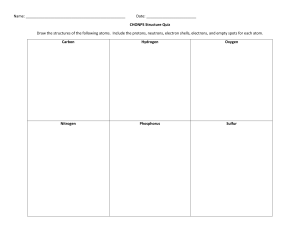
ELEMENTS DEFINITION • A chemical element is a substance that cannot be broken down by chemical means. Although elements aren't changed by chemical reactions, new elements may be formed by nuclear reactions. • Elements are defined by the number of protons they possess. Atoms of an element all have the same number of protons, but they can have different numbers of electrons and neutrons. Changing the ratio of electrons to protons creates IONS , while changing the number of neutrons form ISOTOPES. Cont'd • There are 118 known elements. • Examples of Elements:Any of the types of atoms listed on the periodic table is an example of an element CONT'D • Examples of Substances That Are Not Elements • If more than one type of atom is present, a substance is not an element. Compounds and alloys are not elements. Similarly, groups of electrons and neutrons are not elements. A particle must contain protons to be an example of an element. Non-elements include: • water (composed of hydrogen and oxygen atoms) • steel • electrons • brass (composed of multiple types of metal atoms)