GEOS254Lec6
advertisement

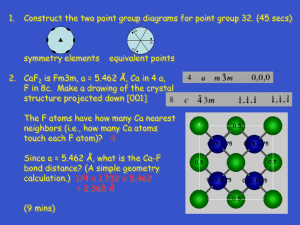
Crystallisation from melts Why do crystals form? What controls their shape and size? Three aspects to the growth of a crystal are Nucleation: formation of a stable nucleus Diffusion of material to the nucleus Growth of the crystal by adding atoms The slowest of these three aspects generally controls the shape and size Different types of crystals If diffusion is slow, the crystal grows spikes to get at the new material needed & forms a dendrite or a skeletal crystal If nucleation is the restriction (a) spherulites can form (many radiating crystals grow from one nucleus or (b) a few large crystals If growth is the restriction (slow crystallisation) then well formed crystals develop Perovskite (Ca,Na,Fe,Ce)(Ti,Nb)O3 A very dense mineral that is cubic with six oxygens around each Ti/Nb and Ca between each four octohedra. It occurs as a minor mineral in syenites Deep in the mantle calcium and magnesium silicates take on this dense form. Model is for MgSiO3 (Si =yellow) Mineral - Melt interface Computer simulated interface between perovskite (MgSiO3) on left and melt. The silicon atoms (yellow) have six oxygens around them (green) in the crystal but only four in the melt. There is a very complex layer at the interface The shape of grains is generally an attempt to minimise the number of unsatisfied bonds associated with boundaries Steno strikes again: the law of constant angles between faces. Steno of stratigraphy fame noted that even though different crystals of the same mineral look different the angles between the faces are constant. This reflects the symmetry and shape of the building blocks. All the quartz crystals have the same faces on a stereo net A) “artists impression of: • (a) nucleus of a metal • (b) nucleus of an organic crystal • (c) nucleus of a long-chain molecular liquid B) Nucleus must reach a critical size before it is stable. The excess energy associated with the surface of the nucleus must be < the energy reduction resulting from the crystal bonding C) Nucleation is commonly on an existing substrate (heterogeneous) NUCLEATION Why a small cluster of atoms may not form a stable nucleus Eight atoms have only 50% of bonds, the 64 atoms have 75% of all the bonds It is a balance between the lower energy of the crystal bonds and the higher energy of the interface. Stable only when XS surface energy is lower Every crystal has imperfections or defects a) cubic lattice b) lattice with an edge dislocation formed by having a half plane of atoms above the dislocation c) a screw dislocation (left handed in this case) DISLOCATIONS Growing a crystal Atoms attach better at a step (Y) than on a plane Screw dislocations produce steps that the crystal uses to grow Re-entrants along twins also help because they are sites of dislocations Crystal faces are: Parallel to lattice planes in which there are strong bonds and across which there are few or weak bonds Biotite (001) faces lack only the few bonds through potassium ions 3 Types of Twinning Growth twins that form as the growing crystal adds on a layer in a different orientation Deformation twins when shear rotates the lattice into a twin orientation (only a very modest strain involved) Transformation twinning as a crystal changes symmetry as it cools (e.g. microcline and cordierite) Some twins are not seen at all under the microscope and some only as a re-entrant (e.g biotite) Sanidine and orthoclase phenocrysts seem to invariably have a simple twin. Why might this be so? Option A: It is groups of atoms in twin orientation that grow large enough to form a stable nucleus Option B: Trace elements that are concentrated in the first stage of growth (Ba,Sr) allow a twin to form Option C: ??????? SIMPLE TWINS