Biol 155 Human Physiology
advertisement
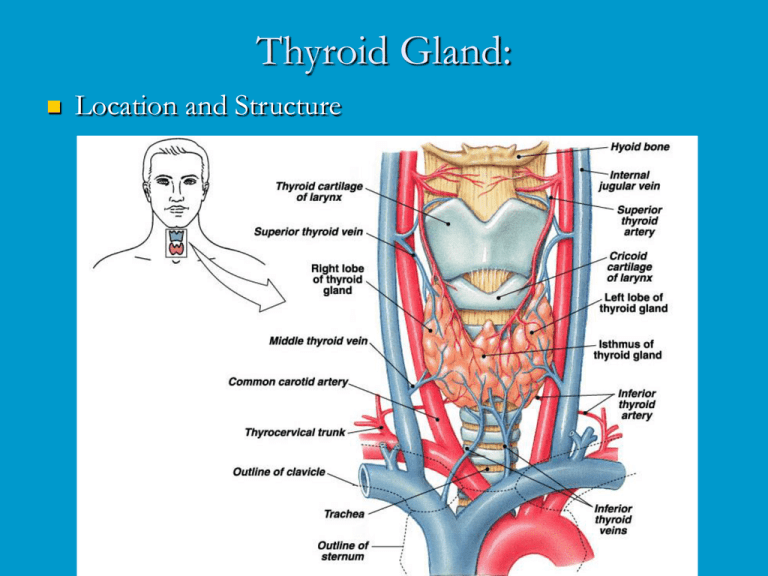
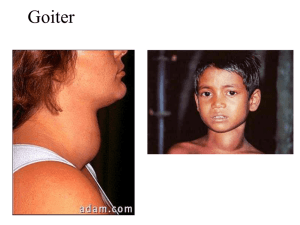
Thyroid Gland: Location and Structure The largest pure endocrine gland in the body, located in the front of the neck, on the trachea just below to the larynx. Its two lobes are connected by a median tissue mass called the isthmus. Internally, it is composed of about 1 million of round follicles. The walls of each follice are formed by cuboidal and squamous epithelial cells called follicle cells, which produce thyroglobulin (glycoprotein). The lumen of each follicle stores colloid, which consists primarily of molecules of thyroglobulin. The follicular epithelium also consists of parafollicular cells, a separate population of endocrine cells that produce calcitonin, a hormone involved in calcium homeostasis. Thyroid hormones (THs) The two THs contain iodine and are called thyroxin (or T4) and triiodothyronine or (T3). T4 and T3 have a very similar structure as each is made up of two tyrosine amino acids linked together and either 4 or 3 atoms of iodine, respectively. T4 is the main hormone produced by the thyroid and T3 has most if not all of biological activity as all target tissues rapidly convert T4 to T3. Except for the adult brain, spleen, testes, and the thyroid gland itself, THs affect all other types of cells in the body where they stimulate activity of enzymes especially those involved in glucose metabolism Increase metabolic rate in target tissues, which increases body heat production (calorigenic effect). THs also are critically important for normal growth and development of skeletal and nervous systems and maturation of reproductive system. Synthesis of thyroid hormones: Formation and storage of thyroglobulin. This process takes place in follicle cells and the final product is packed into vesicles, their contents are discharged into the lumen of the follicle and become a major part of the colloid. Iodide trapping and oxidation to iodine. To produce functional iodinated hormones, follicle cells accumulate iodide from the blood. A protein pump (iodide trap), located on the basal surface of follicle cells, actively transports iodide into follicle cells where it is oxidized and converted to iodine (I). Iodination. Once formed, iodine is attached to tyrosine amino acids which are part of the thyroglobulin. Iodination of one tyrosine produces monoiodotyrosine (MIT), iodination of two tyrosines diiodotyrosine (DIT). Coupling. Then enzymes within the colloid link MITs and DITs in a highly specific fashion, as a result two DITs linked together result in T4 , while coupling of MIT and DIT produce T3. Coupling (cont.) Interactions between two DITs are more frequent so more thyroxin. At this point both thyroid hormones are still attached to thyroglobulin molecules in the colloid. Colloid endocytosis. Colloid droplets containing iodinated thyroglobulin are taken up by follicle cells by endocytosis. These combine with lysosomes to form phagolysosomes. Cleavage of the hormones for release. Within the phagolysosomes, the hormones are cleaved from the thyroglobulin by lysosomal enzymes. The free hormones then diffuse through the basal membrane out of the follicle cell and into the blood stream. Transport and regulation of release: Most released T4 and T3 immediately bind to plasma proteins, of which the most important is thyroxinbinding globulin (TBG) produced by the liver. Binding proteins protect T4 and T3 from immediate degeneration by plasma enzymes, also they allow T4 and T3 to reach target tissues, often located a significant distance away from the thyroid gland. Decreasing blood levels of thyroxin trigger release of TSH from the anterior pituitary, which stimulates the thyroid gland to produce more thyroxin. Actual transmission EM: Pathology of the thyroid gland function: Both hypo- and hyperactivity and of the thyroid gland can cause severe metabolic disturbances. In adults, hypothyroidism is referred to as myxedema. Symptoms: Low metabolic rate, poor resistance to cold temperatures, constipation, dry skin (especially facial), puffy eyes, lethargy and mental sluggishness. If hypothyroidism results from lack of iodine the thyroid gland enlarges to form a goiter. Severe hypothyroidism during the fetal development and in infants is called cretinism. Symptoms: A short disproportionate body, a thick tongue and neck, and mental retardation. The condition is preventable by thyroid hormone replacement therapy. However, once developmental abnormalities and mental retardation appear, they are not reversible. Hyperthyroidism: The most common form of hyperthyroidism is Grave's disease, believed to be an autoimmune disease. The immune system produces antibodies that mimic TSH, which bind to TSH receptors and permanently switch them on, resulting in continuous release of thyroid hormones. Typical symptoms include metabolic rate, sweating, rapid and irregular heartbeat, nervousness, and weight loss despite adequate food intake. Often, exophthalmos, or protrusion of the eyeballs, occurs caused by the edema of tissues behind the eyes followed by fibrosis. Treatments include surgical removal of the thyroid gland (very difficult due to an extremely rich blood supply) or ingestion of radioactive iodine (131I), which selectively destroys the most active thyroid cells. Hyperthyroidism and Grave’s Disease Comparative aspects of thyroid function: Originally, it is thought that animals secreted iodinated proteins as a defense mechanism (panel A on fig. 8-3 Norris). Iodinated molecules were secreted from all over the body, including the mouth. Iodinated proteins secreted in the mouth were ingested and digested. The iodinated tyrosines and thyronines were absorbed. Eventually, the secretion of iodinated proteins was limited to the interior of the mouth (panel B). Iodinated proteins are found in a great number of species, including molluscs, arthropods and even some algae. Cells secreting the iodinated proteins started to collect together in the base of the mouth (panel C). This arrangement is seen in the amphioxus. Cells then congregated into open pits in the base of the mouth (panel D). Seen in the ammocetes larvae of the lamprey. Finally, these pits were internalized and the cells began secreting iodinated tyrosines and thyronines directly into the blood (panel D). This transformation is observed during the metamorphosis of the lamprey larva into the adult form. Pituitary control of thyroid function: This seems to have evolved much later on. There are no thyrotrophs in the agnathan pituitary. Thyrotrophs probably evolved later on, most likely from gonadothrophs. This is supported but the similarities in the hormone structures between LH, FSH and TSH. In elasmobranchs, thyrotrophs are located adjacent to the gonadotrophs. Structurally, thyroptophs and gonadotrophs resemble one-another closely. TSH, LH, and FSH all feed back negatively on both gonadotrophs and thyrotrophs in teleost fishes. This doesn’t happen in mammals. Teleostean gonadotropins have no effect on mammalian thyrotrophs. In non-mammalian vertebrates, there is a strong interaction between the PRL axis and the thyroid axis. PRL antagonizes the response to thyroid hormones in a number of species. In amphibians, TRH does not seem to regulate TSH secretion. Rather, it appears to stimulate PRL secretion. TRH levels are relatively high in the amphibian brain. Agnathan fishes: No true thyroid gland has been identified. However, there are follicle-like structures that concentrate iodine. Thyroid hormones have been detected. No definitive functions have been ascribed to the thyroid hormones. However, T4 levels have been shown to decrease sharply in lampreys when the free-swimming, filterfeeding larva metamorphoses into the parasitic adult. Iodinated glycoproteins have been suggested to play a similar role in metamorphosis in some inverts (e.g.. Cnidarians) Chondricthyan fishes: Very little is known about thyroid function in sharks and rays. However, they do have a well developed pituitary-thyroid axis. Distinct thyroid hormones are found. There are distinct thyroid cycles which coincide with the reproductive cycle. TH thought to be involved in promoting gonadal maturation. Thought to be involved in regulating metabolism during migration. In at least one species, increased TH induces migration. Elevated TH levels seem to induce elevated O2 consumption during embryological development, but the response is lost later in development. A number of inconclusive studies have attempted to show positive TH control of O2 consumption, but conclusive results have been elusive. However, another developmental role for TH has been identified. Development of neurosecretory activity in the hypothalamus is accelerated by treatment with T3 and T4 in the oviparous dogfish. Chondrostean fishes (sturgeons): Preliminary evidence suggests that circulating TH levels peak during spawning behaviour. These elevated levels may also be associated with migration. Injection of TH can reverse the gonadal degeneration seen in captive sturgeon. This suggests a direct link between thyroid activity and reproduction, but further studies need to be done. Teleost fishes: Thyroid tissue is found in diffusely distributed follicles, spread throughout the head region (fig. 8-6). TRH and somatostatin are found in the hypothalamus and appear to have the normal functional effects on thyroid hormone secretion. Exception is the salmonid fishes where TRH seems to function as an inhibitory neurohormone. T3 is the primary form that is secreted (unlike mammals). Thyroid hormone seems to be involved in salt water adaptation, regulation of migratory behaviour, and metamorphosis (in fish with a distinct larval stage). Sarcopterygean fishes: T3 is involved in survival through a drought. During aestivation, the metabolism all but stops and water is conserved. During water deprivation, T3 levels drop significantly. When water is provided, T3 levels rise and metabolic rate increases. Amphibians: Both T3 and T4 are present. Both are involved in regulation of reproduction and metamorphosis. Both regulate moulting and general growth. During metamorphosis, TH influences gene activity in the metamorphosing tissue. T3 stimulates apoptosis in the tails of anurans. T3 is also involved in modulating the water drive seen in newts. Thyroid function in reptiles: Some reptiles have been shown to have circulating MIT and DIT. Isolated thyroid sections secrete MIT and DIT in response to TSH in a turtle (Geochemys reevsii), a gecko (Geckko gecko), and a snake (Elapha rachiata). There are 5 major roles for thyroid hormones in the reptiles. Reproduction: Thyroid function is positively correlated with a number of reproductive events in lizards and turtles. Elevated thyroid activity (as assessed by histological evaluation) is associated with spermatogenesis, ovulation, and mating in a number of lizard species, as well as at least one turtle and one snake species. In the lizard Lacerta vivipara thyroidectomy causes premature ejection of most eggs and the remaining retained eggs fail to develop. However, there is no evidence that thyroid function is associated with gestation in live bearing lizards. Temperature selection: In snakes, thyroid function is greatest during warm periods and lowest during hibernation (as seen in the common Garter snake. In most temperate lizards, the relationship is the same, high in warm seasons and low during cold seasons. Artificially changing ambient temperature changes thyroid function. However, in a number of snakes and lizards, the highest level of thyroid function is associated with reproduction, not environmental temperature. In lizards from tropical climates, environmental temperature does not seem to have any effect on thyroid function. Injecting thyroid hormones has been shown to cause selection of a warmer temperature. Thyroid function and O2 consumption: This relationship is temperature-dependent. In animals tested, manipulation of thyroid hormones, TSH, or thyroidectomy have little effect on O2 consumption at 20oC. However, if temperature is raised to 30oC, then O2 consumption is increased by eloevated thyroid function. A similar effect is seen on reptile tissues in culture at 30oC. Moulting: Snakes and lizards respond in opposite ways. In lizards, moulting is stimulated by increased thyroid hormones and blocked by thyroidectomy. Administration of PRL potentiates the stimulatory effect of thyroid hormones. In snakes, the situation is different. Thyroidectomy increases the frequency of moulting and administration of thyroid hormones stops moulting completely. Growth: Some studies have suggested a possible link between thyroid hormones and growth, but very few studies have been done in this area. More studies need to be performed before a positive correlation can be established. Thyroid function in birds: The thyroid axis is well developed in birds and emerges early in embryonic development. For example, in domestic chickens, the thyroid responds to inject TRH by day 6 of development. Plasma levels of thyroid hormones are detectable by day 13 and levels rise to a maximum by day 20. Thyroid and reproduction. Domestic birds require thyroid for gonadal development. T4 is required for testicular development and thyroidectomy or administration of goiterogenic compounds will induce gonadal regression. However, in many wild species such treatment will induce a precocious gonadal development. Probably due to the breeding program for domesticated birds. Thermogenesis and O2 consumption: Thyroid hormones are directly involved in cold adaptation. As in mammals, thermogenesis is closely linked to O2 consumption. Some wild birds exhibit elevated thyroid function during late autumn and winter. Thyroidectomy of adult birds depress their ability to generate heat. Treatment with thyroid hormones stimulates O2 consumption Carbohydrate metabolism and growth. Thyroid hormone elevation reduces the glycogen stores in the liver, increases plasma free fatty acids and causes hyperglycemia. There is evidence to suggest that there is a significant interaction between thyroid hormones and GH. Release of GH is reduced when thyroid hormones are elevated. Thyroidectomy causes depression of growth in birds. Moulting Thyroids hormones produce stimulatory effects on skin and feathers, that are normally thought of as being associated with moulting. Moulting is usually regulated along with reproduction. Unlike lower vertebrates, thyroid hormones may act in a permissive role in mouloting. Migration: Migratory birds seem to have more active thyroid glands than non-migratory birds (within and between species). May be involved in inducing elevated metabolic rates associated with the strenuous act of migration.