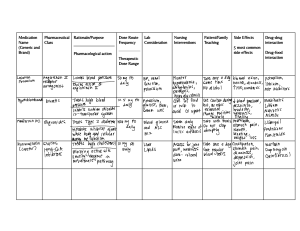
Kimberly Middleton, PharmD PGY2 Oncology Pharmacy Resident UMHM/SCCC Drug Forum #1 – Alkylating Agents Alkylating agents: • Nitrogen mustards (bendamustine, chlorambucil, cyclophosphamide, ifosfamide, melphalan) • Nitrosoureas (carmustine, lomustine) • Alkyl sulfonate (busulfan) • Ethylenimines (thiotepa) • Lurbinectidin, trabectedin Mechanism of action: • Binds alkyl groups to DNA which allows cross linking of base pairs leading to DNA damage and cell cycle arrest • Bendamustine has a benzimidazole ring (purine analog) which demonstrates only partial cross-resistance with other alkylating agents Class effects: • Infertility and risk of secondary malignancies Agent Indications Dosing Dose Adjustments Bendamustine (Bendeka, Belrapzo, Treanda) CLL, NHL, R/R HL, MM (salvage), waldenstrom macroglobulinemia 70-120 mg/m2 Renal: CrCl <30 mL/min not recommended CLL, HL, NHL, iIdiopathic membranous nephropathy, necrobiotic xanthogranuloma, Waldenstrom macroglobulinemia • 0.1-0.5 mg/kg (increase to max of 0.8 mg/kg) • 6-10 mg/m2 Chlorambucil (Leukeran) Hepatic: AST or ALT ≥2.5x ULN or total bilirubin ≥1.5 not recommended Renal: • CrCl ≤50 mL/min: 75% of dose • CrCl <10 mL/min: 50% of dose Toxicities: hematologic and skin reactions Caution with concurrent chemotherapy/radiation within 4 weeks Dosage Forms / Administration IV • Bendeka: Over 10 minutes regardless of indication • Belrapzo, Treanda: Over 30 mins for CLL, over 60 mins for NHL • Concentrations, storage, preparation, and administration vary between formulations Oral tablet • May be administered as a single daily dose • A 2 mg/mL oral suspension may be prepared with tablets PK/PD Emetic Risk Adverse Events Interactions Comments/Notes • Hepatic metabolism via CYP1A2 to active metabolites (M3, M4), and via hydrolysis to low cytotoxic metabolites (HP1, HP2) • Half-life: Bendamustine: ~40 mins, M3: ~3 hrs, M4: ~30 mins Moderate • Myelosuppression (nadir in 3rd week) • Dermatologic toxicity (rashes including SJS) • Hepatotoxicity (usually within first 3 months) • TLS • Progressive multifocal leukoencephalopathy (PML) • Infusion reactions • Peripheral edema, weight loss, constipation or diarrhea, N/V Mod/strong CYP1A2 inhibitors – consider modification (ciprofloxacin, smoking, omeprazole) • Belrapzo and Bendeka are CI in patients with hypersensitivity to polyethylene glycol 400, propylene glycol, or monothioglycerol • PJP prophylaxis • Consider premedication (antihistamines, antipyretics, steroids) for previous grade 1-2 reactions • Complete absorption from GI tract (>70%); reduced with food • Extensive hepatic metabolism primarily to active metabolite Minimal to low • Myelosuppression (BBW) • Fertility effects (BBW) – probably mutagenic/teratogenic and infertility • Secondary malignancies (BBW) • Seizures: with nephrotic syndrome and high pulse doses • Skin reactions No significant interactions Kimberly Middleton, PharmD PGY2 Oncology Pharmacy Resident UMHM/SCCC Cyclophosphamide (Cytoxan) ALL, AML, BC, CLL, CML, HL, mycosis fungoides, MM, neuroblastoma, NHL, ovarian, retinoblastoma, sarcomas, GVHD, HSCT, lymphodepleting therapy prior to CAR T-cell therapy, Merkel cell carcinoma, ovarian, germ cell tumors, PCNSL, SCLC • IV: 75 mg/m2 to 2,000 mg/m2 • Oral: 1 mg/kg to 100 mg/kg • Lower doses in benign conditions and higher doses in transplant Renal (depends on indication): • CrCl 10-29 mL/min: 75-100% of normal dose • CrCl <10 mL/min: 50100% of normal dose Hepatic: • Bilirubin 3.5-5 mg/dL or AST/ALT >3x ULN: administer 75% of the dose • Bilirubin >5 mg/dL: avoid use Toxicities: • Hematologic, cardiotoxicity, hemorrhagic cystitis Ifosfamide (Ifex) Testicular cancer, bladder, cervical, sarcomas, NHL, HL, thymomas and thymic cancers, ovarian 1000-5000 mg/m2 Renal: • Consider if CrCl ≤ 60 mL/min Hepatic: • Bilirubin >3 mg/dL: 25% of dose Toxicity: hematologic and encephalopathy IV/PO • Protocol specific infusion rate • Administer oral in the AM • Ensure patients drink adequate fluids (~2-3 L) with frequent urination during infusion and for 1 to 2 days after • High dose (SCT): hyperhydration and mesna administration IV • Administer over at least 30 minutes (infusion times vary) • Given with hydration and mesna to prevent bladder toxicity (phenylacetic acid mustard) • Half-life: ~1.5 hours; active metabolite ~1.8 hours • Metabolized hepatically to active metabolites (acrolein) • Half-life: 3-12 hours • Metabolism: Hepatic to active metabolites (including acrolein) • Half-life: 7-15 hours (dose dependent) • Excretion: urine • Peripheral neuropathy Moderate to high (>1500 mg/m2 IV) Moderate to high (≥2 g/m2/dose) • Myelosuppression (nadir at weeks 1 and 2 and recovery after ~20 days) • Hemorrhagic cystitis • Cardiotoxicity (high doses – DLT in SCT) • SIADH- (typically with high doses (>2 g/m2) • Pulmonary toxicities • Hepatotoxicity/VOD (age <3 years old at increased risk) • Wound healing impairment • Major substrate of CYP2B6 • Minor substrate of CYP2A6, CYP2C19, CYP2C9, CYP3A4 • Hemorrhagic cystitis DLT (BBW) • Neurotoxicity (BBW): onset within a few hours-days and generally resolves within 2-3 days after discontinuation; higher risk in hypoalbuminemia, renal dysfunction, and high-dose antiemetic therapy • Myelosuppression (BBW) • Nephrotoxicity (BBW)/Fanconi-like syndrome • Hepatotoxicity (SOS) • Major substrate: CYP3A4 • Minor substrates: CYP2B6, CYP2C19, CYP2C8, CYP2C9 • Monitor therapy with moderate and strong CYP3A4 inhibitors and inducers Hydration for urotoxicity prevention • Starting ≥ 4 hours prior to treatment and continued for at least 24 hours after Mesna • Activated in kidneys and binds to acrolein • Dose (as % of cyclophosphamide dose) may vary • Administered as 24-hour infusion starting with treatment and continued for at least 24 hours after • May also be given in divided doses every 4 hours • CNS toxicity / encephalopathy – treat with methylene blue Kimberly Middleton, PharmD PGY2 Oncology Pharmacy Resident UMHM/SCCC Melphalan (Alkeran, Evomela) MM, high dose conditioning treatment prior to HCT, ovarian cancer, amyloidosis, HSCT, HL, regional perfusion in solid tumors • IV: 30-200 mg/m2 • PO: 4-9 mg/m2; 0.150.25 mg/kg; 10 mg (for 4 to 10 days) • Based on actual body weight Carmustine (BiCNU) Brain tumors, HL, MM, NHL, mycosis fungoides, conditioning regimens for autologous SCT 20-600 mg/m2 (higher doses 300-600 mg/m2 in SCT) Brain tumors 75 – 130 mg/m2 Lomustine (Gleostine) Busulfan (Busulfex, Myleran) CML conditioning regimen prior to allogeneic HCT, HSCT conditioning regimens, polycythemia vera, essential thrombocythemia Glioblastoma: 8 wafers (7.7 mg each) implanted intracranially Conditioning regimens: • 130-160 mg/m2 once daily • 0.8 mg/kg every 6 hours or 3.2 mg/kg daily (total dose 3.2 to 12.8 mg/kg) Renal: • No clear consensus, different general recommendations based on route/indication • For conditioning regimens can consider 140 mg/m2 for SCr >2, CrCl < 30, frail, old age • SCT at SCCC/UMHC: 140 mg/m2 if SCr >2 and CrCl <30 Toxicity: hematologic Renal: • CrCl 46-60 mL/min: 80% of the usual dose • CrCl 31-45 mL/min: 75% of the usual dose • CrCl ≤30 mL/min: Consider use of alternative drug Renal: • CrCl 46-60 mL/min: 75% of normal dose • CrCl 31 to 45 mL/min: 70% of normal dose • CrCl ≤30 mL/min: Avoid use Toxicity: hematologic (leukocytes/plt) N/A IV/PO • Alkeran (60 mins stability) infuse over 15 to 20 mins • Evomela infuse over 30 mins (SCT) • Administer oral on empty stomach • Hepatic metabolism primarily via hydrolysis • Oral bioavailability reduced with high fat meal • Half-life: IV: ~75 mins, oral: 1.5 hours Moderate to high (≥140 mg/m2) IV, wafer implant, topical • Hydrolysis, renal elimination Moderate to high (>250 mg/m2) • Achieves high CNS concentrations • Metabolized hepatically • Hepatic metabolism enzymatic conjugation with glutathione • Crosses BBB and achieves high CSF concentrations • Infuse over a least 2 hours • High dose (>150 mg/m2) can cause hypotension, headache, or flushing during infusion due to ethanol content • Oral capsules • Administer on empty stomach to reduce N/V • Only administer enough capsules for one dose IV, oral tablet • IV preferred IV: Over 2-3 hours via a central line • Once daily dosing should be run over exactly 3 hours to get PK labs • Pulmonary toxicity • Myelosuppression (BBW): myeloablation with high doses (≥ 140 mg/m2) • GI toxicity: N/V, diarrhea, mucositis (DLT in SCT-especially GI) • Pulmonary toxicity • Hepatotoxicity (usually transient) No significant interactions • Cryotherapy to prevent mucositis • Give with hydration to prevent accumulation and renal precipitation • Myelosuppression (BBW) • Pulmonary toxicity (BBW) – usually delayed onset • Hepatotoxicity (increases in LFTs) • Renal toxicity • Infusion reactions • Wafer implants: seizures, meningitis, intracranial HTN No significant interactions Pulmonary toxicity risk factors: • Prior bleomycin, smoking, chest radiation, concomitant cyclophosphamide Moderate to high • Myelosuppression (BBW) • Hepatotoxicity • Pulmonary toxicity (may be delayed ≥6 months or longer) • Renal toxicity Minor substrate of CYP2D6 Moderate • Myelosuppression (BBW) • Hepatotoxicity (SOS) • GI toxicity • Mucositis • Pulmonary toxicity (avg onset 4 years; 4 months-10 years) • Skin discoloration • Seizures (Keppra for prophylaxis recommended) • Acetaminophen (hold 72 hours and at least 24 hours after, SCCC/UMHC policy is hold 72 hours pre & post) • Metronidazole • Azoles (voriconazole, Posaconazole, itraconazole) • More effect on myeloid cells than lymphoid cells • PK dosing to a target AUC based on TDM recommended Low dose: 2 to 6 mg orally daily Thiotepa (Tepadina) HSCT for CNS malignancy, leptomeningeal metastases (intrathecal) HSCT: • 150-250 mg/m2/day for 3 days starting 9 days prior to transplant • 50 mg/m2/dose every 12 hours for 6 doses N/A Oral (HSCT only): • Multiple tablets may be placed into an empty gelatin capsule for high doses IV • Infusion times vary by protocol or dose Kimberly Middleton, PharmD PGY2 Oncology Pharmacy Resident UMHM/SCCC • Phenytoin • Extensive hepatic metabolism to the major active metabolite TEPA • Good CSF penetration • Excreted via urine and sweat Low • Mucositis • CNS toxicity (headache, confusion, drowsiness, somnolence, hallucinations, coma, seizures) • Dermatologic: skin discoloration, pruritus, blistering, desquamation, and peeling • Myelosuppression • Hepatic SOS • Major substrate of CYP3A4 – avoid with strong inhibitors and inducers • Inhibits CYP2B6 – administer cyclophosphamide prior to thiotepa • Myelosuppression • GI toxicity (N/V, constipation, diarrhea) • Hepatotoxicity (increased LFTs) • Decreased serum albumin, magnesium, sodium, glucose • Peripheral neuropathy • Myelosuppression • Capillary leak syndrome • Cardiovascular effects (including HF) • Hepatotoxicity • Rhabdomyolysis (monitor CPK levels prior to each dose) • Minor substrate of CYP3A4 – avoid with strong inhibitors and inducers Leptomeningeal: 10 mg Lurbinectidin (Zepzelca) Metastatic SCLC 3.2 mg/m2 once every 21 days Toxicities: • Hepatotoxicity during treatment (withhold and resume at the same dose or reduced depending on grade severity) • Hematologic IV • Infuse over 60 minutes • Metabolized hepatically by CYP3A4 • Half-life: 51 hours Moderate Trabectidin (Yondelis) Soft tissue sarcoma, ovarian cancer (platinum sensitive) Sarcoma: • 1.5 mg/m2 once every 3 weeks Ovarian: • 1.1 mg/m2 every 3 weeks Hepatic: • Reduce dose for moderate to severe impairment IV • 24-hour continuous infusion for soft tissue sarcoma • Administer doxorubicin liposomal first in ovarian cancer; infuse over 3 hours • Metabolized hepatically by CYP3A4 • Half-life: ~175 hours Moderate Toxicities: • Hepatotoxicity, hematologic, capillary leak syndrome, cardiotoxicity, elevated CPK • Major CYP3A4 substrate, minor P-gp substrate – avoid with strong CYP3A4 inhibitors and inducers; monitor therapy with moderate • Excretion in sweat → skin care protocol