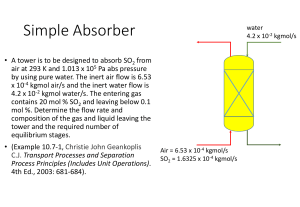
Problems
advertisement

Exercises
Problem-1
• The ozone (O3) concentration in urban areas with
photochemical smog problem reaches a value of 0.1 ppm
over a 1-hr period. Determine by what percentage this
level exceeds the ambient air quality standard of 160
μg/m3 if the temperature is 25°C.
Solution
Step 1
𝜇𝑔
(𝑝𝑝𝑚) ∗ 𝑚𝑜𝑙𝑒𝑐𝑢𝑙𝑎𝑟 𝑤𝑒𝑖𝑔ℎ𝑡 ∗ 1000
C
=
3
𝑚
24.5
[ Note: molecular weight in g/mol]
C
𝜇𝑔
𝑚3
=
(0.1) ∗ 48 ∗ 1000
24.5
= 195.91𝜇𝑔/𝑚3
Step 2:
195.91 − 160
=
∗ 100% = 22.44%
160
Problem-2
• Find the height above sea level where the
pressure is 90 kpa for an atmosphere pressure
of 101.3 kpa and an air density of 1.23 kg/m3.
Solution
Step1:
• dP = - (Rho) g dZ static equilibrium
Step 2:
Integrate from 0 to z:
(101.3-90) kPa = 1.23 kg/m3* 9.81 m/s2* (z-0)
• z = 0.9365 kPa*m3/(kg*m/ s2)
[note- N = kg*m/ s2, N/m2 = Pa]
z = 936.5 m above sea level
Problem-3
• Determine the potential temperature at 1100
ft if the pressure and temperature are 980
mbar and 690 F respectively.
Solution
θ = T (P0/P)R/Cp
θ = T (P0/P)0.288
Where, P0 = 1000 mbar
Given:
T = 690 F = 20.560C = 20.56 + 273 = 293.56 0K
P = 980 mbar
θ = 293.56 * (1000/980)0.288
= 295.27 0K
Problem-4
• During a field program carried by a utility in Ohio, the
atmospheric lapse rate on July 29, 2009 was found
constant up to 800 m. At ground level the pressure (Po) is
1020 mbar and the temperature To is 15oC. A radio
sound measurement indicates that at some elevation z
the pressure and temperature are 975 mbar and 11.5oC
respectively. Determine the atmospheric temperature
gradient dT/dz in degree Kelvin per meter.
Solution
EQUATIONS
• dp = -(Rho)gdz static equilibrium
• p= (Rho)RT
ideal gas law
• dT/dz = b
lapse rate definition
• T –T0 = bz
for constant lapse rate, initial z = 0
dT=b dz
UNITS
• J = N*m
• N = kg*m/s2
• T0 = 15 + 273 = 288 K
use absolute temperatures
• T = 11.5 + 273 = 284.5 K
Solution
Step 1:
Calculate ideal gas law constant for air in appropriate units:
R(air) = Ru/MWair
= 8.315 kJ/(kgmol*K)/(28.96 kg/kgmol)
= 0.28712 *103 (kg*m/s2*m/(kgmol*K))/(kg/kgmol)
= 287.12 m2/s2/K
Step2:
Divide the static equilibrium equation by the ideal gas
law equation and substitute dT/b for dz from the lapse
rate definition.
dp/dz = -(p/RT)*g
dp/p = -(g/R)*dz/T
or, dp/p = -(g/Rb)*dT/T
Lapse rate b = -(g/R)*ln(284.5/288)/ln(975/1020)
= -{(9.81 m/s2/(287.12 m2/s2/K))}*{(-.012227)/(-.045120)
= -0.926 K/100 m atmospheric temperature gradient
Problem-5
• Calculate the equilibrium-incremental
increase in surface temperature from 1850 to
2050 due to carbon dioxide. The
concentrations for CO2 are as follows:
• 1850: 280 ppm
• 2050: 562 ppm (estimated)
• CO2 Emission Calculation:
CO2 = (Pi)*(Foi)*Ci
–
–
–
–
P: Amount of fuel
FO : Fraction oxidized
C : Average carbon content
i: Particular fuel type
• Change in radiative forcing:
For CO2
ΔFCO2 = K3 ln [(co2)/(co2)0]
» K3 : 6.3 (W/m2)
• Surface temperature change:
ΔTs = ΔF * λ
• ΔTs: Surface temperature
• ΔF: Radiative forcing
• λ: Climate sensitivity factor (0.57 in 0C/W/m2)