RCT (drug) - Johns Hopkins Medicine
advertisement

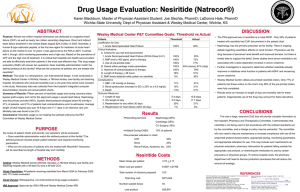
Presenters for Journal Club: James Cooper Eugenie Shieh Aaron Schueneman Tim Niessen Introduction • Nesitiride developed as a novel IV vasodilator in decompensated heart failure • Recombinant brain natriuretic peptide • Alternative to inotropic therapy • 1st randomized double blind trial of Nesitiride in decompensated heart failure Study Outline • Hypothesis: Niseritide improves dyspnea and PCWP (vs IV NG or placebo) • Study Design: Randomized Controlled Trial • Setting: 55 community and academic hospitals in U.S. • Participants: 489 pt w/ dyspnea at rest from decompensated HF • Data Collection: dyspnea scale, followed by measurement of PCWP at 3 hours • Primary Outcomes: – Change in PCWP – dyspnea at 3 hrs • Secondary Outcomes: – Safety – 30-day re-hospitalization rate, 7-day and 6-month mortality Methods Inclusion Criteria Exclusion Criteria • • • • Dyspnea at rest due to dCHF (class IV) severe enough to require hospitalization and IV tx Cardiac etiology of dyspnea = PCWP ≥ 20 + 2 of the following – JVD – PND or 2-pillow orthopnea – Abdominal discomfort due to mesenteric ischemia – CXR c/w dCHF • • Acute decompensation of chronic HF, gradual worsening of chronic HF, new acutely dCHF Allowed prior and current use of dobutamine and dopamine • • • • • SBP < 90 mmHg Cardiogenic shock or volume depletion Contraindication to IV vasodilator Acutely unstable clinical status that would not permit a 3 hr placebo period Use of IV NTG that could not be withheld Mechanical ventilation Anticipated survival <30-35 days Methods Investigator’s clinical decision No RHC RHC Randomization 3hr NTG nesiritide placebo Crossover to double blind treatment 3-48 hr same same NTG nesiritide Same as left Results: Patient Characteristics • • • • • • • 84% had class III/IV HF prior to presentation Vast majority had evidence of fluid overload ACS accounted for 12% of presentations 85% had EF < 40%; mean EF 27% 21% had creatinine > 2.0 mg/dl 47% had diabetes mellitus Mean SBP 121mmHg; 18% < 100mmHg; 22% > 140mmHg • Mean PCWP 28mmHg; • Mean cardiac index 2.2 L/min/m2 • Groups well matched Primary Endpoint #1: Change in PCWP Primary Outcome #2: Change in Dyspnea Primary Outcome #2: Change in Dyspnea Results: Secondary Endpoints • More headache in NTG group (20% vs. 8%) • No difference in number of ischemic events, hypotensive episodes or arrhythmias in first 24h • Duration of hypotension > in nesiritide (2.2 vs. 0.7 hours) • 30-day re-admit rate nesiritide vs. NTG: – 23% vs. 20% for all causes – 7% vs. 13% for CHF specifically • 7-day mortality: 0.5% NTG vs. 1.5% nesiritide • 6-month all cause mortality: – NTG 20.8% (95% CI 15.5% - 26.5%) – Nesiritide 25.1% (95% CI 20.0% – 30.5%) Author Conclusions • Nesiritide rapidly improves hemodynamics as measured by RHC better than placebo and better than nitroglycerin. • Nesiritide subjectively improves dyspnea, compared to placebo and is no different when compared to nitroglycerin. Strengths • Large, multicenter, with heterogeneous patient population • Randomized. • Placebo then crossover to active controls • Double-dummy blinded. FDA Approval JAMA 2006;296:1877 Dissent “Nesiritide was approved on the basis of a single trial in which surrogate end points were assessed three hours after administration… In my view nesiritide has not yet met the minimal criteria for safety and efficacy” Topol, NEJM 2005 353:114 Lies, Damned Lies, and Statistics • Endpoint selection: – Surrogate outcome measures. – “hard” vs subjective outcomes / blinding – Clinical vs statistical significance • Placebo vs head-to-head comparisons & “standard therapy”: NEJM 2011;365:32-43