biogeochemical cycle
advertisement

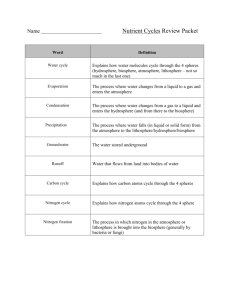
CYCLES OF MATTER The Water Cycle Nutrient Cycles The Carbon Cycle The Nitrogen Cycle The Phosphorus The WaterCycle Life depends on the recycling of chemicals. Chemicals pass back and forth between organic matter and the abiotic components. A biogeochemical cycle is the cycling of elements of ecosystems, chemical compounds, and other forms of matter which are passed from one organism to another and from one part of the biosphere to another. It connects biological, geological and chemical aspects of the biosphere. Water Chemical Properties + + H H Two atoms of hydrogen One atom of oxygen o - What Does Water Move Between? Water moves between the land, oceans, and atmosphere. Water vapor is water molecules that enter the atmosphere as a gas when they evaporate from the ocean or other bodies of water Evaporation The process by which water changes from liquid form to an atmospheric gas Condensation Heat from the sun changes water into water vapor Water vapor produces droplets of water that form clouds Transpiration The process by which water enters the atmosphere by evaporating from leaves Precipitation Results when the water droplets are large enough and the water returns to Earth’s surface The form may consist of rain, snow, sleet, or hail Rain or snow falls from clouds Forms of Precipitation Condensation The movement through plants The Clouds form Transpiration Precipitation The rain falls Evaporation The vapor rises Water Cycle Song (to the tune of “She’ll Be Coming Around the Mountain”) Water travels in a cycle, yes it does. Use pointer finger to draw large circle in air. Water travels in a cycle, yes it does. Repeat above motion. It goes up as evaporation, Raise arms at side with palms up. Forms clouds as condensation, Brings hands together above head, forming large cloud shape with arms. Then falls down as precipitation, yes it does. Slowly lower arms at side with palms down, fingers moving. The Carbon Cycle Carbon Cycle Importance of Carbon Found in all living organisms Found in the oceans, air, and certain types of rocks Less than 1 % circulates within the biosphere Four Process of the Carbon Cycle Photosynthesis and Cellular Respiration; decomposition of plants and animals Release of CO2 into the atmosphere by volcanoes Burial of carbon rich remains of organisms, their conversion into coal and petroleum (fossil fuels) through the pressure of overlying earth Human Activity: mining, burning of fossil fuels, cutting and burning down of forests Steps of the Carbon Cycle Carbon is present as carbon dioxide in the atmosphere Released in the air by volcanic activity, burning of fossil fuels, and decomposition of organic matter and cellular respiration Plants take in CO2 and use it to produce high energy sugars (carbohydrates) during photosynthesis Carbohydrates are passed along the food chain to other organisms Steps of the Carbon Cycle Carbon is found in the oceans, along with calcium and oxygen, in calcium carbonate (CaCO3), which is produced by many marine organisms Calcium carbonate can also be formed chemically in certain marine environments; it accumulates in marine sediments and in the bones and shells of organisms They eventually breakdown and the carbon returns to the atmosphere References Missouri Science Teaching and Education Partnerships Prentice Hall Biology by Miller and Levine Phosphorus Cycle Phosphorus Cycle Importance of Phosphorus Components of nucleic acids (DNA and RNA); life sustaining molecules Components of biological membranes Absorbed by plants from the soil or water and bind the phosphate into organic compounds Phosphorus Facts Not common in the biosphere Remains mostly on land in rock and soil minerals, and in ocean sediments Exists in the form of inorganic phosphates Released by the breakdown of rocks and sediments Some is washed into rivers and streams, where it dissolves Eventually ends up in oceans where it is used by marine organisms Some phosphates stay on land and cycle between organisms and the soil Organic phosphates move through the food web, from producers to consumers, and to the rest of the ecosystem The Nitrogen Cycle Importance of Nitrogen All organisms need nitrogen to make amino acids, the building blocks of proteins Several different forms of nitrogen occur naturally in the atmosphere Nitrogen gas (N2) makes up about 78% of the Earth’s atmosphere Nitrogen-containing substances are found in wastes produced by many organisms as well as dead or decaying ones; examples are ammonia (NH3), nitrate ions (NO3), and nitrite ions (NO2) Importance of Nitrogen Nitrogen also exists in different forms in the oceans and other large bodies of water Human activity adds nitrogen to the biosphere in the form of nitrate (a major component of plant fertilizers) Steps of the Nitrogen Cycle Only certain types of bacteria can use the form of nitrogen gas Bacteria that live in the soil, on the roots of plants called legumes, can convert nitrogen gas into ammonia (NH3): process is called nitrogen fixation Other bacteria convert ammonia into nitrates and nitrites Steps of the Nitrogen Cycle Once nitrates and nitrites are available, producers use them to make their own proteins Upon death, decomposers return the nitrogen back to the soil as ammonia The ammonia is taken back up by the producers Other bacteria convert the nitrates back into nitrogen gas: process is called denitrification Denitrification releases nitrogen back into the atmosphere