NO in ARDS
advertisement

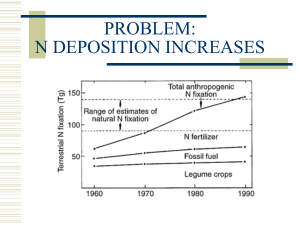
Nitric Oxide Ventilation in ARDS Muhammad Asim Rana ► Nitric oxide was formerly known as endotheliumderived relaxing factor (EDRF). It is one of the nitrogen oxides ("NOx") and is synthesized within cells by an enzyme NO synthase (NOS). This enzyme catalyses the oxidation of L-arginine to Lcitrulline, producing NO, which diffuses into vascular smooth muscle, activating guanylate cyclase which in turn converts guanosine triphosphate into cyclic guanosine monophosphate (cGMP), causing vascular relaxation. NOS is present in two forms: ► The constitutive form (eNOS) Present in vascular, neuronal, cardiac tissue, skeletal muscle and platelets, producing small quantities of NO continuously. Here NOS is Ca2+/calmodulin dependant and is stimulated by cGMP. ► The inducible form (iNOS) Present in endothelium, myocytes, macrophages and neutrophils, which produces relatively large quatities of NO after exposure to endotoxins in sepsis. Following induction high levels of NO produced may form cytotoxic radicals and cause capillary leakage. Effects Cardiovascular Nitric oxide is a potent vasodilator. Shear stresses in vessels increase NO production and may account for flow dependant vasodilatation. Endothelial NO inhibits platelet aggregation. In septic shock the overproduction of NO results in hypotension and capillary leak. NOS inhibitors have been investigated experimentally in the treatment of sepsis. Respiratory ► Important basal vasodilatation in pulmonary vessels is provided by endogenous NO and this may be reversed in hypoxia. Nitric oxide inhibits hypoxic pulmonary vasoconstriction and preferentially increases blood flow through well-ventilated areas of the lung, thereby improving ventilation: perfusion relationships. Neuronal ► Nitric oxide appears to have a physiological role as a neurotransmitter within the autonomic and central nervous system. Proposed roles include modulation of the state of arousal, pain perception, apoptosis and long term neuronal depression and excitation whereby neurones may “remember” previous signals. Peripheral neurones containing NO control regional blood flow in the corpus cavernosum. Gastrointestinal ► NO is a determinant of gastrointestinal motility and appears to modulate morphineinduced constipation. Genitourinary ► Nitric oxide may play a role in sodium homeostasis in the kidney. Immune ► Macrophages and neutrophils synthesize NO which can be toxic to certain pathogens and may be important in host defence mechanisms. Hematological Platelet aggregation is inhibited by NO. Basic Concept ► One hallmark of ARDS is severe hypoxemia caused by physiologic shunting and ventilation/perfusion (V/Q) mismatching. Inhaled vasodilators, particularly nitric oxide can selectively dilate vessels that perfuse well ventilated lung zones, resulting in improved V/Q matching, better oxygenation, and amelioration of pulmonary hypertension. MECHANISM OF ACTION ► Nitric oxide relaxes vascular smooth muscle by binding to the heme moiety of cytosolic guanylate cyclase, activating guanylate cyclase and increasing intracellular levels of cyclic guanosine 3',5'-monophosphate, which leads to vasodilation. When inhaled, pulmonary vasodilation occurs and an increase in the partial pressure of arterial oxygen results. Dilation of pulmonary vessels in well ventilated lung areas redistributes blood flow away from lung areas where ventilation/perfusion ratios are poor. Inhaled vasodilators ► Inhaled vasodilators (green circles) preferentially dilate the pulmonary vessels that perfuse functioning alveoli (white circles), recruiting blood flow away from poorly ventilated units (black circles). The net effect is improved ventilation/perfusion matching. ► In addition, inhaled vasodilators have few systemic effects and rarely cause hypotension because they act locally and have short half-lives. ► Inhaled Nitric oxide (NO) has been wellstudied in patients with acute lung injury and ARDS. ► Inhaled NO has beneficial physiological effects, but there is little evidence that patient outcome improves. This is illustrated by the following clinical trials: ►A well-designed multicenter trial randomly assigned 385 patients with moderate to severe acute lung injury (P/F ratio ≤ 250 mmHg) to either placebo or inhaled NO at 5 ppm. The acute lung injury was not caused by sepsis, and significant nonpulmonary organ dysfunction was absent. Inhaled NO induced short-term improvement of oxygenation; however, there was no improvement in the duration of mechanical ventilation, 28-day mortality, or one-year survival. ► Another multicenter double-blind trial randomly assigned 177 with ARDS to receive increasing concentrations of inhaled NO or placebo. Inhaled NO improved oxygenation modestly, but was not sustained. There was no difference in 28-day mortality, although this was not a primary end point. The modest improvement of oxygenation detected in this trial caused some to argue that further investigation of inhaled NO for ARDS is not warranted, although this view is not universal. It has also been hypothesized that NO may have benefits unrelated to improved V/Q matching, including 1.antiinflammatory properties, 2.antiplatelet activity, and 3.effects which diminish vascular permeability ► Dosing ► Inhaled NO is typically administered at a dose between 1.25 and 40 parts per million (ppm). ► It has been used continuously for days to weeks, with interruptions or attempts to discontinue therapy resulting in worsened oxygenation and increased pulmonary artery pressure. ► However, there is evidence that patients treated with continuous inhaled NO might become sensitized, such that lower doses improve oxygenation and continued higher doses have little or no effect. Metabolism ► Nitric oxide combines with hemoglobin that is 60% to 100% oxygenated. Nitric oxide combines with oxyhemoglobin to produce methemoglobin and nitrate. Within the pulmonary system, nitric oxide can combine with oxygen and water to produce nitrogen dioxide and nitrite respectively, which interact with oxyhemoglobin to then produce methemoglobin and nitrate. At 80 ppm the methemoglobin percent is ~5% after 8 hours of administration. Methemoglobin levels >7% were attained only in patients receiving 80 ppm. PHARMACODYNAMICS / KINETICS ► Absorption: Systemic after inhalation ► Excretion: Urine (as nitrate) ► Clearance: Nitrate: At a rate approaching the glomerular filtration rate. Storage ► NO is stored in aluminium or stainless steel cylinders which are typically 40 litres. These contain 100/1000/2000 p.p.m. nitric oxide in nitrogen. Pure NO is corrosive and toxic. Administration ► The drug is injected via the patient limb of the inspiratory circuit of a ventilator. The delivery system is designed to minimise the oxidation of nitric oxide to nitrogen dioxide. Monitoring ► Chemiluminescence and electrochemical analysers should be used and are accurate to 1 ppm. Potential harms ► Inhaled NO may produce toxic radicals. However, it is unknown whether the toxic radicals are more harmful than ongoing exposure to high fractions of inspired oxygen. ► Methemoglobin and NO2 concentrations may increase when high doses of NO are given(500-2000 ppm of NO), and the concentration of both should be monitored frequently. ► Inhaled NO is associated with renal dysfunction. ► Inhaled NO has immunosuppressant properties that, in theory, could increase the risk of nosocomial infection. ► NO can cause DNA strand breaks and base alterations, which are potentially mutagenic. SUMMARY ► Management of acute respiratory distress syndrome (ARDS) is supportive, aimed at improving gas exchange and preventing complications while the underlying disease that precipitated ARDS is treated. ► Potential ARDS-specific therapies like inhaled NO have been studied; however, they have not been shown to improve clinical outcome and, thus, cannot be recommended for routine care. Predictors when to use Inhaled NO ► Inhaled NO does not improve oxygenation in all patients and the factors that determine responsiveness are uncertain. ► One retrospective study found that patients with septic shock responded to inhaled NO less frequently than patients without sepsis or septic shock. ► A different study reported that a high baseline pulmonary vascular resistance and responsiveness to positive end-expiratory pressure (PEEP) predicted a positive response. ► So the decision lies with treating Intensivist about starting a patient on inhaled NO keeping in view the potential benefits and harms of such therapy. Acknowledgements ► Dr. Mostafa Adel ► Dr. Omar Alsayed ► Dr. Ahmed fouad ► Dr. Ahmed Hossam ► Dr. Ahmed Rajab ► Dr. Sameer Ibrahim ► Dr. Bashir Ahmed ► Dr. Sayed Afzal Thank You