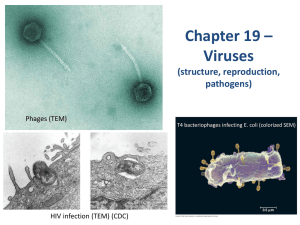
Coronaviruses
Chapter 40
Structure and Composition
•
•
•
•
•
•
•
•
Enveloped
Spike proteins resemble solar corona
or crown
120-160 nm
Positive-strand RNA (27-32 kb)
Cytoplasmic replication
Budding into ER and Golgi
Notoriously difficult to propagate in culture
High frequency of recombination
Cause colds and severe acute respiratory
syndrome (SARS)
•
•
•
Classification
Family Coronaviridae
Genus Coronavirus
Genus Torovirus
Replication
Details are largely unknown because viruses are difficult to grow
in cell culture
Mouse hepatitis virus is model for coronavirus replication
•
•
•
•
•
•
•
•
•
•
•
Viral spike proteins mediate attachment
•
Aminopeptidase N is a cell receptor target for many coronaviruses
Endocytosis is thought to mediate infection
After uncoating, the viral genome (mRNA) is translated to produce RNA
polymerase
Subgenomic RNAs are synthesized for each viral polypeptide
Genomic RNA is cosynthesized with nucleocapsid
•
Results in nucleocapsid binding immediately to genomic RNA
Progeny virus buds from ER and Golgi and are packaged into vesicles
Vesicles travel to and fuse with plasma membrane, releasing viral particles
from cell
•
Coronavirus Infections
Pathogenesis
Limited knowledge
Highly species-specific
Typically mild upper respiratory infections (“colds”) that remain
localized
Exception: SARS
Immunity is not durable
Many people become resusceptible after a few years
Laboratory Diagnosis
ELISA - may not discriminate past infections
HA
PCR
Virus isolation is difficult (often impossible) and requires great
expertise
•
•
•
•
•
•
•
•
•
•
•
•
Initial
outbreak in
SE Asia Respiratory
Severe
Acute
Hong Kong and Singapore first reported
Disease originated in China
Originally thought to be from wild game
markets
•
•
•
•
•
•
Syndrome
Palm civet cat (which isn’t a cat) - Paradoxurus
hermaphroditus
Raccoon dog (which isn’t a dog) - Nyctereutes
procyonoides
It is a bat virus
Chinese horseshoe bats (Rhinolophus
sinicus)
No virus isolation
•
•
•
•
•
Amplification of coronavirus RNA from anal swabs
Serology
It is highly-similar, but not identical to SARS-CoV
•
•
Mutations have most likely occurred in
transmission from bats to civets to humans
Reverse genetics of SARS-CoV and some bat
SARS CoV
Coronavirus Phylogeny
Chymotrypsin-like protease (3CLpro), RNA-dependent RNA polymerase
(Pol), spike (S), and nucleocapsid (N)
Coronaviruses Are Bat
Viruses
SARS Pathogenesis
•
•
Virus is transmitted by respiratory and fecal routes
Infection is mediated by human angiotensin-converting enzyme 2
(hACE2) receptor
High expression
Lung alveolar epithelial cells
Intestinal enterocytes
Low expression
Blood vessels (virtually all organs)
Pneumonia
Cause of death is lung failure
•
•
•
•
•
•
•
Pulmonary Inflammation of SARS
Pathologic findings of lung tissue sections. A: Pulmonary congestion and edema (H&E stain, original
magnification x100). B: A mild degree of interstitial lymphocytic infiltration. Intra-alveolar organizing
exudative lesion was occasionally found. Detached atypical pneumocytes indicated by arrow (H&E stain,
original magnification x200). C: Atypical multinucleated pneumocytes were occasionally identified. Definite
viral inclusion was not apparent (H&E stain, original magnification x400). D: Fibrin thrombi were frequently
noted in small pulmonary arteries and arterioles (H&E stain, original magnification x200).
Is SARS an Immunopathogenesis?
Criterion
Evidence in SARS
Precedent in other viral infections
Worse disease with
decrease in viral load
Controversial; viral titres, measured
in nasopharyngeal-aspirate
samples, decrease as clinical
disease worsens; but high viral
loads have been detected in lungs
and immune cells after death
MHV-induced demyelination increases as virus is
cleared; MHV-3-induced hepatitis correlates with
macrophage activation and not viral load; and IBVinduced nephritis is detected in chickens with very low
viral loads
Macrophage or DC
infection
Infection is abortive but induces
expression of pro-inflammatory
mediators
MHV and FIPV productively infect macrophages
Macrophages are present in large
numbers in infected lungs
In MHV infection, macrophages infiltrate the CNS
coincident with demyelination (thought to be the final
effector cell); and in FIPV infection, macrophages are
the main cell type in granulomas and are crucial for
pathogenesis
Controversial; anti-inflammatory
mediators might contribute to
delayed viral clearance
MHV-3-induced FGL2 expression is crucial for liver
necrosis; in MHV-JHM-infected mice, IFN- is required
for CD8+ T-cell-mediated responses; and in FIPV
infection, increased cytokine concentrations are present
in blood and tissues during exacerbation of disease
Macrophage infiltration
into sites of
inflammation
High concentration of
pro-inflammatory
mediators in serum or at
site of infection
Inhibition of type I IFN
Shown using isolated macrophages,
induction in infected
DCs and fibroblasts
cells
Lymphopenia and
neutrophilia
MHV does not induce type I IFN expression
Present in most severe cases; and
In FIPV infection, lymphopaenia is present during
lymphocytic infection has been
clinical relapses; and in MHV-3 infection, lymphopaenia
detected
is present and lymphocytic infection has been detected

![Info. Speech Packet [v6.0].cwk (DR)](http://s3.studylib.net/store/data/008110988_1-db39bdd1f22b58bf46d9a39ab146e2e3-300x300.png)