Percent Yield Quiz: Limiting Reactant & Yield Calculation
advertisement

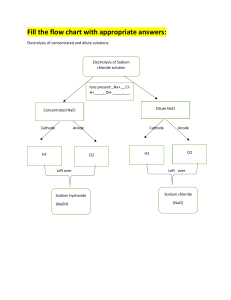
Ch 9 Percent Yield Quiz Name____________________________________________ Answer the questions given the following balanced chemical equation: 2 Na(s) + Cl2 (g) → 2 NaCl (s) a) Given 25.00 g of each sodium metal and chlorine gas, identify which is the limiting reactant and the excess reactant. Show all work. No credit will be given without work. b) If 17.5 g of NaCl are produced in the experiment, what is the percent yield of NaCl? Show all work