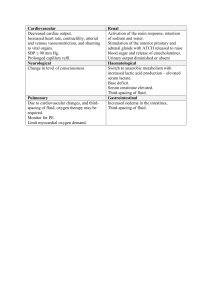
Electrolyte notes Anions Negatively charged electrolytes. calcium (Ca2+) An important body electrolyte ion having two positive charges (divalent cation). cations Positively charged electrolytes. dehydration Body fluid volume (especially plasma volume) deficit caused by fluid intake or retention below what is needed to meet the body’s daily fluid needs. diffusion Movement of particles (solute) across a permeable membrane from an area of higher particle concentration to an area of lower particle concentration (down a concentration gradient). electrolytes (ions) Solute particles that express and overall electrical charge (positive or negative). Also known as ions. extracellular fluid (ECF) The compartment composing fluids outside of the cells (interstitial fluid and plasma volumes). facilitated diffusion Movement across a cell membrane that requires a membrane-altering system. filtration Movement of fluid through a cell or blood vessel membrane because of hydrostatic pressure (water pressure) differences. fluid and electrolyte balance Regulation of body fluid volume, osmolarity, and composition; the regulation of electrolytes by the processes of filtration, diffusion, osmosis, and selective excretion. hydrostatic pressure Pressure exerted by water molecules against the surfaces (membranes or walls) of a confining space. Also known as water pressure. hypercalcemia Total serum calcium level above 10.5 mg/dL or 2.62 mmol/L. hyperkalemia Serum potassium level higher than 5.0 mEq/L (mmol/L). hypermagnesemia Serum magnesium level above 2.6 mEq/L or 1.07 mmol/L. hypernatremia Serum sodium level over 145 mEq/L (mmol/L). hyperosmotic/hypertonic Fluid with an osmolarity greater than 300 mOsm/L. hypervolemia Fluid overload. hypocalcemia Total serum calcium (Ca2+) level below 9.0 mg/dL or 2.25 mmol/L. hypokalemia Serum potassium level below 3.5 mEq/L (mmol/L). hypomagnesemia Serum magnesium (Mg2+) level below 1.8 mEq/L or 0.74 mmol/L. hyponatremia Serum sodium (Na+) level below 136 mEq/L (mmol/L). hypo-osmotic/hypotonic Fluid with an osmolarity less than 270 mOsm/L. hypovolemia Lower than normal circulating blood volume. insensible water loss Body water loss that has no mechanisms for control, including losses through the skin, the lungs, GI tract, salivation, drainage from fistulas and drains. interstitial fluid The part of extracellular fluids present between cells, also called the third space, that includes blood, lymph, water in the bones and connective tissue water, and the transcellular fluids. intracellular fluid (ICF) The compartment composing fluids inside of the cells (cellular fluid). isosmotic/isotonic Fluid that has the solute (particle) concentration (osmolarity) within the normal range for human body fluids, 270 to 300 mOsm/L. magnesium (Mg2+) A divalent cation that is stored mostly in bones and cartilage. obligatory urine output Minimum amount of urine output per day needed to excrete toxic waste products (400 to 600 mL). osmolality The number of milliosmoles in a kilogram of solution. osmolarity The number of milliosmoles in a liter of solution. osmosis Movement of water only through a selectively permeable (semipermeable) membrane to achieve an equilibrium of osmolarity. potassium (K+) The major cation of the intracellular fluid (ICF). sodium (Na+) The major cation (positively charged particle) in the extracellular fluid (ECF). solute Particles dissolved in the solvent of body fluids. solvent The water portion of body fluids.