CSF Tau Protein Phosphorylated at Threonine 231 as a Potential
advertisement
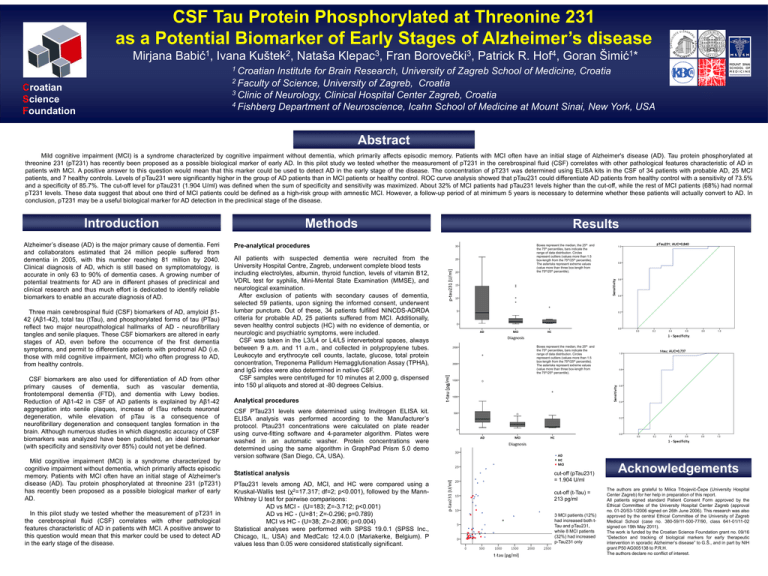
CSF Tau Protein Phosphorylated at Threonine 231 as a Potential Biomarker of Early Stages of Alzheimer’s disease Mirjana 1 Babić , Ivana 2 Kuštek , Nataša 3 Klepac , Fran 3 Borovečki , Patrick R. 4 Hof , Goran 1 Šimić * 1 Croatian Institute for Brain Research, University of Zagreb School of Medicine, Croatia 2 Faculty of Science, University of Zagreb, Croatia 3 Clinic of Neurology, Clinical Hospital Center Zagreb, Croatia 4 Fishberg Department of Neuroscience, Icahn School of Medicine at Mount Sinai, New York, USA Croatian Science Foundation Abstract Mild cognitive impairment (MCI) is a syndrome characterized by cognitive impairment without dementia, which primarily affects episodic memory. Patients with MCI often have an initial stage of Alzheimer's disease (AD). Tau protein phosphorylated at threonine 231 (pT231) has recently been proposed as a possible biological marker of early AD. In this pilot study we tested whether the measurement of pT231 in the cerebrospinal fluid (CSF) correlates with other pathological features characteristic of AD in patients with MCI. A positive answer to this question would mean that this marker could be used to detect AD in the early stage of the disease. The concentration of pT231 was determined using ELISA kits in the CSF of 34 patients with probable AD, 25 MCI patients, and 7 healthy controls. Levels of pTau231 were significantly higher in the group of AD patients than in MCI patients or healthy control. ROC curve analysis showed that pTau231 could differentiate AD patients from healthy control with a sensitivity of 73.5% and a specificity of 85.7%. The cut-off level for pTau231 (1.904 U/ml) was defined when the sum of specificity and sensitivity was maximized. About 32% of MCI patients had pTau231 levels higher than the cut-off, while the rest of MCI patients (68%) had normal pT231 levels. These data suggest that about one third of MCI patients could be defined as a high-risk group with amnestic MCI. However, a follow-up period of at minimum 5 years is necessary to determine whether these patients will actually convert to AD. In conclusion, pT231 may be a useful biological marker for AD detection in the preclinical stage of the disease. Introduction Alzheimer’s disease (AD) is the major primary cause of dementia. Ferri and collaborators estimated that 24 million people suffered from dementia in 2005, with this number reaching 81 million by 2040. Clinical diagnosis of AD, which is still based on symptomatology, is accurate in only 63 to 90% of dementia cases. A growing number of potential treatments for AD are in different phases of preclinical and clinical research and thus much effort is dedicated to identify reliable biomarkers to enable an accurate diagnosis of AD. Three main cerebrospinal fluid (CSF) biomarkers of AD, amyloid β142 (Aβ1-42), total tau (tTau), and phosphorylated forms of tau (PTau) reflect two major neuropathological hallmarks of AD - neurofibrillary tangles and senile plaques. These CSF biomarkers are altered in early stages of AD, even before the occurrence of the first dementia symptoms, and permit to differentiate patients with prodromal AD (i.e. those with mild cognitive impairment, MCI) who often progress to AD, from healthy controls. CSF biomarkers are also used for differentiation of AD from other primary causes of dementia, such as vascular dementia, frontotemporal dementia (FTD), and dementia with Lewy bodies. Reduction of Aβ1-42 in CSF of AD patients is explained by Aβ1-42 aggregation into senile plaques, increase of tTau reflects neuronal degeneration, while elevation of pTau is a consequence of neurofibrillary degeneration and consequent tangles formation in the brain. Although numerous studies in which diagnostic accuracy of CSF biomarkers was analyzed have been published, an ideal biomarker (with specificity and sensitivity over 85%) could not yet be defined. Mild cognitive impairment (MCI) is a syndrome characterized by cognitive impairment without dementia, which primarily affects episodic memory. Patients with MCI often have an initial stage of Alzheimer's disease (AD). Tau protein phosphorylated at threonine 231 (pT231) has recently been proposed as a possible biological marker of early AD. In this pilot study we tested whether the measurement of pT231 in the cerebrospinal fluid (CSF) correlates with other pathological features characteristic of AD in patients with MCI. A positive answer to this question would mean that this marker could be used to detect AD in the early stage of the disease. Methods Pre-analytical procedures All patients with suspected dementia were recruited from the University Hospital Centre, Zagreb, underwent complete blood tests including electrolytes, albumin, thyroid function, levels of vitamin B12, VDRL test for syphilis, Mini-Mental State Examination (MMSE), and neurological examination. After exclusion of patients with secondary causes of dementia, selected 59 patients, upon signing the informed consent, underwent lumbar puncture. Out of these, 34 patients fulfilled NINCDS-ADRDA criteria for probable AD, 25 patients suffered from MCI. Additionally, seven healthy control subjects (HC) with no evidence of dementia, or neurologic and psychiatric symptoms, were included. CSF was taken in the L3/L4 or L4/L5 intervertebral spaces, always between 9 a.m. and 11 a.m., and collected in polypropylene tubes. Leukocyte and erythrocyte cell counts, lactate, glucose, total protein concentration, Treponema Pallidum Hemagglutionation Assay (TPHA), and IgG index were also determined in native CSF. CSF samples were centrifuged for 10 minutes at 2,000 g, dispensed into 150 μl aliquots and stored at -80 degrees Celsius. Results Boxes represent the median, the 25th and the 75th percentiles, bars indicate the range of data distribution. Circles represent outliers (values more than 1.5 box-length from the 75th/25th percentile). The asterisks represent extreme values (value more than three box-length from the 75th/25th percentile). Boxes represent the median, the 25th and the 75th percentiles, bars indicate the range of data distribution. Circles represent outliers (values more than 1.5 box-length from the 75th/25th percentile). The asterisks represent extreme values (value more than three box-length from the 75th/25th percentile). Analytical procedures CSF PTau231 levels were determined using Invitrogen ELISA kit. ELISA analysis was performed according to the Manufacturer’s protocol. Ptau231 concentrations were calculated on plate reader using curve-fitting software and 4-parameter algorithm. Plates were washed in an automatic washer. Protein concentrations were determined using the same algorithm in GraphPad Prism 5.0 demo version software (San Diego, CA, USA). Statistical analysis PTau231 levels among AD, MCI, and HC were compared using a Kruskal-Wallis test (χ2=17.317; df=2; p<0.001), followed by the MannWhitney U test for pairwise comparisons: AD vs MCI - (U=183; Z=-3.712; p<0.001) AD vs HC - (U=81; Z=-0.296; p=0.789) MCI vs HC - (U=38; Z=-2.806; p=0.004) Statistical analyses were performed with SPSS 19.0.1 (SPSS Inc., Chicago, IL, USA) and MedCalc 12.4.0.0 (Mariakerke, Belgium). P values less than 0.05 were considered statistically significant. cut-off (pTau231) = 1.904 U/ml cut-off (t-Tau) = 213 pg/ml 3 MCI patients (12%) had increased both tTau and pTau231, while 8 MCI patients (32%) had increased p-Tau231 only Acknowledgements The authors are grateful to Milica Trbojević-Čepe (University Hospital Center Zagreb) for her help in preparation of this report. All patients signed standard Patient Consent Form approved by the Ethical Committee of the University Hospital Center Zagreb (approval no. 01-20/53-1/2006 signed on 26th June 2006). This research was also approved by the central Ethical Committee of the University of Zagreb Medical School (case no. 380-59/11-500-77/90, class 641-01/11-02 signed on 19th May 2011). The work is funded by the Croatian Science Foundation grant no. 09/16 “Detection and tracking of biological markers for early therapeutic intervention in sporadic Alzheimer’s disease” to G.Š., and in part by NIH grant P50 AG005138 to P.R.H. The authors declare no conflict of interest.