Chapter 1-1
advertisement

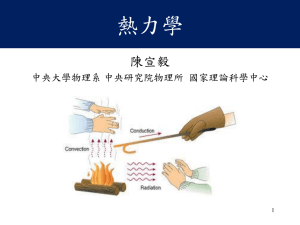
Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. PowerPoint Presentation for Instructor’s Online Learning Center Thermodynamics An Engineering Approach Fourth Edition Yunus A. Çengel Michael A. Boles CHAPTER 1 Basic Concepts of Thermodynamics General overview • Mechanical Engineering – Mechanics – Energy – Systems – Design The over arching goal is design of products to meet societal needs. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki • Thermodynamics – A part of the Energy component of mechanical engineering. – Governs all energy consuming and transforming devices and system. Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics The engineering framework THEORY DATA JUDGMENT (DESIGN) Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. FIGURE 1–5 Some application areas of thermodynamics . Air vents Diet 1-1 Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. FIGURE 1–7 The definition of the force units. Check for dimensional homogeneity! 1 lbm will have a gravitational force on it of 1 lbf on earth. 1-2 Beginning ideas and concepts... Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Macroscopic vs. microscopic viewpoints... A collection of atoms within a container, each with a unique velocity. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Energy in a microscopic description 1 Energy of each atom = e m V 2 2 Number of atoms = N N mV Total Energy = eN 2 Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki 2 N ke1 i 1 Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics The macroscopic description The energy in both cases is the same, E. In the macroscopic description, atomistic concepts are disregarded. How we describe the system chosen for study requires careful selection of properties that are based on observable, measurable quantities. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics The macroscopic viewpoint... Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics State of a thermodynamic system. Enumeration of all of its properties. In macroscopic thermodynamics, the properties of system are assigned to the system as a whole. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Properties in macroscopic thermodynamics m 0 lim Density, Limit of the macroscopic model and assumptions. Molecular and atomic effects are important Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki (m3) Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics How does the state of system change? Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Energetic interactions System Boundary System Surroundings Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Energy Flow Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Basic definitions... Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics System Closed System Mass Flow dm dt 0 Open System Mass Flow Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics System and surroundings Universe = Systems +Surroundings Surroundings Closed System System Boundary Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics System and surroundings Surroundings Mass flow Control Surface Open System ( Fixed space or volume) Open systems have mass flow across their boundaries. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Interactions between system and surroundings... Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Closed system Surroundings Heat System Work Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Open system Surroundings Mass Flow Heat System Power Mass Flow Usually look at rates. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Example: Piston and cylinder - a closed system m Piston Gas at pressure, p Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Example: The gas turbine engine - an open system Fuel Flow In Combustor Air Flow In Shaft Work Output Compressor Work Out Exhaust Gases Out Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. FIGURE 1–17 A control volume may involve fixed, moving, real, and imaginary boundaries. A control volume is an open system 1-5 Key concepts and terms Closed System Open System System Boundary Surroundings Universe Thermodynamic Properties Thermodynamic State Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Equilibrium states and properties Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Properties Properties: •Temperature •Pressure •Volume •Internal energy •Entropy System The system can be either open or closed. The concept of a property still applies. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Classes of properties • Extensive – MASS – VOLUME – ENERGY • Intensive – TEMPERATURE – PRESSURE – DENSITY Specific Properties ADDITIVE OVER THE SYSTEM. NOT ADDITIVE OVER THE SYSTEM. Continuum approach is valid if system is large compared to distance between molecules Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics State • At a given state, all the properties of the system have fixed values. • If the value of one property changes, the state changes. • If no properties are changing, then it is at equilibrium Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Equilibrium • Thermodynamics deals with equilibrium states. • No driving forces • Mechanical equilibrium – No change in pressure with time • Thermal equilibrium – Temperature is same throughout system Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics Equilibrium • Phase equilibrium – No change in the amounts of different phases • Chemical equilibrium – Chemical composition does not change with time. Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics State Postulate • The state of a simple compressible system is completely specified by two independent, intensive properties • Simple compressible system: – Absence of electrical, magnetic, gravitational, motion and surface tension effects. – Independent if one property can be held constant while another is varied Instructor’s Visual Aids Heat Work and Energy. A First Course in Thermodynamics © 2002, F. A. Kulacki Chapter 1 Module 1 Slide ‹#› Energy, Heat and Work Introduction to Macroscopic Thermodynamics