England: Ukminingventures.com Streak: White Right: Blackdene Mine, Weardale, England: Ukminingventures.com Cleavage: Perfect octahedral cleavages in four directions, and typically forms cubes, octahedra and rarely dodecahedrons. Specific Gravity: 3.1 Class: Halide, Dana Number: 9.2.1.1 Common Impurities: Y, Ce, Si, Al, Fe, Mg, Eu, Sm Point Group: 4/m 3bar 2/m Optical Data: Isometric, n=1.433 Both the element Fluorine and the term “Fluorescence” comes from Fluorite. Fluorine was successfully isolated by separating the Calcium from the Fluorine. The deposits in Illinois, where the majority of commercial Fluorite is mined, were formed as hot water flowed up into the competent limestones layers, which are below a sandstone formation. Mineral deposits formed in this manner are called hydrothermal deposits. The hot water flowed up along weak areas in the rocks of this area. These weak areas are associated with the faults of the Wabash and Reelfoot rift zones. These two rift zones are also responsible for earthquakes such as the famous series of quakes at New Madrid, Missouri. The southern Illinois fluorite deposits were probably formed between 100 and 250 million years ago. The name Fluorite comes from the Latin word, “fluere”, which means, “to flow”. Two Fluorine cations replaces the Carbonate cation (CO3) in Calcite, which then creates Fluorite. It can also form during normal faulting and fluid fills the cracks. Fluorite also forms with Spinel and Calcite in hornfels, high temperature granties and other similar contact metamorphic rocks. Another environment that Fluorite is found with are epithermal veins with barite and galena, but the most common are the lode hydrothermal veins with zinc and lead. Fluorine is often found with Pyrite, Sphalerite, Calcite, Quartz, Dolomite, Chalcopyrite, Galena and Barite. The map below shows the area where Fluorite is most commonly found in the United States. Fluorite also has properties of Thermoluminescence. This means that when Fluorite is heated it can glow. This process, however, is a one shot procedure, so one sample can show it and never show it again. Fluorite mining in the United States started around the 1800’s, near Illinois and Kentucky. Most of the large scale mining operations where done underground at depths of 1500 feet (dnr.state.il.us). Open pit mines replaced underground mines and Illinois and Kentucky totaled about ninety percent of the mining operation in the United States. Large scale mining in Illinois ended in 1995 when the mining of Fluorite no longer became profitable. Rogerly Mine, Weardale, England: www.mineral-auctions.com Extraction and Environmental Impacts: “Although some industrial processes can make use of fluorite directly, releasing elemental fluorine from the mineral fluorite requires a fair amount of processing. Hydrofluoric acid (HF) an important raw material for the chemical industry and the source of elemental fluorine gas, is produced by the following reaction: Fluorite (CaF2) + sulfuric acid (H2SO4) = 2 HF + CaSO4 (anhydrite) The reaction is typically carried out in horizontal revolving kilns that are heated externally. Finely ground fluorite and a slight excess of strong sulfuric acid are mixed in a hopper and introduced at one end of the kiln. A large collector pipe at the other end captures the HF and other gases released by the reaction, and the solid anhydrite is extracted by a screw drive. The hot gases the come out of the reactor consist of about 95% HF, 4% air and 1% other impurities that include H2SO4, SiF4, H2O, CO2 and SO2. Pure HF boils at 19 degrees Celsius and this relatively high boiling point is used to purify the gaseous mixture by distillation. Theoretically, 1.95 tons of pure CaF2 will yield one ton of HF, but impurities in the fluorite ore and the sulfuric acid generally require that greater tonnages of both be used in the reactors. To obtain elemental fluorine, anhydrous hydrogen fluoride undergoes electrolysis to release hydrogen and fluorine gas at the cathode and anode, respectively”. (http://www.isgs.uiuc.edu/faq/ggfaqs/GGQ52.html). About ninety percent of the Fluorite that the United States uses is imported from Mexico, China and Brazil. The Fluorite that was extracted from the United States primarily came from Illinois, which became the state’s mineral because of it’s economic values. Fluorite is also used to extract the Fluorine to produce Hydrofluoric acid (HF). HF is used in refining mineral such as aluminum and uranium and is a component of rocket fuel. Mines that have been exhausted have been left as scars upon the land. Exhausted mines leave large tailings piles of limestones and fluorite that have been left by companies that have abandoned the sites. The pits themselves are filled with water and become sinkholes. There are no hazards in mining fluorite, nor have there been any large consequences on the land, when mines are abandoned. While Fluorite is non-toxic in rock form, Hydrofluoric Acid (HF) is the strongest acid. Use caution when handling it. There is ongoing research on the effects of Fluorite mining and water quality. Regions where Fluorite is found: Purple: Rosiclare, Ill. , Cave-In-Rock, Ill., Putnam County, Hardin County, Ill., Putnam County, Ky., Musquiz, Coahuila, Mexico. Clear: Clay Center, OH., Toledo, OH. Mipimi, Durango, Mexico, Wilberforce, Ontario, Silverton District, Colorado. Green: Madoc, Marmara, Huntingdon, Hastings County, Ottawa, Canada. Namibia. Rogerly, Mine, England. Pink: France, Switzerland Blue: Yuogangxian, Hunan Province, China, Mexico. . Hill-Ledford Mines, Illinois. www.northstarminerals.com http://www.isgs.uiuc.edu/servs/pubs/geobits-pub/geobit4/assets/geobit4fig.gif Denton Mine, Illinois: www.northstarminerals.com References: http://mineral.galleries.com/minerals/halides/fluorite/fluorite.htm Jamison Brizendine and Lauren McCollough sift through a Tailings Pile in Mahoning Mine, Illinios. Photo Coutesy of Meg Streepey. http://www.isgs.uiuc.edu/servs/pubs/geobits-pub/geobit4/geobit4.htm. http://www.uwgb.edu/dutchs/PETROLGY/Fluorite%20Structure.HTM http://www.minerals-n-more.com/Fluorite_Info.html http://en.wikipedia.org/wiki/Fluorspar http://www.mii.org/Minerals/photofluor.html Left: Fluorite “Window”, Mahoning Mine, Illinois: www.mineral-auctions.com http://dnr.state.il.us/mines/education/indus2.htm Right: Musquiz, Coahuila, Mexico : www.mineralauctions.com http://geoinfo.nmt.edu/publications/memoirs/34/home.html Far Right: Okarusu Mine, Otjiwarongo, Namibia: www.mineral-auctions.com http://www.isgs.uiuc.edu/faq/gg-faqs/GGQ52.html). http://www.mineralminers.com/html/fluminfo.htm http://www.museums.udel.edu/mineral/mineral_site/displaycollection/Halides/100125_D3631.html Chesterman, Charles. National Audubon Society of Minerals, 1979, Chanticleer Press. Johnson, Ole. Minerals of the World, 2004, Narayana Press, Denmark.