Flo-=6;T*
I
Z2
i
!
a
THEN[tl!ODYl\lA[tl|IOS
HIPOLITO B. STA. MARIA
COIVTENTS
Preface vii
Chapter
1 Basic Principles, Concepts and Defrnitions I
Mass, Werght, Specilc Volume and Density; Spe-
- Weight, Pressule,
cific
Conservation of Mass.
2 Conservation of Energy Zg
Potential E_1ergy, Kiletic Energy, Internal Energy,
$eat, Work, Flow Work, Enthalpy, General EnergT
Equation.
3 , The Ideal Gas 87
Constant, Specific Heats of an tddal Gas.
4 Processes of Ideal Gas 5f
Isometric Process, Isobaric process, Isothermal
Process, Isentropic Process, polytropic do""sr.
5 Gas Cycles 81
Camot Cycle, Three-process Cycle.
6 Internal Combustion Engines gg
Otto Cycle, Diesel Cycle, Dual Combustion Cycle.
7 Gas Compressors ll5
Single-Stage Con pression, Twestage Compression,
Three-Stage Compression.
8 Brayton Cycle 16l
PREEACE
The purpose of this text is to present a simple yet rigorous
approach to the fundamentals of thermodynamics. The author
expects to help the engineering students in such a way that
learning would be easy and effective, and praetical enough for
workshop practice and understanding.
Chapters 1 and 2 present the development of the first la'ar
of thermodynamics, and energy analysis of ope:r systems
Jhapters 3 and 4 give a presentatign of equation of state and
;he process involvingideal gases. The second law of thermody-
namics andits applications to different thermodynamic cycles
are discussed in Chapters 5 and 6. Chapter ? deals with gas
compressors andits operation. Chapter 8 develops the Brayton
eycle which can be omitted if sufficient time is not available.
The author is grateful for the comments and suggestions
received from his colleagues at the University of Santo Tomas,
Faculty of Engineering.
The Author
vll
Basic Ppq"iples, Concepts
1
I and Definitions
Thermodynamics is that branch of the physical sciences
that treats of various phenomena of energ-Jr and the related
properties ofmatter, especially of the laws of transformation of
heat into other forrns of energy and vice versa.
Systems of Units
Newton's law states that 'the aceeleration of a particular
body is directly proportional to the resultantforce acting on
and inversely proportional to its mass.o
hE, F= D8,
"- m
k
it
k =+F
k is a proportionality constant
Systenns of units where k is unity but not dimensionless:
cgs system: I dyne forcre accelerates 1 g mass at
1 cm,/s2
mks system: 1 newton force accelerates I kg mass at
I m./sz
fps system: 1 lb force accelerates 1 slug mass at l Nsz
l--t;]*ldyne I t -* i*l newton [T,,'*-l-'r,0"
/777r/7mrV /7furm,h n77v77v?rrvr
1 cm./s2 _+
1m/s2
1&,/sz
t=r,4'cm-cyne.s"
o=t#;p
Systems of units where k is not unity:
k=rw
47
If the same word is used for both mass and force in a given
system, k is neither unity nor dimensionless.
1 Ib force acceierates a I lb mass at 32.L74 fVs2
1 g force accelerates a I g mass at 980.66 cm/s2
L kg force accelerates a 1 kg mass at 9.8066 m/s2
f-.,.-f*
,0, l- t ,. l-. t u
,
[-t u*. f-, nr'
d7mzm'V /72zv7m77 /7V7v77v77v7
r=f,a
1 poundal = (1 lb_) (1 fVs2)
F is force in poundals
# tr mass in pounds
a is acceleration in ftls2
32.L74 fVsz----+ 980.66 cm"/s2 -------> 9.8066 mlsz --'-+
k=
[T**
l*
/7V7V7mV ',0,
rz.tllthP k = e80.66-*F k = e.80668#
Relation between kilogram force (kgr) and Newton (N)
k=1k#
k = e.8066
Therefore, t
k# = e.8066 H#
L fVs2 --------+
m
l(
.U =r-8.
ks .m
Ets"
1 pound = (1 slug) (1 fvsz); 1 slug = 1 lb" s2
-lr-
F is force in pounds
is -ass in slugs
S
K
a is acceleration in fl;/s2
1kg"= 9.8066 N
Relation between pound psss (lb-) and slug
k=1#
Therefore,
k= 32.r74ffi
t*5& = 82.r74ffi
L slug = 32.L74Lb
A unit of force is one that produces unit acceleration in a
body of unit mass.
I poundal
:.._l
E
The mass of a body is the absolute quantity of matter in it.
The weight o,f a body means the force of gravity F, on the
lrody.
mFF"
k =t=gwhere g = acceleration produced by force F*
a = acceleration produced by another force F
Acceleration
I
I
Mass and lVeight
I fvs2 --)
AL or near the surface of the earth, k and g are numerically
,.r1rr:rl, so are m and F-
1(
Problcms:
lb .ft
--'l- J
ls-PI,l
S'=
0.4e tu.ll+se.o#-l = 222.26
=l
K
l.Whatistheweightofa66-kg-manatstandardcondi-
s Bo.b+
tion?
Solution
^"J
t*tfufl
I = 9.8066 m/s2
m=66k9-
'L
F"ok Fto.lF'
mo=-?-=
Bosg_.-
F "f*J
g,,,
= 1435.49 g-
? = (o ro,r"er fz.rt- U|nu-r rtrJ = 1459.41 g,"
2. The weight of an object is 50 lb. What is its mass at
standard condition?
Total mass = mr + m2 + na + m4 + m5
= 500 + 843.91 +222.26 + 1435.49 + 1459.41
= 446t.07 g^
Solution
r rb!rf-
FK
* =d-=
Fo
(b) Total mass = 446L.0J g^
g= 32.L74ftlsz
F, = 5o lbr
453.6
lb.rrl
fztz+14s'j
32.L74 ft
So lb_
(t') Total mass
-
= g.EB lb-
ils
9'83 ]!-o'
32.174;ifis
= 0.306 slug
P
3.Fivemassesinaregionwheretheaceelerationdueto
grr"itv i. 30. 5 fVs2 are as follo**t m, is- 500 g of masq rq, y^eighs
[oo eim, weighs 15 poundals; mo weight-g.lli mu is 0'10 slug
;i *]',r. trnuf iu theiotal mass expressed (a) in grams, 16) in
pounds, and (c) in slugs.
4. Note that the gravity acceleration at equatorial sea level
rr s = 32.088 fpsz and that its variation is - 0.003 fps2 per 1000
(a)
l't, :rscent. Find the height in miles above this point for which
llr:, gravity acceleration becomes 30.504 fps2, (b) the weight of
,r lsivcn man is decreased by \Vo. (c) What is the weight of a 180
I I r,,,
rn an atop the 29,131-ft, Mt. Everest in Tibet, relative to
por r r L'?
Solu,tion
g = (30.5 fVsz) (12 in/ft) (2.54 cm/in) = 929'64 cmls2
,\til tr tion
(;r ) change in acceleration = 30.504
(a) mz =
F't [roo4frro.uuM
=
e2e.64
4
this
+
= 843.91 g;
- 32.088 = * 1.584 fps2
p:; = 528,000 ft or 100 miles
llcight, h = - I lP* fps'
0.003
- -T0008
+T
(b) F = 0.9b Fg
-t
Specifrc Volume, Density and Specifrc Weight
Let Fg = weight of the man at sea level
.a
FF=
-ag
0.95 F" F"
a =g
The density p of any substance is its mass (not weight) per
unit volume.
____q
I
h
I
rl=D
rv
a = 0.959 = (0.95) (32.088) = 30.484 fps2
-L 'Fg
The specific volume v is the volume of a unit mass.
g = 32.088 fps2
" --
t.,
lt
(30.484 - 32'088) fps'z=
_ o.oosTS;r
b34,6z0 ft or tOt.B miles
-Tmorr
(c)
,vF
g=
a
8
Since the specific weight is to the local acceleration of
gravity as the density is to the standard acceleration,Tlg= pk,
conversion is easily made;
29.1.31 ft
Tk orY ='fr
os
P='g
r_.6 F8
g = 32.088 fps2
m = 1801b-
a = 32.088 fps' -
o =T-=
V1
mp
The specificweightTof any substance is the force of gravity
on unit volume.
F
ma
----
-1
rIto 1"1 {}l
fTdriil [0'003
At or near the surface of the earth, k and g are numerically
cqual, so are p and y
fpsz] = 32'001 fpsz
tlso lb-l pz.oor&l
#=179.03
32.174F"1T"
Problems
r _^ ^^ ,,
lbr
1. What is the specific weight of,water at standard condi.
tion?
Stilution
g = 9.8066 m/sz
*_pg
I- -
kg_
P = 1000 n5.
[*,SE**d
E- e.8066ffi#
kgF
= looo mo
ry
Pressure
2. Two Iiquids of different densities (p, = 1500 kg/m3,Pzi^
500 kg/m3) are poured together into a 100-L tank, frlling it' If
the resulting density of the mixture is 800 kg/mt, frnd the
respective quantities of liquids used. Also, find the weight of
the mixture; Iocal g = 9.675 mps2.
The standard reference atmospheric pressure is 760 mm
Hg or 29.92 in. Hg at 32"F, or 1"4.696 psia, or 1 atm.
Measuring Pressure
Solution
1. By using manometers
mass of mixture, mm = pmvm = (800 kg/m3) (0'100 m3) = 80 kg
I
(a) Absolute pressure is greater than atmospheric pressure.
mt+m2=mm
po
PrVt+PrV,=D-
q = 80
V, + V, = 0'100
1500 Vr + 500
p = absolute pressure
Po = atmospheric pressure
D
'lt p" = gage pressure, the pres'
sure due to the liquid
I
I
(r)
Q)
column h
solving equations (1) and (2) simultaneously
p = Po+Pg
Vt = 0'03 mg
(b) Absolute pressure is less than atmospheric pressure
Ve = 0'07 m3
m, = P,Vr = (1500 kg/m3) (0.03 m3) = 45kg
P=Po-P,
mr= prY2= (500 kglm3) (0.07 m3) = 35 kg
The gage reading is called
vacuum pressum or the vacuum.
weight of mixture,
re-=x"=@
e.8066*#
=?8.esksr
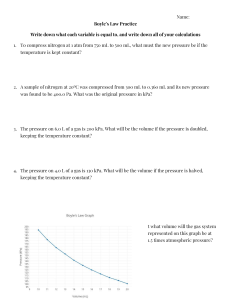
I ll"y using pressure gages
A Jrrt:ssure gage is a device for
rilr,,1||llr rt ng gage pressure,
'l'lrin picture shows the
rrr,vr.rn(.1)t, in one type ofpres!, I r r
. l::ll{(', k nown as the single-
I r r lrr. p1i r13.. 'l'hc f'luid enters the
lnlrr, llrrrrrrglr t,lrc thrcnded
,
',,rur.r'lrorr. A$ t.hc prOssur:e
Fig. 1 Pressure Gage
I
ry_
increases, the tube with an elliptical section tends to straighten,
the end that is nearest the linkage toward the right. The linkage causes the sector to rotate. The sector engages a small
pinion gear. The index hand moves with the pinion gear. The
whole mechanism is of course enclosed in a case, and a gpaduated dial, from which the pressure is read, and is placed under
the index hand.
Solution
["*S
pr=*#= FuuS
', kg-'4
' N.sz
(30 m)
= b48,680 N/mz or b43.6g pps(gage)
(p=po+p")
+Pt
Atmospheric Pressure
,=O,P=Po)
A barometer is used to measure atmospheric pressure.
-P, V
(p=p"-pr)
Absolutet Pressure
(p=0,Pr=P")
Gage Pressure
po
I
P=Po+Pg
--T--- Pr=*-A-=:6l
_ F" 1V yAh-
P.=Y\
ps
P, = Tb,
=ry'=*
Problem
A 30-m vertical column of fluid (density 1878 kg/ms) is
located where g = 9.65 mps2. Find the pressure at the base of the
column.
IO
Where ho = the height of column of liquid supportedby atmospheric pressure {
l)roblems
1. A vertical column of water will be supported to what
lrcight by standard atmospheric pressure.
Absolute Pressure
Solution
P=Th
At standard condition
yh"-* h = ho * hr, the height of column of liquid supported
-by absolute
pressure p.
\* = 62'4lblfts Po = 14'7 Psi
;-l
T ..-rr
lu.z
*l
lt++'#l
,t'= p,, - L----:n!-!_--!t"! = 33.9 ft
t;
If the liquid used in the barometer is mercury, the atmospheric pressure beconoes,
P" = THshs = (sp S)H, (T*) (h")
62.4Y
-'- ft3
trg.ol
Thespecificgravity(*pg')ofasubstanceistheratioofthe
spccifrc weight of the substance to that of water'
Fz.+
H rL'" i',1
1728H
^{
sps=T
po = 0.491 h" l4
2, The pressure of a boiler is 9.5 kg/cm2. The}arometric
pressure of the atmosphere is 768mm of Hg. Find the absolute
p".*r,r"* in the boiler. (ME Board Problem - Oct' 1987)
where ho = column of mercury in inches
then, ps = 0.491 n-
Solution
Pg = 9'5
kg/cm3
h
and, p =0.491 hP-=
ho = 768 mm Hg
ln."
At standard condition
l)roblems
T* = 1000 kdmt
l. A pressure gage regrsters 40 psig in a region where the
l,irrometer is 14.5 psia. Find the absolute pressure in psia, and
'rr kPa.
po = (ynr) (h") = (sp gr) nr(T*) (h")
(13.6)
Fooo
S to.?68 m)
c!*
10.000
'm'
_ 1.04 kg
cm-E
Srilution
p = 14.5 + 40 = 54.5 psia
= po * p, = 1.04 + 9.5 = 10.54#
t-t k-+'r newton [ , "[-ft, ,0,
/Tnvrnh
a = I m./sz
l2
/vTTvvmmiV
a=1fUs2
1T-t-
lkgn =
1+
Solution
E KgJ P.
= 0.06853 slug
(a)p = Po * Ps = 14.7 + 80 = 94.7 Psia
Pr=
= FS][tr'fl =8.28$
ao Ps]L
= S.A4atmospheres
t7 Psla
r,.
I':t. | --:af,m
F,lbf
h = 9.92 in. Hg abs
a = 3.28 Nsz
t = ff = (0.06863 slug) [.za {l= o.zzas tb,
lrg = 2o in.
P = 0.491 h
h"= Z9.tilt".
llth' -1f-
$..
p
=
(0.491) (9.92) = 4.87 psia
J
1 newton = 0.2248Ib"
p8 = 4.7 psi vacuum
1.1b" = 4.4484 newtons
(1rb)
rl4
ln' =
ps = (4.7 esi)
F**H lrr.ut;]
ln-
P = 10 psia
r
o"_l
l:8e5;-s!
=32,407 Ps(gage)
114= osgs\
mo
(rl)
h =15in.
h = 29.92 + 15 = 44.92 in. Hg abs
= 375,780 Pa or 375.78 kPa
2. Given the barometric pressure of L4.7 psia (2g.g2 in. Hg
abs), make these conversions:
(a) 80 psig to psia and to atmosphere,
(b) 20 in. Hg vacuum to in. Hg abg and to psia,
(c) 10 psia to psi vacuum and to Pa,
(d) 15 in. Hg gage to psia, to torrs, and to pa.
(1 atmosphere = 760 torrs)
t4
P, = 0'491 h,
=[r"H F"!F*'H
= 50,780 Pa(gage)
15
.lF
I'empcraturc
It follows that,
1. Derive th. r.l:rtion between degrees Fahrenheit and degrees Centigrndo. (FlE Board euestion)
1Fo=1Po
and
100"c
T212.F T
lc.-1K"
tl
*uu
*r".
1 ,r"" I0".
t.F -32 .=
_ t"C-0
212 -
n
2. Show that the specific heat ofa substance in Btu/(lb) (F")
is numerically equal to caV(g)(C").
Solution
lbb: o
I t"C + 32
toF =
r
Btu
(lb) (r")
o
toC =
5( t.F
I
- 32)
, Absolute temperature is the temperature measured from
absolute zero.
Absolute zero temperature is the temperature at which all
molecular motion ceases.
Absolute temperature will be denoted by T, thus
TbR = t.F + 460, degtees Rankine
TK=t"C+z71,Kelvin
Degrees Fahrenheit ("F) and degrees Centigrade ("C)
indicate temperature reading (t). Fahrenheit degrees
iFJ) and
Centigrade degress (C") indicate tempertu""
or differ"h"ogu
ence (At).
180 Fb = 100 C"
1p"-5g"
9
1 C. =!-1l,"
o
Btu - cal
-Ir-IEXD
=IG'(E
. Conservation of Mass
'l'lr. law of conservation of mass
,tr ttr.ltltl.e.
'l'lr,r. rluantity
states rhat mass is inde-
of fluid passing through a given section is
,'r\ r'n t)y fne lOfmUla
V=Au
-: VAu =Aup
III = i__
v v-
Wltcrc V = volume flow rate
A = cross sectional area ofthe stream
l) :, ilvcrage Speed
rir ,., m:rss llow rutc
16
t7
F7---
\t
I
I
4=ff =Erf,El a,E4zftz I
Applying the law of consewation of mass'
=' *T-
- - \- =-n;
T
2. A 10-ft diameter by 15-ft height vertical tank is receiv-
ArDrpr = \rtrPz
ing water (p = 62.1 lb/cu ft) at the rate of 300 gpm and is
discharging through a 6-in ID line with a constant speed of 5
I
I
I
Problems
1. Two gaseous stre?ms enter a combining tube and leave
section:
single mi*trrr". These data apply at the entrance
as a
-fot
6rr" gur, A'r= 75 in,z, o, = 590 fps,-vt] 10 ft3llb
For the other gas, A, = 59^i1''.:T, = 16'67 }b/s
-
:j:rlil"ffJrr;,'frh'iisfilTil;1lo' I
P" = 0.12lb/ftg
At exit, u.. j 350 fPs, v, = 7 ftaAb'
Find (a) the speed u, at section 2, i- 'd
ft) the flow anii area at the exit section'
Solution
\
I
rs,
f___ _ _]=
t__
I l=:-:_-_*--l -l-,
I F'--=- -:-1J tiu'
e""" =-f, (10)2 = 78.54 ftz
tu'",=il'i,=ffi
=4oorps
r\lirrur lr,,w rate enreri", =
(b)
.
Aru,
-[.'9!d=2604+
mr = --vr = --------r6Tt3-
ib
rh, = rh, + rh, = 26.04+ 16'6? = 42'?1+
18
=
[rr fi
[ffi]
r
r\t,r'* tuwrateleavins=Aup=
z4so.\
? Bd'F.uo*J F +
= ru*
S*
Mass change = (3658 - 2490.6) (15) = 17,511 lb
volume ch^nge =
17'51-l:-!b
62.1#
Decrcased in height =
ffi#
= 282 ft'
= 3'59 ft
Water level after 15 min. = 7.5 - 3'59 = 3'91 ft
(decreased)
Review Problems
1. What is the mass in grams and the weight in dynes and
in gram-force of 12 oz of salt? Local gis 9.65 m/s2 1 lb- = 16 oz.
Ans. 340.2 g-;328,300 dynes; 334.8 g,
2. A mass of 0"10 slug in space is subjected to an external
vertical force of4 lb. Ifthe local gravity acceleration is g = 30.5
fps2 andiffriction effects are neglected, determine the acceleration of the mass if the external vertical force is acting (a)
upward and (b) downward
Ans. (a) 9.5 fps2; (b) 70.5 fps'?
3. The mass of a given airplane at sea level (g = 32.1 fps2)
is 10 tons. Find its mass in lb, slugs, and kg and its (gravital.ional) weight in lb when it is travelling at a 50,000-ft elevation.
'l'he acceleration of gravity g decreases by 3.33 x 10-6 fpsz for
r,rrch foot of elevation.
Ans. 20,0001b-; 627.62 slugs; 19,850lbr
4. A lunar excursion module (LEM) weights 150[r kg, on
r.rrrth where g = 9.75 mps2. What will be its weight on the
rrrrrface of the moon where B. = 1.70 mpsz. On the surface of the
,noon, what will be the force in kg, and in newtons required to
',,'ttlerate the module at 10 mps2?
Ans. 261.5 kg; 1538.5 kgr; 15,087 N
,l-r. The mass of a fluid systenis 0.311 slug, its density is 30
ll,/l'1,:r and g is 31.90 fpsz. Find (a) the specific volume, (b) the
(c) the total volume.
"1,,'r'ific weight, and
Ans. (a) 0.0333 ft3Ab; (b) 29.75 lb/ft3; (c) 0.3335 ft3
{;. A cylindrical drum (2-ft diameter, 3-ft height) is filled
*'rllr :r tluid whose density is 40lb/ft3. Determine (a) the total
,,,lrrrno of fluid, (b) its total mass in pounds and slugs, (c) its
,'1r'r'rlit: volume, and(d) its specific weight where g = 31.90 fps2.
Ans. (a) 9.43 ft'; (b) 377.21b; 11.72 slugs; (c) 0.025 ft3l
lb; (d) 39.661b/ft3.
'i A wuathcrman carried an aneroid barometer from the
! r "t, ir l llrxrr to tris ofl'icc atop the Sears Towcr in Chicago. On
20
2l
the ground level, the barometer read 30.150 in. F,Ig absolute;
topside it read 28.607 in. Hg absolute. Assume that the average
atmosphdric air density was 0.075 lb/ft3 and estimate the
height of the building.
Ans. 1455 ft
8. A vacuum gauge mounted on a condenser reads 0.66 m
Hg.What is the absolute pressure in the condenser in kPa when
the atmospheric pressure is 101.3 kPa?
Ans. 13.28 kPa
9. Convert the following readings of pressure to kPa absolute, assuming that the barometer reads 760 mm ltrg: (a) 90 cm
Hg gage; (b) 40 cm Hgvacuum; (c) 100 psiS; (d) 8 in. Hg vpcuum,
and (e) 76 in. Hg gage.
Ans. (a) 221..24 kPa; (b) 48 kPa; (c) ?90.83 kPa; (d)
74.219 kPa; (e) 358.591 kPa
10. A fluid moves in a steady flow manner between two
sections in a flow line. At section 1:A, =10 fLz,Dr= 100 fpm, v,
= 4 ft3/lb. At section 2: Ar- 2ft2, pz = 0.201b/f13. Calculate (a)
the mass flow'rate and (b) the speed at section 2.
Ans. (a) 15,000lb/h; (b) 10.42 fps
Consenration of Energy
Gravitational Potential Energy (P)
The gravitational potential energ:y of a body is its energy
due to its position or elevation.
p=Fsz=ry
AP
=
P, - P, =
ff@r- zr)
AP = change in potential energy
Datum.plane
11. If a pump discharges 75 gpm of water whose specifrc
weiglit is 61.5 lb/ft3 (g = 31.95 fpsz), frnd (a) the mass flow rate
in lb/min, and (b) and total time required to fill a vertical
cylinder tank 10 ft, in diameter and 12 ft high.
Ans. (a) 621.2lblmin, (b) 93.97 min
Kinetic EnergT (K)
The energy or stored capacity for performing work pos'
Hrls$ed by a moving body, by virtue of its momentum is called
kinetic energy.
K=#
nK=4-K,=fttoi-ui)
AK = change in kinetic energy
22
23
qT
Internal EnergY (U' u)
Flow lVork (Wr)
Internal energy is energy stored within a body or substance
by virtue of the r"ti.rity an-cl configuration of its molecules and
ol thu vibration of the atoms within the molecules'
Flow work or flow energry is work done in pushing a fluid
across a boundary, usually into or out of uy*L-.
u = speci{ic internal energy (unit mass) Au = tlz - ul
"
13orr nrll
lr'_
lVr=Fi=pAL
;1=Area of Sur.face
Wr=PV
fJ = mu = total internal energy (m mass) AU = Uz - Ur
Work (W)
l"ig. 3
FIow Worh"
work is the product of the displacement of the body and the
component of the force in the direction of the displacement.
w,r.k is energy in transition; that is, it exists only when a force
is "moving through a distance."
Work of a Nonflow SYstem
Cylinder ---.
The work done as the
piston moves from e to f is
Final Position of Piston
dW=F,d*=(pA)dL-pdv
Piston
At ea = .zl
I
'"**F
which is the area under the
curve e-f on the pV plane.
Therefore, the total work
done as the pistonmoves from
lto2is
AW, = change in llow work
Ideat (e)
lleal is energ'y in transit (on the move) from one booy or
'::1"11.1'ry1 to another solely because of a temperature difference
I'r'l wr:err the bodies or
nV
Fig. 2
which is the area under the
curve 1-e-f-2.
woRK ot EXPANSIoN.
The area und.er the curue of the prrcess on the pV plnne
rcpresents the work d'one during a nonflow reuersible process.
Work done by the system is positive (outflow of energy)
Work dnne on the system is negatiue (inflow of energy)
24
systems"
u{-_.
,{,.-.
t) is poslfiue when heat is added to the body or system.
(l is negatiue when heat is rejected by the body or system.
Classificati.on of Systems
r I t A r'lrr.se d' system is one in which
'
w =Jlndv
AW,=Wr,-Wrr=pr%-FrV,
mass does not cross its
l,or r ntlaries.
' r .\ r | (
system is one in which mass
'r,t'n
crosses its bounda-
Cnnservation of Energy
|1,, l.riv ol r:orrservation of energy states Lhat energy ls
:. r.ti, I r r'rtlr.tl ttttt't/t,St,nlyeCli l,, f u:,1 l;rw ol'l.lrr:r'modynarnics states that one
fornt oI
:::i:':. , !tttt \. ltt. (..,ttIt('t.l((1. i.n.l.O U.nOthCf.
SteadY Flow EnergY Equation
of steady flow system'
Characteristics
-
i. There is neither accumulation nor diminution of mass
within the sYstem'
2. There is neitier accumulation nor diminution of energy
within the sYstem
3. The state of"the working substance at any point'in the
system remains constant'
Problems
t. During a steady flow process, the pressure of the working substance drops from 200 to 20 psia, the speed incneases
from 200 to 1000 fps, the internal energy ofthe opeh system de.
creases 25 Btu/lb, and the specific volume increases ftom I to
8 ftsnb. No heat is transferred. Sketch an energy diagram.
Determine the work per lb. Is it done on or by the substance?
Determine the work in hp for 10lb per *io. (t hp = 42.4Btu/
min).
Solution
peia p, = 20 psia
o, = 200 fps
rlr = 1000 fps
vc=8 ffnb
n vr=lfts/lb
pr = 200
Kl
Fig.
4 Energy Diagram of a Steady Flow System
Energy Entering System = Energy Leaving System
P, + K, + Wr, + U, + Q = Pa* t-l Wl"+ U" + W
d=l"P+ak+l-wr+aU+W
(SteadY Flow Energy Equation)
W,,
II,
2
Au=-25Btu/lb Q=0
Energy Diagtam
,F, + K, + W' + U, + A,=Pr+
4 + W* + U, + W
llrrnis I lb2
lr"3 ]
EnthalPY (H, h)
fluids
Enthalpy is a composite property applicable to all
and is defined bY
h=u+pv and H=mh=U+PV
The steady flow energy equation becomes
+K'+H'+Q-l;..?J*ril*
ffL
fi, ,lf = Offiimi=le.e?r+b
W,, l',v,
llr V.l -*
26
= o.8o
E*'ii,lE-Hl
= sz,o2
Bfi
(20) (r44) (8)
= 2e.6rff
778
27
-T'r-(a) Basis f lb'?n'
Kr+Wrr=Iq+W,r+Au+W
0.8 + 3?.02 = 19.9? + 29.61 -25 +
w = 13.24
K,=S= ,Cffio,, =3'20ff!
W
(1100)2 = Z+.t7 BJu
,q =*= (z',) (32.174)
(778)
ff,0t,
rb-
t-
w:
lr s24ffi["*il
L-
Wr, = PrVr =
= 3,12 hp
(200) (144) (2.65)
779
--'-- #E
= 98.lC lb_
42.4(mi#)hp)
wrz= PzYz=A+#@=s+'z+ff
turbine bt 200
2. Steam is supplied to afully loaded 100-hp
ftsAb and u'.=^19'0 fp*'
priu *itft
= 116'bT nl"/lb,"t, ::'1U
"r at r prl" *ilrt * J ozs Btunb, Y,=-29! ft3Ab and
is
Exhaust
-=
turbin is L0
rioo fps. tne heat loss from the steam in the
"glJu. il;;ipor""tiur enersy
change and determine (a) the
K, + Wr, + ur + Q- IL + Wo + u, + W
;t.20+ 98.10 + 1163.3 + (-10) =24.L7 + 54.42 + 925 + W
Fl{
w= 251ff
*o"t p"" tU steam and (b) the steam flnw rate in lb/h'
Solution
L163.3 Btunb v, = 2'65 ftsnb
p, = 200 psia
u, =
p, - l Psia
u" = 925 Btunb
vr= 294 fts/l.b
u, = 400 fPs
u, = 1100 fps
Q = -10 Btu/lb
(b) Steam flow =
(roo hp)
r
251 Btu
---
Eru-l
P544lrr) trro)
E;
r
= 1014
+
:t. An air compressor (an open system ) receives 272kgper
rr r r
l of air at 99.29 kPa and a specific volume of 0.026 m3/kg. The
nr r" llrws steady through the compressor and is discharged at
W=t00hp
frrllf l-r kPa and 0.0051 mslkg"The initial internal enerry of the
,r r rrr | 594 Jlkg; at discharge, the internal energy is 6241ilkg.
'l'lrr.<'rxrling water circulated around the cylindercanffis away
.l:ul:t .f/kg of air. Thc change in kinetic energ"y is 896 J&g
rr{ n.nso. Sketch an enerry diagram. Compute the work.
/r+Kr+ Wr, + Ur + Q=/r+ Iq + Wo + U, + W
2B
29
Solution
P, = 99.29 kPa
v, = 0.026 m3/kg
u, = L594 J/kg
Q = -4383 Jlkg
h = 272 kg/min
Pz = 689.5 kPa
vz = 0.0051 m3/hg
uz= 6241J/kg
AK = 896 J&g
r4
wo
u2
Solution
fr = 2270 k'elmin
= 0.1524 m
Pr = 82,740Pa
p
1000 kg/mg
q == 0.1016 m
275,800 Pa
Pz r
dr
C
1
EnergY Diagrom
y'r*Kr+ W., + U, + Q=/r+ 4 + Wo + U, + W
Basis 1kB-
Area at entrance, A, =
t (0.1524F = 0.01824 mz
Area at exit, Ao =ftO.rOro)2, = 0.00810? mg
IIPe
,![I F
W,, :p lvr = €9.29'm1.l
).026
'l; 2.583 kJ&e
ol =
KS
b-l l=
mil
kl\i r
,Ia- = 3.51.6lnl/kg
t0.00
005 ,0il
wn =pPzv
68e.
I
'o
mz
'zYz=
m
L
t_
F
t-
lI
2270k9^
'J
- U*
Pr-l= #
H1r,r,if at entrance, Dr =
;1
u2-+ w
+Q= AK+'wlzf2z* uz
vflr
\w.
t*1 -1 +G
ilgxrcd at exit, D, =
q4--4. 383 = 0.896i+333.516
;16-+ 6.2,
6.24 1+W
2.582 + 1. 594
'
[oootrl P'0t824 {
m
m
2270160
(1ooo) (o.oo81o7)
=2.074m1s
= 4.667 mls
llnHis 1 kg-
\{ = - 10.g6H
t- kr-l l- _ ke_l
w - j_- to.se6gJ
K, =;ik=
Vzztry)
4. A centifugal pump operating under steady flow condi'
E
I
II
IL
tions delive rs 2,270 t glmin of water from an initial pressure of
82,740Patoa final p"essore of 2?5,800 Pa. The diameter of the
inlet pipe to the pump is .15.24 cm and the diameter of the
ilischaree prpe is 10.16 cm. What is the work?
30
Fffi
N.m
= 2.151q;
(4.667Y
K
=D? = (zxit= to.gg T.'" -DEks-
\[I = - 2954*
i
Q.orni]'
w
-l.
t21o*'
,,
., =E =;;El)'vr
82.24+,.rts
= oL''*
kgm
3l
Basis 1kg,
Pr=?= fs.eooof'(B
[-,'"ffiE*H
.4_
5. Aturbine operates under steadyflow conditions, recei
iag steqm at the following state: pnessure 1200 kPa,
tue 188"C, enthalpy 2785kJ/kg, speed 33.3 m/s and elevati
3 m. The steam leaves the turbine at the following
pressure 20 kPa, enthalpy 25L2 klkg, speed 100 m/s
elevation 0 m. Heat is lost to the surioundings at the rate of 0.
hVs. If, the rate of steam flow throughthe turbine is 0.42
what is the power output of the turbine in kW?
zr=3m
zz= 0m
ks
2
P
= 0.55a4 K
s
q=;i =1#f =,hIs.o00E
a-
-o.zey
;F-
= {).6eo5H
Pr+Kr+hr+Q=%+4+ L+W
Pr+Kr+hr+Q=4+\+W
0,0!fg4 + 0.5544 + 2785+ ({.690b) = b.000 + 2bt2 + W
W = ZG?.gg
*_
k;
roT.eEl 19.42fl
W = 112.52 kW
E
IKg
4=2512H
ur=33'3fl
u, = 100*
'
4
w=, l:^- ^ hrl I-
Solution
h. = 2?85
fm_l
Kl= m- L33.3g:l
kI
W = -4b8.1ffn
= 0.0294
.TT.F
'E-E
Kr+Wrr=Iq+\{Io+W
2.L5L + 82.74 = 10.89.+ 275.8 + W
m)
'
l&I
Q = -O.29 s
32
fi = 0:4#
88
T
Review Problems
1. Assuming that there are no heat effects and no fric'
tionaleffects,nnatnekineticenerg]andspeedofaS220.lb
starr wfth the steady flow
;;d**; iiiar, 778 ft,from rest.which
are inelevant'
deleting energy terms
a;;til,
fPs l
. - ? ,:"?l:..
Ans. 224
The discharge conditions are 0.62 ms/kg,-100 kpa, and 270
m/s. The total heat loss between the inlet and discharge ie g
kJlkg of fluid. In flowing through this apparatus, does the
specific internal energy increase or decrease, and by how
rnuch?
' 2. A reciproc"ti"e di"pressor draws in 500 cubic feet per
it
mir'rte of air whose density is 0.0?9 lb/cu ft and discharges
*iiit au"sity of 0.304lUcu ft' At the suction' p, = LS.psia; at
"
in the specific-internal
ait"ftt"g",
Pz = 80 psia' The increase
the air by
enerm/ is gAS Btudb anrl the heat transferred from
in
ri et"nU. Determine the work on lhe air Btu/min
u"a irittp. Neglect change in kinetic energy'
Ans. 56.25 hP
;
;ft
In a steady flow apparatus, L3b lc.I of work is done by
-6..
each kgof fluid. The specific volume of the fluid, p""*s.r"*, und
speed at the inlet are 0.37 mslkg, G00 kpa, and 16 m/s. The inlet
is 32 m above the floor, and the discharge pipe is at floor level.
Ans. -20.01 kJ/kg
7. Steam enters a turbine stage with an enthalpy of 862g
k.l/hg at 70 m/s and leaves the same stage with an entharpy of
:ltl46 kr&g and a velocity of L2a n/s. calculate the work done
l,y the steam.
Ans. 776.8 kJ&e (ME Board Problem - Oct. 1996)
3. Steem enters a turbine with an,enthalpy of 1292B,h,1|b
an enrhalpy of 1098 Btu/tb. The transferred
hp for a
heat is 13 Btu/lb. what is the work in Btrlmin and in
flow of 2 lb/sec?
Ans. 512.3 hP
*dl;;;;;h
4. A thermodynamic steady flow system receives^4'56
n"ii where n1 1JBQ0 T?.Y'= 0'0ll-8:-]1
p"" Li"
"i"aod ,r, = 17.16 k nte' The fluid leaves the sys
i.-= tii J",
ui u t"""aary wheie Pz = 551'6 kPa, v, = 0'193 m3/kg' o, =
DurFs pasiage through tbe sv
%. ="sz.eo uttfite
inu nnid receives 3,000 J/s of heat. Determine the work'
Ans. -486 kJ/min
;;ffi
5. Air flows steadily at the rate of 0'5 kg/s through qn
compressor, entering at 7 mls speed, 100 kPa pressure
0.95 m3/kg specific volume, and leaving at 5 m/s, 700 kPa,
0.1"9 m34rg. The internal energy of the air leaving is 90
greater t[an that of the air entering. Cooling water in
io*p""rror jackets absorbs heat from the air at the rate of
kW. Compute the work in kW.
Ans. -122 kW
l
it4
lfl-r
3 The rdeal Gas
An ideal,gas is ideal ronly in the sense that it conforns rc
llrc simple perfect gas laws.
Boyle's Law
lf' the temperature of a given quantity of gas is held
,,rr'l,irnt, the volume of the gas varies inversely with the
rrl*rolute pressure during a change of state.
l or V=9
V* pp
pV=C or prV, =prYz
Charles'Law
r I r lf' thc pressure on a particular quantity of gas is held
,,,*irt;rrrl., t,hon, with any change of state, the volume will vary
rlirr.r tly :rrr lhc absolute temperature.
V,."1
or
V=CT
v (:
L-IL
' or q=q
'r'
r,:r ll tlrr.volurnc of a particular quantity of gas is held
will vary
,f i* e' | !r' 1ri lli,' lrllsll utC te mpe ratUfe.
, r,1 1e | ;1 1, l . | | rr. r r, wi th nny change of state, the pressure
,tt
-7
P-T
or
or
fr=c
used, the pressure was 200 psia and the temperature was 85oF,
P=CT
(a) What proportion of the acetylene was used? (b) What
volume would the used acetylene occufiy at L4.7 psia and fl0'F?
R for acetylene is 59.35 ft.lb/lb."R.
t=+,
Equation of State or Characteristic Equation of a
Perfect Gas
Be = rllass of acetylene used
Tr =90oF+460=550'R
Pz = 200 Psia
Tz =85oF+460=5451R
pV
T =mR
volume of dr,r* =
pV = mRT
ml= PrV, =
pv =RT
(unit mass)
m
T
R
English units
ffiffi
V
T
ft3
lb_
oR
m3
kg
K
R
= 0.6545 cu ft
(25cD $44) (0.6545)
RT,
= absolute pressure
= volume
= specific volume
= maSS
= absolute temperature
= specific gas constant or simply gas constant
}F
frr = rlrBss of acetylene initialiy in the drum
Pr = 250 Psia
+=ry =c,aconstant
V
v
(a) Let
oz = ltrass of acetylene left in the drum
Combining Boyle's and Charles' Iawg,
where p
Solution
(59.35) (550)
= 0.7218Ib
o-E= (200,)=911)!9.6j45)
lb
"'""-- -(54b) = 0.b828
mz =
ifq'=
ms -
ml mz= 0.72L8 - 0.5828 = 0.1390Ib
Acetylene used =
(bgsb)
#i = 3+# = 0'1e26 or re'26vo
tlr) p, = 14.7 psia
'f.=80oF+460=540oR
i
SI units
E
N
;t
Problems
1. A drum 6 in. in diameter and 40 in. long
acetylene at250 psia and 90"F. After some ofthe acetylene
I
-t
3rl
Vr=
m EltTr
Pa
(b=e.Bit t5+01
\roils$l
' (r4.7)
= 2.'0b fr3
(L44\
:l 'l'lrc volume of a 6 x 12-ft tank is 339.3 cu ft. It contains
sir rrl '.1(X) psig and 85"F. How many l-cu ft drums can bc fillcd
l' rru 1rrr1.f :rnd 80'F if it is assumed that the air temperasturtt
irr llrr' lrrrrh remains at 85"F? The drurns have been silting
€*,iurrl rrr l.hu atmosphere which is at 14.7 psia anrl [t0"1"
;t 1)
r
Solution
Solution
Let
Dr = IIlBss of air initially in the tank
Dz = rnoss of air lelt in the tank
Ds = mas$ of air initially in the dmm
ha = rnsss of air in the drum after filling
20,000 kg
l
I
Pz = 50 + 14.7 = 64.7 psia
EH" = mass of Helium
v = volume of the balloon
+
ms
(2L4.7)(r44) (33J.3)
P,Vr
_*-(SmtGaSI
= 360.9Ib.
RT, =
RT, -=
-z= R"S
IDo
(64;3),(l*t)=(?gie'3)
(53.34) (545t-
mass of air that can be used = 860.9
p.v.
(r44) (1)
'GBiJAIGadf
'ff (t4.7)
=
Itor the air
R = 287.08
= 108.? lb
- 10g.? = 252.2Ib.
p-V
101.32bV
'nu=ili" = tffirl
=l'2oolvkg
f','t lltt'heliUm
= o'0735 lb
mass of air put in each drum = 0.323b
"p=
E__
P, = 101,325 Pa
&r" = 2,077.67
2#
- 0.0?gb = 0.25Ib
= 1009
3. It is planned to lift and move logs from almost inaccessible forest qery by means of balloons. Helium at atmospheric
pressure (101-.325 kPa) and temperature 21.1oC is to be used
in the balloons. What 6inims6 balloon diameter (assumo
spherical shape) will be required for a gross lifting force of 20
metric tons?
#R
P11" = 101,325Pa
"'o= Sf =ttf#[]l*}$i =o'3235rb
Numberof drums filled
J
T,=21.t +273=294.iK
For the drums
40
= "mass of air displaced by
the balloon
EH.
For the tank
m3 =
mr
Po = 50 + 14.7 = 64.7 psia
Tn=80+460=540R
Tr=8S+460=545oR
[l=
I€t
I
psia p, = 14.? psia
545.R
T, = 80 + aOO = b40R
Pr = 200 + 14.7 = 214.7
Tr = 85 + 460 =
T""=21.1 +278=Zg4.lK
,,,
_
rrrrl,,=
Pn.v = _101,325 V
ffiT""
qOngZffim =0.1658Vkg
fr,=DH,+20,000
V =0.1658V +,20,000
V = l9,BB7 mJ
.l rf = 19,337
.l
1.200f
1
r = 16.6b m
d - 2(16.65) = 3B.B m
4l
G{
4. TVo vessels A and B of different sizes are connected by
a pipe with a valve. Vessel A contains L42L of air at2,767.92
solving equations L and 2 simultaneously
kPa, 93.33oC. Vessel B, of unknown volume, contains air at
68.95 kPa,4.44"C. The valve is opened and, when the prcperties have been determined, it is found that p- = 1378.96 kPa,
t- = 43.33'C. What is the volume of vessel B?
Vs = 110.4 liters
Specifrc Heat
specific heat of a substance is defined as the quantity
_ _The
of heat required to change the temperature of unit mase
through one degree.
In dimensional form,
Solution
For vessel A
c__*
Po= 2,767.92 kPa
In differential quantities,
Yn= L4?liters
TA = 93'33 + 273= 366'33 K
c^ or dQ=mcdT
e= ;ffif
For vessel B
nrr<l for a particular masg m,
Ps = 68'95 kPa
a=* !'.ar
I
TB =4.44+273=277.44K
(The specific heat equation)
For the mixture
ll llrr: mean or instantaneous value of specific heat is used,
P- = 1378.96 kPa
Q = mc
T- = 43.33 + 273 = 316.33 K
(constant specific heat)
III,,,=IIIO*IIIU
p-v*
* bY!
RT_ RTn RTu
(13?8.e6)V
^ (2767.s2) (yLD , 68.e5 VB
p^V^
4.36 V- = 1072.9 + 0.25 Vu
V-=142+Vn
= mc (T, - T,)
!'u,
l-
(1)
I'orrnltnt Volume Specifrc Heat (c,)
^uI
Volume
(
lorrstant
I
Q"=aU
I
Qu = mcu (T2
I
(2)
- Tr)
, ---l
a,
42
4:l
-y'r
Relation Between cn and c,
Constant Pressure Specifrc Heat (co)
Qn
mco (T, -Tr)
Qn
AU+W=AU+
Fromh =u+pvandpv=RT
dh = d11+ RdT
al
pdv
-l\
codT = c"dT+RdT
co -c,+R
= AU+p(%-Vr)
= Ur-ur+pz%-prV,
Q, = I{-H'=AH
Qn
g
c" =Eh
-B
^'p -k-l
Ratio of Specific lleats
lfroblems
1. For a certain ideal gas R = 2b.8 {t.lb b..R and k - f.09
(r) What are the values of co and c,? (b) What
mass of this gag
worrld occupy a volume of l5 cu ft dt ZS psia and gO"F? (c) lfgO
lll.rr are transferred to this gas at constant volume in (b), what
nrr. the resulting temperatur,e and pressure?
c
k=d:>r
Internal Energy of an Ideal Gas
Joule's law states that "the change of internal energy of an
ideal gas is a function of only the temperature change." There.
fore, AU is given by the formula,
Htilution
AIJ = rtrc" (T2 _ Tr)
""',, =
whether the volume remains constant or not.
., t
Eiil
ll,r V
tIt
44
lScuft, p=75psia T=80+460=b40o3
pV _ (75)
(1114) (rb)
-6ffi
-= ffi=
=11'631b
r
whether the pressure remains constant or not.
oro.aotffi
=
AH = ECo (Tz - T1)
a
= su.4z**"
,. -% = T3# 0.868#"
Enthalpy of an Ideal Gas
The change of enthalpy of an ideal gas is given by
formula,
* = #ig
r I tf
'ilr
n,c" (T, _ Tr)
I t.63 (0.3685) (T, _ 540)
4{t
E T
where:dQ = heat transferred at the temperature T
AS = total change ofentropy
Tz = 547"R
Pz = Pr (Tuftr) = 75
(5471540) = ?6 Psia
2. For a certain gas R =320 Jll<g. K and c, = 0.84 kJlkg. K"
(a) Find co and k. (b) If 5 kg of this gas u4dergo a reversible non
flow oonstant pressure process from V, = 1.133 m3 and Pr = 690
kPa to a etate where tc = 555"C, find AU and AH.
as--fu
as =
-lftl ; mc hr _&
T1
(constant specific heat)
Solutlon
(a) cp = c" + R = 0.84 + 0.32 = 1.16
kI
IFF
'l'emperature-Entropy Coordinates
dQ = TdS
k= &+1= f# + t ='t.3st
cY
a2
Q = jTds
(b)r-
I
pr[.
(6901909[!.133)
= 488.6 K
=
=
'r mR
(5) (320)
'The area under the curve
ofthe process on the TS plane
represents the quantity of
heat transfered during the
AU = rnc, (T, - T1) = 5 (0.84) (828 - 488.6)
= 1425.51r.I
process."
AH = trrcn (Ts - T1) = 5(1.16) (828 - 488.6)
= 1968.5 k I
I lt lrr.r Enerry Relations
Entnopy (S, s)
Entropy is that property of a substance which
constant if no heat enters or leaves the substance, while it
work or alters its volume, but which increases or dimini
should a small amount of heat enter or leave.
The change of entropy of a substance receiving (or deli
ing) heatis defined by
dS=
46
F
-2
or As =JF
I
12
-)VdP=W+AK
I
(Reversiblesteadyflow,AP= 0)
"The area behind the
curve ofthe process on the pV
planes represents the work
ofa steady flow process when
AK * 0, or it represents AK
when W' = 0."
47
-{
Any process that can be made to go in the reverse direction
by aninfinitesimal change in the conditions is called a nrersible
process.
Any process that is not reversible is irreversible.
Review Problems
1. An automobile tire is inflated to g2 psig pressurs at
60"F. Alter being driven the temperature rise to
zb"F. Determine the final gage pressure assuming the volume remaina
constant.
Ans. 84.29 psig (EE Board problem)
2. If 100 fts ofatJnospheric air at zero Fahrenheit tenperacompressed to a volume of 1 fts at a temperaiuoe or
lrlrj""
?00oF, what will be the pressure of the air in psi? Ans. 2109 psia (EE Board problem)
3. A 10-ft3 tank co-ntains gas at a pressure of b00 psia,
l.rnperature of 8b"F and a weight of 2b pounds. A part
oithe gas
w^s discharged and the temperature ind p""**"
.t
to
70"F and 300 psia, respectively. Heat was applied "og"d
and the
I.rnperature was back to 8b"F. Find the nnd weight.
volume,
nrrrl pressure of the gas.
Ans. 1b.48 lb; 10 fts;808.b psia (EE Board problem)
4. Four hundred cubic centimeters of a gas at ?40 mm Hg
alr"lut'e and 18oc undergoes a proc€ss uotit ttre pr?ssune
lp.rmes 760 mm Hg absolute andihe temperature 0"c.
what
tr l,hc final volume of the gas?
Ans. 36b cc (EE Board problem)
fi. A motorist equips his automobile tires with a relief-tlpe
uo that_the pressure inside the tire never
will exceed 240
::]u,:(sage).
ll'^
He starts
wilh
pressru€
a
of
200 kpa (gage)
1tlp
e.rrrl rr uemperature of 2B"c in the tires. During
the long drive,
lf*r l.mperature of the air in the tires reaches-g8"c. nich
tire
xrrrlrrins 0.11 kg of air. Determine (a) the mass
of air escaping
eer lr l.ire, (b)
lhe pressure of the tire when tfre tempe""t"""
relrrr.rrH to 28"C.
ArrH (a) 0.006,1kS; ft) 192.48 kpa (gage)
{i A 6-m3 tank contains helium at 400 K and is
F,nr rrl,mospheric pressure to a pressure of 240evacuated
mm
Hg
remaining in the tank;
f kf rrrrrHs of helium pumped
out, (c) tfre tempei*ui" of tfr"
l€*r'rrrr^g helium falls to 10"C. What is the pi*u*rr"" in kpa?
te, urrrn. I)etermine (a) mass of helium
48
49
Ans. (a) 0.01925 ke; ft) 0.7L23 ks; (c) 1.886 kPa
7
. An automobile tire contains 3730 cu in. of air at 32 psig
and 80"F. (a) What mass of air is in the tire? ft) In operation,
the air temperature increases to 145''c .If the tire is inflexible,
what is the resulting percentage increase in gage pressure?
(c) What mass of the 145"F air must be bled off to reduce the
pressure back to its original value?
Ans. (a) 0.5041 Ib; (b) 17'53Vo; (c) 0'0542lb
4 Processes of Ideal Gases
-
8. A spherical balloon is 40 f,t in diameter and surrou
by zrir at 60"F and29.92in Hg abs. (a) If the balloon is filled
hydrogen at a temperature of 70"F and atmospheric pressure'
what iotal load can it lift? (b) If it contains helium instead of
hydrogen, other conditions remaining the same, what load can
itlift? (c) Helium is nearly twice as heavy as hydrogen. Does it
have half the lifting force? R for hydrogen is 766.54 and for
helium is 386.04 ft.lb/lb."R.
Ans. (a) 2381 lb; (b) 2209 lb
9. A reservoir contains 2.83 cu m of carbon monoxide
6895 kPa and 23.6"C. An evacuated tank is filled from I
reservoir to a pressure of 3497 kPa and a temperature
Lz.4}C,while tfe pressure in the reservoir decreases to 62
kPa and the temperature to 18.3"C. What is the volume of
tank? R for CO is 296'.92 J/kg.K".
Constant Volume process
An isometric process is a reversible constant volume proc.gs- A constant volume process may be reversible or irreiersrlrle.
2T
I
T_ I
I
Pz
I
'l
Hl
Ans. 0.451 m3
F-_sz
10. A gas initially at 15 psia and 2 cu ft undergoes a
to 90 psia and 0.60 cu ft, during which the enthalpy in
by 15.5 Btu; c" =2.44Btunb. R". Determine (a) AU, (b) cn,
(c) R.
Ans. (a) 11.06 Btu; (b) 3.42 Btunb.R'; (c) 762.4ft.lVlb.
11. For a certain gas, R = 0.277 kJ/kg.Kandk= 1'
(a) What are the value of co and c,? ft) What mass of
gas would occupy a volurire 6t O.+ZS cu m at517.l'l kPa
26.7'C? (c) If 31.65 kJ are transferred to this gas at
volume in (b), what are the resulting temperature and
sure?
Ans. (a) A.7214 and 0.994 kJ/kg.R"; (b> 2'M7
Fig. 5. Isometric Process
(;r) Relation between p and T.
Tt
Pz
It
Pr
;fr- =-
(b) Nonflow work.
,'2
W.=JpdV=0
(c) 43.27"C, 545.75 kPa
50
5l
(c) The
For reversible nonflow, Wn = 0'
For irreversible nonflow, Wo + 0'
W = nonflow work
!d = steadY flow work
change of internal energy'
6{J = rtr'c" (T2 - Tr)
(d) The heat transfened'
Q = Itrc'
l': oblemg
(Tz - Tr)
l.TencuftofairatS00psiaand400.Fiscooledtol40"F
(b) the
*t <.onstant rroto*". Wnat are (a) the( final pressure, heat'
(e) The change of enthalPY'
6tl =
"oittatpy,
Hululion
(0 The change of entroPY'
lS = mc"h
(g) Reversible
tralsferred
the change of internal energy' d) the'
ana (0 ihe change of entropy?
i,,, ,.r," .frurrg" of
w o rh, (c)
mco (T2 - T1)
ll
ft
I
I
steady flow constant volume'
ta) ( =16+AK+AWr+W"+AP
2
v
W"=-(AWr+AK+AP)
Pr
Tr
T2
i0 cu ft
300 psia
400+ 460= 860'R
140+460=600"R
t z-- += Ag#q = 2oe psia
W"=-AWr=V(Pr-Pr)
(AP=0'AK-0)
/2
&)- -llVdP=W"+lK
-V(Pz-Pr)=W"+AK
v(Pr-Pr)=W"+AK
llr)
W=0
Ir
"' = S'=
I
l##li6?#) =g'4?tb
,\lI= mC"(Tr-Tr)
. (s.4L7) (0.1?14) (600 - 860)
v(Pr-P')=w"
166 = 0)
(h) Ireversible nonflow constant
V
V
-420 Btu
volume process'
r,tr (,f
mc" (T, - Tr) = -420 Btu
Q=AU+W"
53
Tr=60+273= 333K
(e) AH = mcn (T, - Tr)
(a) ,p _ T,p,
= (9.417) (0.24) (600 - 860)
= -588 Btu
(0
os = -...1o
'
'2
Pr
=
gPS652 = 999 K
DOI.O
(b)"vv - R
377 =1b0g-J==
kg.K"
k-l -= 7.25-1-
$lr
AU= mc, (T, - Tr)
= (1.36) (1.508) (999 - 333)
= 1366 kJ
= (e.4tz) (0.1?14) t" 333
= -0.581H
W"=Q-AU=105.5-1366
2. There are 1.36 kg of gas, for which R= 377 J/kg'k a
k = 1.25, that undergo a nonflow constant volume process
pr = 551.6 kPa and t, = 6OC to p, = 1655 kPa. During the proc
tlie gas is internally stirred and there are also added 105'5
of heat. Determine (a) tr, (b) the workinput and (c) the
= -1260.5 kJ
(")
(1.36) (1.508) l"
ls = mculn l"
i=g
Tr =
q99
=2.2ffiY
ofentropy.
:t. A group of 50 persons attended a secret meeting irr rr
Solution
2
//
/
k = 1.25
R = 377 Jlke.k
m = 1.36 kg
Q = 105.5 kJ
Pr = 551.6 kPa
Pz = L655 kPa
,,u,rrr which is 12 meters wide by 10 meters long and a ce ilirrll
ill ,l rneters. The room is completely sealed off and insulrtl'r'rl
l,lirr.lr Jrerson gives off 150 kcal per hour of heat and occultit'r, rr
vnl11111o of 0.2 cubiC meter. The room has an
initial presstrrc ol'
lo t tt hPa and temperature of 16"c. calculate the roortt lcrrr
ll)f't4 )
1u r ;rlrrre after l0 minutes. (ME Board Problem - April
lit,l rr lion
z rl
ll/Pr
ll/
ll/r,
I l','
L
z
= 101"3 kPa
= 16 + 27:f . ',tt{lf l(
Vg
lrlr
t-r4
c, = 0.1?14
#.
= 0.1714# =
W= (-1 hp) (h) =r(-lhp) (0.74G kWhp) (h) (8600 n/lr
0.r7r4ffi
= -2685.6 k I
Q = (50 persons) (150 kcaVperson.hour) = 7500 kcal/h
a = AU+W
volume of room = (L2) (10) (3) = 360 m3
AU = Q - W = -850 - (-2685.6) = 1835.6 kI
volume of air, V = 360 - (0.2) (50) = 350 m3
AU = mc" (AT)
-4 =(0.28708)
. = RT,
,(191,31(l5ol
(289)
mass of air, m
AT = -AU. =
= 427.34kg
a = Ll-ruooealt-l9
h llliO hl
I = rzsok.ul
DC"
1250 = (427.34> (0.1714) (T, - 289)
T, = 306'1 K
5. A closed constant-vorum,e system receives r0.5 lr.I of
k.mperature is 400 K. Gn Board problem _ April
2T
I
t
the tank loses 850 kJ/h of heat. Calculate the rise in
ture of the tank after I hour, assuming that the process
at constant volume and that c" for water is 4.187 kJ/(kg) (
lt = 2b9.90 J(ks) (K)
I
p, = _344 kPa Tr = 278 K
V-0.06ms
Tz=400X
,/
1
vs
Solution
p,v
(0.06)
ke
q _= id:t500n?s) = 0'2857
c.
,\lr
I
/
Vs
Irreversible Constant Volume Process
a = (-850 kJ/h) (1 h) = -€50 kJ
56
:1 = 0.6SgS kJ(kc) (K)
I
4. A l-hp stirring motor is applied to a tank contai
22.7 kg of water. The stirring action is applied for I hour
l
f
lg, l"ggg)
Solution
tz = 33.1"C
I
rffi5.6 kJ
(22.7 kS) @.t87 kJ/kg.C") = 19.3 C"
lrrrddle work. The system.coSt-ains o*yg"r, at B44kpa, 2?g K,
rr.d occupies 0.0G cu m. Find the t eat (gain or loss)
#e nnat
a = mc,T2-Tr)
-l
'l
r
(344)
_
mc" (T, - Tr)
Q.2857) (0.6595) (400 - 278)
22.99 kJ
AU+W
22.99 + (*r0.5)
t2.49 kJ
fr7
(g) Steady flow isobaric.
Isobaric Process
-
(a)Q=AP+AK+AH+W'
An isobaric process is an internally reversible prccess of
substance during which the pressure remains constant.
W =-(AK+Ap)
W" = -aK
(AP = 3;
N\
\s\:i\
.2
(b) - JVdp = W + aK
I
0=W"+AK
W" = -aK
Fig.6. Isohric Process
(a) Relation between V and T.
Tz Vz
Tr=vi
(b) Nonflow work.
W"
t2
{,ndV = F(V2 - Vr)
l'roblems
A certain gas, with c, = 0.b29 Btu/lb.R" and R 96.2 ft.lV
.lh."R,
l. expands
=
g0"F
from b cu ft and
to 15 cu ft while the
remains
constant at lb.b psia. Compute (a) T", (b) AH,
trrcsgutre
(r') AU and (d) AS. (e) For an internally
reversible'nonflow
f r'ocess, what is the work?
Solution
(c) The change of internal energ:y.
T
l
(d) The heat transferred.
__>_2
AH = rlc, (T, - Tr)
58
p = 15.5 psia
V, = 5cuft
% = l5cuft
T, = 80+460=540"R
vc
(e) The change ofenthalpy.
aS = mcohfr
/
,/
Q = mcn (T, -Tr)
(f) The change ofentropy.
2
/
AIJ = rDC" (T2 - Tr)
,^)'r', =1:,=
g+lP =r620R
.
'r'\,, =
ffi i##ffif)
=o.2r48rb
51)
= mce(Tz _ Tr)
= (0.2148) (0.529) (1620_ 540)
= 122.7 Btu
(c' c" co-R=
=
0.b29-W=0.40ss#S
(n\
2. A perfect eas
a value
of R = 319 .2 Jlkg.lfurrrtt
\1s
lt
*" iaggJ-fi;ik;
of this gas ar c''r.rlrrrrl
,fiTre):f: jli.i?Ttlmrnlm{:m1t,'i,i,t?,,,,,
r.2G. If 120 kJ
Solution
AU= mc, (T2 _ Tr)
= 1.26
= 2.27 kg
= 319.2 J&g.K
a = f20 kW
Tr = 32.2 + ZZg BO5.Z
K
k
= (0.214s) (0.40$;(1620 _ b4o)
= 94 Btu
(d)
os = mcorn
ftI
= (0.2148) (0.52e) h
=
m
R
ffi
(a) co
Btu
=*
-(1.2gxo.a1e2)= t.b46e
a = mco (T, - T,)
0.1249 oR
f{_
kg.Ku
r20 = (2.27) (r.b469) (T, _ g05.2)
(e) \=
p(% - v,)
=
Ta
= s39.4 K
(r5.5) (144) (15 - 5)
778
(b) aH= mco (T2 _
28.7 Btu
(c) cv
=
Tr) = l20 kI
h=ffit$ =r.22??#h
AU- mc, (T, - Tr)
(2.27) (r.2277)(33e.4 _
=
(d) W
305.2)
95.3 kJ
= p(%- V,)' = plg,_
-ITl =mR(Tr*T,)
^LP, --tri]
= (2.22) (0.8192) (Js9.4 _ g0s.z)
= Z4.Zg kJ
-Fr
Isothermal process
G) Steady flow isothermal.
isothermal process is an internally reversible
constant
temperature process of a substance.
(a)Q = Ap+AK+AH+W
w"=e-Ap-AK
W"=Q
(AP-0,4K=0)
.2
ft) - JVdp = W + aK
'i!:{t
From pV = C, pdV +
F-o'-{
Vdp -_ 0, dp
-,!'uoo=-l;,i
I
Fig. Z. Isothermal process
=
- pdv
-v-
#l j
/2
=
oou
I
P'\1n
(a) Retation between p and V.
PrVr = Pz%
W"=W"
ft) Nonflow work.
(AK = 6;
f2
)2
r
{v
w" = Jpav=l$Y= Cln5= n,v,rr
vr
' *v,
(c) The change of internal energy.
^s=+-mRrn$j
.t''ir drops fr.om g0 p.i" tol
the pressurc orr
gsic. For
"rilJ"lr",,
lfru ipaV and the work of a
the-_ JVdp;ndllie *o"k of
a steady llow
f 'r , !,
'.,,:, rluring which AK = 0, ("i e, iai aU
oS.
il;fi,;liii
*= -nrrn&
n
I l)uring an isothermal process at ggoF,
*r,,r.11;i[lls process,
Q= N + W" = p,Vrln
(e) The change of enthalpy. Y r
(f) The change of entropy.
'r'olrlcms
i,,,i1ll1v1y process, (b)_d:,tennile fal
(d) The heat transfenred.
AH=9
f
rr tt,
AU=9
62
-w
Pz
Tl
r
t pV,=[
,'ul
I
\l
\\.2
T
1*--__r.__2 m
pl
I
-L
V
Pr
88+460=54fi,,lt
8tb
80 psia
+ 14.7 = 1.9.? 1lsi1
r.t
(a)
lndv = p,V,tnV' = mRT r"
Vr
v2
*Pz
Q = Prvrlo q
= tltt#ftQ t" f# = 42L.2Btu
V,
In vl =
"r- Uft =
W,= jOaV=42l.2Btu'
%
= €-1.80
q
jvap = p,V,ln .f, = 42L.2Btu
(c) a = ryt *W"= 421.28tu
(b)
(d)
= 0.1653
v, = (0.1653) (0.30r) = 0.0498 m3/s
P,t, - (b86) (0.
--To:oa#l) =3542kPa
AU=0
AH=0
(e) m=
m#oO =-r.80
(b) Since AP = 6 and AK
(t)ns=
3=W=0.2686#
+=
#
= 0, W" = lV" = e = -B1Z kJ/s
=-1.ob8kJ/r(.s
AH=0
2. During a reversible process there are abstracted 317
kJ/s from 1.134 kg/s of a certain gas while the temperature
remains constant at 26.7'C. For this gas, cD = 2.232 and c"
1.713 kJ/kg.K. The initial pressure is 586 kPa. For
nonflow and steady flow (AP = 0, AK = 0) process, determine (
Vr,% and pr, (b) the work and Q, (c) AS and AH.
i,',, r r' l.hird the inlet pressure and the inlet pressure is zoz
Solution
111'plrplible. (EE Board Problem
:l
Air flows steadily through an engine at constant tem_
rrl,'re,4_09
K.Find the workperkilogram ifthe exitpressure
u'r
r
kpa.
Arrarrrro that the kinetic and potential energy variation is
- April lggS)
tlnlttlitttt
a=. -317 kJ/s
fi= 1.134 ks/s
Pr=
,n
586 kPa
26.7 +273=299.7
\
Pr
p, =$
(a) R - cp c, = 2.232 - 1.713 = 0.5L9 kl/kg.K
64
= 400K
= 282.08 kJ(ke) (K)
Pr = 2O7 kPa
T
R
'\2
vs
\i. = _*xTl=
pr
tT
\ l)V=C
(1.134) (0:5_U)) (299.7)
= 0.301 m3/s
586
V
R't'I -_.(9,?87_q8) gog)
l),
207
= 0.5547 m,t/kg
(;5
W = prvrl"
(c) Relation between T and p.
t=nrvr1nfl
k-1
12 [p,l r-
(20?) (0.5547) ln 3
=
= 126.1 kJ
q = LP-'l
2. Nonflow work.
Fromp\A=C,p-C1r-r
IsentroPic Process
adiabatic process'
An isentropic process is a reversible
A reversible adiabatic is one
Adiabatic simply *"t"t-"theat'
W" =
,2
rz
,2
CV+dV=
C
lpdv=J
{ V-ndV
t'Itl
Integrating and simplifing,
of constant entroPY'
wn
pvn=9
l-k
l-k
'fhe change of internal energy.
.pv=Q
tJl
AIJ = ncu (T2 - Tr)
\
I
'l'he heat transferred.
Q=0
'l'hc change of enthalpy.
Fig. 8. IsentroPic Process
1.
AI{ = mcp (Tz * Tl)
Relation among P, V, and T'
'l'lrr: change of entropy.
(a) Relation between P and V'
ns=0
P'VI=PrVb=C
I iI r.rrrly flow isentropic.
(b) Relation between T and V'
From p,VT = pr$u,td
T,= lvt-
T,
(i(;
q =+'
,,,r(c,.AP+AK+AH+W"
we have
wo,,_-AP_AK_AH
k'l
W. -AH
I
LqJ
r \l'
O, Al( = 0)
67
T-
E
(lr) _ p,V, (800) (t44)(100)
.2
(b)- lVdp=W"+AK
t'
m=
1-L
LetC=pIVorV=Cpk
'.2.1
- t'lVap =!C pk dp
k (P'v' - P'v')
l-k
= r. f'nav
i
Problems
1. From a state defined by 300 psia, 100 cu ft and 240"
helium undergoes andisentropic process to 0.3 psig. Find (a)V
and tr, (b) AU and AH, (c)JpdV, (d) -5vdp, (e) Q and AS. Wha
is the work (f) if the process is nonflow, (g) if the process i
steady flow with AK = 10 Btu?
=l5'eelb
AII = ms, (f, * Tr) = (1b.99) (1.241) (211.8 _70{)= _9698
AL.I = mc, (T,
Integrating and simPlifYing,
- t'fiao' -
ftfr=-6f6ffi
tt')6av
tstu
- Tr) = (15.99) (0.74b) (211.S - 200) = _5822 Btu
= &!;f,J'
=ffi
= b822 Btu
rrlt *!Vdp = kjpdV = (1.606) (b822)= 9698 Btu
lr,)a=0
As-- 0
rlr a = AU+W"
W"= -AU= 1-5822) =b822 Btu
Irir JVdp = W" + AK
1Xj9g=W"+10
Solution
W" = 9636 31rt
Pr = 300 Psia
Pz= 0.3 +'l'4.7 = 15 psia
V, = 100 cu ft.
T, = 240+46A=700'R
'.', An adiabatic expansion of air occurs through
h
a nor,zlt,
"rrr ll28 kPa and ?1oc to 1Bg kpa. The initial kinetlc energy i"
..'11lr1lible. For an isentropic expansion,
compute the spcr:if i.
section.
.r,lrnnr), temperature and speed at the exit
titi rr lion
s
I
(a)
\ = v, H$t= 1oo[,!9f
1'666
I
= 608.4 rtg
1.666-1
k-1
l?r -'l-k-
T
lr2
-2--T^'Lpil
I
r.-_T r.666
= 7001__{q_l = 211.8'R
\ pVk= 6
\
\z
828 kPa
7L + 273 = i|44 l(
138 kPa
Lsool
t"= -248'7"F
68
(il)
k-r
r.4_l
-k
tnl
T"=T,
- ll2l
'Lpil
=
-.-1.4
344lHgl
= 206 K
18281
;,,\
it>\
't.h^I
tz= -67oC
",
=
#,
_ (0.287q8X344) = 0.1193 m'/ks
., // i,
22Q..,
'Zzt
75yty:;
'iivr2i
lI
-
ve = vr
-
= 0.429m'/ks
[g'l. = 0.1198 lHgl'n
LprJ
11381
Ah = cp (T, * Tr) = 1.0062 (20G - 344) =
Fig. 9. Polytropic Process
-188.9 kJ/kg
A =&*aK+Ah+/"
Itelation among p, V, and T
AK--Ah=136,900J/kg
(a) Relation between p and
AK=4-^r=*
D2r= (2k)(AK) = zf
1Jz
V.
P,vi = Prvi
r
ffil
(b) Relation between T and
1rg,966S ) = 277,800 m
V.
To /-vJ "-t
= 527.1m/s
T =1q1.
t, t li.elation between T and p.
*.1
L
Polytropic Process
r-lP.
l:-€-
I
t_^ t--l
Ra 'r',
-lp.
A polytropic procebs is an internaliy reversible
during which
pV" = C and prVl = prVl = p,I"
rn
Le
r-
I
I
I
Nonflow work
It,
where n is any constant.
I
(paV = PrY, - P,V, - mR
" ,'l-n
'l'hc change of internal
AIJ = mcu (T, - T1)
70
(T, - T,)
energy
4. The heat transferred
(b)- Juao=W"rAK
a = AU+W-
I
,2
- ,fvao = {&t:!&
=
-n
JPdv
T_n-.
= mc" (T2 - T,) +
mR-(T, - Tr)
1-n
Ic -nc +Rl (r2-r,)
= *Lffj
[c - nTl
= - lffl
polytropic process,
(r'?-rr)
= ,n." f-!- I}_l
"-j (T, _ T,)
Lr -
a = mc. (T, - Tr)
l'-t -;l
cn = cu
lfrl , the polytropic specific heat
D.
I'rohlems
The change of enthalpy
AH = mcp (T2 - Tr)
The c.hange of entropy
t0Ib of an ideal gas, whose
3X"^1u: and
It l.^
40 ft.lbnb.R
cop = o.-zs
__:_ etju.&1;;;;;;il
_vwrv.r!, luau6,cs suate Irom
l#;;zu
lrlr;r and 40'F to 120 psla
ra and 340"F. Determine (a)
dY, (? -
n, (f;4g
urr4
il{t (g) rf the pi"*,, i ,iuuav
;ll,l
l,'rv !ilil,-(11'9:l"ljf
<luring which AK= 0, whaf is w"i]wuut
i. axirw"s J;it1
\Vlr;rI is the work fo, u
f
"o"n*-p."i"rrZ
Se
ilution
l',
ilO psia
m = 10lb
ffn 120 psia
l"
,10 + 460
R=40**
= 500"R
l'" it4o + 460 = g00"R
cp = o.2b
#
n_l
AS=mc ln It
"T,
l),
=T'
Tr
l),
7. Steady flow polytropic
(a)Q=AP+AK+AH+\
w"=Q_AP_AK_AH
w = Q_AH
(AP=0,aK=g;
n-l
liio J_ _ g00
:ro
b00
I -
tr I
ln6=ln1.6
tl
l 0.4700
rr =-1.7918
rr-
n = l.Bbo
72
'/3',
(b) c, - cp R = 0.25 -
#=
0.1986
m
(h) W" = JpdV = -433.3 Btu
AIJ = DCu (T2 - Tr)
= (10) (0.1986)
(800 - 5oo)
= 595.8 Btu
\,
AH = mcp (T2 - T1)
= (10) (0.25) (800 - 500)
= 750 Btu
(c)
k = 5=
^9'^4 =r.25s
q 0.1e86
2. Compress 4 kg/s of COrgas polytropically (pVr.z = C)
{ro3 pr = 103.4
!lu,-t, = 60oC to-tr- zzT.C.Assumingideal gas
tction, frld pr,
ry, e;lS (a)g.as ionflow, (b) as a stleady flow
where
AP
l)rocesg
= 0, AK =
Solution
Pr = 103.4 kPa
AS
?= (10) (0'0541) r"ffi= 0'2543+#
= -c" lt d,
(d)Q = mc"(Tr-Tr)
=
(10) (0'0541) (800 - 500)
L62.3 Btu
(e)Jnav-
eE+*L)-ffi
= -433.3 Btu
(0 -JVap = nJRdV = (1'356) (-433'3) = -587'6 Btu
Tr = 60 +273 = 333 K
fi=4\gs
T, =227 +Z7B = b00K
trr ) Nonflow
*#,
o, = o,
= (10s.4)F$$] = r184.e kpa
[+..|
L rl
Lgo'-l
w = ,hR %u __,4),0,1T16):900 - 33o
KJ
= -631.13 ;-s
c =c ll-d
" "Ll-ul =ro.osorffi;]
(g) W" = -fVdP = -58?.6 Btu
AK
74
= -JVap = -587"6 Btu
= -0.2887 []*
IT,
TIIF'
7. If 10 kg/min of air are compressedisothermally from p,
=, 96 kPa *{Vr.= 7.G5 ms/min to p, = 620 kpa, find tie worh,
:he change ofentropy and the heat for (a) nonflow process
.b) a steady flow proce-s-s_with or = lb m/s and u,
='60
(a)
-tBZ0 kJ/min, _b. gbo kJK.min;iU)_f
Ans.
and
Js.
386.9kJ
min
5 Gas Cycles
8. One pound of an ideal gas undergoes an isentropic
pf9c9s9^fr9m gb.B psig and a volume of 0.6 {tr
to a final volume
of 3.6 ft3. If c^ = 0.1,^2{3nd c, - 0.098 Btunb.R,
----'--' what
a.eia)
\
'!-asw *rv
(b) pr, (c) AH'and (d) W.
Ans. (a) -2€.r"F; (b) 10.09 psia; (c) _21.96
(d) 16.48 Btu
t'
9. A certain ideal gas whose R = 22g.6 J/kg.K and c- = 1.01
HAg.X expands isentropically from lbt? kFa, ie8"t t" gO
kPa. For454 glsof this gas determine, (a)W",
fljV'i.iAU
(s) AH.
Ans. (a) 21.9 kJ/s;(b) 0.0649b m'/s; (d) 80.18
kJ/s
-
10. A polytropic process ofair from lbO psia, 800.F, and 1
occurs to p, = 20 psia in accordance with pVt.g - C. Determir
t,
ft) lU, AH and AS, (c) JpaV and JVap. 1
9)
-%,Compute the heat from the polytropic splcific heat and cl
by the equation Q = AU + fpdV. (e) Fina tne nonflow work
(f) the steady flow work for AK 0.
=
Ans. (a) 17.4"F, 4.71t ft3; (b) -2b.8f Btu, -86.14
0.0141Btu/"R; (c) 34.4f Btu,44.78 Btu; (d) g
Btu; (e) 34.41Btu; (0 44.?B Btu
*d
-
Fleat engine or thermal engine is a closed
system (no mass
only heai
*itr,
rts surrounding and that operates in cyclls.
""a -"rr.
Illements of a thermodinemic heat engine
with a fluid as
I lrr. working substance:
a working substance, matter that receives
heat, rejects
.lu,rrl,I and
does work;
2. a source of heat (also called a hot body, a heat reservoir,
,r'.;ust source), from which the working zubstancei*.*iuuc
.r'osses its boundaries) that exchanges
lrlrr [;
3. a heat sink (also called a receiver, a cold body, just
or
rrrrk), to which the working substance can
reject rr""i; *a
4 ' an engine, wherein the
working substa'nce
*""r.
lr. lurve work done on it.
"rr"h"
A thermodynamic cycle occurs when the
working fluid of a
rv'l.t'm experiencer, u.ly.*,ber of processes that
Jventuaily
nrlrrrn the fluid to its initial state.
Cycle lVork and Thermal Effrciency
11. The work required to compress a gas reversibly accon
ing to p[r'ao = C is 67,790 J, if there is no flow. Detennine A
3"d Q if the gas is (a) air, (b) methane.For methane, k 1
R = 518.45 J/kg.K, c, = 1.6lg7, co= Z.lB77 kJ/kg.K'-
Ans.(aiso.gi KI, -ro.esokl;ruiog.bo kJ,
=
- 4.zgkJ
(1.
QA
= heat added
Qn = heat rejected
W-
net work
ftl
Available energy is that part of the heat that was converted
into mechanical work.
Unavailable energy is the remainder of the heat that had
be rejected into the receiver (sink).
The Second Law of Thermodynamics
AII energy receiued as heat by a heat-engine cycle cannot
conuerted into mechanical work.
Work of a Cycle
(a)W=IQ
W=Qo+(-Qn)
(Algebraic sum)
W=Qo- Q*
(Arithmetic difference)
(b) The net work of a cycle is the algebraic sum ofthe
done by the individual processes.
W= LW
Operation of the Carnot Engine
A cylinder C contains m mass of a substance. The cylindor
head, the only place where heat may enter or leave the subgtance (system) is placed in contact with the sounoe of heat or
hot body which has a constant temperature Tr. Heat flows from
the hot body into the substance in the cylinCler isothermally,
l)rocess l-2, and the piston moves from tr' to 2'. Next, the
t:ylinder is removed from the-hot body and the insulator I ie
placed over the head of the cylinder, so that no heat may be
l,ransfemed in or out. As a result, any further process is
ndiabatic. The isentrppic change 2-3 now occurs and the piston
moves from 2' to 3'. When the piston reaches the end of the
sl.roke 3', the insulator I is removed and the cylinder head is
placed in contact with the receiver or sink, which remains at a
ronstant temperature T". Heat then flows from the substance
t,rr the sink, and the isothermal compression B-4 occrut while
tlrc piston moves from 3'to 4'. Finally, the insulator I is again
lllnced over the head and the isentropic cor.npression 4-1 ret,urns the substance toits initial condition, as the piston moves
ftom 4'to 1'.
W=Wr-r+Wr"r+W'n+..
The Carnot Cycle
The Carnot cycle is the most efficient cycle concei
There are otherideal cycles as effrcient as the Carnot cycle; but
none more so, such a perfect cycle
forms a standard ofcomparison
for actual engines and actual cycles and also for other less effisient ideal cycles, permitting as
to judge how much room there
might be for improvement.
H'
m
Fig. 11. The Carnot Cycle
82
n
Vm
Fig. 12 Canrot Cycle
Anulysis of the Carnot Cycle
(ln = Tl (S2
- Sr), area l-2-n-m-1
(1,, = T3 (S4
- Ss), area B-4-m-n-B
83
-TB (Ss - S. ) =
w- Qn -
*Tr (S2 - Sr)
Q* = Tr (Sz
- Sr) - Ts (S2 - Sr)
(Tl - Ts) (S2 - S1), arca L'2-3'4'l
e=
o"= - r;s;;r
\[ = A^ - a- = mRTrtnt mRTrh
g=
e = ---Erl
The thermalefficiencye is definedas the fractionoftheheat
cycle that is converted into work
; supplied to a thermodynamic
Work from the TS Plane
g=
+-v
(Tr - Ts) mR ln
w
a;
t
fvl
,V,L
mRT. k
Tt-Tt
-T,
Work from the pV plane.
W = IW = Wr_, + Wr-, + Wr-n + Wr-,
= mRTrfn f
w = p,v,l"
V.-V3
Qn = mRTrln 1; = -mRTrln
t
w- (Tt - Tr) mRl"
W (Tr - T3) (Sz - Sr)
Tr-T,
Q^
Q* = -mRTrt"
i
t. &+: :J,+ p,v,rnf,.&tJ{.
From process 2-3,
T3 l-v, l*-'
T =Lv'J
Mean Effective Pressure (p_ or mep)
P-=W
VD
From process 4-1,
T,
-l-v,J.-'
11 -lfJ
but Tn = Ts and Tr =T2
- | =
therefore,l V"-k-r
LqI
then, &
%
=vr
v,
84
Vp = displacement volume, the volume swept by the piston
rr one stroke.
Mean effective pressure is the average constant pressure
l,ir:rt, acting through one stroke, will do on the piston the net
work of a single cycle.
Ratio of Expansion, Ratio of Compr.ession
I,)xpansion ratio
vglute,3t
end of expansiql
-.,
the
= volumeattheffiili
ft5
Point 1:
Isothermal exPansion "atio
IsentroPic exPansion
=
,:-
t
naRT. (2) (53.34) (960)
vr = -E-
VL
ratro = 1;
Point 2:
Overall exPansion 'utio = h
Compression ratio =
% =
of
EH*#
lsothermal comPression ratio =
Isentropic compression ratio'
#
Pg= p,
rr. = Y^-
1;
--*
[:t:^ = 11ee.7''b-d
L.aJ
l-sso-l
= 24.57 psia
mRT" (2) (53.34) (530) = 15.72 f13
=-(24,s7) ( lll*4)
%= -Ti
V
Point 4:
ratio rn is the compression
ratio
Problems
1. A Carnot power cvcle operates on 2 lb
{l*j::?*ii?
'ffi
b;:.
"ttffi
n""q;l"^:l
Ho"n
#
ri*i,
:l: *,".':t'H :f
;bd n* ilu^* 11:, : 11 :'-' i;1'":ff31
l'ffi:S J'";n#J'#*ff;;il;
?qif vorume at the end
rD rvu ]'"'b' - - ffilT.f#H;
lX"-lff
?xpallsrv[
' isothermal process,
proce
nS durine an
-.";^- rlt
G) lP
isothermal compression,
$"t"q: - ^r ^-aanoinn rlrrrine
(h)
iil'6::?.i' 6Ji";fi:ie
ffil,3
:, g*:
:** or"*pansion,
$""#"ffi:T and
ft;;rr iutio
h[fi;
lll,?*,$*;1il*
,
the mean effective Pressure'
[q = (1b.?2)F-ffi =
v4=
v,
(a) \
= 7.849 ftg
(b)
= mRln
^s,-,
t=
2.84e rtg
Q.%19 h*ffi = o"oeoz{fi
(c) Qo = Tr (AS) = (960) (0.0952) = 91.43 Btu
(d) QR - -T, (AS) = {530) (0.0952) = - 50.46 Btu
(e) W = Qn - Qn = 91.4g -50.46 = 40.97 Btu
Solution
m= 2lb
Pr=
Tr=
400 psia
Pz=
199.7 Psia
Tr=
ft(;
+=ti$ffit#,=8.b61 na
Point 3:
v
Overall comPression tutio = \t
The isentropic compression
most commonlY used'
= -I4OOXI4;JI= = L.778 ft,3
960'R
530'R
(o
o'4481 ot
" l[=4s
a^ fl'43- =
=
(8)I*oth""-al
44'8Lvo
expansion ratio =
* =ffi =,
87
s
o
15
t
E
E
<r
oq
,-E!vv a EF
osoo{r
6lIOti€rO
ilillllttl
lF{
+t9{
el\
o
t:
Eg
€E
gh
oi
o?
|r)
fi
OJ
tl
Hla
cl
a
a
Io
ca
-q
tl
o,
rt
<l
I
19
t=---r
6It=
I l*l
tl
{.r
r;
rRlR
t'
ll
*{
6
q
ll
fl
a
le."le''*.
tl
I
l-lvfl
o
FIE
lolol
'€lC'l'
o
-l
N
E{
h
(0
#
tl
oO
ilo
-l ;€
I
c,a
(a
o
tl
3lv
allt?
<{ lv
st3
ro
!i{
-l-,:
tl
tl
RIE
-lu
,RlAl
il'r*;
ll
>1+
dltr
g
drlF
1l
Ef
MI
to9
rt
G
-lH
lj
-@
eqq
ct?
i./
)!c
f;
HJ_l-d
co
I-
^:t{
-t,'
'II
,
qolH
(ol
@l '
rlq
il
lF-l
"ql
'. lro
Frl
AIA
vtyJ
I,_ 116l
il"
vll
sv.:v
coA
F'lro
rO
lOrt
@la tll
I
Fl€
I cld
E
r
NlOIr
+
fl a
tl
II
rlatA,I lJlrl
-*
,-()
lt
.ol
rtE
[E! E
:;
d le o
of;E-EAES8
o
if Fil" * dF" H g e iAE*q
,r
r
T
""
{
H €H,FEd
"o o''
g i 'EHd i'
B*EE,
?€:3+
I E'E:E
Ei gt
s
E
fi;JE
;
3
HE
' 3 TB.
H-ti-L$* EiiE}
0r
rn c\r
c..i
c
.H
d
a
g
M X'a
Fa
rr:<
FB
€€ bo
r.E
ronoJ
t-O<r
q
cO
lr:
ll
l<t
^lFr
.X
$E
9ii
.ab
6I.i
E;O
tssd
.5x a_ R
<EE od
9.X B3
..EE
o
u)
$
illlll
C- CQ rO
Er
sa
ts
,fa
rb
F{
.d
ddt' dE *
"olQ
ll
O/?
--r
g{l!o colY
\Itr
rril\ FIA
,
('JI
f.ll
:l\
c-lFi
-o
b0d
l^lrO
'liil
.d. d
>"b
3l-l
r
l€lv
IcE
lrrll
Bl
ti'
5 t>,'E
(
|lr
|I
IpI,
lxrl
IOA
laBl;
lHrl
IO)
lAd
lv
.E
IV
I
I
I
I
I
I
II
I
Qe
= (m) (c") (T3 = 37.63 Btu
Tr) = (0'1382) (-0'6808) (540 - 939'9)
cp = c, + R = 0.7442+ 0.2969 = 1.0411
KI
EAIF
c_ 1.0411
ro={=yiffi,=1'3ee
en= mRr.rn{=,Wt"*h
Point 1:
n
v.
'' =
- -IT, -
&
= -27.82FJttt
'!{ = Qo - Q* = 37.63 -27.82 = 9'81 Btu
(2.5) (q396e) (e50)
= 0.8522 m3
827.4
Point 2:
o -A sz _o.osrdlg
As.ir:fl=-bao
Qn = mco (T, - Tr)
=
-132.2 = (2.5) (1.0411) (T, - 9b0)
(9.8!X179)- = B.lb psi
w
p-=ql172=
ffi-v'LvEe'
Tz = 899'2 X
gas with- R = 2963 Jfte)
2. T\vo and a half kg of an ideal
kPa and a
(K) and c" =6i++i r'"lltr'?Xrc11i a-ryJt:y""
9f 127 't
heat at constant pres'
temperatrfe b6Fc *J*t 132.2 kJ of
C to a point
to nJis =
"f
sure. The e""1;it""-"d;a*a "tto"ails
back to its
bring-tle
wil
p"ot"tt
where a constant volume
100 Hz'
for
poier in kW
original ttateS;t"rttil; er;q' *d the
e:
%=
u,F,] = (0.8b22)ffi21 = 0.8066 mg
Point 3:
r, = r,
H]"''
=
rsro.rlffi"u-' = 880.e K
Solution
Qo = mco (T, - Tr) + mcv (Tr - T3)
v
Pr=
rF
11 -
Q*=
827.41,Pa
677 +273= 950K
- 132.2 kJ
Qn = (2.5X-{.4435X886.9 - S99.2) + (2.5>(a.7442)(950- 886.9)
Qn = 131 IGI
'![
= Qo-Q*=131 -L32.2=-L.2kJ
w-
if
r#iFosgfl =-12okw
1) |
Review Problems
l.ThbworkingsubstanceforaCarnotcycleis8lbofair.
is.9 cu ft
The volume at the feginning of isothermal expansion
the
during
tn" pressure is 360 psia. The ratio of expansion
*a
is
uaaiuo" of heat is 2 and the temperature of the cold body
(h)
(g)
(0
the
P-,,
;0"F, Fi;J (a) Qe, o) QR, (c) vr, (d) pr, (e) vn, pn,
(i)
the
and
process'
isenlropic
u*purrsion duffng the
6 fnternal Combustion Engines
ratio of
overall ratio of comPression.
Ans. @) gia.a, Btu; (b) -209.1 Btu; (c) 63.57 99.ft; (d)
25.(/-p*iu; t"> ef.Zg cu ft; (f) 51.28 psia; (g) 13'59 psia;
(8) 7.06
(h) 3"53;
in
2. Gaseous nitrogen actuates a Carnot power -cycle
cycle,
whict the respective iolumes at the four corners of the
Vri
rt"*frtg ;tlnetUegittning of the isothermal expansion' arg cvcle
L
3
r57'7
zza.r+!, *1 Yr :
ib. iit i; v, = 1 4.bI L, v
Jhc
"Z
Determine (a) the work and (b) the
it"it.
t<.1
of
receives zi.r
Internal combustion-engine'is a heat engine
deriving its
power from the energy liberated by
the exploJion oi" *l*trr"
of some hydrocarbon, in gur*o.r, or vaporized form,
with
atmospheric air.
Spark.Ignition (SI) or Gasoline Engine
mean effective Pressure.
Ans. (a) 14.05 kJ; (b) &'91kPa
Erh06l
the thermal efficiency of the carnct cycle in
3. show
-of thatisentropic
compression ratio rk is glven
the
terms
bvg=l- .
1.
L-l
rk
4. Two and one'halfpounds of air actuate a cyclecomposed
urith n
=
of the following pro"u*t"*t polytropic compressiol Y'
known
The
3-1'
1.5; constant pressure 2-3-; constant volume
Btu' Determine (a)
au1,a *", p, = i0 p.iu, t, = 160'F, Q* = -1682
plane' in Btu;
i^
iul th;;;;k'of the cvcle'using the pV
(e)
efficiency, and
(J) ""a
. -: -.
'-' Q^,' (ai tne thermal
(a) itzo'R,4485'R; (b) 384'4'Btu; (c) 2067 Btu;
t-
p-'
- Arrr.
Infoh ttrcb
Comprarrlcn Strol.
Ittr.u!t lkol.
Fig. lB. Four-stroke Cycle Gasoline Engine
A cycla beginr wilh the intoke slroke or fhe pirlon move3
down the cylinder ond drows in o fuet.oir
mixlure' Next, the pisron compresse3 rhe mitture
whire rnoving up ri,. iyiiJ"r.-iiri.'i"o
or n.
comprersion ttroke. fhe spork prug ignites rhe mixrure.
Br:rning gq!es puth ,he pirton down for fho
piston rhen,o"1, ,p the cytinde-gJ", prrhrg
rhe'burneJ
ori!"rins
i".ilTrili?ii;lte
for",
*,o
(d) 18.60%; (e) 106.8 Psi
1'0et
5. Athree-process cycle of anideal gas'.forwhi*.htr=
compresisentropic
an
*aI." = 0.804 lr,yl*e.K', tl-tTlt"FibyIiPa. A cbnstant volume
t
sion 1-2 from rog.a"kpa, 27 "C 1060g. 1
3:l 11ll n= L'Zcomplete the cvcle'
p"".*t Z-S and a
(a) Qa, ft) W'
Circulation ir rtiuiv raL of o.go5 kg/s, compute
"
(c) e, and (d) p-.
Ans. (a) 41.4 k'ys; &) - 10 kJ/s; @\ 24'157o; (d) 19'81
kPa
92
The four-stmkg cycre is one wherein four strokes
of the
piston, two revolutions, are required to complet"
u.y.l".'
*-ftti*t
9:i
Otto Cycle
The Otto cYcle is the ideal prototype'of spark-ignition
engines.
,V
wnere
"* =vr.,
the isentrcpic compression ratio
Derivation of the form ,la for
e
Process l"-2:
5_
Tr- t-rl-l
LVol
'
T, = Tr"oo-t
(2)
Process B-4:
FiS. 14. Air-standard Otto CYcle
&
I-v;l*''
=F
T=
Air.standardcyglemeansthatairaloneistheworking
medium.
L-l
(3)
Substituting equations (2 ) and (3)
Tn-T,
'-E4rffi
a - ,
e = 1_n+
-t
Analysis of the Otto CYcle
= mc" (T, - Tr)
(Tn- Tr)
Qn = mc, (T, - Tn) = -mc"
\{ = Qn - Q* ' BC" (Ts - Tr) - BC' (T4 - Tr)
Qe
e=fr=ffi
r-#+F
'rr
e = 1-+
rl
94
tI
T, = Tn"*
1-2: isentroPic comPression
2'3: constant volume addition of heat
3-4: isentmPic exPansion
4-1: constant volume rejection of heat
e=
Lr*J
rz
(1)
IVorh from the pVplane
W = IW = Pr%'- 9rV, * O,? - O, -%
Clearance volume, per cent'clearance
"*=f=q;r=Hg6
_l+c
".*c
in equation (t)
lrr
where s = p€r cent clearance
% = clearance volume
Vn = dsplacement volume
(a) Point t:
v, =
s"-
= 1.oar
$
Point 2:
Ideal standard of comparison
*rt
fV
p, = prLfrJ
Cold-air standard, k = 1.4
Hot-air standard, k < 1.4
The thermal elficiency of the theoretical Otto cycle is
Tr=Tt
1. Increased by increase in r*
2. Increased by increase in k
3. Independent of the heat added
= P, (r*)h = (13) (5.5;r.e = 119.2 psia
l.l
el
= \ (r*)h-r = (590) (b.b;r.s-r = ggB.9.R
tz = 523.9,F
The average family car has a compression ratio of about 9:1.
The economical life of the average car is 8 years or 80,000
miles of motoring.
Problems
1. An Otto cycle operates on 0.1 lb/s of air from 13 psia and
13trF at the beginning of compression. The temperasture at
the end of combustion is 5000oR; compression ratio is 5.5; hotair standard, k = 1..3. (a) Find V' p2, t s, ps, V3, tn, and pr. (b-)
Compute Qn, Qj,'W, e, and the corresponding hp.
Solution
m=
^k
k=
Pr=
Tr=
Ts=
96
(0.1x€-.94)l_5eo)
0.1 lb/s
o.o
1.3
13 psia
130 + 460 =
5000"R
li
l'6=81
v-z-t
= 5.8 = o.Bob6 &i
=
s
Point B:
%=%=0.3056ts
Point 4:
l-ti : r'r
r. = 4Li-J
=(boo)m"'
tr = 2538"tr'
o, =
t
[+J=
(2ee8)H= 66.r psia
= 2998"R
R =(h)c=
\u'f cv =
L11
Qo = rhc" (T,
Btu
53.34 ==0.22tv'o'c'o
(zzgfitm
c'=
l6.R"
-=*+ =ffi=o'o43e6lce
- Tr) = (0.1) (0.2285) (5000 - 983.9)
Qn = sr.zz ntrt
s
Qp = rhcu (T, - Tn) = (0.1) (0.2285) (590 - 2998)
Qn = -55'03 Btu
"*
ry
=f= tdi%to =,,
(a) Point 2:
v, 0.0!
' =T=
#
s
W = Qo - Q* = 91.77 - 55.03 ; 36.75
E*=m =o'8444*k
' "'
T, = Tr"*t't = (805) {ll;t't-t
o =W =3!'75=0.4005 ot4A.O1Vo
tl
Pz =
(36.?5 BtuX60+)
W'=
'smrn
n'*t#ftnr
= o'003455 m3
Pr{
= (101.8) (tt;
= 6g9 K
t'e
= 2blg lipa
Point 3:
=52hp
Q^ = mc" (T, - Tr)
2. The conditions at the beginning of compression in an
Otto engine operating on hot-air standard with k ='1.34, are
101.3 kPa,0.038 m3 and lz'C.The clearanceisL0%oand 12.6hI
are added per cycle. Determine (a) V' T*P* T3, Ps, Tn atd p.'
(b) W, (c) e, and (d) p-.
12.6 = (0.04396) (O.UU)(TB
- 689)
Tg = 1028 X
Ps =
Solution
r,ltJ= (2518)
t8rfl = BZbzkpa
Point 4:
t =t{W"'=r&l'],r*r{*J
P, = 101.3 kPa
V, = 0'038 mg
Ti=32"C +273 =306
n,
=n,ffi:r,91]ruzuaftl'
1.t4.1
=455K
= 16l kPa
(b) Qn = mc" (T1- T1) = (0'04396)
(0'8444) (305 - 455)
Q* = -5'57 kJ
W = Qn * Qn = L2'6-5'5? =
?'03 kJ
, \ e=q=
- W - 7.99-= 0.558 or 55.87o
(c)
12S-
(d) p.
12.6
=#" = #T,= o55s - oso3455
(b)
(")
= 364.7 kPa
Fig. 16. Air-standard Diesel CYcle
1-2: isentropic comPression
2-3: constant-pressure addition of heat
3-4: isentropic expansion
4-1: constant-volume rejection of heat
or Diesel Engine
Compression-Ignition
Analysis of the Diesel CYcle
Qn = mcn (Ts - T2)
Q* I -." (T, - Tn) - -DC, Tn -
ln|!l.
Sl.ok.
ComF.trlon
ComF'trlon
Sftok'
?ow'r Stlol'
Fig. 15. Four-stroke Cycle
A cycle begins with the
Crh!urt Sitol'
Diesel Engine
intake stroke when the
piston moves
iil;t;*:: j*:t:-::f".1fit:11
and draws'"t1':-:ini'".-""ussion
downanddraws"ilffi
down
stroke' the tem''
when o' is
:H3:j!rye?tio"'
Htr
1l
ll:
n*J,ffi";
*iift ttt" hot air and
it
-i*"t
iniected into the
"tU"a"1
'U'"
rra$tru Prvuuvv- -"
exhaust
burnsexplosivelv'e;'";;;;'"'*:Jg1*;if
burns explosrvery'
ror the Power strt,k". During the::f,1f'l
do*o
gases
ffi;"tfit"oo
burned
forces the
;;Jt;
#";
piston
the
*t*k",
""d
Tr)
W = Qe - QR = mcn (T, -T, ) -DC" (T1 - Tr)
"=frW
.
e = 1-
T.-T
Fd:fJ
(4)
€=1where
"*
=F the comPression ratio
out of the cYlinder'
""
=
+, the cutoffratio
l0l
Point 3 is called the cutoffPoint.
Derivation of the fornula for e
efficiency ofthe Diesel cycle differs from that of.th* ( )r,r.r,
-The
cycle by the bracketed factor".o'1 . This factor i*iit*,,vu
trFT
greater than 1, because r" is always greater
than l. Thus, lirr rr
particularcompression ratio rn, the otto cycle
is more efficiont.
However, since the Diesel eigirr" compresses
air only, thr,
compression ratio is higher than in an otto engine.
An actual
Diesel engine with a compression ratio of lb is
mo"e efficierrt
than an actual otto engine with a compression ratio of
9.
Process 1-2:
'- *k-l
T"=Lv^,l
lv,
q
k-l
T, = Tr"*
I
(5)
Relation among rLr r.r and r" (expansion ratio)
Process 2-3:
t-
e
ft={; =f"
rk-
Ts = Trrrk'tr.
(6)
L% -L
-%
t =[+][q
' \=f"f"
Process 3-4:
Problems
t=F;-'=m-'=*'
Tn=Trrnk-l
Tr = Trr"k
H
(7)
1' A Diesel cycie operates with a compression ratio of l3.b
and" with a outoffoccuring at 6vo of the stroke.
state 1 is defined
ta psia and 14OF. Foithe hot-air standard with t<
!f
= f .ga ana
for an initial I cu ft, comp-ute (a) tz, p2,,.Uz,tsn
po,
,rrl-tn, {b)
%,
Q*, (c) w, (d) g uttd p-. (e) For aratlof"ciic,riauon irrooo.r-,
compute the horsepower.
Solution
Substituting equations (5), (6), and (7) in equation (4)'
T.t"ni.-e=1-m\f-'r--ffii)
'.,'4^ L
rn = 13.5
= 1.84
p, = 14 Psia
Tr=140+460=600'R
y, =lcuft
. 1 f-t"*-rl
e=r-,r-rlq:11l
r02
Io;l
i
c,
R
=FIf
53.34
= (078) (1.34 1)
-
=OrOtUffi
(b) QA = DCo (T3 - Tr) = (0.063) (0.2702) (2545 - iaga)
Qe = 18.57 Btu
cn = kc" = (1 .34) (0'2016) = 0'2702
ffi"
Qn = mc" (T, - T.) = (0.063) (0.2016) (600 -72i:l1r)
Qn = 8.52 Btu
(14) (144]jp
p,V, _
* = alf
= (b&lr+,1 (buu) = o.68o rb
(a) Point 2:
(c) W= QA- Qn = 18.57 -8.52= 10.05 Btu
(d) e = W = f0.05 = 0.54L2 or 54.L2Eo
v,=1fS = 0'0741 ft3
V,
1
=#x
T, = Tr#-1 = (600) (13.5)1 31-t = 1454oR
tz = 994oF
a^ 18.57
P- = (10.05) (778) = 58.64 psi
(l -.0:,0741) (144)
(e)
pz = prrr.k = (14) (13.5I'34 = 457.9 psia
Point 3:
% = V, + 0:06VD = % + 0.06
% = 0.0741 + (0.06)
..
(Vl -V2)
(1 - 0.0?41) = 0.1'297 ftc
0.L297
- r\il = G454) i,^g?A
= 2545"R
r, = Trl_C
ft''l
nin-l
[""ir*f
fo*
w_
42.4 lltu
'= 287 hp
min.hp
2. There are supplied 317 kJ/cycle to an ideal Diesel
engine operating on227 g air: p, = 9?.91 kPa, t, = 48.9oC. At the
end ofcompression, pz = 3930 kPa. Deteruineia) ro, (b) c, (c) r",
(d) W, (e) e, and (f) p-.
Solution
m = 0.227 kg.
P, = 97.91 kPa
Tr = 48.9 + 273 = 321.g K
Pz = 3930 kPa
Qo= gf7 kJ/cycle
\------(
\\
t, = 2085'F
4
\
Point 4:
rn =
I
r, l_sf'' = (2545) lli2gfl '''n-' = 12?1"R
LvrI
L1J
tr = 811oF
r (45't.e)
oo., IgJZgZl'''n = 29.7 psia
o. = n,lt'J =
[-T.]
r-v-r
l)oint 1:
v --r
'l-
mRT.
ll
.:l
* (0.227) (0.28708) (32r.e) = 0.2143 mg
97.9r
10s
ryPoint 2:
(d) QB - &c, (T,
1
Qn = -136.9
1.1
u, =
urffl
= (0-2143)
ffi
0.0153 m3
IJil
Hfl1f = sz4lK
Point 3:
Qn = mco (Ts - T2)
3r7 = Q.227) (1.0062) (T3 - 924.4)
T, = 2312I(
Im|
v, = vr,if i= (().olb3) lW1= o.oB8B mg
L-2)
P24A
kI
W = Qo - QR = 317
(e) e =
Tr=T, lo;l+'=(821.e)
- Tr) = (0.227)(0.2186) (B zt.g -tt6t )
- fg6.g = lg0.l kJ
P= lao.t = 0.b6g1 or 56.glvo
QA 317
1fl P- =g=
l0o.l _= 9ob kpa
vD =.w
vr_%=o-zr+s:00rog
DuaI Combustion Engine
In modern compression ignition
engines
constant during the.combristio" p"o"ess the pressure is not
manners illustrated in the ng"*.-ili;*J but varies in the
il ffi* ol" *
combustion can be conside*dt";il;ach
a constant-vorume
process, and the late burning,
u *;rilunt-pressure process.
Point 4:
,,=*b{'=(rrrr) B*?H" = 1161k
(a)
-V--o.oi^re -'
"'* =vr=0.2143_14
1+c
(b) f,=-*c
1+c
1r
I4t
=-
c
c = 0.0769 or 7.69Vo
-c v^
0.0383- t2.50
(c) f = -!iL =--:-::=
v, 0.0153
106
Fig. tZ. Air_Standard Dual Cycle
l-2: isentropic compression
2-B: constant_volume addition
of heat
3-4: constant-pressure addition
of heat
4-b: isentroplc expansion
5-1: constant-volume rejection
of heat
Analysis of Dual Combustion Cycle
Qo = mc, (T, - Tr) + mcp (T. _ fr)
Procesg B-4:
Q* = me, (T1 - T6) = -mc" (Tr - Tr)
W = Qe - Qn = mc" (\
g='W= mc" (T, - Tr) + mc, (T, - Tr) - mc, ('t'o - T,)
e=l-
\/v t
^g
tg
Tn = Trrr
mc, (T, - Tr) + mco (Ta - T, )
\QA
tn
4a
il= f,=""
- Tr) + mco (T1 - Ts) - DC" (T6 - Tr)
t'lr;{"
, (lt)
(8).
Tu = Trr*'t-l ror.
where
=S, the pressure ratio during the consant volume
""o P, ' poii"" of co-U"stio"
Tu= Tpor"r
v
rr =titr, the compression ratio
,2
or.
too"otuting equatirins (9), (10), (11),
\r
r.' =#, the cutoffratio
Y3
Th'b thernal,efficiency of this cycle lies between that of the
ideal Otto qnd the ideal Diesel.
Derivation of the formula for e
Proccss 1-2:
(r2)
€=l-
l-T
o=l-
*L
and (12) in equation
Problems
the *tllpg d:op-p."*rsion in
.bustion
L. At
an ideal dual comcvcle, the w.orki"ng n"ia-ir i ru
The compre*io.l
"irri"i-iijT#"
i il"- p"*rru*
99:F..
at the end of ""a
:.ilIor n*ullrito
tlre
constant
volume addrtion
;;i""#;;#;
added 100 Btu uA* th;,;il;;ilpor*,ro
"*
expansion. Find
(a)
(b)
T" -lv,l
-k-1
q=LrJ /
T" = Trr*I'r
ro,
Process 2-3:
r", (c) the percentage cfearence, (d)
e, and 1e) p_.
t=#="
T, = Trrrk-t rn
(10)
r0g
*'
Point 5:
Solution
m = llbair
p., = 14.1 psia
T, = 80+460=540oR
pa = 470 psia
rk= 9
t, = t
l+ln.'=
L_'I-J
(rnru)
E&1" = 1082"R
+!y = L.54
P=g=
Pz 305.6
(a) r^
Qr-n = 100 Btu
L.Zr
v, !g!tg
1.576 =
" =t=
(b) r
(c)r.-1+c
*c
Point 1:
u,=-3l'-=%#ffi#=la186rt3
9=1+c
c = 0.125 or ]'Z.EVo
Point 2:
v. 14.186
%=t=-t-=
(d) QA =
1.576ft3
= (1) (0.1?14) (1999 - lB00) + 100 = 219.8 Btu
rir-'l k-l
Tr= T, l+
I = (540) (9) ''n-' = 1300R
L'rJ
Qn = (mXc"XT, - Tu) = (1X0.1714X840- 1082)
l-v,l*
W 219.8-e2q o'5773 0r
^=Q;=
57 '73Vo
--fts-=:: =
l, = n,l_if = (14.1) (9) 1'4 = 305.6 psia
w =
P*=V,-%
Tr=T, [pJ
ffi
(126.e) (778\
=54.3?psi
2. An ideal dual c'ombustion cycre operates on 4b4 g of air.
At the beginning ofcomp_ression, the airis at g6.b3 p",?g.g"c.
t
Itet ro - 1.5,,r..= 1.!-0, an{ r* 11. Determine (a)
the percentage
=
LF;J
Point 4:
Qr-n = (m) (co)
= -92.9 Btu
"
Point 3:
('lea.rance, (b) p, V, and T at
each corner of the cycle, tc) e-n,
(d) s, an6 (e) p-.
(T. - Tr)
100 = (1) (0.24) (T4
Solution
- 1999)
I'
Tn = 24J.6"R
v. = v,R] = o.b?o)
il0
Q-, + Qr.n = (m) (e") (T, - Tr) + 1oo
f+f
= 1.905 ftg
'f-\.
t,\:
,-/i
4
A'
'/ -""
,2'
m = 0.454kgof air
P, = 96.53 kPa
T, = 43.3 + 273 = 816.3 K
rp = l'5
r" = 1'60
rr = ll
ill
W = Qr - Q* = 474-L95.7 = 278'3 kJ
(a)-rk--1+c
w 278.3 = 0.5871 or 58.7lVo
c
"=6o= 474
1+c
11 =-;
278.3
w
(e,p_=Vr5,=
= 716.8 kPa
o.427L 0.03883
g = 0'10 or IUVo
-
mRT, (0.454) (0.28?08) (316'3) = e.427r ms
(b)Vr=-p;=re
vt- --11
o.42t]- o.oB88B m3
*,
=
vr =T;-=
l-v-lr'-r
,, = t,FJ*-'= T, ("n)
p, = n,
*-'= (316'3) (11)'n-' = 8254K
I-vlF = pr(roy = (96.b3) (11) ''n =2770'81.Pa
ft'1
ps = (Pz) ("n) = (2??0'8)
(t'5) = 4156'2 kPa
,, = r,fog = (82b.4)
ffi
Vn = (Vr) (r.) = (0'03883)
=
K
'288.1
(t'60) = 0'06213 m3'
l-ri-l
rn = t'L+l= (1238'1) (1"6) = Le81
K
I-vln', = (1e81)
e16.2 K
,, = -r.LirJ
Bm''n-' =
pu =
l-m-l (e6.53) e1g.?
=27s .6 kpa
p,l+l=
'L'
d
316'3
(c) Qe - (m) (c") (T, - Tr) * (m)
(cn) (T4
(0.454X0.?186X1238'1 - 825'4) +
=
- T3)
(0'454X1'0062X1981-1238'
l)
= 474kJ
(d) QR = (m)(c"XT, -Tu) =
(0'454X0'?186X316'3 - 916'2) = 195'?
I t:l
-l
Review Problems
hot'air standard
1. An ideal Otto engine, operating on5'the
At the beginning of
with k = 1.34, h^t ;;;;;;tfi
ratiJof
psia and
cu ft' the pressure is 13'?5
;;;;t;;;irt" uor"-"is 6 Ouring
the constant'volume heatthe temperature i. fOO"f'
(d)
uaJJp"t cvcle' ritta (u) c' (b) T" (c) p" e'
t"g, il;'Bl"
^t"
and (e) p-.
Ans,
7 ""s Compressors
(a) 257o; (b) 5209"R; (c) 639'4 psia; (d) 42'14Vo;
(e) 161.2 Psi
operates
2. An ideal Otto cycle engine 'lrrtlnll%o clearance
The
i"Lx" !tut". is 100'58 kPa' 37'7oC'
on 0.227 kg/s of
is 110 kJ/s' For hot-air
energy released d;l;;;*bustion
(a)
p'
V' and T at each corner'
standard with k = isi,-"o-pute
(b) W, (c) e, and (d) P-'
t"1t':f:
*'r., o
kPa;
;6. + x, zazo.t r<P a, 5s2,1K 19 1'71
kPa
301'1
(b) 52'7 kJis; (c) 47 '9LVo;(d)
is from 14'7 psia'
3. In an ideal Diesel engine compression
Btu/cvcle are added as heat'
80"F, 1.43 cu ft to 5d0;tt* i"hi" tu
and find (a) T" V2' T3'
Make computatio,', f* cold-air standard
and (d) the hp for 300 cvcles/
v3, Ta, and pn, ft)
mrn.
ft3' 2113:l' 0'1&6 ft3' 890'I
Ans. (a) t4?9"R,0'1152
gt"; (e) 60'637o' 39'9 psi; (d) 68'
"ii
a";.*Ai;.idig
i.pili
029?qm'hl:9:*
Operation of Compressor
Discharge
Di5charge
Valve
Intake
Valye
Compressbn
w;i;;""Jp-'
zi.ipui^;(Ujg'Z
l rtruenlionol Diogrom without Clearance.
Fig. 18.
v
Conuenttonal Diagram witn Clearance.
hp
eycle with the overall value
4. For an ideal Dieselgi'g
and p,"'
kPa'
1.33.' r,- = 15, r. =2.l,Pr=
^Anr. 35-89 kPa, 602 kPa
of k =
find P2
5. State 1 for a dual combustion engine is pJ = 1 atm
t, Joo.g;Cfrn = 18; a! th9 "i*{*::"Y?L::t:*",?fr
;t;J,"o;;J;ilp*til" i' zogr kPa'-r" = 1'5' tsase on l kg/
with k = 1-31,.deiermine 1")!l-^P:1
;ilil;i-;r standard
(b) p,
andr at each corner point on the
;;;;i;;.""ce,
v,
(c) W, (d) e, and (e) P-'
ilJ.*-a);.EEq";&) 0.e443 m, Q'!szjo^3i *9q
;;4.; n, i ilio.zK, 0.0?869 Ti' ?^19e;3.*
(e) 900
f.p"pZO.g K; (c) 803.5 kJ; (d) 57'a3%;
114
Fig. t9
Figure 18 shows a conventional indicator card for
a compressor without clearance. As the piston
starts the stroke 4-r,
the inlet valve opens and gas is drawn into
the cylinder arong
[he line 4.'1. A-t point 1, th; piston starts
ttr"
ui"nr.", u,l
va ves being closed, and the gas is compressed"e1,r*
along the curve
t-2. Atz,the discharge valve opens und th";;pGfigas
is
<lclivered to the receiver.
The events of the d"iagr"m with clearance are
l'lrose with no clearance, except that
the same as
since trre piston J* ,rot
lirrce.all the gas from the cylirrdu" at the pr"rrrrr"-o.,
tfr*
rcmsifilg gas must re-expand to the intake p"urr".*,
irL*r,
it 4, before intake starts again. without
clearance, th* ioi r-o
Il5
r^, p,V,,
Preferred Compression Curves
(lob) (6)
"'=f;4= (ozmtGoo = 5.722 kg/min
The work necessary to
drive the compresor decreases
as the value of n decreases.
Polytropic compression and
values of n less than k are
brought about by circulating
cooling water.
(a) Isentropic compression
f- r-r
w- E# I rp,t- -l
Llp;j
T
I
Comparison of work for
Isothermal and for Isentropic
=
Compression.
(1.67) (105) (6)
1-1.67
t.67-
T_
'630y1 rsz
ffi
=
I
105,
- 1652 kJ/min
Another solution:
Heat Rejected
k-l
The heat rejected during compression 1-2 is,
Qr-, = mrcr, (T, - Tr)
T2
l&l*
= (300)
--,r
^t
lP,
I-'J
= 615.6 K
I
w = -AH = -fi'c e (T2 - Tr)
Problems
1. A rotary compressor receives 6 m3/min. of a gas (R = 410
J/kg.K,c- = 1.03 kJ/kg.K, k = 1.67) at 105 kPa, 27"C and delivers
it at 630 LPa. Find the work if compression is (a) isentropic' (b)
polytropic with pvt'r = C, and isothermal
Solution
= - (5.122) (1.03) (615.6 - 300) = - 1665 kJ/min
(b) Polytropic compression
w =+Fffi.,1*
=
vf=
Tr=
Pr=
Pz=
6 m3/min.
27 +273 = 300 K
105 kPa
630 kPa
I
f-
(1.a) (rOs) (0)
1-1.4
l1f3gl
1.4-l _.1
" -11
=
- 1474 kJ/min
Another solution
#
iogol'n
Tr=T, l-p]
= 300
_ l.,l-l
LEJ
118
"'-t
F'ql
= 500.5 K
il9
off = !#}.r-ffi
c- l'oq = 0.6168 ry
cv =f =
kg.k"
L.6z
rh' =
cn= c [-t -rr-l= 0.6168 I r.oz - r.el = 4.41G8 kI
"Lr*l [-T:T-.a J
[sF
r,=r,
=
I
I
\if =
-afr* 6=-th'co(Tr-Tr)+ fi'co(Tz-Tr)
= -(5.L22) (1.03) (600.5 - 3oo)
+ (5.L22)(-0.4163) (500.6 - S00)
_r.32_r
L*J'=(E4o)
L#t= =64r.eeR
s c"v U-qJ
k-l -=(o.lzr4)' fILl
co =
Ll-tful =-o.oaze ffi
AH = drbp (Tz
- Tr)
= (22.05) (0.24)(641.9 - 540)
=-1486kl/min
= bB9.B Btu/min
a -nqTr-T,)
(c) Isothermal compression
= (22.05) (- 0.0429) (er.g _ 540)
w = p,tf r"[*-l
LP?-i
=(105)(6)rn
n r*
= 2*.o'rb/min
= * 96.4 Btu/min
o* = m'* u'J
tffi
ffi
= - 1129 kJ/min
2. A centrifugal comprcssor handles 300 crr ft per ninute
of air at t4.7 psia and 80"F. The air is compressed to 80-psia.
The initial speed is 35 fps and the final speed is 1?0 fps. If the
compressionis polytropic with n = 1.32, what is the work?
Solution
6 =#*ar(+ali+W
w = Q-aK-aH
= - 96.4 _ tZ.Z_ bBg.B
=
f;= 300 ctu
Pr = 14.7 Psia
Pz = 30 Psia
Tr=80+460=540R
u, = 35 fps
u, = 170 Ss
- fl47.gBtu/min or _ lb.2g hp
Volunetric Effidiency
Conventional volumetric effciency
=
ffi
n,=$=kX
"VDVD
Displacement volume Vo
l,he piston in one etroke.
is the volume swept by the
face of
l4r
1
The clearance ratio or per cent cleararrce, c =
t,
then,D"=1+c-c [+-]t
Free Air
Free air is air at normal atmospheric
conditions irr n
particular geographical location.
Problens
LP'J
If the compression process is isentropic, let n = k.
r' j twin-cylinder, double-acting compressor with
a crear=
ance of ,vo handles 20 ms/min. of
nitiogen from roo i.i", az"c
Uo
ggrypression.ana urp""Jio" .r" p"fyt""pil
!Z!
n = 1.30. Find (a) the work, (b) the
hialre5ected, and (c) the bore
vo ={ortN
.itf,
^H*.
and stroke for I"b0 rpm and UD
where:
D = diameter of piston
L = length of stroke
N = number of cycle completed per minute
N = (n) (1) (number of cylinders), for
single'acting compressors
N = (n) (2) (number of cylinders), for
n
= f .gO.
Solution
PVt's -
double-acting compressors
= compressor speed, revolution per min., rpm
V; = 20 m3/min.
"
= 100 kPa
= 725 Wa
Tr = 37+273=Bl0K
Pr
P2
e=\Vo
n = lbO rpm
IID = 1.39
A single-acting compressor makes one complete cycle in one
revolution.
A double-acting compressor makes two complete cycles in
one revolution.
(a) W
=T#[A* -_l
Fie. 20. Single-acting Compressor
, Pision rinRs
7l ,''"on.
Connecting rod
nk pin
,-- Crank
! Crankshaft
-J/
Crosshead
Wrist pin'
t
L
Crosshead guard
Y
= -5023
mrn
(b)n, =1+c*c l-pJ F
l!-,J
Fig. 21. Double-acting Compressor
722
l2:l
I
= 1 + o.ob -
(o.ob) lzzq-l
p-T 3ld 381 mm, respectivery with a percentage cre'r'rrr.o
5?o, rf su'oundins air ar* it r00 kFa
zi-.c *hJio 'f
fi
tt,,,
compression and expansion processes are pVr.s
""a _ C. Dutor,r,,,,u
(a) Freg air capacity in mtZs.
iU) power of the **pr"rro" i" f, W
(ME Board hoblem Oct. 19S6)
-
"'Llo0l
= 0.9205
n.' 20
vo=n'.,=o8Do5= = z+.ss
4
Solutian
P, = 100 kPa
T =293K
t, = Vo * V, = Vo + cVu = Vo (1 + c)
25'60 4
= (24.38) (1 + 0'05) =
-T
*,=*=#Hffi=27.''*t
It
C= f%o
P=$SSmm
L = 381mm
+. n=150rpm
Pr = 97.9 kPa
Tr=300K
r
Fz{lst = 48s.7 K
rn
,, -= t, I-Cl+ : (s'o)
\!,rvl
[ool
l_n, |
o, =
."ffi $.7442)Fffi#:l 4'4b'#
=
=
(a) n" = 1 +
6r-, = rhrc" (T, - Tr)
[#J* =
I + 0.0b-(0.0b)
vD =-tDpLN =f, {o.essft0.Bsl) (r50)
= (27.83) (-0.2456) (489.7 - 310)
Y
= {ZZsmrn
(c) vo ={nrlN =tD',(1.3 D) (r50) (2) (2)- 612.6
c-.
= s.osz
V;= (n,) (Vo) = (0.9094) (b.6bz) = 5.r+a
O'#
o. = vr
F,]hl
= (b r44)
m]*=0.e0e4
-'
#
t+rtffi = 4.els#or 0.082$
24.38= 612.6 D3
(b)w =T#'tre,J*:,]
D = 0.3414 m or 34.14 cm
L = (1.30) (34.14) = 44.38 cm
;
t
ri
2:. A single.acting air compressor operates at- 150 rpm with
initial condilion of air at 97.9 kPa and 27"c and discharges the
air at 3?9 kPa to a cylindrical tank. The bore and stroke are 355
ll
;
= (1.3) (97.9) (b.
1- 1.3
t26
I
KJ
= - 800.3 mrn
or 13.34 kW
W=
"-
and discharges it at a pressure of 85 psia. The air handled is
0.25 cu ft per cycle measured at discharge pressure. If the
compression is isentropic, frnd (a) piston displacement per
cycle, and (b) air hp of compressor if rpm is 750.
(ME Board Problem - March 1978)
Solution
Pr =
,Pz =
_1J
=
= 96hp
3. A single-acting air compressor with a clearance of 6Vo
takes in air at atmospheric pressure and a temperature of 85oF,
-IF-to,/
'?i#iffifiea- [ia,z/ 'l
4' A single-acting compressor has a volumetic
of 87vo and operates at 500ipm.
effici'rt,y
Il trk"r in air at 100 kpa nrrrl
iil
esc\argel jr ar 600 kpa.
ai, rraodted is o .i * p,,.
l[CA!
mrn
measured at discharge condition.
If the comii#io' i,
isentropic, find (a) piston d;;pi;;;;;t
per
stroke in cu m, and
(b) mean effective pressure
in kpa.
(ME Board p"otrem :ep"riliilal
Solution
14.7 Psia
85 Psia
0.25 ft,3lcYcle
%=
T, = 85+460=545oR
= 100 kPa
600kPa
=
fz
V2 = 6 ms/min
Tr = 3O+273=B0BK
Pr
(a)r,=r,H* =(545) [q* = 900"R
(%###
-' #,,
=
=
_J
= o.o68z4 ib/cycte
ni RT. (0.06374X53.34X545)
=o'87i4ftvcYcle
v,=ff=ffi
D"=L+c-c [r;tt -1+0.06-(0.06) l-ar lfi = 0.8499
LP'-J
h47l
ra) vi
=o,Fno =
+
" q, o.87
126
= 21.58 m3/min
mrn
T'
_ 24.8mrn
=3f;3# = 1'o3o n3lcYcre
tbl V; = (0.8754) (750) = 656'6 ft3lmin
Lrooj
v^ =&=?rs = 24.8 It
UOO
v"
(6) looo l'''
(h) w=
,-L
0.M9G
stlgkes =
stroke
mln
++lZ+r+
r-k l\p,/ -
I
t",,7
_@ffi@Kml*_!
(b) Barosetric pressure at 6000
New intake pressure, pr*
Y
= -sosa.g mln
= ll.Zg psia
New discharge pressur€, pz*
bob3.g
n
rn=_li{_=
vD 24.9 = 208.8 kpa
ft = 1r.?g psia or 23.gg in rlg
= g0.B + ll.Zg = 102.0g psia
New volumetric efliciency,
DvN = 1 + o.o6
6. A compressor is to be designed ntith 64o clearane tn
handle 500 cfin of air at L4.7 pcia and 70pF, the state at the
beginning of compression stroke. The compression is isentropic
to 90.3 peig.
(a) What displaoement in cfu is neessary?
iU) f tU" co*presso"is used at an altitude of 6000 ft and if
the initial temperature and dischargp pressure remain the
same as given in (a), by what percentage is the capacity of the
@mpressor reduced?
(c) WUat snouldbe the displacement ofacumpressor at the
altitude of 6000 ft to handle the sa-e mass of air as in (a)?
-(0.06)
r
ffiff"o = o.77e|
fr:
New capacity, Vi* = @.7795)(6tB)
= 472.8mln
Percentage decreased in cqpacity
5010:j[r?.8
(c) pr = 14.7 psia
Vi = 500 cfu
=
= 4.44Vo
R, at 6000 ft = 11.78 psia
T, at 6000 ft = 530"R
Tr = 530'R
Solution
q
14.7 psia
90.3 + L4.7 = 105 psia
500 ft3/min
Tr
70+460=530"R
Pr
V, at 6000 ft = capacity to handle the same mass
of air
as in (a)
vD at 6000 ft = displacement volume to handle
the same
mass of air as in (a)
-,=#,=
Vl at 6000 ft = q+{H00) = ozs.g 4*
1+c-"[fl*
Vo at 6000 ft =
ffi= 800.4 g
*', lr0ilfr = 0.8156
=I+0.60-(0.0_.1;14.fl
y-=Yt==5=ry==
-=orgq-
'o- o"
0.91b6
min.
l2{,
Compressor EfficiencY
Adiabatic overall efficiency
is
ideal work
In general, effrciencY = actual work
^{. Mechnnical EffrciencY
The mechanical elficiency of a compressor is
- indica@
n*
If the compressor is driven by a steam or internal combus'
tion engine, the meehanical efficiency ofthe compressor system
is
"-'-
indicated work of compressor
indicated work of driving engine
B. Compression. EfEciencY
adiabAtic ideal work
,,oc%
.. =
Isothermal overdll efficiency
is
- isotherlpel ideal *"*
or%
o^,
Polyhbpic overall efficiency
no, = (n-) (n")
is
= Sltpolvtmpic
ideal worli
Adiabatic compression effieiency is
adiabatic ideal work
-c - indicated work of compressor
S-
Isothermal compression efficiency is
isothermal ideal work
-'t -- indicated
work of compressor
Polytropic compression effrciency is
oolvtropic ideal work
= indicated work of compressor
"p
c. Overall Effrciency
Overall elficiency is
no = (mechanical efficiency) (compression efficiency)
130
Indicated workjs the work
done in the cylinder.
Brake work or sh"n *o"r.lr
tn" i"* delivered at the shaft.
Adiabatic compressio"
ciencycommonryused.c;p;;i;;;ffi
"E"i"i.r r, ,t " compression effrmean adiabatic compressi;
"tr;;y;h";;;*,wo.,td
"ffi";;;
Problems
J
1. A twocylinl":f:gl:__actils air
compressor is direcily
coupled to an electric motor
*rrrririg at 1000 rpm.
Other data are as follows:
Size of each cylinder, lbO
mm x 200 mm
Clearance
f OZ.of Jirpfacement
"?\-9,
Exponent (n)
for both comp.e5ri""
process, 1.6
Airconstant,k= t.{
Air molecular mass, 29
""J
*-expansion
Calculate:
(a) The volume rate of air delivery in terms of standard
air for a delivery pressure of 8 times ambient pressure
under ambient conditions of 300 K and 1 bar.
(b) Shaft power required if the mechanical efficiency is
81%. (ME Board Problem - April 1984)
Solution
2. A 12 x 14_in., dollle-acting air compresor
with 6.6*"
clearance operates at lS0 ,p*,
ari*ing air at l'.'pnin en'
dischargin g.it at 62' p; i;thu .91
9,u^ _":"d
n"".rion an d ex pH I r,
sron
processes are polytropic
with n = l.Bi. Determini i"l tfru
volume of free air irirnarea
if atmospheric condi.
tions are 82'F and r+.2 psia,
,r,,
indicated work of the-.o-p."rror
iitit" compression e-fficiency
is 87Vo, and (d) the ideal *ort .
pJ;i;;e,
?tiil t";;fiffi;i"l
Solution
pr = lbar=100kPa
Pz= g
Pr
P" = 14.7 psia
T = 82"F+460=542"R
Pr = 14.5 psia
Tr=85oF+460=b4b.R
o
(a) vo =tryLN ={to.rso)'?(0.200x2x1000) = ?.06e
#
I
tr, = I * .
-.pf
Lru
r
(a)n"=1+c-c lP,-LI'
= 1 + 0.10 - (0.10X8)t = 0.?332
l&i
Vl= rr"Vo = (0.?332X7.069) = 5.183#ot 0.0864 m3
vD =4'-D'?LN =
S
(b)w=T#R)*
-l
(1.6) (100) (0.0864)
1-1.6
Shaft power =
27.2L
ffi
= r.ob5 - o.obb
m]* =
0.8e2,4
t H' frq (1b0x2) = 274.e crm
Vf = (o,) (V;) = (0.8924) (214.g)=
248.8 cfm
(v/
(P,) (r")
(542)
9'" - --In"Jnt-= 84!€I(14.s
-liz:7t6) = 240'6 cfm
[t,*-t]
= 33.59 kW
= 27.ZlkW
(b) \ir = Vn *
% = Vo + cVo = Vo(l + c)
= (274.9) (1 + 0.0bb) = 290.02 cfm
.
m, =
P,V,
1i1; =
Q!.5) O44) (2s0.02)
(53.34) (545)
= 2o.ss
lP
mln
r, = r,
[t]"
co = c"
= 545EH
F=; =
(o.tz14)
\51
= ?88"R
ftfrfl= - o'3025ffi
3. There are compressed g.4g kg/min of oxygen by a g!,0€
x E5. 5 6-cm, double -actin g, motor d"irre' co-p"essor
oporetlnf
at L00 rpm: These data apply: Fr = 101.9b kpa, t, Z$.ZA
inE
=
p,'310.27kPa. compression and expansion
polyt"opic wt&
n = 1.31. Determine (a) the con-uentional volumetricefliclency,
"t"
heat rejected, (c) the work; and (d)the XW inpui by tfd
9ltlt.
driving motor for an overall adiabatic elficiency of ittir.Solution
= (20.83) (-0.03025)
(788 - 545)
.,-o'' Btu
= - IDO.I ::::'
mtn
OYt'a
*C
D'=
.br
(c) iV,"",, =
k4{&fiq* - rl
r-K L\pr/
fr'=
4)F_V:
'vo '
-J
Pr=
Pz=
Tr=
L = 0.3556 m
8.48 kg/min
101.35 kPa
310.27 kPa
26.7 + 273 = 2gg.7 K
(1.4) (14.5) (144) (245.3) t7g) * -r
=@lrr+.rt
)
BtP
(a) v, =fD,tN
= - 1185 mln o" -27.97 hP
adiabatic
,n.i@
vf=+=W=6.bu#
ideal wor!
o"
- -*:!.
(b) 12 = r,l+4-
=ryreil{-']
(1.34) (14.5) ]44)(245.3) lTsz-t'*g -
=@l\r+si
Blu or - 27 -29 hP
= - 1157 mln
#
0.9227 or gl.z7vo
vo W^
"=*2.068 =
Indicated work =H#= 32:15 hP
(d)w
=t0.Bbb6), (0.sbr6i (100) (z) = 2.068
-
il
Lrrl =
eee.7) t!-lq€fl+#= Beg.b K
." =.,p3J = (0.6beb)
]
L101.351
H$H
= -0.1808 kJ(ks) (K)
D"=l+c-c F;r+
l-F;l
135
0.9227=1+c-cl
work input by the driving motor
I-gro.2?l#
Ll0L5il-
Multistage Compression
c = 0.0573 or 5.737o
r
Multistagingis simply the compression
more cylinders in place of a singffitinaerof the gas in two or
como"Jrro". l, iu
usedin reciprocatingcompressors
in order to(l) save power, (2)
limit the gas discharge temperaru"q
differential per cylinder. 4 ------r -
rrt3
V, = Vo (1 + c) = (?.063) (1 + 0.0573) = 7.468 -*
mrn
,- p,v, (101.35\ tn aRR\
= 20.41 hW
;;?JiilililJ;;:r"""
kg
'l',=ffi=idffiffi=e.717 ;ff
rvater
Q,-, = rhrcn (T, - Tr) = (9.717) (-0.1808) (390.5 -ZggJ)
in
water out
I-r
mln
= _159.5 ^1
(c) W= nth'RT,
T.n
+
l(tl -rl
IIP cyUnder
= (131) (8.48) (0.25ee) (zss.7> [7 srO.ZztttJil - .'l
Tl\lolsb/
-:l
Y
= -846.1 mln o" -14.1 kW
(d)w,"*=qPR)*-!
[121s.2711fH
=(1.3eb) (8.48) (0.25ee) (2ss'7) l!0135i
g-
= -..309.b mrn or -14.49 kw
'
'^oc
-
adiabatic ideal work
brake work
' = 14'49 = 20.41 kW
DraKe wofK
0J1
Cards,
IiS ?2. Conventional
'rwo-Stage,
No pressure Drop
v
_Fig.,23. Conventional Cards,.
Two-Stage, with pressure Diop
The figures abov-e-show the bvents
ofthe conventional cards
of a two-stage machin", *itl ifr*
nigh pressure (Hp; srpe.posed on the low pressure (Lp).
suition il th; ilp.ji"a*"
begins at A and
pry"Vai;
in. Compression t-2
occurs and the gas is
The discharged gas
passes through the $yharc.ei
interc*te"
cooled by circulating
water
G
Ii"*r
"*ir-".
through the interc*t." ".rd"is
i"U"r. Co"uu"tio'Jfi,"it
i,
t:f7
entering the
Pr = P' --
el
rrpcvrindeTiu.ir,?,u-g*g;;^iil:;tt*mi*""1i$
assumed that the
gas leaving the intercool:l
'*
Hft
*u*kil*t=P**T'*'-**fr
r
must reexpand F-E
fromtheGuuv^'--ot Iearance and
Heat Tlansferred in Intercoolor
The heat rejected in the intercooler is'
c
; each cylinder because
pe (LP cvlinder)'
iirp tvu'ii"'i"*a
cylinder
W of the loLPlessure
Pressure cYhnoer
!\f =
=
+ W of the high
Problems
of multistage
adjust ll:.o*tution
to
practice
Itis common
works are donejn the
work tbr comcompressor, *o
tr'uiipii#;;*y:f
ti":*imum
cvlinders, p"u"""Jiil"t oru
"^"'otf
*: :liiiT:H:ftff#Til:
"
pressine . gi*'u" q;"iG
of P, = Pr =.P*' weltave
p,= yTF*'-
i,
i
I
for minimum
intermediate pressure
work
sane' tlre t?la\work
of eachcvlila"iillh"
work
cvlinder' or
the
since
the workin each
#;;;tJtwice
for the two-stage
-1\
;1
='+Pfel*
1-n
l9'/ r
w= "iffiLft,? _J
2nm'Rr,f-1P,$
A pressure drop
"ide
in the intercooler could
t*o-ri"gr.oilpr"rrion when the intercooler cools the air
to the
tions
: of part (b).
ial liow much heat is exchanged in the intercooler?
*p'"ttiin efficiency of 78Vo' what
(e) For *
""*"ff-is required?
driving motor outPut
#trf,*{=+[tlt'i
I
l.Therearecompressedl'1'33m3/minofairfrom26'7"C'
are 8Vo'
L03.42kPa to 821.36 kPa' All clearance
(a) Find the isentropic power and piston displacement
for a single stage cornpresslon'
required
--=ft)-u*ing
the,"-, a""t , nnd the minimum ideal work for
initial
---6 temPerature.
Fi"h trr" di-splacement of each cylinder for the condi-
l;,,h toitrat of the HP stage' or
where: P, =
Qt" = m'cn (T, - T')
the intercoolor
where m' is the mass of gas passing through
byifrgif .ili"der and delivered bv tho
i Jro tfr" mass clrawnin
HP cylinder).
l#,Kkl*-1.#[ft]*-tr
#T- = i- ;d
l'rtrHFllrlr tllrtll
Solution
vf=
Pr=
Pz=
rT
rl
-
11.33 m3/min
103.42 kPa
827.36 kPa
26.7 + 273 = 299.7 K
be spread on each
oi this ideal value'
Pressure droP
Pr=P,*--T--
139
r =IilFR)*
_(1.4) (108.42)
l
ftzgz.szttft;l
N-mtz-t-il-J
(i,l.BBi lTga.BqtY/
1-1.4 -
=
(1.a)11s3.a2) (11.33)
1-1.4
L\ 1o&42l -
|
- 1416 #
mln o" -28.6 kW
Tqtal work - (2) (23.6) = -47.2 kW
- 3327# ot -55.45 kw
(c)n"=L+c--c l-&1 + =1+0.08-(0.08)
= 0.9119
LP'l
tr"=1+c-c
vnrp=#=## =12.42#
' lezz'361.r
=1+0.08-(0.08)h1ffi1
*' = n#, =,+ffiffi$?, = 18.62
tr. 11.33 _r^*o *t
vo=#= mffi
-'"'"Y min
,l-=- -,BT€ - (13.62) (0.2q2q81j299.7) 4.006 T3
=
'3
292.52
Pa
mln
V;
r/
vnur
(b)
4006
rn3
=;jf = ffig = 4.393;fr
p
(d) Qrc = th'cn (Ts
Pr
Pa
103.42 kPa
- Tr)
(13.62) (1.0062) (299.7403.4)
827,36 kPa
(e) Outpur of driving motor
:
p,=y'[];=@
**=+#F)*
#
292.52kPa
I
= _ 1427
l&I-
min
=!7:? = 60.5 kW
0.79
lb/min of air from l4.B psia and gb,r to a final pressurer tf I gn
psia'. $e lormal barometer is 29. g in. Hg and the tempern t rr ro
is 80"F. The pressure drop in the intercooler is B paiand th,
temperature of the air at the exit of the intercooler is g0,,1., tho
speed is 210 rpm and pVt.er = C during compregeion und
expansion. The clearance is E% for both cylinders. Ths temperature of the cooling water increase by iA F". Find (a) the
volume offree air, (b) tlie discharge pressure ofthe low pr*rruro
t4l
cylinder for minimum work, (c) the tempprature at discharge
from both low pressure and high pressure cylinders, (d) the
mass of cooling water to be circulated about each cylinder and
through the.intercooler, (e) the work, and (f) if, for the low
pressure cylinder, IJD = 0.68 and if both cylinders have the
sam: stroke, what should be the cylinder dimentions?
m
90lb/min
po
(29.8) (0.491) = 14.63 Psia
To
80+460=540oR
Pr
L4.3 psia
Tr
90+460=550oR
Pr
185 psia
Low pressure cylinder
=
(143t(144) -'
ffi|$)
=
r-* I + 0.0b{0.0b) Fzslt
c-clfil
=
= 0.9178
vn =*=
rB98 cfm
0.9173 =
' ru ;€g
V, = VD (1 + c) = (1393) (1 + 0.0b) = L46Z cfm
;, =$1f= tra,'f*ggxpzr = 1oB rb/min
Heat to water = Heat from air
1Zg2 cfm
(rh*) (c,") (At*)
tfffi#ffi#P = 12Bo crm
(b) p- = ilFm, =J043) (185t= 51.4 psia
pz= 5!.4+&= 52.9 psia
rh*=----414-l f Btu\ (18F")
\6F/
+
r42
[*{*
= (bbo)
t#.f
#
Intercooler
,n ,rr
l-p, |
(550) [Bzd #'
''"" -= ,767 oR
t,
- ,,trR^\
- t,b:l
=
=
Lffi]
=37.5 lb
mrn
High pressure cylinder
.ll"
(c) ps = 51.4 -9= +g.gpsia
= er.z
678 Btu
a* = BZ.E
r, = r,
[a s]
= -675 Btu/min
6'RTr _ (90) (bB.B4) (bb0)
Pr
dhi
Q"z = frrc" (T, - Tr) = (10S) (-0.0302 ) (767 - b50)
l" Vi*:--l
v" =
*-ffi = (0.1?r4)Htf = -{.0302
D" = I +
Solution
(a) Vr=
,
(d) c, =
'#' = 7G7'u,
Q," = rir,co (\ - Tr)
= (90) (0.24) (bbo
- 767) =+osz Blt
mln
l4lj
mass ofcooling wate" =
{y
4L2.3 D2 = 400.5
lb
D = 0.986 ft or 11.88 in.
= 260.4 min
L = 15.01 in.
(e) Low pressure cylinder
Three-Stage Compression
nrh'RT,
\i/.
"LP -= l-n l7gt+ _ il
L\prl
]
(1.34) (e0) (53.34) (550) 1-t52.effi
=@l\ra.si
-jl
'1
=
-
IP cylinder
LP cylinder
B!t'
b2G5
mrn = -L24.2hp
Fig. 24. Three-Stage Compression
Total work, fr = (2) (-124.2) = *248.4 hp
pV=C
pV"=C
(0 Low pressure cylinder
py
y^D44
=3.D2LN =!pe (0.68 D) (210) (2) = 224.3 D3 cfm
Condltlone for nlnlnum rork
1) wr,p = wrp -
PV"=C
2-P,
224.3 D3 = 1398
-PV" = C
L = (1.84) (0.68) = L.25 ft or 15.01 in.
High pressure cylinde
":
p,
(49.9) (144)
e)TS =T3 =Tl
I
D = 1.84 ft or 22.08 in.
v _ fi'Rr3 _ (eg) !5giq1) (?50)
%p
Fig. 25. conventiorrut cu"arlThree-stage, No pressure Dr'p
,,1'T,r,
il =,,p'or. f&\.'-l = --1*-L\&i
Zluf+If+l+- l=-I-n-[\d/
l-n l\Pr/
-l
nm'Rro
11
= 36?.4 cfm
\r'D --i;n-,- gal = 400'5 cfm
Ufr*
V^u44
=ID2LN =3D2 (t.zb) (210) (z) = 4Lz.g D2 cfm
Pr P" Pn
P, =F, = P,
I
P, = (PrPr)
2
P, = (P,Po)
2
(1)
I
(2)
ft)T3-Tr=BgbK
Solving equations (1) and (2) simultaneously,
n-l
p,=\/ir'p, and p, =t6trJ
T
/&\ -- =,ruc /3ss'olff/ = 411 K
'z = Trrgf/
"no (ib3;)
1-n [gf#-il
l\P'r l
3nm'Rr,
Problem
Air is compressed from 103.4 kPa and 32"C to 4136 kPa by
a three-stage compresor with value of n = 1.32. Determine (a)
the work per kg of air and (b) the heat rejected in the intercool-
Heat rejected in the first intercooler,
Qrc= m'co (\ - Tr)
= (1) (1.0062) (305 - 4rr) = -106.2 kI
Total heat rejectred = (Z) (_t06.7)
= _218.4 kJ
ers.
Solution
p
m
lke
Pr
103.4 kPa
4136 kPa
32"C + 273 = 305 K
Po
Tr
(a) p, = (p,,pu)*= fioa.aX (4136j#= 353.6 kPa
,.,
_ 3nm'RT, 7&.*
vY-
l-n
-1
l\P,)"-1.J
L.IZJ
-
(3) (1.32) (1) (0.28708) (305) l/353.6\ r'32-11
r
It|
1-1.31
_1
l]103.4/
I
= - 376.2 kJ
t47
IT
il
Review Problems
5. From a testjf an.air compressor
,t
'
i
1. A reciprocating compressor handles 1000 cfm of air
measured at intake where P, = 14 psia and t, = 80"F. The
discharge pressure is 84 psia. Cdlculate the workifthe process
*
of compression is (a) isothermal, (b) polytropic with n L.25,
and (c) isentropic.
Ans. (a) -109.5 hp; (b) -131.7 hp; (c) - 143 hp
6. An air compressor with a clearance of 4Vo compresses
;
I
I
f
t
cm
3. A double-acting compressor with c = 7Vo draws 40 lb per
minute of air atl4.7 psia and 80"F and discharges it at 90 psia.
Compression and expansion are polytropic with n = 1.28. Find
(a) the work, (b) the heat rejected, and (c) the bore and stroke
for 90 rpm and UID = L.25.
Ans. (a) 77.68 hp;(b) -1057 Btu/min; (c) 18.96 x23.70
in'
4. A 14 x L2-in., single'cylinder, double-acting air compressor wit}'5.5Vo clearance operates atL25 rpm. The suction
pressure and temperature arc14 psia and t00oF, respectively.
The discharge pressure is 42 psia. Compression and expansion
processes are polytropic, with n - 1.30. Determine (a) the
volumetric effrciency, (b) the mass and volume at suction
conditions handled each minute, (c) the work, (d) the heat
rejected, (e) the indicated air. hp developed if the polytropic
compression efficiency is 75Vo, and (f) the compression effrciency.
Ans. (a)92.7Vo;(b) 247.8 cfm,L6.72lblmin; (c) -18.93
hp; (d) -175.7 Btu/min; (e) -25.24 hp; (f) 77.42Vo
l4 f]
o-ut"i,r.,a,
capacity, 800 cfm; suction it t+.2 psia;
disch;d;;;
iio pri,,;
indicated work of
compressor,'i5S
frp;
indicated
work ol.
lhe
steam engine, IZ2 hp^aCal..rlute
(u) tt u.";p;;i""im.i"n.y
and (b) the overall efficiency.
Ans. (a) 90,06Vo; (b) Bt.t6qo
14.73 ms/min of airfrom gz kpa, z7ic
2. A twin-cylinder, double-acting, compressor with a clearance of \Vo draws in oxygen at 450 kPa, 17"C and discharges it
at 1800 kPa. The mass flow rate is 20 kg/min, compression and
expansion are polytropic with a = 1.25. Find (a) the work, (b) the
heat transferred, and (c) the bore and stroke for 100 rpm and
llD = 1.20.
Ans. (a) -40.23 kW;(b) -829 kJ/min/ (c) 2L.71x25'76
driven direcily by a
rt"uq engine, the following data and resurts
**
I'
to 462r<pa.If the overail
adiabatic efficiency is 6rvo, d"t"r-i.r"
the indicated horsepower of the directly connected
driving steam engine.
Ans. 91.89 hp
7. Methane is compressed in a two-stage, double_acting
compressor which is electricaily driven
at rbb rpm. The row
pressure cylinder (3_0. E x Bb, b cm)
receive, O. S6
pe r-mirrute
of air at 96.b3 kpa,4B.B"C, *Jtfr"
""
x
!20..3 35.5 cm) discharges til" -"th* e at 7t7.06 kpa. The
isothermal overall efficiencyi szq,%-.inanu
and the kwoutput
of the raotor.
;
hish;;;iJ.r]ioa""
" Ans. 8O.02Vo,90.g6Vo
+
i,
i'i
A tw-ostage compressor with a clearance
of *Voreceives
^^ ,9:
80lumin
of air at 14 psia and 8E"F and dcrivers
ii
pri".
The comp,ressions
1.g0, and"i1io
the inter_
cooler cools the air Td-polyt*pi;;th
"
Uact to ar"i.. ri"Jfrl the worL, (bj li" rruut
transferred in the various processes,
i.ith" ;;;il,f#;^.irer"_
gtage m achine, (d) the correspondiog percentage
s avin g for the
two-stage_machine, and (f) tle
water to be circulated
_
-asiif
through the intercooler if its t"*p"i"l.rre
rise is 15 F".
Ans. (a)-17_1.0 hp; (b) -Soz.S Bru/min;
l.l _igie stu/
min; (d) _196 hp; (e) t2.4EVo; (ft igo lb/mi;
8 The Brayton Cycle
Operation of a Simple Gas T\rrbine power plant
Combustor
To
'*'/
Generator
lr?:
Compressor Turbine
fi:-r-:i-::::i::a
1.,:".:':::Sinki.,'
r...: r:: i : :r't..i: ... .:l
F------
J
Open Cycle
Q*
Closed Cycle
Fig. 26 Diagrammatic Layout of Gas Turbine Units
Air continuously enters the compressor 1. After
compression, it enters the combustors, som'e
of it going u"o,rrra tfru
outside of the comhrrstion chamber proper
and the remainder
fulnish]1* oxygen for burning the fieljwhich ir-.orrti*orrrfv
injected into the combustioniha-ber.
Because of their temrise, the gases expand and enter the
ryTlure
turbine
3'
After expansion through the turbine, the exhaustin state
t. ilrt:
atmosphere is in some condition 4. In
an ordinary powor'r:r.t.
arrangement, the work of the turbine W,
is g"*i ,,,r,,,,gt, t,.,
drive the compressor W
delive.
U.ut ,, *,rrL W,, i,,',t,.iu,,,
_and
say, a generator or proptlllrlr;
W, __ W,, I W,. Arr ,,*..il,,,,l H()r(.(,
of power is needed to si lrrt :r
liirH l.rrr.l'r'rrrr. rrrril..
Derivation of the formula for e
Process 1-2:
T =H"=FJT
T2 =
Fig.27. ,Air-standard Brayton (Joule) cycle
.
Tr"an.t
(2)
rok-t = ro Y
(3)
L-2 isentroPic conPression
2-3: constant-pressure addition of heat
3-4: isentroPicexPansion
4-tr; constant-pressure rejectionofheat
Analysis of the BraYton CYcle
Qo = mco (Tr - TJ
Q* - DCo (Tr - T4) =
*nrco (T4 - Tr)
L-l
t=Fl*=
l-p
LF
Ta = Tn"*tt
(4).
k
S = Q^ - Q* = mco'(Ts - TJ - mco (Tn - T,)
Substituttuig equations (2) and (4) in (1).
e = W = mc.(Tr-Tr)-mco(Tn-Tr)
€ =1-
q@
e=
+,-+
1_ rg
(1)
- t2
1
.:=
r-r
r
f
1-#I
"J
e-1- -11 =1-t-
Total compressor work, W"=& -AH
"o*
W.= -mco(T,
rL
where rk =
v
+,
the comPression ratio
rppr= -P, I ttre pressure ratio
Total turbine work,
W, ={-AH
W, - -mc, (T. - \)
W, = mco (T, - T,)
W--_
.,].
Net work, W or W" = W, -
Point 3:
W"
Vg = v2
Problems:
= e.7z)
4.s4 rtsnb
tt.]
L
f+#fl =
Point 4:
Bray-
an air-standard
1. The intake of the compressor of90oF'
The compression
and
ton cycle is 40,000;;;it;sia
turbine inletis 1440"F'
ratio, rr = 5 andth-;;;;;'i""3t11"
is 15 psia Dgterminl ll: ""'
The exit pressure oiiftJi""tine
pressure'
;;;;,;#;al efficiencv and the mean effective
I
t--*
v=v
l&l =@.94)ll+Z8lt"=
.4 ,.LpJ
\^rvrlLlbIz4.Tfttnb
f-,, -lt -l
r
L'rJ
124:Ll
^
r.= r, i+l- = (leoo) l#l'o'=
eesoR
= 40,000 cfm
Pr = L5 Psia
T1 = 550'R
T3 = 1900'R
p4 = 15 psia
r. = S
=5
v2
v1
Compressor work, W = -{o (Tz - Tr)
= - (0.24) (L047 - 550) = - 119.8 Btu/lb
Turbine work, W, - co (T, - T.)
= (0.24)(1900 - 99S) = 216.b Btu/lb
K
Net work, WB = W, - W" = 2L6.5 - 119.8 = g7.2 Btu/lb
n =*,t
Point 1:
v1
(15x144X40,000) _
=
--T5rilx550t
2945lb/min
Pz
T2
I ll4
f
Heat added, Q
2945
vl
13.58
2.72fbs/,b
= -=
rkD= --=- =
142'8 Psia
= Prf**-t = (15X5)" =
= Trr*k-r =
(550X5)1:a 1= 1047"R
= c, (T, - Tr)
= (0.24) (1900 -1047) = 2A4.7 Btu/lb
0.4748 or 47.48?o
= +Qo= V2204.7 =
v, 4o.ooo = 13.58 fta^b
--=
- IiI
Point 2:
v2
= 6751 hp
= W_Q945)
42.4
p.
=g
=
vD= =-luv,
-%
(97.2) (778)
6+7 _ zlz) tt,t,n
_
= 23.89 psi
2' There are required 2288 kwnet from
r,rrr'rrrr* rrrrrl
lirr'prrmpi.g of crude oil from thc Nrrth Arrrrrkrrrr
'11:rs ,.rr,1*, .i*
thc
gg"n?l-r
compressor
scction at
'|'irt'(!r'$
kPr, ltzH ti, rrr* lr*ee
IFE