pptx
advertisement

GE 11a, 2014, Lecture 2
Minerals and rocks; the composition and materials of the earth
Symmetry is key to understanding mineral structure, but needs to be
understood as something different from ‘shape’.
Some symmetry elements are permitted (and common)…
Others are (usually) forbidden…
They key distinction can be understood as coming down to success or failure to
‘tesselate’ without leaving gaps
An early reasonable-seeming (but wrong) idea
Macroscopic cubes (and so forth)
are made of microscopic cubes
But we know that the chemical ‘entities’ that make up crystals are actually
molecular structures that are not symmetric shapes like cubes or hexagons. How do
they make such regular shapes?
e.g., crystals and molecules of insulin
crystal
monomer
The answer to this mystery is that low-symmetry objects can ‘fit’ together
into high-symmetry arrangements
Monomer
Hexamer
Crystal
X-rays have wavelengths similar to inter-atomic spacing in crystals, so passing them
through crystals leads to diffraction with a pattern that reveals crystallographic symmetry
Manganese Silicide
The magic ratios for ‘packing’ of cations and anions
Common oxide cations
Na, K
Fe, Ca, Mg
Si, Al
B, C, N
C
“Closest packing” arrangements—
a good starting concept for most
oxide and sulfide minerals
Evidence suggesting I’m not lying to you
These cation/anion units can share anion corners, edges or faces to make
larger ‘superstructures’
The framework of silicate minerals are regular polymers of SiO4-4 tetrahedra
Combinations of silicate ‘polymer’ structures and metal-oxide octahedra can create
diverse structures. E.g., sheet-like micas:
The difference between silicate minerals and glasses
Quasi-crystals — the wrong side of the symmetry tracks
Diffraction pattern of Icosohedrite
Quasi-crystals — the wrong side of the symmetry tracks
‘Ordered but not periodic’
— local centers with 5, 7, 11 or other
forbidden rotational symmetries
– But no translational symmetry
Solve the ‘space filling’ problem in a
fashion analogous to a penrose tiling:
Earth contains a great diversity of mineral and rock types — at least 10x that known
from other planets and early solar system bodies
Silicates
Clays
Sulfides
Minerals
Halides
Oxides
and Hydroxides
Carbonates, Phosphates
Sulfates, Nitrates, Borates
Clastic sediments
(sands, silts, clays)
Chemical sediments
(salts, some clays)
Steno
Rocks
Igneous
(silicate melts)
Metamorphic
Rocks
(‘cooked’ versions of other kinds)
Leibniz
How did we end up at this mix of elements as the ingredients for the crust?
A start to understanding the composition of the earth is to look at the sun,
which contains most of the atoms in the solar system
‘Fraunhofer lines
The solar wind
The Genesis mission
Genesis: an experiment to directly collect the solar wind
The Genesis sample collection module after ‘landing’
Picking through the pieces
A consensus view of the chemistry of the sun
Features that demand an explanation:
• H and He are by far most abundant elements
• Li, Be and B are anomalously low in abundance
• Overall ~ exponential drop in abundance with increasing Z
• Even Z > odd Z
• Fe and neighbors are anomalously abundant
“Hydrogen as food’ hypothesis: Burbidge et al., 1957
(built on ideas of Gamow re. nucleosynthesis in big bang)
I. H burning
positron
H + H = D + + + +
(rxn. discovered by H. Bethe, 1939)
D +H = 3He + …
3He
3He
neutrino
photons
+ 3He = 4He + 2H + …
+ 4He = 7Be + …
(and similar reactions to make Li and B)
Products quickly decay:
Timescale ~ 10-16 s
{
7Be
+ e- = 7Li
7Li + P = 8Be
8Be = 2.4He
Stuck; no way to elements
heavier than B
“We do not argue with the critic who urges that stars are not hot enough for this process; we tell him to go and find a hotter place.”
A. Eddington
Willie Fowler, Salpeter and Hoyle
Show the solution is the following reaction in red giant stars:
4He
+ 4He + 4He = 12C
Opens possibility of many similar reactions:
12C
+ 4He = 16O
16O + 4He = 20Ne
20Ne + 4He = 24Mg
Collectively referred to as ‘He burning’
“Would you not say to yourself, 'Some super- calculating intellect must have designed the properties of the carbon atom,
otherwise the chance of my finding such an atom through the blind forces of nature would be utterly minuscule.' Of course
you would . . .. A common sense interpretation of the facts suggests that a superintellect has monkeyed with physics, as well
as with chemistry and biology, and that there are no blind forces worth speaking about in nature. The numbers one calculates
from the facts seem to me so overwhelming as to put this conclusion almost beyond question.”
F. Hoyle
Advanced burning:
origin of the 2nd quartile of the mass range
12C
+ 12C = 23Na + H
16O
+ 16O = 28Si + 4He
CNO cycle
12C
+ P = 13N = 13C
13C + P = 14N
14N + P = 15O = 15N
15N + P = 12C + 4He
The E process (for ‘Equilibrium’): why the cores of planets are Fe-rich
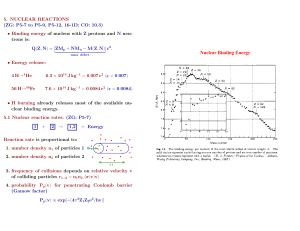
A quasi-equilibrium between proton+neutron addition + photo-degradation
Promotes nuclei with high binding energy per nucleon
Neutron capture as a mode of synthesizing heavy elements
Occurs in environments rich in high-energy neutrons, such as super-novae
Features that demand an explanation:
• H and He are by far most abundant elements
H primordial; He consequence of 1˚ generation H burning
• Li, Be and B are anomalously low in abundance
Consumed in He burning
• Overall ~ exponential drop in abundance with increasing Z
Drop in bonding energy per nucleon w/ increasing Z
• Even Z > odd Z
Memory of He burning
• Fe and neighbors are anomalously abundant
Maximum in bonding energy per nucleon at Fe
These factors are directly responsible for the fact that terrestrial planets
are made of silicates and oxides (‘rocks’) with magnetic Fe cores.
The next clue comes from primitive meteorites
FOV ~ 2 cm
FOV ~ 2 mm
Primitive meteorites look a lot like the sun
(minus the gas and all the hotness)
N
The separation of volatile from other elements can be easily understood as a result
of condensation from an initially hot gas
The earth like planets formed in the inner solar system, which didn’t cool enough
to accrete thick envelopes of ice and gas before the nebula dissipated
How did the earth get any volatiles at all?
A conceptual model for the secondary origins of
volatiles in asteroidal bodies
But primitive meteorites are diverse; how are we to know which
is most like the earth?
letters indicate compositional fields of various
types of primitive meteorites
Earth is somewhere near here
Much of the diversity in meteorite composition reflects
variations in oxidation state of solar nebula (H2O/CO ratio)
How do we guess the composition of the bulk earth if both terrestrial rocks
and meteorites are so variable?
Broad groupings of elements in geochemical processes
The earth’s mantle is mostly chondritic, but depleted in moderately volatile
elements (K, Na)
1
Silicate earth
CI chondrites
Are they simply missing, or hiding somewhere in the earth?
We’ll revisit this question later
The earth’s mantle is also depleted in siderophile elements (Ni, Cu, Au)
Silicate earth
CI chondrites
0.1
Are they simply missing, or hiding somewhere in the earth?
We’ll revisit this question later too