Oral Anti-diabetic Medications
Mario Skugor, MD FACE
Endocrine and Metabolic Institute
Cleveland Clinic
CLASSES OF ORAL ANTIDIABETIC MEDICATIONS
Insulin
Insulin
secretaguages sensitizers
Incretin
AlphaCNS acting
based
glucosidase
therapies inhibitors
Long acting
Liver
PPD-4
inhibitors
Sulphonylureas
Metformin
Short acting
Muscle and Fat
Meglitinides
Thiazolidinediones
Acarbose
Bromocriptine
Miglitol
In development
Other
Dual PPAR gamma agonists
Bile acid sequestrants
SGLT-2 antagonists
Orlistat
CLASSES OF ORAL ANTIDIABETIC MEDICATIONS
Insulin
Insulin
secretaguages sensitizers
Incretin
AlphaCNS acting
based
glucosidase
therapies inhibitors
Long acting
Liver
PPD-4
inhibitors
Sulphonylureas
Metformin
Short acting
Muscle and Fat
Meglitinides
Thiazolidinediones
Acarbose
Bromocriptine
Miglitol
In development
Other
Dual PPAR gamma agonists
Bile acid sequestrants
SGLT-2 antagonists
Orlistat
Canagliflozin just approved.
METFORMIN
French lilac – used in folk
medicine for centuries.
Synthesized in 1920s
In 1950 Metformin was used to
treat influenca and noted to
lower blood glucose to
physiologic levels.
METFORMIN
• In 1957 first clinical trial of diabetes treatment was
published in France
• Approved in 1958 in UK, 1972 in Canada and in
1995 in US.
METFORMIN
• Suppression of hepatic gluconeogenesis through
activation AMP-activated protein kinase (AMPK).
• Improves glucose uptake in muscle and fat.
• Causes weight loss in some individuals.
• Improves menstrual cycle and fertility in PCOS
• May improve NASH.
• May reduce risk of range of different carcinomas
METFORMIN
• Comes in 500, 850 and 1000 mg pills.
• Extended release pills are available (but more
expensive)
• Usual dose is 1000 mg twice a day.
• Main side effects is abdominal cramping and diarrhea
• Metformin extended release is better tolerated by
some patients
• Even mild renal failure (Cr>1.4 in males and >1.5 in
females) is contraindication for use
Metformin – Bottom line
• Clearly the first line.
• Cheap
• Improves physiology
• Has other benefits.
• Unfortunately, significant proportion of patients has
contraindications or cannot tolerate it.
SULPHONYLUREAS
• Marcel Janbon and co-workers discovered
hypoglycemic effect of sulfonylurea in 1942.
• They were studying sulfonamide antibiotics and
discovered that the compound sulfonylurea
induced hypoglycemia in animals
Sulphonylureas
• First sulfonylurea for treatment of DM introduced in 1955.
– General structure:
Sulphonylurea
• First generation – Binds to the proteins in the blood.
– Tolbutamide
– Chlorpropamide
– Tolazamide
– Acetohexamide
– Carbutamide
Sulphonylurea
• Second generation – Not bound to serum proteins.
– Glipizide
– Glyburide (glibenclamide)
– Gliclazide
– Glibornuride
– Gliquidone
– Glisoxepide
– Glyclopyramide
Sulphonylurea
• Third generation
– Glimepiride
SUFONYLUREAS
Sulfonylurea
• Advantages
– Fast acting
– Once a day dosing
– Gliclazide may be particularly beneficial
• Disadvantages
– Risk of hypoglycemia
– Weight gain
– Possible problems with ischemic preconditioning
Glicilizide
• Inhibits platelet aggregation
• Associated with lower mortality from malignant
neoplasms.
• Improves repair of DNA damage caused by oxidative
stress in tissue cultures.
Sulfonylureas – Bottom line
• Fast acting
• Older ones are cheap
• Do not improve physiology
• Hypoglycemia is significant risk
• Require strict regime of diet
Meglitinides
• Nategelinde
• Repaglinide
• Act on same potassium channel as sulfonylurea but
bind to different part of the molecule.
• Short acting – taken 0-30 min before meal.
• Risk of hypoglycemia is small
Meglitinides – Bottom line
• Useful in small number of patients for relatively short
period of time.
• Allow for some flexibility in timing of the meals.
TZD-s – actually Pioglitazone
• The proliferator-activated receptor gamma (PPAR-γ)
and to a lesser extent PPAR-α agonist in the muscle,
adipose tissue, and the liver.
• Pioglitazone reduces insulin resistance in the liver and
peripheral tissues.
• Pioglitazone decreases the level of triglycerides and
increases HDL without changing LDL and total
cholesterol.
Pioglitazone
• Pioglitazone - 15 - 30 - 45 mg pills
• Peripheral edema is main side effect.
• More hospitalizations for CHF in studies with all
TZD-s
• Effect is maintained when combined with metformin
and incretin based therapies.
Thiazolidinediones and bladder cancer.
Colmers IN, et al. CMAJ 2012I:10.1503
TZD-s and fracture risk
Toulis KA, et al. CMAJ, 2009, 180 (8) 841-842
TZD-s and fractures
TZD-s are associated with fractures in females over 50
years of age.
In men risk is increased if TZD-s are used with loop
diuretic
Bilik D, et al. JCEM. 2010 (10) 1210.
Bottom line on Pioglitazone
• Benefits are still higher than risks.
• There is some evidence that lower dose is not
associated with risk of bladder cancer.
• However – at this time Metformin and PPD-4 inhibitors
are clearly ahead of Pioglitazone as choices for
treatment.
Incretins
• Gut-derived hormones, secreted in response to nutrient ingestion,
that potentiate insulin secretion from islet cells in a glucosedependent fashion, and lower glucagon secretion from islet cells
• Two predominant incretins:
– Glucagon-like peptide–1 (GLP-1)
– Glucose-dependent insulinotropic peptide (GIP)
(also known as gastric inhibitory peptide)
• Incretin effect is impaired in type 2 diabetes
– Known as GLP-1 deficiency
Incretin system and DPP-4 physiologic action
Native GLP-1 is rapidly degraded by DPP-IV
Human ileum,
GLP-1 producing
L-cells
Capillaries,
DPP-IV (Di-Peptidyl
Peptidase-IV)
Double immunohistochemical staining for DPP-IV (red)
and GLP-1 (green) in the human ileum
Adapted from: Hansen et al. Endocrinology 1999;140:5356–5363.
Glucagon-Like Peptide–1 Normalizes
Postprandial Hyperglycemia in Patients
with Type 2 Diabetes
Healthy subjects
T2DM patients
Infusion
Infusion
300
300
Plasma glucose (mg/dl)
Plasma glucose (mg/dl)
Liquid meal
250
200
150
Placebo
100
50
0
–1
GLP-1 [7-36 amide]
1.2 pmol/kg/min
0
1
2
Time (h)
3
4
Nauck MA et al. Acta Diabetol. 1998;35:117-129.
Liquid meal
250
Placebo
200
150
100
50
0
–1
GLP-1 [7-36 amide]
1.2 pmol/kg/min
0
1
2
Time (h)
3
4
Slide Source:
Lipids Online Slide Library
www.lipidsonline.org
Continuous Glucagon-Like Peptide–1
Infusion Reduces Appetite over 6 Weeks
500
*Satiety
*Fullness
400
Mean (SE)
AUC for
300
Visual
Analogue 200
Score (mm)
vs Time (h)
Time (wk)
*Prospective food intake
*Hunger
100
0
*p<.05
0
1
6
Time (wk)
All data for patients treated with glucagon-like peptide–1 (n = 10).
No changes in these parameters were observed in the saline group.
Zander M et al. Lancet. 2002;359:824–830.
Slide Source:
Lipids Online Slide Library
www.lipidsonline.org
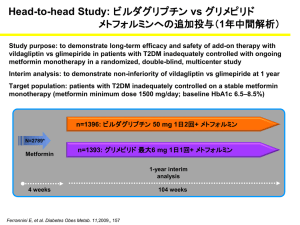
Glycemic Control with GLP-1 Receptor
Agonists in Head-to-Head Clinical Trials
Trial:
Size (N):
Study length (weeks):
LEAD-61
464
26
DURATION-12 DURATION-53 DURATION-64
303
254
912
30
24
26
0.0
LIRA
EXN QW
A1C Change (%)
EXN BID
*Significant difference
vs comparator GLP-1
receptor agonist
-0.5
-1.0
-1.5
-2.0
-0.8
-0.9
-1.1
*
-1.3
-1.5
-1.9
*
-1.6
*
-1.5
1Buse
JB et al. Lancet. 2009;374:39-47 | 2Drucker DJ et al. Lancet. 2008;372:1240-1250 |
3Blevins T, et al. J Clin Endocrinol Metab. 2011;96:1301-1310 | 4Buse JB et al. Presented at
47th EASD Annual Meeting, Lisbon, Portugal, 14 September 2011.
Slide Source:
Lipids Online Slide Library
www.lipidsonline.org
Comparison of Incretin Modulators
GLP-1 Analogues
DPP-4 Inhibitors
Administration route
Injection
Oral
GLP-1
Sustained
Meal-related
Effect on A1C
Effects on body weight
Nausea,
Rare: pancreatitis
(Well tolerated)
Nasopharyngitis, skin rashes,
Stevens-Johnson syndrome
Side effects
-cell function
GLP-1=glucagon-like peptide–1; DDP-4=dipeptidyl peptidase–4
Slide Source:
Lipids Online Slide Library
www.lipidsonline.org
DPP – 4 inhibitors
• Sitagliptin
• Saxagliptin
• Linagliptoin
• Alogliptin
• Vildagliptin – marketed in EU
• More in development
– Gemigliptin
DPP4 inhibitors
• All taken once a day
– Sitaglipitin 100 mg daily
–50 mg if Cr 1.7-3.0 for men and 1.5-2.5 for women
–25 mg in ESRD
– Saxagliptin 5 mg per day
–2.5 mg in renal impairment
–2.5 if taken with cytochrome P450 inhibitors
(ketoconazole)
– Linagliptin 5 mg daily
Sitagliptin - example
DPP-4 inhibitors
• Side effects are minimal
• Acute pancreatitis is seen
– Linagliptin 15.2/10.000 patients
– Placebo 3.7/10,000 patients
– Saxaglipin – No data but some postmarketing cases
are reported
– Sitagliptin – There is 88 cases in 2.5 years in
postmarketing reporting.
DDP-4 inhibitors – Bottom Line
• Very well tolerated
• Improve physiology
• Expensive
• So far, no serious adverse effects with long term use.
• No increased risk of pancreatic caner.
Alpha-Glucosidase inhibitors
• Acarbose - 25, 50 or 100 mg tablets
• Miglitol - 25, 50 and 100 mg tablets
– They block intestinal enzyme breaking sugars to
monosaccharides.
– This slows down and blocks some of carbohydrate
absorption.
– Postprandial peak is diminshed and Hba1c
improves.
Alpha-Glucosidase inhibitors
Alpha-glucosidase inhibitors
• Taken with each meal.
• Side effects are flatulence and diarrhea
• This can be diminished with low carb, high fiber diet
and slow titration of the dose form 25 mg to 100 mg
per day.
• Very low risk of hypoglycemia
Alpha-glucosidase inhibitors – Bottom line
• Very useful if tolerated
• Relatively cheap.
Bromocriptine
• Bromocriptine mesylate given within 2 hours of waking
up in the morning improves glycemic control by
unknown mechanism.
• It is given in escalating dose starting with 0.8 mg and
increasing by 0.8 mg every week to maximal tolerated
dose.
• Therapeutic dose is between 1.6 to 4.8 mg per day.
• Very low risk of hypoglycemia.
• About 25% of patients experience some nausea.
Bromocriptine
Bromocriptine
Bromocriptine – Bottom line
• Useful if tolerated.
• Still expensive
• Many patients are discuoraged by need to use 2-6 pills
at once (In US only 0.8 mg pill is available).
Bile acid sequestrants
• Colesevelam
• Cholestyramine
• Colestid
– Mechanism is unknown
– In db/db mice these drugs increase metabolic
utilization of glucose in peripheral tissues which
corelates with decrease in muscle long chain
acylcarnitine content
Meissner M, et al. PLOS 2011 (6)11 e24564.
Coleselavam
Cholestyramine
Colesevalam
Colesevalam
Orlistat
Effect on weight
Effects on Hba1c
Targeting the Kidney
Chao EC, et al. Nat Rev Drug Discovery. 2010;9:551-559.
Renal Glucose Transport
Chao, EC & Henry RR. Nature Reviews Drug Discovery. 2010;9:551-559.
Rationale for SGLT2 Inhibitors
• SGLT2 is a low-affinity, high capacity glucose transporter
located in the proximal tubule and is responsible for 90% of
glucose reabsorption
• Mutation in SGLT2 transporter linked to hereditary renal
glycosuria, a relatively benign condition in humans
• Selective SGLT2 inhibitors have a novel & unique mechanism of
action reducing blood glucose levels by increasing renal
excretion of glucose
• Decreased glycemia will decrease glucose toxicity leading to
further improvements in glucose control
• Selective SGLT2 inhibition, would also cause urine loss of the
calories from glucose, potentially leading to weight loss
Brooks AM, Thacker SM. Ann Pharmacother. 2009;42(7):1286-1293.
Canagliflozin
Metformin + Canagliflozin Dose-Ranging Study
Mean Baseline
A1C (%)
7.71
8.01 7.81 7.57 7.70 7.71 7.62
*
*
*
*
*
Rosenstock J, et al. Abstract 77-OR. ADA 2010.
*
*P˂.001 vs. placebo calculated using LS means
Changes from Baseline in Body Weight
in Phase 3 Dapagliflozin Studies
Placebo
Dapa 2.5mg
Dapa 5mg
Dapa 10mg
Wilding JPH, et al. Abstract 78-OR. ADA 2010; Strojek K, et al. Abstract 870. EASD 2010;
Ferrannini E, et al. Diabetes Care. 2010;33(10):2217-2224; Bailey CJ, et al. Lancet. 2010;375(9733):2223-2233.
Canagliflozin Trials
• Symptomatic genital infections in 3-8%
canagliflozin arms
– 2% placebo
– 2% SITA
• Urinary tract infections in 3-9% canagliflozin arms
– 6% placebo
– 2% SITA
• Hypoglycemia in 0-6% canagliflozin arms
– 2% placebo
– 5% SITA
Rosenstock J, et al. Abstract 77-OR. ADA 2010.
Treatment and cancer risk
metformin
sulphonylurea
metformin + sulpha
insulin
No treatment
Treatment and colorectal cancer risk
metformin
sulphonylurea
metformin + sulpha
insulin
Treatment and pancreatic cancer risk
metformin
sulphonylurea
metformin + sulpha
insulin
ADOPT and RECORD Trials
ADOPT and RECORD Trials
METFORMIN
METFORMIN
Are all sulphonylureas the same?
Retrospective cohort study
Glibenclamide treated N-378
Gliclizide treated N-190
5 year follow up
Cancer mortality higher for
glibenclamide after
adjustments for age and sex,
BMI, metformin and insulin
treatment- HR 3.56 (1.1-11.9)
Are all sulphonylureas the same?
Matched case-control study
195 diabetic patients with incident malignancy
195 matched diabetic patients with no malignancy
Matched for sex, age, BMI, duration of diabetes, Hba1c,
smoking and alcohol abuse
Exposure to antidiabetic drugs over last 10 years was
analyzed.
Are all sulphonylureas the same?
Possible mechanism
New Classes Presently in Development
• Long-acting GLP-1 receptor agonists
• Ranolazine
• 11 Hydroxysteroid Dehydrogenase (HSD)- 1 inhibitors
• Fructose 1,6-bisphosphatase inhibitors
• Glucokinase activators
•
•
•
•
G protein-coupled Receptor (GPR)- 40 & -119 agonists
Protein Tyrosine Phosphatase (PTB)- 1b inhibitors
Camitine- Palmitoyltransferase (CPT)- 1 inhibitors
Acetyl COA Carboxylase (ACC)- 1 & -2 inhibitors
• Glucagon receptor antagonists
• Salicylate derivatives