Draper Catalogue of Stellar Spectra
advertisement

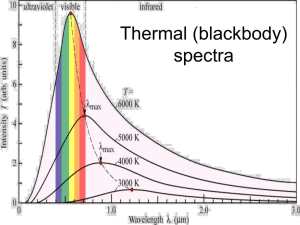
Stars were originally classified according to their spectra (strength = width of their absorption lines) either from a purely observational perspective or with an incomplete understanding (i.e., educated guess) of how their spectra depend on their physical properties. Today, we understand that the (optical) spectra of stars depend on their (photospheric) chemical composition (abundance of different elements), effective temperature, gas pressure (surface gravity), and also phenomena occurring above the photosphere such as stellar winds or chromospheric/coronal activity. The Harvard scheme classifies stars according to their effective temperatures. The Morgan-Keenan scheme extends the Harvard classification to further classify stars according to their luminosities. Learning Objectives Spectral Lines in Solar and Stellar Spectra What does the strength of spectral lines indicate? Classifying Stellar Spectra Secchi classes Harvard classes Morgan-Keenan classification Physics of the Formation of Stellar Spectral Lines Excitation of atoms Ionization of atoms Widths of spectral lines Learning Objectives Spectral Lines in Solar and Stellar Spectra What does the strength of spectral lines indicate? Classifying Stellar Spectra Secchi classes Harvard classes Morgan-Keenan classification Physics of the Formation of Stellar Spectral Lines Excitation of atoms Ionization of atoms Widths of spectral lines Solar Absorption Lines Recall that, in 1802, the English chemist and physicist William Hyde Wollaston passed sunlight through a prism (like Newton and many others had done before him) and noticed for the first time a number of dark spectral lines superimposed on the continuous spectrum of the Sun. By 1814, the German optician Joseph von Fraunhofer had cataloged 475 of these dark lines (today called Fraunhofer lines) in the solar spectrum. He labeled the William Hyde Wollaston, 1766strongest lines A to K, and weaker lines with lower-case 1857 letters. Fraunhofer determined that the wavelength of one prominent dark line corresponds to the wavelength of yellow light emitted when salt is sprinkled in a flame. Today, we know that this dark line is produced by the sodium atom, and is in fact a doublet but was spectrally unresolved at the time. Joseph von Fraunhofer, 1787-1826 Solar Absorption Lines Note that some of the spectral lines (e.g., O2, H2O) are produced by absorption in the Earth’s atmosphere (such lines are called telluric lines). Solar Absorption Lines Among the most prominent absorption lines are the sodium (Na) doublet, the first atomic species to be identified on the Sun. Solar Absorption Lines Among the strongest absorption lines are iron (Fe) lines. Solar Absorption Lines The singly-ionized calcium (Ca II) H and K lines also are particular prominent in the solar spectrum (Ca = Ca I, Ca+ = Ca II, Ca++ = Ca III, etc.). Solar Absorption Lines Of course, the hydrogen (H) Balmer lines also are prominent. Solar Absorption Lines Of course, the hydrogen (H) Balmer lines also are prominent. Solar Absorption Lines Given that absorption lines of Na, Fe, and Ca are roughly as prominent as H, does this mean that Na, Fe, and Ca are roughly as abundant as H on the Sun? Cosmic Abundance of the Elements in the Solar System Atoms of Element per 106 atoms of Silicon (Si) Solar Absorption Lines Given that absorption lines of Na, Fe, and Ca are roughly as prominent as H, does this mean that Na, Fe, and Ca are roughly as abundant as H on the Sun? No Cosmic Abundance of the Elements in the Solar System Atoms of Element per 106 atoms of Silicon (Si) Solar Absorption Lines What then does the strength of absorption lines in the solar spectrum indicate? Cosmic Abundance of the Elements in the Solar System Atoms of Element per 106 atoms of Silicon (Si) Solar Absorption Lines What then does the strength of absorption lines in the solar spectrum indicate? In (smaller) part abundance, and in (greater) part … Cosmic Abundance of the Elements in the Solar System Atoms of Element per 106 atoms of Silicon (Si) Stellar Absorption Lines Notice of that the strength of Balmer absorption lines change with the effective temperatures of stars. What do we need to understand to predict the relative strength of Balmer absorption lines across different stars? Stellar Absorption Lines What do we need to understand to predict the relative strength of Balmer absorption lines across different stars? How the number of hydrogen atoms excited to the n = 2 level depends on the gas temperature. Solar and Stellar Absorption Lines What then does the strength of absorption lines in the solar and stellar spectra indicate? In (smaller) part abundance, and in (greater) part how atoms are excited and ionized to produce the observed spectral lines. Solar and Stellar Absorption Lines Why are solar/stellar lines seen in absorption rather than emission? Kirchhoff’s laws Why are solar/stellar lines seen in absorption rather than emission? Because the outer layer of the Sun/stars (where the absorption lines are produced) is cooler than the inner layer where the light is produced (photosphere). Structure of the Sun Why are solar/stellar lines seen in absorption rather than emission? Because the outer layer of the Sun/stars (where the absorption lines are produced) is cooler than the inner layer where the light is produced (photosphere). Structure of the Sun On the Sun, the temperature drops with height from the interior to the photosphere until increasing in the chromosphere and corona. Learning Objectives Spectral Lines in Solar and Stellar Spectra What does the strength of spectral lines indicate? Classifying Stellar Spectra Secchi classes Harvard classes Morgan-Keenan classification Physics of the Formation of Stellar Spectral Lines Excitation of atoms Ionization of atoms Widths of spectral lines Classifying Stellar Spectra Suppose that you were asked to classify stars according to their spectra, with little understanding of how the strength of their different absorption lines depend on stellar properties. What would you do? Classifying Stellar Spectra Suppose that you were asked to classify stars according to their spectra, with little understanding of how the strength of their different absorption lines depend on stellar properties. What would you do? Presumably you would start by: - looking for strong spectral lines common to all stars - identify the elements that produce these lines Classifying Stellar Spectra Among the strongest and common lines are produced by hydrogen, the most common element in the Universe (75% Hα of all baryons). Near-IR Optical Ultraviolet Hβ Hγ Hδ Hε Classifying Stellar Spectra Among the strongest and common lines are produced by helium, the second most common element in the Universe (23% of all baryons). Classifying Stellar Spectra Among other relatively strong and common lines are the most abundant metals (elements other than hydrogen and helium). Cosmic abundance of the elements: The Secchi Classes During 1860s and 1870s, Father Pietro Angelo Secchi, Director of the Observatory of the Roman College, made the first classifications of stars based on their spectra (from his collection of about 4000 stellar spectrograms): - Class I stars exhibit prominent hydrogen Balmer lines - Class II stars exhibit calcium and sodium lines - Class III stars exhibit broad and complex bands of lines - Class IV stars show prominent carbon lines - Class V stars show lines in emission Pietro Angelo Secchi, 1818-1878 The Secchi classes have been superseded and largely forgotten. Recall that: 1802: Wollaston discovered absorption lines in sunlight 1814: Fraunhofer cataloged 475 lines in sunlight, and identified the strongest line as being produced by calcium 1860s: Foundations of spectroscopy established by Bunsen and Kirchhoff 1880s: Wavelengths of Balmer lines at optical wavelengths precisely determined The Secchi Classes The Secchi classes: - Class I stars exhibit prominent hydrogen Balmer lines - Class II stars exhibit calcium and sodium lines - Class III stars exhibit broad and complex bands of lines - Class IV stars show prominent carbon lines - Class V stars show lines in emission The Harvard Classes The modern classification of stellar spectral types dates back to Edward C. Pickering of the Harvard College Observatory and his assistants, Williamina P. Fleming, Antonia Maury, and most importantly Annie Jump Cannon. This classification system is sometimes referred to as the Harvard system, familiar to all: Oh Be A Fine Girl/Guy Kiss Me (coined by Cannon). Edward C. Pickering, 1846-1919 Williamina P. Fleming, 1857-1911 Antonia Maury, 1866-1952 Annie Jump Cannon, 1863-1941 The Harvard Classes In the 1880s, Pickering began a survey of stellar spectra at the Harvard College Observatory using the objective-prism method. This method uses a prism placed in front of the telescope to disperse light, and has the advantage of simultaneous spectral measurements of multiple astronomical sources with wide wavelength coverage but low spectral resolution. Illustration of spectra taken with an objective prism The Harvard Classes In the 1880s, Pickering began a survey of stellar spectra at the Harvard College Observatory using the objective-prism method. This method uses a prism placed in front of the telescope to disperse light, and has the advantage of simultaneous spectral measurements of multiple astronomical sources with wide wavelength coverage but low spectral resolution. Actual spectra taken with an objective prism Illustration of spectra taken with an objective prism The Harvard Classes A first result of this work was the Draper Catalogue of Stellar Spectra, published in 1890. This work was later extended to become what is known today as the Henry Draper Catalogue, where stars are labeled according to their number in this catalogue (e.g., Betelgeuse is HD 39801). Henry Draper was a pioneer of astrophotography (his father made the first photograph of the Moon through a telescope). In 1872, Draper succeeded in taking the first photograph of a stellar spectrum that showed absorption lines. Upon his untimely death, his widow funded the Henry Draper Medal for outstanding contributions to astrophysics, and an endowment used to finance the compilation of the Henry Draper catalog. Henry Draper, 1837-1882 The Harvard Classes In the 1890s, Fleming labeled stellar spectra with capital letters according to the width of their hydrogen absorption lines, beginning with the letter A for the broadest lines. She divided the Secchi classes I to IV into more specific classes, given letters from A to N. Also, the letters O, P and Q were used, O for stars whose spectra consisted mainly of bright lines (today recognized as Wolf-Rayet stars), P for planetary nebulae, and Q for stars not fitting into any other class. Hδ Hγ Hβ Hα The Harvard Classes Why did Fleming use the width rather than the intensity of spectral lines as a measure of the strength of spectral lines? The stellar spectra were recorded on photographic plates. An example of the spectrum of six individual stars as recorded on photographic plates is shown here. Obviously, it is difficult to quantify the intensity of absorption lines. On the other hand, it is relatively easy to quantify the width of absorption lines. What determines the width of stellar absorption lines? The Harvard Classes Why did Fleming use the width rather than the intensity of spectral lines as a measure of the strength of spectral lines? The stellar spectra were recorded on photographic plates. An example of the spectrum of six individual stars as recorded on photographic plates is shown here. Obviously, it is difficult to quantify the intensity of absorption lines. On the other hand, it is relatively easy to quantify the width of absorption lines. What determines the width of stellar absorption lines? - natural broadening - Doppler (thermal) broadening - pressure broadening The Harvard Classes The sizes of stars on a photographic plate/CCD image depends on the brightness of the star. The Harvard Classes Why did Fleming use the width rather than the intensity of spectral lines as a measure of the strength of spectral lines? The Harvard Classes Why did Fleming use the width rather than the intensity of spectral lines as a measure of the strength of spectral lines? The Harvard Classes Why did Fleming use the width rather than the intensity of spectral lines as a measure of the strength of spectral lines? The Harvard Classes Why did Fleming use the width rather than the intensity of spectral lines as a measure of the strength of spectral lines? The width reflects the absorption depth of a spectral line. The Harvard Classes In 1897, Maury (Henry Draper’s niece) developed a much more complex classification scheme she was using to study the widths of spectral lines. Maury rearranged her classes in a way that would be equivalent to placing Fleming’s B class before the A class. Hδ Hγ Hβ Hα The Harvard Classes In 1901, Cannon negotiated a compromise and based on her classification scheme dropped all letters apart from O, B, A, G, K, and M, and placed B class before A, and O class before B. She also subdivided each class into 10 subclasses from 0 to 9 (e.g., the Sun is G2). Hδ Hγ Hβ Hα The Harvard Classes In 1901, Cannon negotiated a compromise and based on her classification scheme dropped all letters apart from O, B, A, G, K, and M, and placed B class before A, and O class before B. She also subdivided each class into 10 subclasses from 0 to 9 (e.g., the Sun is G2). Cannon’s and her coworker’s work was published between 1928-1924 in nine volumes that became the Henry Draper catalog, listing nearly 230,000 stars. (Cannon was the first woman to receive the Henry Draper Medal, awarded since 1886 by the National Academy of Sciences for outstanding achievement in astronomical physics). The Harvard Classes In Cannon’s classification scheme, visible lines of ionized helium (He II) are detectable in O stars. (An atom’s ionization stage is indicated by a Roman numeral, where I is neutral, II is singly-ionized, III is doubly-ionized, etc.) Note that this photograph is a negative, so that bright lines correspond to absorption lines. The Harvard Classes In Cannon’s classification scheme, visible lines of neutral helium (He I) is strongest for B2 stars. Note that this photograph is a negative, so that bright lines correspond to absorption lines. The Harvard Classes Energy diagram of helium and permitted transitions to the n = 1 and n = 2 states. (In this rendering of the energy diagram, the ground state of parahelium is defined to have an energy of 0 eV). How does the ionization energy of helium compare to hydrogen? Orthohelium Parahelium The Harvard Classes How does the ionization energy of helium compare to hydrogen? Parahelium higher, but orthohelium lower, ionization energy than that of hydrogen. Orthohelium Parahelium The Harvard Classes In Cannon’s classification scheme, hydrogen Balmer lines reach their maximum intensity in the stars of spectra type A0. Note that this photograph is a negative, so that bright lines correspond to absorption lines. The Harvard Classes In Cannon’s classification scheme, hydrogen Balmer lines reach their maximum intensity in the stars of spectra type A0. Note that this photograph is a negative, so that bright lines correspond to absorption lines. The Harvard Classes In Cannon’s classification scheme, visible lines of singly ionized calcium (Ca II) are most intense for K0 stars. (Neutral Ca has 20 electrons.) Note that this photograph is a negative, so that bright lines correspond to absorption lines. The Harvard Classes In Cannon’s classification scheme, broad (molecular) bands are prominent in M stars. Note that this photograph is a negative, so that bright lines correspond to absorption lines. The Harvard Classes A modern description of the Harvard classification scheme. The Harvard Classes Today, we know that Cannon’s classification scheme reflects the effective temperatures of stars, with spectral type O being the hottest and M being the coolest stars. Why, and how, do the strength of absorption lines in stellar spectra depend on stellar effective temperatures? We need to understand how the excitation and ionization of atoms depend on the gas temperature. In Cannon’s days, only how the excitation of atoms depend on the gas temperature was understood. Hγ Hβ Hα The Morgan-Keenan Classification In 1943, William W. Morgan and Phillip C. Keenan published the Atlas of Stellar Spectra comprising 55 photographic prints showing how stellar spectra depend on both stellar effective temperatures and luminosities. They extended the Harvard classification system to include the dependence of stellar spectra on stellar luminosities, giving rise to the present-day system for classifying stars. The Morgan-Keenan Classification Notice that, in the spectra shown below, all six stars have the same effective temperature, yet the widths of their spectral lines (as measured at their relatively diffuse line wings) are different. These stars have different luminosities, and therefore different sizes. Their different spectral linewidths (as measured at their line wings) is caused by their different surface gas pressures. Learning Objectives Spectral Lines in Solar and Stellar Spectra What does the strength of spectral lines indicate? Classifying Stellar Spectra Secchi classes Harvard classes Morgan-Keenan classification Physics of the Formation of Stellar Spectral Lines Excitation of atoms Ionization of atoms Widths of spectral lines Formation of Spectral Lines in Stellar Atmospheres Today, we know that the (optical) spectra of stars depend on their (photospheric): - chemical composition (abundance of different elements) - effective temperature - gas pressure (surface gravity) - presence of above-photospheric phenomena such as stellar winds and chromospheric/coronal activity (beyond scope of this course) The Harvard scheme classifies stars according to their effective temperatures. The Morgan-Keenan scheme extends the Harvard classification to further classify stars according to their luminosities. To understand how the strengths of stellar spectral lines depend on effective temperature, we need to know - how atoms in stellar atmospheres are excited and ionized (through collisions) - how the speeds of atoms are distributed, and the dependence of the speed distribution with temperature - how the excitation and ionization of atoms, and hence the strengths of their spectral lines, depend on temperature Formation of Spectral Lines in Stellar Atmospheres Today, we know that the (optical) spectra of stars depend on their (photospheric): - chemical composition (abundance of different elements) - effective temperature - gas pressure (surface gravity) - presence of above-photospheric phenomena such as stellar winds and chromospheric/coronal activity (beyond scope of this course) The Harvard scheme classifies stars according to their effective temperatures. The Morgan-Keenan scheme extends the Harvard classification to further classify stars according to their luminosities. To understand how the widths of the wings of stellar spectral lines depend on luminosity, we need to know - what processes affect the widths of the wings spectral lines (pressure broadening) - how the widths of the wings of spectral lines depend on gas pressure