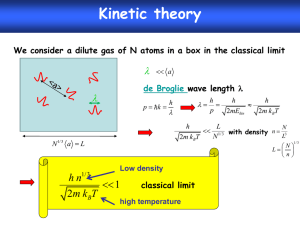
Materials Chemistry and Physics 99 (2006) 329–332 Lead-free (K0.5Bi0.5)TiO3 powders and ceramics prepared by a sol–gel method Mankang Zhu, Lei Hou, Yudong Hou ∗ , Jingbing Liu, Hao Wang, Hui Yan The Key Laboratory of Advanced Functional Materials of China Education Ministry, Beijing University of Technology, Beijing 100022, China Received 23 February 2005; received in revised form 2 September 2005; accepted 31 October 2005 Abstract A lead-free ferroelectric material, (K0.5 Bi0.5 )TiO3 (KBT), was prepared by a sol–gel process. Thermal analysis and X-ray diffraction showed that perovskite KBT powders with a grain size of 100–200 nm were obtained by calcining dried gels above 700 ◦ C. When the calcining temperature was lower than 650 ◦ C, only the Bi2 Ti2 O7 phase was found in the X-ray diffraction patterns. Moreover, KBT ferroelectric ceramics was fabricated by traditional sintering of as-prepared KBT powders. The KBT ceramics sintered at 1050 ◦ C showed a high density (91.2% of theoretic density) and low dielectric loss, better than that prepared by a solid-state reaction method. The temperature dependence of dielectric permittivity of KBT ceramics showed a Curie temperature at 385 ◦ C. © 2005 Elsevier B.V. All rights reserved. Keywords: Synthesis; Ferroelectric materials; Phase transformation; Sol–gel method 1. Introduction Recently, lead-free piezoelectric ceramic materials have attracted attention from the viewpoint of environmental protection. The increasing demand for environmentally benign materials points in the direction of lead-free ceramic materials for different electronic applications, such as ceramic filters and resonators. Among them, (K0.5 Bi0.5 )TiO3 (KBT) is a typical lead-free ferroelectric material of perovskite structure [1–3]. It has a Curie temperature TC of 380 ◦ C higher than that of (Na0.5 Bi0.5 )TiO3 , which is beneficial for the application as piezoelectric filters, resonators and micro-electro-mechanical systems. However, research on KBT has rarely been reported possibly because it is difficult to fabricate high-density KBT ceramics due to the serious volatilization of the K and Bi components at sintering temperatures [4,5]. KBT was synthesized by a mixed-oxide reaction method [6–9]. However, the powders prepared by this method usually feature a high agglomeration and an inhomogeneous particle size as a result of the high-temperature treatment. In comparison with other techniques, a sol–gel process has shown considerable advantages, including excellent chemical stoichiometry, compo- ∗ Corresponding author. Tel.: +86 10 6739 2733; fax: +86 10 6739 2412. E-mail address: ydhou@bjut.edu.cn (Y. Hou). 0254-0584/$ – see front matter © 2005 Elsevier B.V. All rights reserved. doi:10.1016/j.matchemphys.2005.10.031 sitional homogeneity, and lower crystallization temperature due to the mixing of liquid precursors on the molecular level [10,11]. In the present work, the sol–gel method was used to prepare KBT ferroelectric powders of perovskite structure. Through analysis of the thermal behaviour and phase evolution during calcination of dried gels, pure perovskite KBT powders were fabricated at 700–900 ◦ C. KBT ceramic samples were fabricated and its dielectric performances were investigated. 2. Experimental details Reagent grade bismuth nitrate pentahydrate (Bi(NO3 )2 ·5H2 O), potassium nitrate (KNO3 ), tetrabutyl titanate (Ti(OC4 H9 )4 ), acetic acid (CH3 COOH), and ethanol (CH3 CH2 OH) were adopted as the raw materials. In the experiment, bismuth nitrate pentahydrate was dissolved in acetic acid, and potassium nitrate dissolved in CO2 -free distilled water, respectively. The two solutions were mixed and then introduced into a prepared ethanol solution of tetrabutyl titanate with a stoichiometric amount. After stirring vigorously for 2 h, a slight yellow sol was formed. Then, the sol was heated to 70 ◦ C for 12 h to prepare dried gels. Finally, white powders were obtained by calcining the dried gels for 2 h at 700–900 ◦ C. The obtained white powders were pulverized, and pressed into pellets with a diameter of 11.5 mm under a uniaxial stress of 10 MPa, adding 0.5 wt.% polyvinyl alcohol (PVA) as binder. Conventional sintering was performed at a temperature interval of 25 ◦ C between 1000 and 1100 ◦ C for 2 h in a sealed alumina crucible. The sintered specimens were then lapped and electroded with a silver paste for the dielectric measurements. The thermal behaviour of the dried gels were investigated by differential scanning calorimetry (DSC) and thermogravimetry (TG) using a Netzsch 439C thermoanalyzer in ambient condition at a heating rate of 10 K min−1 . The X-ray 330 M. Zhu et al. / Materials Chemistry and Physics 99 (2006) 329–332 diffraction (XRD) analysis was performed in the θ–2θ mode using an X-ray diffractometer (Bruker D8 Advance) with Cu K␣ radiation. The Raman spectra were recorded from a Spex 1403 Raman spectrometer in a backscattering geometry with 488 nm radiation of an Ar+ laser and 100 mW output power. The dielectric measurements were carried out on an automated system consisting of a temperature control unit and a precision LCR meter (Agilent 4284A). 3. Results and discussion Thermal analysis was carried out to investigate the decomposition and phase transformation of the KBT dried gels. Fig. 1 shows the TG–DSC curves of the dried gels in the range 50–900 ◦ C. The TG data show a stepwise weight loss in two primary stages. The first stage appeared in the range 200–300 ◦ C, causing a weight loss of 64%. Correspondingly in DSC curve, exothermal peaks at 200–300 ◦ C were observed, which can be attributed the evaporation of the solvents and the decomposition of metal organics [12,13]. The second stage appeared in a broad range from 400 to 700 ◦ C, leading to a weight loss of 24%. This weight loss may result from the oxidation of the residual organic groups. However, some weak endothermic and exothermic peaks appeared in this temperature region, which may be attributed to the low heat change of the crystallization of Bi2 Ti2 O7 and KBT as observed in the XRD patterns. XRD analysis was carried out to study the phase evolution of the dried gels in the calcinations process. Fig. 2 shows the XRD patterns of dried gels calcined at 400, 500, 600, 650, 680, 700, 800, and 900 ◦ C, respectively. As can be seen in the figure, an amorphous phase was formed at sintering temperatures lower than 400 ◦ C. When the temperature arised to 500 ◦ C, some diffraction peaks appeared. All of them can be assigned to Bi2 Ti2 O7 structure as reported in the JCPDS File No. 32-0118 [14,15]. When the temperature increased to 600 and 650 ◦ C, the height of the diffraction peaks belonging to Bi2 Ti2 O7 increased, while the full width at half maximum decreased due to crystal growth and the improvement of crystallinity. It means that the powders calcined in the temperature region 500–650 ◦ C were cubic pyrochlore Bi2 Ti2 O7 . However, for the powders heated at Fig. 1. TG and DTA curves of the KBT precursor gel dried at about 70 ◦ C. Fig. 2. XRD data of the dried gel heated at different temperatures. 680 ◦ C, obvious change was observed in the XRD patterns. The characteristic peaks of perovskite KBT crystals (JCPDS File No. 36-0339) appeared at 22.56◦ , 31.96◦ , and 39.44◦ , respectively. After further increasing of the temperature to 700 ◦ C and above, only diffractions belonging to perovskite KBT was observed and there was no evidence of a second phase. Besides, the full width at half maximum of the (1 0 1) diffraction peak of KBT powders changed from 0.94◦ at 700 ◦ C to 0.75◦ at 800 ◦ C and to 0.61◦ at 900 ◦ C, respectively. It means that the grain size increased from 100 to 150 nm according to Scherrer’s equation, indicating nanoscale dimensions of the powders. The Raman spectra of powders calcined at 600 and 700 ◦ C are shown in Fig. 3. The Raman spectra of powders calcined at 700 ◦ C show three scattering bands. Through fitting by Gaussian function, nine peaks at 145, 204, 265, 326, 433, 536, 634, 740, and 845 cm−1 , respectively, could be separated out. The results are in good agreement with perovskite KBT as reported by Jones et al. [16] and Kreisel et al. [17]. However, the Raman spectra of powders calcined at 600 ◦ C showed a different pattern. There appeared peaks at 152, 226, 281, 326, 418, 562, 622, and Fig. 3. Raman spectra of the powder heated at 700 ◦ C. M. Zhu et al. / Materials Chemistry and Physics 99 (2006) 329–332 331 Table 1 Dielectric constant εr and dielectric loss tan δ of the KBT ceramics sintered at 1000–1100 ◦ C Sintering temperature (◦ C) εr tan δ Relative density (%) 1000 1025 1050 1075 1100 1652 856 690 570 536 1.020 0.678 0.052 0.132 0.360 81.2 85.0 91.2 88.5 86.7 726 cm−1 , respectively. As reported by Kojima et al. [18] and Wang et al. [19], there is only one mode in the 600–800 cm−1 range for Bi2 Ti2 O7 . So the pattern of powders calcined at 600 ◦ C present characteristic of cubic Bi2 Ti2 O7 . It affirmed that the powder obtained by calcining the dried gels at 700 ◦ C is a perovskite KBT phase. KBT ceramics was fabricated using the powders heat-treated at 700 ◦ C by a traditional sintering process. The dielectric constant, εr , and dielectric loss, tan δ, of the KBT ceramics sintered at 1000–1100 ◦ C were measured (Table 1). The KBT ceramics sintered at 1000 ◦ C shows a εr value of 1652. However, due to the high value of tan δ (1.02), the large εr value is not reliable. As the sintering temperature increased to 1050 ◦ C, the εr value of KBT ceramics was about 690, which is in agreement with the value of 682 of material prepared by a conventional solid-state reaction method [8]. Meanwhile, tan δ of KBT ceramics sintered at 1050 ◦ C was significantly lowered (0.052), only 10% of the tan δ value (0.490) for the samples prepared by a conventional solid-state reaction method [8]. The temperature dependence of εr of the ceramics sintered at 1150 ◦ C was measured at a frequency of 10 kHz (Fig. 4). It shows that the Curie temperature (385 ◦ C) is very close to that of reported data [1–3]. The superior dielectric properties of the KBT ceramics prepared by the sol–gel method can be attributed to the higher relative density of 91.2%, compared to the low density (70%) of a ceramics prepared by conventional solid-state reaction method. However, increasing the sintering temperature above 1075 ◦ C decreased Fig. 5. XRD data of the KBT ceramics sintered at different temperatures. the density of the ceramics and increased the dielectric loss due to the volatilization of the K and Bi components. Fig. 5 shows the XRD patterns of KBT ceramics sintered at different temperatures. It can be seen that, when the sintering temperature is below 1075 ◦ C, only diffractions corresponding to perovskite KBT are observed; however, diffraction peaks belong to K4 Ti3 O8 (JCPDS File No. 41-0167) appeared when the sample was sintered at 1100 ◦ C. The appearance of a second phase may result from the high volatilization of the bismuth element. Thus, the dielectric properties were seriously deteriorated. 4. Conclusions KBT powders and ceramics have been prepared by a sol–gel technique. The gels converted to pure perovskite phase of KBT at 700 ◦ C from pyrochlore Bi2 Ti2 O7 phase at lower temperatures. The obtained KBT powders had a smaller size and even shape than those prepared by a traditional solid-state reaction method. Besides, high-density KBT ceramics can be prepared from the as-prepared KBT powders, and ceramic samples with a relative density of more than 90% were prepared at sintering temperature of 1050 ◦ C. It means that the powders prepared by the sol–gel method can be sintered at a lower temperature, which is beneficial to controlling the volatilization of the K and Bi components. The dielectric properties of the powders are better than those of powders prepared by a conventional solid-state reaction method. Acknowledgement The authors are grateful to the Science & Technology Development Project of the Beijing Education Committee for financial support. References Fig. 4. Dielectric constant with temperature at various frequencies for the KBT ceramics sintered at 1050 ◦ C. [1] T. Takenaka, K. Maruyama, K. Sakata, Jpn. J. Appl. Phys. 30 (1991) 2236. [2] T. Takenaka, T. Okuda, K. Takegahara, Ferroelectrics 196 (1997) 175. 332 M. Zhu et al. / Materials Chemistry and Physics 99 (2006) 329–332 [3] Z.F. Li, C.L. Wang, W.L. Zhong, J.C. Li, M.L. Zhao, J. Appl. Phys. 4 (2003) 2548. [4] C.F. Buhrer, J. Chem. Phys. 36 (1962) 798. [5] T. Wada, K. Toyoike, Y. Imanaka, Y. Matsuo, Jpn. J. Appl. Phys. 40 (2001) 5703. [6] G.A. Smolenski, V.A. Isupov, A.I. Agranovskaya, N.N. Krainik, Soviet Phys. Solid State 2 (1961) 2651. [7] J. East, D.C. Sinclair, J. Mater. Sci. Lett. 16 (1997) 422. [8] J. Suchanicz, Mater. Sci. Eng. B 55 (1998) 114. [9] T. Zaremba, J. Therm. Anal. Cal. 74 (2003) 653. [10] Z. Surowiak, M.F. Kupriyanov, D. Czekaj, J. Eur. Ceram. Soc. 21 (2001) 1377. [11] H.Q. Fan, H.E. Kim, Jpn. J. Appl. Phys. 41 (2002) 6768. [12] Y. Li, J.P. Zhao, B. Wang, Mater. Res. Bull. 39 (2004) 365. [13] S.F. Wang, F. Gu, M.K. Lü, C.F. Song, S.W. Liu, D. Xu, D.R. Yuan, Mater. Res. Bull. 38 (2003) 1283. [14] W.F. Su, Y.T. Lu, Mater. Chem. Phys. 80 (2003) 632. [15] A.L. Hector, S.B. Wiggin, J. Solid State Chem. 177 (2004) 139. [16] G.O. Jones, J. Kreisel, V. Jennings, M.A. Geday, P.A. Thomas, A.M. Glazer, Ferroelectrics 270 (2002) 191. [17] J. Kreisel, A.M. Glazer, G. Jones, P.A. Thomas, L. Abello, G. Lucazeau, J. Phys.: Condens. Matter 12 (2000) 3267. [18] S. Kojima, A. Hushur, F.M. Jiang, S. Hamazaki, M. Takasige, M.S. Jang, S. Shimada, J. Non-Cryst. Solids 1 (2001) 250. [19] S.W. Wang, W. Lu, X.S. Chen, N. Dai, X.C. Shen, H. Wang, M. Wang, Appl. Phys. Lett. 1 (2002) 111.