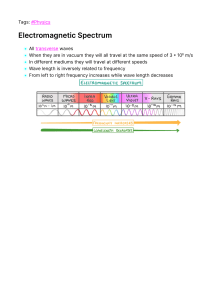
Physics for CSEC® Physics for CSEC® Lancelot Darren Yvette Earl Caesar Forbes Mayers Skerritt 3 Great Clarendon Oxford It University furthers and Oxford © CXC ® The and rights in means, Press, as Enquiries must British Data 6DP, department objective of of UK and the Kingdom University excellence worldwide. the United Oxford in in is certain a of Oxford. research, scholarship, registered other trade mark of by Oxford University registered the the sent authors Nelson published by No the Press trademarks Oxford part of system, prior the have Thornes been 2014 of the Caribbean Examinations Ltd in asserted 2013 University this or permitted by in law, by reproduction any of licence rights outside Department, in may in writing reprographics Rights Press publication transmitted, permission appropriate to the 2014 be reproduced, form or Oxford or by any University under terms organization. scope Oxford of the University above Press, at above. not this circulate same Library this work condition Cataloguing on in in any any other form and you must acquirer Publication Data available 978-1-4085-2245-5 10 9 8 7 Printed 6 in 5 4 Great Britain by CPI Group (UK) Ltd., Croydon CR0 4YY Acknowledgements Cover photograph: Mark Lyndersay, Lyndersay Digital, Trinidad www.lyndersaydigital.com Illustrations Page Thanks are due contributions Although we copyright cases. the and the this If Lancelot the have notied, to for to in drawn made the by GreenGate Publishing Publishing Caesar, development every before Yvette of this effort publication publisher will to Mayers and Earl Skerritt for book. trace this has rectify and not any contact been errors all possible or in omissions opportunity. third party information materials work. artwork GreenGate holders earliest Links include make-up: of countries 2014 are expressly address in © concerning be impose ® retrieval with should Press reserved. a a OX2 ). without or agreed You ® rights edition stored the CSEC (CXC is publishing Forbes published This Press illustrations moral First by Darren Council Oxford, University’s University Original All the education Text Street, websites only. contained are Oxford in any provided by disclaims third party Oxford any in good faith responsibility website referenced for in all at their Contents Introduction Section 1.1 1 1 3 Waves 3.1 Wave 3.2 Describing motion 76 3.3 Electromagnetic 3.4 Light 3.5 Reflection 3.6 Refraction 3.7 Refractive 3.8 Converging 3.9 Diverging Mechanics Base units, derived units and Making 1.3 Variables measurements and Graphical Vectors 1.6 Forces 78 waves 80 2 1.2 1.5 waves measuring density 1.4 Section graph plotting and shadows 82 of light 84 6 analysis and rays 4 of light 86 8 scalars index and total internal 10 reflection 1.7 Moments 14 1.8 Stability 16 1.9 Investigating Section 1 Hooke’s Practice law exam Moving objects and lenses lenses 3.10 Magnification 3.11 Diffraction, 90 and the eye 92 94 18 questions 1 interference and theories 20 about 1.10 88 12 the nature of light 96 displacement–time graphs 3.12 Sound 3.13 Experimenting waves 98 22 1.11 Acceleration 1.12 Newton’s and velocity–time 1.13 Momentum and 1.14 More momentum graphs about Section 1 of motion 1.15 Energy and 1.16 Energy sources exam energy waves 100 3 Practice exam questions 102 26 impulse Practice sound 24 Section law with 28 Section 4 Electricity and magnetism 30 questions 2 transformations 4.1 Static 4.2 Electric electricity 4.3 Uses 4.4 Electric 4.5 Electrical 4.6 Simple 4.7 Series 4.8 Cells 4.9 Resistance 106 32 fields and the Van de Graaff 34 generator 1.17 Potential 1.18 Conservation 1.19 Power 1.20 1.21 energy and of electricity and kinetic energy energy and dangers sinking 1 and Practice energy and Thermometers 2.3 The density current energy circuits questions and and kinetic 3 and states Heat temperature of and temperature 2.6 Changes 2.7 Heating, 2.8 Specific 116 Investigating 4.11 Circuit current and potential characteristics 124 analysis Direct 4 126 Practice current and 4.13 Mains electricity state and and evaporation exam questions alternating 1 current 128 4.14 Mains safety 4.15 Conversion 4.16 Logic circuits 1 4.17 Logic circuits 2 Convection latent heat heat the Caribbean 130 132 a.c. to d.c. 136 62 138 140 66 currents 4 Practice exam questions 2 142 68 radiation thermal 134 of 64 4.18 Permanent magnets 4.19 Magnetic 4.20 Electromagnetism 4.21 Forces 4.22 Electromagnetic 144 70 energy fields 146 transfer principles 2 in 60 Section Applying 122 4.10 54 conduction Infra-red 120 58 cooling 2.12 118 batteries 56 of 2.11 circuits scales 52 matter latent Thermal parallel 50 capacity Section 114 components theory laws 2.5 2.10 transformations and 48 Section 2.9 112 46 4.12 Gas 110 42 exam physics 2.2 2.4 electricity 40 efficiency Thermal Heat, static 38 difference 2.1 of 44 Floating, 2 108 36 Pressure Section Section for 148 72 Practice exam questions from magnetic fields 150 74 4.23 4.24 The d.c. motor induction and the a.c. 152 generator Transformers Section 4 Practice 154 156 exam questions 3 158 iii Contents Section 5 The physics 5.1 Models 5.2 Electrons, of the 5.3 Radioactive 5.4 Nuclear 5.5 A 5.6 Nuclear 5.7 Applications 5.8 The the of the atom atom 160 nucleus and the periodic table decays model of 5 and 164 reactions radioactive decay release Section iv 162 emissions and of of decay half-life radioisotopes nuclear Practice energy exam questions 166 168 170 172 174 176 Glossary 178 Index 182 Acknowledgements 187 Introduction This Study Guide has been developed exclusively On Your Marks activities provide sample ® with be the used both in Caribbean as an and Examinations additional out of resource school, Council by following (CXC ) to examination-style candidates, questions, the and Caribbean with feedback short answer example from an and candidate examiner to essay type answers show where ® Secondary Education Certificate (CSEC ) programme. answers build It has been prepared by a team with expertise could your be improved. understanding, These skill activities level and will confidence in in answering examination questions. ® the CSEC The contents providing Physics you the for syllabus, to are tools and master syllabus. full help the Do key achieve concepts the examination. support included remember on and to you features guidance examination designed to the teaching to your make and refer course learning best it in easier your revise and format! this includes Study Guide electronic developing good is an activities interactive to examination assist CD you in techniques: activities experience sections of and inside are questions and check of helpful the problem Answers can Inside Yourself questions for syllabus requirements T est provide requirements to by are specifically multiple-choice feedback study guide will so designed to examination refer that you you to can areas. included practice your own on the exam work as CD for summary questions, you so that you proceed. which This unique and interactive with combination invaluable of examination support to focused practice help you syllabus will content provide reach your you full ® potential in CSEC Physics. 1 1 Mechanics 1.1 Base and When LEARNING we units, measuring measure the end of this able topic should be state and use units and prefixes 1.2 you derive units measure a shaped T able for the regularly quantity we need to density metres or The length 1.2 m, not of a just desk include should be the given units as, for of the example, 1.2. to: range of Base SI A a units OUTCOMES measurement. At derived quantities density and set of been are of units seven agreed shown units, based on internationally. in T able the They metre, are kilogram known as and the SI second, base has units and 1.1.1. irregularly objects. Derived 1.1.1 units There are These units many can mathematical Quantity Base unit mass kilogram kg length metre m more units all derived be in relationships addition directly and to the from seven the base seven, units. based on definitions. Symbol For example, the speed of an object distance speed is moved second s current ampere A This means that the from the equation: (m) = time time defined unit for speed (s) is a distance divided by a time: −1 metres temperature kelvin K amount mole mol Every of substance For per other candela (m s derived convenience derived luminous second units quantity (and have ). to can be honour been given worked important special out this way. scientists) names. For some example, of the the unit cd for a potential difference derived from the base units would be intensity 2 m −3 kg s −1 A . This unit is given the special name volt (V). ACTIVITY Standard The SI base precise units have definitions or very To derivations. avoid form using standard very form. large In or very standard small form numbers, the number scientists is always use written in x Find out what these are. the is format the of x A × number indicates movement to 10 of , where places to movement the A is a move to the number the between decimal right while point. a 0 A negative and 10 and positive value x value indicates left. 3 4500 is 0.0006 written as 4.5 × 10 × 10 −4 is written as 6 Prefixes Scientists so use are Figure 1.1.1 SI units were used in a often set shown of in need to prefixes T able use very which 1.1.2. This large may set be of or very small attached prefixes to numbers any allows a unit. These quantity all 6 8.4 the in calculations designing Tree Transport Kingston, 2 the needed Half-Way Centre Jamaica. in × 10 watts to be written as 8.4 megawatts or and 8.4 MW. like T able Using units: 1.1.2 Prefix The density scientific cubic of Prefixes for SI units density a material language centimetre this of is the is the how mass much material. per unit mass The volume. there equation is in a In cubic defining Symbol metre density or 12 tera T 10 G 10 M 10 kilo k 10 centi c 10 m 10 micro μ 10 nano n 10 pico p 10 is: 9 giga mass m 6 mega density Represents non- = or ρ = volume V 3 −2 WORKED EXAMPLE 1 −3 milli −6 Find the derived SI unit of density. −9 Mass is measured in kilograms (kg). Volume is measured in cubic 3 metres The (m −12 ). equation defining density shows that density is a mass −3 divided by a volume and therefore the SI unit of density is kg m ACTIVITY Use Measuring the these To measure measure taking technique described on density the the density mass appropriate of using a a regularly balance measurements shaped and for then the object you calculate dimensions of must the volume and using by a pages range to of find the density irregularly shaped objects. a formula. To a measure the measuring rise by the rough cylinder volume WORKED A volume of has an partly the EXAMPLE stone of a irregular full of object, water. The place level the of object the inside water 25 25 20 20 15 15 10 10 5 5 will object. 2 mass of 30 g. It is placed in a measuring 3 cylinder containing 20 cm of water and the level of the water 3 rises The to the 24 cm density of the mark stone (Figure is mass 1.1.2). found using: (g) 30 g –3 = = 3 volume (cm 7.5 g cm 3 ) (24 – 20) cm Figure KEY SUMMARY Use the derived POINTS QUESTIONS 1 1 1.1.2 following units for equations velocity, to work out acceleration the and set appropriate of use base a standard units (SI) for quantities. force. distance velocity Scientists 2 Units for most quantities = are time derived from these base units. change acceleration in velocity = 3 The density of an object is time mass force = mass × acceleration given by density = volume 2 Write these quantities milliamperes, 4.9 using the micromoles, base 2400 unit and standard picoseconds, 0.2 form: 83 gigavolt. −3 and the unit is kg m or −3 g cm 3 1.2 Making Common LEARNING At the end should be select of this able an topic Length you to: a range identify and to of measure random length error error If in use of the calculations measured the have smallest a with a ruler resolution difference in of a or tape 1 mm. reading measure. The the These resolution of instrument an can be length used. needs Callipers gauge to have micrometer be a measured, resolution will have a callipers of or 0.1 mm resolution of a micrometer and a high quality 0.01 mm. number figures and be typically is smaller screw appropriate significant a can measurements instrument can measure. experiments systematic lengths instruments appropriate instrument in measurements OUTCOMES Many measurements Time in answers. Time intervals stopwatches times the To are full generally have more a measured resolution than 0.1 of second, with 0.01 it a digital second may be but, stopwatch. as human unnecessary to Many reaction measure to resolution. measure triggered much are times by more events better precisely, such response as the we can breaking use of a automatic stopwatches light-beam. These have times. Mass Mass a 1.2.1 This micrometer used of a to measure is being the measured known pan Figure is one or, balances is by a balance more often, available, Uncertainty and which by some a compares top-pan with a an unknown balance. resolution of A variety mass of to top- 0.01 g. error gauge wire. When we as ‘true the make Systematic could or the have a Readings leading to it from some may not be exactly instruments uncertainty in the introduce our same error in calculations. error systematic device measurement value’. measurements A a error is usually measurement damaged end the result of technique. so that it some For flaw example, shows all in a the measuring metre lengths as rule 1 mm too short. Systematic true Random 1.2.2 Human a factor reaction when time using is will make the readings shift away from the error error is an unpredictable measurement when the you you take a error introduced reading. into Sometimes this a is because of a technique use such as poor positioning stopwatch. while 4 recorded measurement. Random Figure errors measuring length (Figure 1.2.3). of your line of sight Observer ACTIVITY Observer Use Line of sight Line of a range of instruments sight to take measurements of the Bar Metre properties rule how of Correct reading left-hand Figure end of of True the 1.2.3 A bar line reading right-hand of sight of end Incorrect of the bar of the reading bar due to of right-hand ‘line of a to of measure sheet of errors will cause the be scattered around the b A Student The effects of random error can be reduced by B true 55 °C 55 °C value. thickness paper. a to out error error readings Find end sight’ Student Random objects. performing repeat True readings and finding a mean value. Readings which were slightly too value high will tend to cancel readings which were slightly too low. 50 °C 50 °C Accuracy If a to and measurement be accurate. without precision is It close is repeating not the to the true possible to value know measurement of if several a a quantity reading times it is and is said accurate 45 °C 45 °C calculating Precise a mean value. All possible systematic errors must be avoided. If (but of the measurements taken are close to the mean then you can not that the results are accurate) (but precise readings mean measurement show value of very the is little a measurement difference. They where are all all very of the close 1.2.4 Demonstrating is to a not possible to systematic precise which are a precision accuracy WORKED the EXAMPLE readings. obtain error (Figure both b repeated to The It precise) accurate. and A not be Figure confident Accurate all results (Figure 1.2.4b). accurate which are 1.2.4a). The and very precise Results best but can not also accurate be experiments due accurate produce but results precise. diameter bearing is of small measured callipers giving 6.4 mm. Using for a volume a with value the gives ball of formula an answer 3 of Quoting answers 137.258 mm volume to significant The number reflection of of significant how certain fi gures you are you of quote the in any precision answers of your is a never resolution to two given or in of use more your three significant instruments. significant questions or by In fi gures than general, fi gures unless measuring are sensible answers more should signifi cant by be You the The 1.4 number of volume × 10 is recorded as 3 mm given figures are KEY instruments. 1 SUMMARY the a answer. justifi ed Record figures. 2 should . POINTS Systematic errors QUESTIONS produce amount 1 How can you 2 T able 1.2.1 increase the accuracy of a a constant of uncertainty in measurement? measurements. shows three sets of results for the measurement of 2 the diameter of a metal rod of true diameter 5.0 mm. Which Random a of results are accurate, which are precise and which are errors varying amount in measurements. 1.2.1 3 Set of both? uncertainty T able produce sets a/mm 4.3 5.6 5.5 4.2 4.9 The number of significant 5.1 figures Set b/mm 5.1 4.9 5.0 4.9 4.9 5.1 Set c/mm 4.7 4.8 4.6 4.8 4.6 4.7 of a shown calculation indication in in the of the is the result an uncertainty answer. 5 1.3 Variables and graph plotting Scientific LEARNING investigations connections At the end should be select of this able an topic a you physicist the to: independent effect investigate dependent identify other its effect on highly The of this controlled during be able change to in and in order alter behaviour . one variable In to in an reveal an ideal experiment experiment and see isolation. variables independent experimenter control variables would quantities a variable and between variable Key and are OUTCOMES to variable see what is the effect variable changing which it has is on manipulated the by the experiment. an The dependent variable is the variable which is expected to change investigation in plot an accurate and response explore experiment explaining For EXAM of connection finding cause if you water water, allow s relation ship variable. is of between relationship that were the a these two between variables them and in then relationship. investigating denser it will the be’ hypothesis then the ‘The colder independent a variable as this changing temperature The is of the dependent the quantity water variable you as this would expect to is be what the change you would density in of response be the the to temperature. the one varia ble depend ent the us be tween indepe nden t be manipulating. experim ental invest igate one independent TIP desig n while the by the example, sample would to the graph. an Good altering wellScientists scaled to Control variables and There are usually a wide range of other factors that need to be taken varia ble into contro lling all account during an experiment so that they do not have an effect on other the varia bles. results. designs These ensure are that known these as control factors do variables. not alter Good during the experimental experiment. Graphs WORKED Find out if container cooling. design One of is plot the colour affects Your the of in a rate of experimental should ensure that to the are kept The best ways graph graph to show (Figure then would explanation the can a clear then be try given 1.3.1). relationship If there relationship to explain then the is has how a between clear two been this pattern variables demonstrated. relationship relationship shown is occurs. If an causal. these graph plotting takes time and practice. Use these tips to help constant: produce a Scientists Perfect factors the EXAMPLE temperature of yours. the Use a When sharp pencil and ruler. surroundings The size of the label The volume of you draw the axes make sure you leave enough space to containers liquid in them clearly. the Plot the independent Plot the dependent Plot the graph Use variable on the x-axis containers Air paper as so possible increments possible. 6 variable on the y-axis flow that the data horizontally of 2, 5 or 10 points and (or occupy as much of the graph vertically. their multiples) for your scales if a 8 Clamp 7 6 1– s m/yticoleV 5 4 Thread 3 2 l Maximum 1 a swing angle about of 10° 0 0 10 20 30 40 50 Time/s Figure 1.3.1 A well-drawn up the graph available has clearly labelled axes and the data points fill space. ACTIVITY Bob A pendulum A mass is a simple device that can be used to measure time. Pointer on the end of a thread or wire is allowed to swing centre from side swing to back side and (Figure forth to 1.3.2). the One same complete oscillation at freely is of swing a b position. Stop c The following three factors may affect the period (time it takes b for one complete oscillation). b Investigate each controlling the comparing each of the other three two. factors Collect independently data and plot a while separate graphs Start of the independent variables to the period. One Length Mass Initial of the complete Figure Y ou of the angle will need pendulum of to use the data to produce a graph in the next b + c 1.3.2 Timing a pendulum POINTS QUESTIONS about An the following hypotheses and outline a independent variable to investigate them. For each one identify variable, dependent variable and variable is is changed an investigation to clearly see independent that basic during experiment the + topic. a Think a displacement 1 1 = bob KEY SUMMARY oscillation thread any if this change has any other effect. variables you will have to control. 2 a The greater the current in a light bulb the brighter it will A dependent variable b The c The further away from a sound you are the quieter it will the force you put onto a spring the more it which is is a measured seem. to larger variable be. determine the effect of will changing the independent stretch. variable. 2 T able 1.3.1 shows the data from an investigation into 3 resistance. Plot a graph using this T o make the T able effect of affects 0.0 0.1 0.2 that only altering independent 1.3.1 Potential sure data. 0.3 0.4 0.5 the the variable outcome of the 0.6 experiment, other factors have difference/V to Current/A 0.00 0.60 1.02 1.57 2.07 2.52 3.01 be These carefully are controlled. control variables. 7 1.4 Graphical A LEARNING graph can be analysed to analysis find the relationship between the two OUTCOMES variables. At the end should plot be a of this able topic you to: straight Graphs line of best a set of determine straight the line analyse a gradient of of simple a the graph graph best relationship two to between If the a line the data not ACTIVITY collected the between period the length of to identify the lines or curves) variables. relationship that pass These through lines or follow close to of and of graph best points may large Y ou these fit with appear (Figure an not fit may also in the or straight Draw pattern the above of line, line and results. uncertainties made results a number errors have be 1.4.1). equal random anomalous to mistakes when in drawing you so can use ruler that below These it may recording the line of passes the produced a line. be due during the best data. Do fit. 60 a the pendulum or use 50 the data T able provided in T able 1.4.1. 1.4.1 2 Length/m Period l T 2 /s 40 2 Δy 0.20 0.10 0.40 0.15 0.60 0.20 0.80 0.25 1.01 0.30 1.21 0.35 1.41 0.40 1.61 m/tnemecalpsiD 0.05 Point to Plot a graph length 20 of comparing the square of pendulum the Describe the in (T 1 2 3 4 5 graph. Figure 8 5.0 s 0 ). relationship the = with period Time/s shown 8.0 − 3.0 the 0 = 10 2 the 55.0 − 20.5 35.5 m be Δ x = = checked 30 1.4.1 Drawing a line of best fit 6 7 8 9 to the when relationship swing the use a points experiment. investigating (straight on unexpectedly pendulum’s graphs dependent lines points draw Some you onto and points. through data drawn the variables. the are independent describe to Use fit the shapes Straight the relationships points between linear fit Lines through showing Gradients The gradient between To find the the Draw a read Use a (for the can be used to find a mathematical relationship EXAM two are triangle the points widely example, in It right-angled meets that change line variables. gradient: large, triangle points of two to on the spaced points find x-value triangle the line and that lie on the of best those on change the in graph fit. whose so If that values are the easily it y-value (Δy) and the grap h helps . join the a to draw grap h ‘insi de’ select gridlines). the easier cur ve d the possible, is TIP from cur ve . paper Y ou grap h a Ro ta te arou nd mus t as a if neve r serie s of do t-t o-do ts . (Δx). Δy The gradient (m) is given by m = Δx Intercepts Sometimes this case the the line two of best variables fit will are in pass directly direct through proportion the to origin. each In other: 1 independent If the as line linear . (c) and does The the variable not line ∝ pass will through cut relationship dependent the the y-axis origin at between the = c y variable a the point relationship known variables will be is described as the intercept of the form: x y mx + Figure where y is the dependent variable, x is the independent variable, m 1.4.2 You can pattern the gradient and c is the intercept on the sometimes find a is y-axis. by plotting x 1 against y Graphs showing other relationships T able The points on a graph may also follow a curve. This may 1.4.2 Examples which also variables represent a Instead plotting of simple mathematical a graph relationship comparing the x between and y the variables of relationships occur in between physics variables. directly, Relationship Produces line 1 a graph straight- when 2 functions such as plotting y or may show the relationship more clearly. y x ∝ y x plotted against y x plotted against y x plotted against 2 2 x ∝ x ∝ y 1 1 y SUMMARY There is a mathematical illuminance that relationship (brightness) source. Data are of a light provided in between source T able and the the light distance from KEY graphical techniques to find out if POINTS 1.4.3. 1 Use y QUESTION this relationship is A to 1 illuminance 1 ∝ or illuminance line of best fit is used either: show the between relationship two variables. ∝ 2 distance distance 2 A straight passing T able line of through best the fit origin 1.4.3 shows Distance/m 0.5 1.0 1.5 2.0 2.5 3.0 16.0 4.0 1.8 1.0 0.6 0.4 3 Curves direct can proportionality. also show –2 Illuminance/cd m mathematical relationships. 9 1.5 Vectors There LEARNING At the end should be of this the between two classes of quantity scalars in physics: scalars and vectors. OUTCOMES able describe are and a topic A scalar quantity A vector has magnitude (size) but no direction. you quantity has magnitude and direction. to: difference scalar and Comparing a a scalar and a vector vector Distance and displacement are both lengths which can be measured quantity in use a vector diagram the resultant of two to metres: find vectors. Distance is reference a scalar to the Displacement object For is example, walk 5 of if metres distance your is from of 1.5.1 Vector calculations in predicting of how far object moves without walk quantity. (original 10 metres towards your However, It is a measure of how far an position). northwards, starting your point turn you displacement around have is 5 and covered metres a north point. quantities such as mass from and each volume, simply add together or are other: hurricanes. Adding a mass of 40 kg to a mass of 30 kg 3 WORKED an the subtracted paths is are Scalars, used vector origin metres. Combining Figure a its you starting It direction. back 15 quantity. EXAMPLE 1 Removing 40 cm gives a mass of 70 kg. 3 of water from 100 cm of water will always leave 3 60 cm Find the resultant of the acting Figure 1.5.2. on the ball water. three Vectors, forces of such as velocity and force, cannot be simply added together. in To find the resultant of vectors you must also take their direction into account. 5.2 N 6.1 N Parallel vectors 4.8 N If Figure the vectors are parallel to each other, then you can add or subtract 1.5.2 to Resultant = 5.2 N + 4.8 N find a resultant. Always give the direction of the resultant in your − answer. 6.1 N = 3.9 N to the right Non-parallel The parallelogram parallel F vectors adjacent R 2 to vectors: be sides added by of (F rule and parallelogram allows drawing the a us to scale ). The find (in lengths The red) of F 2 magnitude. the diagram parallelogram F 1 their θ the rule resultant (Figure (R) of the and F are two vectors the in by 1 Figure 10 1.5.3 The parallelogram rule the diagonal of the parallelogram, non- T wo two vectors proportion to 2 is P F two 1.5.3). represent 1 resultant of shown in green. represented WORKED What is EXAMPLE the 2 resultant of the two vectors shown in Figure 1.5.4? R 50 N y 75 N θ Figure 1.5.4 x Check the that other (Figure the two vectors sides 1.5.5). of are drawn the Measure to the same parallelogram the length and and scale. the angle of Then draw Figure 1.5.6 diagonal the diagonal, SUMMARY and use the scale factor to find its value in 1 1 2 0 QUESTIONS newtons. Y ou follow the instructions N below 50 N in a treasure hunt. 15° Calculate the total distance 75 N travelled Figure 1.5.5 then Vectors at right angles 2 the vectors are at right angles to each other, you can 40 m south and Draw a the Where and theorem and trigonometry. The two vectors of a right-angled triangle with the resultant can be of found the triangle (Figure 1.5.6). The size of forming the two east. to find velocity use acting a on river, a as boat shown in the Figure hypotenuse 10 m diagram of north, 70 m form crossing sides 50 m then finally scale final Walk east, resultant vectors Pythagoras’s two the displacement: 1.5.7. hypotenuse using: 2 2 hypotenuse = adjacent = x 2 2 R 2 + opposite 2 + y –1 The angle of the resultant vector can be found 5 m s using: –1 2 m s opposite tan θ = y = adjacent x y –1 which leads to the relationship: θ = tan ( ) x Figure WORKED EXAMPLE 3 KEY A remote-controlled followed by a toy is movement instructed of 60 m to west. move What 80 m the toy after this is the POINTS south displacement 1 of Scalars of displacement: 2 R R 2 = = have 80 can 2 + 60 √10 000 = be not of direction. added or They subtracted simply. 2 = but 10 000 m 100 m 2 Vectors have magnitude Direction magnitude movement? (size) Magnitude 1.5.7 both and direction. displacement: Addition of vectors has 60 –1 θ = tan = ( 80 ) 37° to take into direction of account the the vectors. 11 1.6 Forces Forces LEARNING acting on an object can change its shape or the way it is OUTCOMES moving. At the end should be of this able describe the topic you to: Describing effects of simple As forces acting on forces are find the vector quantities (they have size and direction) they are objects represented forces resultant of a set of direction of in diagrams the force as and arrows. the The length direction represents of the the arrow size of gives the the force. forces The calculate object the from weight its gravitational mass field of unit of and the Example When are se t objects When or TIP ques tions = on the the weig ht Earth 10 N kg at is differe nt = 1.6 N kg two you ques tion air surfaces grip be touching past types depending against objects for the force to exist, these forces forces move These resistance Floating to contact movement. each each other, of forces are on where they forces known attempt as to prevent friction, originate from. drag, This gives other. experience upthrust from the fluid in which they float. Non-contact o ther forces (e.g. In field places some exist situations between the objects are not in direct contact but forces still them. g Planets are Electrons held in orbit around the Sun by the forces of gravity −1 ), check is need as g Moon sure (N). Howe ver , grav itatio nal stren gth are where −1 . newton forces objects Most the forces described this invo lving is strength. Contact EXAM force an so mak where e the same are forces bound cause to atoms attraction by electromagnetic and repulsion in forces. The magnets. se t. The nucleus of Combining The an force is is held together by strong nuclear forces. forces combination resultant atom of force. all In of the many forces situations acting you on will an object find that is the called the resultant zero. Weight Weight is attraction The a force which between weight of the the acts on object object an and depends object the on because of the gravitational Earth. two factors: the mass of the −1 object and the gravitational field strength (g = −1 W 12 (N) = m (kg) × g (N kg ) 10 N kg on Earth). WORKED Figure EXAMPLE 1.6.1 shows a 1 ACTIVITY tug-of-war. Which team is winning? Use the some plasticine effects Stretch Squash Roll it then of it to explore forces. until it snaps. it. into roll a ball the shape ball and along the desk. 400 N Figure + 600 N 300 N + 700 N Throw Rub it ball against along the the desk to wall. feel 1.6.1 the Forces to the left Forces to the right = 400 N = + 300 N 600 N + = 700 N 1000 N = forces are balanced so the Draw 1000 N resultant is frictional what what The a it does force see plasticine. diagrams to explain happens. explore magnets EXAMPLE and the zero. Now WORKED force to 2 the have Arrange effects on them each so that bar other. they repel or attract. A student of mass 45 kg jumps off a diving board into a deep pool. After the dive the student then floats on the surface of the Try to that a Calculate the weight of the the student. Draw W = balance one magnet so pool. the N-pole N-pole force of floats the above other. diagrams for these mg situations. −1 = b Draw he a falls size of 45 kg diagram and as these × 10 N kg showing he floats forces the on where Air = 450 N forces the acting surface of on the the student water. Give as the possible. resistance 450 N 450 N SUMMARY QUESTIONS 1 landing 450 N Figure The lunar which 1.6.2 a visited mass would KEY Earth POINTS of module Moon 15 000 kg. this and the weigh on the on had What the Moon? −1 (g = 1.6 N kg ) Moon 1 Forces 2 The are vector quantities. 2 resultant same 3 The overall weight of a force effect of an of is the object is a single other the force forces product which would have the combined. of the mass and If the lift field strength. (W = largest the What is could lift mass Earth the is you largest when could 150 kg. mass standing you on the the gravitational on Moon? mg) 13 1.7 Moments The LEARNING turning When At the end should be of this able determine force effect of a force OUTCOMES a force acts on an object it may cause a turning effect, known as you the moment the force of the force. This turning effect depends on the size of to: the acting topic moment about a of applied and the distance from the pivot or point of rotation. a fulcrum Calculating moments The of (pivot) combine moments resultant moment to find a moment perpendicular a M analyse to find systems forces in and is (N m) the to the = F product of the force and the pivot: (N) × d (m) equilibrium distances. Combining When WORKED force distance EXAMPLE 1 there moments are WORKED several forces EXAMPLE acting a resultant moment can be concrete slab found. 2 500 N 2.5 m 400 N 2.2 m m c 0 4 25 N Pivot Figure 1.7.1 Figure A long nut spanner using a is force used of to 25 N. turn What T wo is the moment of the 1.7.2 a force workers 1.7.2). the spanner is held 40 cm using large lever to lift up a (Figure Worker pivot. A applies Worker B a force applies a of force 400 N of at 500 N a distance at a 2.2 m distance from 2.5 m nut? from M (N m) = F (N) × d M M = = 25 N × the pivot. What moment turning effect produced? moment = (400 (worker A) + moment (worker B) × 2.2) + (500 × 2.5) = 880 + 1250 10 N m acting Clockwise and 2130 N m against each other can be subtracted in a similar way. anticlockwise Anticlockwise When describing anticlockwise 1.7.3 total 0.4 m Moments Figure the = = Clockwise is (m) Total 14 a from the the are when are the action used to of moments, describe the the terms direction of clockwise action and (Figure 1.7.3). Moments and equilibrium ACTIVITY When an object is in equilibrium it is not accelerating or rotating. The Test two equilibrium There The is no conditions resultant clockwise force moment the principle is acting equal on to the the object. anticlockwise moment. by suspending its centre centre (a second of these points is called the principle of of EXAMPLE works Try Metre 400 N from through best) and masses various masses balanced 3 200 N moments ruler the then on either moments side. WORKED a hole suspending The of are: until combinations the (Figure ruler is 1.7.5). or 500 N half-metre 0.4 m 0.6 m ruler d d Wire d 1 loop 2 R Slotted Pivot mass hanger Slotted m masses m 1 2 Pivot (nail hole centre Figure is in the distance equilibrium d of the (Figure third child from the pivot if the a seesaw in there is no Calculate 1.7.4). each clockwise overall resultant two moments = anticlockwise the side the facts the acting weights can be used and to find pivot the distance point. the values of the force R should find that the ruler and only balanced when the d upwards R The moments using masses the clockwise The of moments. is distance principle force You These the equilibrium: from Testing moments of 1.7.5 seesaw on For through ruler) 1.7.4 Figure Find of force = moments Clockwise (0.6 m R 400 N are + also moments × must equal 200 N the + sum 500 N of = the downward forces: the are equal to moments. 1100 N balanced. = anticlockwise 500 N) = (0.4 m 300 N m = 160 N m × moments 400 N) + + (d × 200 N) 200d N m KEY 200d moments anticlockwise = 300 – POINTS 160 1 A force can cause a turning 140 d = = effect 0.7 m (moment). 200 2 SUMMARY QUESTIONS What are the two conditions required for an object to be moment of product the perpendicular the An pivot. object is of a the to 3 1 The (M in force the is force and distance = Fd) equilibrium in when the clockwise equilibrium? moments 2 Three people 4.0 m. the Sketch three of a equal weight diagram people could sit on showing sit so that a the the seesaw of possible seesaw total length positions would be where balanced. are anticlockwise there is no equal to the moments overall and resultant force. 15 1.8 Stability Objects LEARNING are attracted At the end should find be the of this able topic each you to: centre of gravity for of the of billions and seems gravitational of forces call this to pull particles separately the come weight from a of we of the each of which use the single a Earth. Instead single equivalent object point as in a of is whole. the considering resultant This object. This single is called a centre of gravity objects compare objects the these force The of by force the range composed OUTCOMES the using centre of stability the centre of gravity is the point from which the weight appears to act. of position of Finding gravity. As the will the weight always directly This of be in above idea (Figure can centre an object gravity acts equilibrium the be 1.8.1). of centre used A to lamina of when it the is centre of gravity suspended from an a object point gravity. find is from a the thin centre sheet of of gravity for material in a lamina any shape. ACTIVITY Second Make near point several the Suspend it can small edges the of holes the lamina rotate freely. lamina. so that When it Stand comes to gravity is rest the directly centre below of the First point point of suspension. Centre Card WORKED of EXAMPLE of gravity suspension. draw Why do carry a some very tightrope long flexible walkers Plumbline pole lowers and of be pole lower held centre increases the and can their that (a mass string on with the end) through it The of ends downwards centre small plumbline. gravity stability. bend the so of line Repeat at Figure 1.8.1 Finding gravity gravity the of a centre of least and line centre process two a the pencil point following (This the the Use from suspension pole? a The a more the must of to of pass gravity.) using points of suspension. triangular The point where the lines meet lamina even further (Figue 1.8.2). is Three-dimensional Three-dimensional They draw can lines where you diagonal Figure 1.8.2 A tightrope be and would lines objects are suspended see the where expect joining middle of centre of gravity. objects they for the a little from a more a meet. The regularly corners uniform difficult number of a of cube ruler). to points centre shaped walker sphere, 16 also the of gravity objects meet, investigate. but the (e.g. it’s is hard where centre to usually of a the Stability An of object standing gravity above is the beyond on directly base. the If base the it a table above object will will the be point is topple tilted gravity gravity An object when if that can released tilting the the is centre be said object the centre of of gravity be results and in in is acts, gravity i.e. moves of Pivot the return stable the centre gravity outside will its force Centre Pivot tilted to when reaction Centre of If the 1.8.3c). of Pivot 1.8.3 equilibrium and (Figure Centre Figure in where base to of its the original equilibrium. centre of object gravity This being it will topple. position occurs moved upwards. If the object falls over with a slight push then the object was in Figure unstable lowers equilibrium. and so the This object happens falls into a when the position centre where it is of 1.8.4 During gravity more feet stable. of and these them Designing A stable car has for object a wide a judo competitors match spread bend down. actions more the their Both make stable. stability has a low centre wheelbase and of low gravity centre and of a wide gravity to base. make A it racing more ACTIVITY stable when gravity it might travels topple around corners. A car with a higher centre of Balance over. a flat they fall SUMMARY or Why only 2 does fill a the Describe Which and chest top how laboratory 3 of range can over. be tilted You can objects see how before tilt the on far they of drawers become more unstable if is you could objects in gently by pushing tilting the object surface the side itself. you find the centre of gravity for a KEY in Figure unstable 1.8.5 is in stable equilibrium 1 equilibrium? POINTS The centre object the is 2 3 The of the weight appears to centre of object from its at an which object gravity suspending of centre the of act. by Stability gravity point of found depends 1.8.5 from drawers? stool. the which Figure of and QUESTIONS by 1 a surface different an on of can be the points. object the position of gravity. 17 1.9 Investigating Hooke’s law Extension LEARNING OUTCOMES The At the end of this topic increase called should be able describe the to its original the force acting on and the its compare spring the caused force is by a stretching removed the force spring is may shape and size. 17th century, the Robert Hooke found that there was a simple between the extension of the spring and the force acting extension it. of The different a extension on of When a relationship spring length extension. relationship In between the to: return in you extension of a spring is proportional to the force acting on it. springs. Expressed mathematically F where the force is F is the spring effectively a to spring a is is a the extension spring the of stiff is: kx The measure law spring the and k constant by one stiffness of is a constant represents metre. a the The spring. A known size spring high of as the constant spring spring. EXAMPLE loaded 20 cm What is stretch indicates WORKED from x constant. required constant A force, = Hooke’s to the with a 5.0 kg mass and its length increases 45 cm. spring F = k = constant of this spring? kx –1 F 5.0 kg × 10 N kg 50 N –1 = = x b How long is 0.45 m the spring if – a 0.20 m load of = 200 N m 0.25 m 7.0 kg is applied? –1 F Figure x 1.9.1 = 7.0 kg × 10 N kg = = 0.35 m –1 k The spring 0.2 m, EXAM Mak e sure Hook e’s’ the law that the Limit you limita tions law as well as of Hooke’s new by length is 0.35 m from its original length of 0.55 m. proportionality law only applies proportionality. If the up until load is a point increased called after the this limit point of the spring itself . will continue proportion 18 the extended TIP remem ber to so has 200 N m to to extend the force but this applied. additional extension will not be in Elastic If the limit spring is stretched even further then it will become permanently Beyond This deformation called The the limit is of elastic happens elastic graph force means in will when 1.9.2 The Up the return spring to shows spring proportionality). limit). not to is its original stretched length. beyond a This point limit Figure applied. it this It the follows extension Hooke’s continues point the to of law stretch spring is a up up able to spring to to when point point return P E to a m m/noisnetxE deformed. elastic Elastic if the force is removed. Beyond the elastic limit the (the Increasing the permanently force (the its 0 original spring force/N will Figure be limit P Stretching length limit E 1.9.2 Stretching a spring deformed. ACTIVITY: FINDING Investigate a spring Suspend a spring Measure and Add a Add more Calculate Remove small THE using from record mass masses the its and SPRING the a technique stand at extension as described shown in here. Figure 1.9.3. length. record one CONSTANT a mm the time, for each new length. recording the Spiral length each spring 10 time. length. 20 Pointer is the followed loaded may masses during beyond be one by one unloading. the elastic If limit to check not, and that then the the the last same spring few data pattern was 30 Slotted Plot a graph and find of the load (x-axis) against total extension scale (y-axis) Load 50 The the gradient gradient of this using graph a line of best or stretching force fit. is: Figure 1.9.3 Investigating extension Δy masses 40 invalid. mm Hanger points Hooke’s law = load Δx 1 and so the spring constant is given by k = gradient Investigate the extension of a rubber band using the same process. To what Hooke’s extent do the spring and the rubber band obey law? SUMMARY QUESTIONS KEY 1 1 Calculate the length of a spring of original length 50 cm constant 40 N m Hooke’s the when a mass of 2.0 kg is Use the data in T able 1.9.1 to determine the spring a that spring proportional on it. (F to = the load kx) constant. 2 T able of it. acting 2 states extension suspended is from law and −1 spring POINTS The spring constant for a 1.9.1 spring to Force applied/N 0 1 2 3 0 3.2 6.4 9.6 4 5 12.8 16.0 the produce force unit required extension. 6 3 Length/cm is Stiffer springs have a larger 19.2 spring constant. 19 Section SECTION 1 What 1: SI 1 Practice units are Practice exam used for questions the exam questions 1 following 4 Find the resultants quantities? using a Mass a 16 km east b Time b 27 km west c Length d Speed e Density f The g Pressure 5 The Pythagoras’ mass and meteorite of a of these and 20 km and 50 km of measured Calculate south the Complete this table measurements a and standard small sample shown density in of of the the sample. force of with north as cm 2 displacements theorem. volume are diagrams. moment 1 descriptions the 3 cm 3 25 25 20 20 15 15 10 10 5 5 of appropriate prefixes forms. Description Using SI prefixes standard unit and …….kN 5 …….nm 7.2 form 3 a force of 5000 × 10 g N newton on −7 a length seven of × 10 m hundred 6 and An expedition to the planet Mars is being twenty planned. The landing module has a mass of nanometres 50 000 kg. a current of 30 …….mA a Draw a labelled force diagram showing milliampere this 990 μg module Mars. resting Include the on size the of surface the of forces. −1 (g = 3.7 N kg ) Mars 3 Use a these scale diagram combinations to of find the forces. resultant of b The closest and Earth spacecraft a days how approach is 56 can fast between million only must carry it Mars kilometres. supplies travel in If the for order 600 to 30 N to Mars run 50 N b 20 N 40 N 40° c 20 N 40 N 30° 2 5 N 20 and out? back before these supplies get 7 On a iron building girders. site To a crane is counteract used the to lift turning heavy 9 effect A group the of students stretching of a were spring asked and to investigate elastic band. 3 a concrete mounted position 2.0 m on of and balance block, the this of other block 10.0 m loads mass of 2.0 side can from of be the different × 10 the kg, crane. adjusted pivot is in They The and between order measured then recording to shown in the the extension elastic the the by of loading extension, the spring masses producing and the results table. sizes. Load/N Spring Elastic length/cm length/cm 3 The mass lifting a b of cable the is girder is connected Where should the placed for crane the What is while staying always the the has in × 10 3.0 m concrete largest to 4.2 to be load from block in the equilibrium be kg connected and the the pivot. 0 5.5 8.0 1 6.2 8.9 2 6.9 10.4 3 7.6 12.2 4 8.3 14 5 9.0 15.8 6 9.7 17.6 7 10.4 19.4 8 11.1 20.9 9 11.8 22.2 be equilibrium? crane if the can lift load 3.0 m from pivot? 3 2.0 × 10 3.0 m kg Pivot a Plot a suitable comparison graph of the allowing extensions the of the spring 3 4.2 × 10 kg and b During the to c 8 Describe how to find the centre of gravity the elastic. which elastic the part band of the extension stretching was proportionally load? Determine the spring constant for the of spring. an irregularly shaped metal sheet of uniform 10 A single spring was used to suspend a block thickness. of a mass 40 kg. What is The the spring spring extended constant for by 0.5 m. this spring? b How so can that two the combination i twice ii half identical spring be for arranged the is: that that springs constant of of a a single single spring spring? 21 1.10 Moving objects and displacement–time graphs Key LEARNING At the end should be of this able topic Displacements you define in key definitions describing and distances to: The definitions OUTCOMES the (starting used motion displacement point) in of a an object stated is the distance it is from the origin direction. of The total distance an object travels is not always the same as the objects describe object the by motion of interpreting displacement–time an a displacement, as the pendulum motion. during one 1.10.1 Units and motion the minute object but may move swinging its backwards will travel displacement will symbols used and forwards never be very during distance large. The speed of an The average object speed is for the a distance whole it covers journey (or in part a particular of that time. journey) is in given equations Unit or considerable velocity by: distance Quantity a graph. Speed T able A x Symbol average speed = or v = time distance m x displacement m s t −1 The SI unit of speed is the metre per second (m s ) but kilometres per −1 hour time s (km h ) and miles per hour are also used. t At different points an object may be travelling faster or more −1 speed m s v or c slowly than average. The speed at any particular time is called the −1 velocity m s v or u instantaneous speed −2 acceleration m s a The velocity of an object is Displacement–time Displacement–time from the origin over the in a particular direction. graphs graphs a speed period represent of time. the The distance time is of an plotted object on the 100 horizontal axis The in graph m/tnemecalpsiD the sprinters the blue while Figure are sprinter the displacement 1.10.1 from took the can be starting longer than is used line the plotted to at the determine any red on time. sprinter It to vertical how also far axis. away shows reach the that 100 m 50 finishing The line. velocity can be found from the gradient of a displacement–time graph: change Δs velocity 0 0 5 = displacement change Δt 10 in = in time Time/s Figure 1.10.1 The displacement sprinters during a of two WORKED EXAMPLE race What is the velocity of the red sprinter as shown in 100 m Δs –1 Velocity (m s ) –1 = = Δt 22 Figure = 10 s 10 m s 1.10.1? More complex represented as changes journeys, by a graph involving (Figure in the gradient: steeper the gradient The A zero A negative gradient towards (flat gradient the the line) changes 1.10.2). greater indicates (downward starting in velocity, These the zero slope) can changes be will be shown velocity. velocity. will indicate movement back point. 300 D 250 C SUMMARY QUESTIONS m/tnemecalpsiD 200 B 1 Calculate blue the sprinter velocity in of Figure the 1.10.1 150 A and the and C taxi of during Figure parts A 1.10.2. 100 2 Sketch 50 a more displacement a sprinter speeding 0 0 100 200 300 the race realistic graph showing up at and the then for him start of slowing Time/s down Figure 1.10.2 The graph analysed T able 1.10.2 Journey shows to Describing part give the the During 0–60 s moving B The the at taxi journey of of a the first a taxi through shown in a T able town. It can seconds of the the taxi taxi is The speed. 60 How line that seconds. the on the From graph the and 60 s 120–240 s This taxi moves speed is at a steady slower than speed for again. part A of The the journey. D Finally, so to has means the taxi stops again. The 240–300 s the the slope but graph a line 120 s The taxi of is the taxi on has the this has passed the line. movement slopes the is upwards not line is showing increasing. line on displacement shallower the taxi shows displacement horizontal. The he finish 1.10.2. 60–120 s C after 100 m be taxi sixty for a movement steady stops of information motion Description A the the is graph not is changing moving. on the slope than travelling graph is graph for more is upwards part A. This slowly. horizontal again and so stopped. KEY POINTS Acceleration 1 When is an object unrealistic would as changes it accelerate shows for velocity instant several it accelerates. changes seconds in and, The velocity . instead graph In of reality a sharp above the taxi change, graph would show a curve as the speed displacement–time shows the object over Similarly the sprinters would accelerate from the The starting they reached a top speed and then slow after the period of of an time. velocity finishing found of from an object the can gradient of blocks a until a graph changed be gradually . movement the 2 displacement–time A displacement–time graph. line. 23 1.11 Acceleration and velocity–time graphs Acceleration LEARNING OUTCOMES The At the end of this topic acceleration of an object is the rate of change of velocity: you –1 should be able change to: in velocity (m s ) describe object the by motion find the object find an the where a using of a – u = (s) t is the which initial the velocity, object v is the changed final velocity and t is the time velocity. an travelled the u during graph distance object v or an graph acceleration using the of interpreting velocity–time ) = time (m s –2 acceleration WORKED EXAMPLE 1 A accelerates by −1 rally car to 25 m s from a standing start in 15 s. graph. What is Noting the acceleration that the initial of the velocity car? is 0 gives: –1 –1 Velocity/m s v – u (25 – 0) m s –2 a = = = 1.7 m s 30 t 25 X X X Y 15 s X 20 X X Velocity–time 15 graphs X X 10 A X second way to represent the motion of an object is through a X 5 0 velocity–time X graph. On these graphs: X X 0 2 4 6 8 10 the time is represented the velocity on the x-axis Time/s Figure 1.11.1 A simple is represented on the y-axis. velocity–time Figure 1.11.1 compares the movements of two cars. Car X accelerates graph uniformly seconds –1 from but 0 then to 10 seconds. reaches a Car steady Y accelerates speed. It is for the important first to six notice Velocity/m s −1 that car Y does not stop. It keeps travelling at 18 m s until the end 6 of B X 5 the test. X 4 Using velocity–time graphs to find acceleration 3 A C The 2 1 0 acceleration can be found from the gradient of a velocity–time graph: X Time/s X X change Δv E D –1 acceleration = in velocity = X change Δt in time –2 –3 WORKED Figure 1.11.2 Motion phase of A a the toy. toy In phase B is is at a constant the acceleration of car X throughout the test, shown in the Figure toy 2 is What accelerating. EXAMPLE During 1.11.1? velocity. –1 During phase C the v toy – u (25 – 0) m s –2 a decelerates. Phase D = = = 10 s t that with is the a toy is now negative moving moving velocity; backwards it and As accelerating. phase comes 24 E the to Finally, toy rest. 2.5 m s shows with displacement–time graphs, velocity–time during slows and several phases of motion (Figure 1.11.2). graphs can show EXAM Using an velocity–time graphs to find the distance travelled by T ake TIP care no t to confus e object the You can area find the beneath broken the line into shapes WORKED The the down simple distance on a simple cannot be EXAMPLE simple travelled of a an velocity–time shapes used and the object graph. the area area may by measuring This area can calculated. be the be Where differe nt grap hs. T hey simila r but differe nt motion may they look repres ent quan tities estimated. 3 velocity–time motion by two robot. graph What in Figure distance 1.11.3 does it –1 represents Velocity/m s travel? 4.5 The graph is broken down into three simple shapes: 4.0 triangle A, rectangle B and triangle C. 3.5 The areas are calculated: 3.0 1 Area A = 1 × base × height = 2 × 5 × 4 2.5 =10 2 2.0 Area B = Area C = base × height = 10 × 4 =40 1.5 1 × 6 × 4 =12 2 1.0 The total area is 10 + 40 +12 which represents a distance 0.5 travelled of 62 m. Time/s 0 0 Figure 5 10 15 20 25 1.11.3 ACTIVITY Attach ticker vibrating regular Let the pin tape or intervals object to pen an object. which (usually fall, Thread produces 50 allowing dots a per a the dot tape on through the paper a at second). pattern of dots SUMMARY to be made on 1 the Calculate the acceleration of strip. the QUESTIONS This pattern reveals the distance the object has travelled graph th of a second and so the velocity and in each part of the in 1 each toy acceleration in Figure 1.11.2. can 50 be 2 calculated. a Calculate distance toy Ticker Object in the total travelled Figure by the 1.11.2. timer b released What is the final 5 displacement of the toy? Speed 4 KEY POINTS Clamp to hold base of Ticker 1 tape stand The acceleration can be found of from an object the 5 3 4 gradient of a velocity–time 3 2 graph. 1 0 0 2 Time 2 Soft The distance travelled by an pad 1 Ticker tape object can be found using Go with dots the Figure 1.11.4 Investigating falling using a ticker tape timer area under velocity–time the line of a graph. 25 1.12 Newton’s laws of motion The LEARNING Greek keep At the end should be of this able topic the you on to: is state and scientist apply an object faster his an the laws of observation of in If TIP his and does is as the force was be moving object on it. (v tends ∝ to However, The required F). greater He come based to Aristotle rest the to this force idea unless performed there no his idea Galileo, and the when, idea hundreds was found to of years be later, wrong. tests Isaac there mean girl are fo rces that no ideas called about the laws forces of and movement. Some motion. motion resultant force acting on an object it will remain at velocity. object will remain continue unmoving) to Roll a in straight a on to move in a straight line at constant speed change unless acted behaviour is on by called a resultant force. This inertia it. marble Note along line that a smooth unless it desk and encounters frictional forces are you will see something acting and that that these it continues produces will slow a force the there marble fo rce. alwa ys the of our are first objec ts, and developed law. o ppos ite fo rces (Figur e a velocity. ge t the third e differe nt the is reluctance resul tant paire d on to abou t and no t The no t becaus no by discoveries law there (or confus ed equa l an acting verify out fully key constant caref ul Just would that force to carried First law that constant situations. of Be thought a a Newton EXAM at Newton’s motion were range moving object obvious experiments three Aristotle OUTCOMES down. T he This act such first cause law an leads object to to the idea that it is always resultant forces which accelerate. chair 1.12 .2a). Second The law of acceleration acting on the of motion an object object and is proportional inversely to proportional the to resultant the mass force of the object. This statement leads to the force = mathematical mass × relationship: acceleration or F = ma −2 When force mass is is called WORKED expressed the in newton EXAMPLE kg (N) and in acceleration honour of in m s Newton s the unit of work 1 7 A cruise ship of mass 1.5 × 10 kg is pushed by an engine 4 force of 2.3 × 10 N. The water resistance acting on the ship 4 1.2 × 10 N. What is the acceleration of the ship? 4 The resultant force acting on the ship is (2.3 − 1.2) × 4 = 1.1 × 10 N. 4 F 1.1 × 10 N –4 Figure 1.12.1 The acceleration of a ship a = = = 7 depends the 26 on its resultant mass force. and m 1.5 × 10 kg 7.3 × 10 –2 m s 10 N is Third law of motion a Reaction When an equally sized When chair. in an The object sit Sun wobble detect the your force a chair force (Figure an the equally B then on B exerts force an A. acts force object downwards equal to your on the weight but 1.12.2a). Earth sized These planets weight reaction (Figure on object direction your a 1.12.2b). large on opposite produces exerts some a direction exerts also in on chair upwards Earth exerts force you The A which force stellar orbiting keeps on the wobbles other it in Sun orbit, causing have been but it the to used to stars. Rockets Weight Gases they from burning expand. downwards These and fuel hot the are expelled gases rocket gain gains from the base momentum an equal as and of a they rocket are opposite as b pushed momentum Sun upwards. Earth As the fuel is used and leaves the rocket, its mass decreases and the F Figure acceleration of the rocket will increase as a ( WORKED EXAMPLE 1.12.2 Examples of the third law = m ) in action 2 5 At launch, The a the rocket What is produces the Remember the thrust The initial the minus of a of of resultant the the a rocket maximum acceleration that weight mass and force the of rocket force = mg = is × 10 kg. acting launch? on the rocket will be weight. rocket 7.80 is given by: × −1 10 kg × 10 N kg 6 = 7.80 6 F 7.80 13.5 MN. at 5 W fuel 13.5 × 10 × 10 N 6 N – 7.80 × 10 N –2 a = = = 7.3 m s 5 m 7.80 × 10 kg 5 b One fuel of minute but the still into the flight produces rocket at this the the rocket same has thrust. used What is 1.0 × the 10 kg of acceleration stage? Figure 1.12.3 An is Now use the reduced mass of the rocket to find the Ariane used 6 13.5 × 10 rocket launch system satellites acceleration: from F to French Guiana. 6 N – 6.80 × 10 N –2 a = = = 9.9 m s 5 m 6.80 × 10 kg KEY 1 SUMMARY POINTS The first shows forces 1 Draw a law of motion QUESTIONS diagram showing a ladder leaning against a wall, that are resultant needed to cause so acceleration. that the someone ladder, can wall climb and up it. Draw all of the forces acting on 2 floor. The second shows 2 A skydiver just of before 1000 N mass 60 kg opening just after. her experiences parachute Calculate the two instants. (g and air an =10 N kg ) resistance air acceleration −1 these an of resistance of the that law F = of motion ma 400 N 3 of skydiver at The third shows in law that equal of motion forces and always opposite act pairs. 27 1.13 Momentum and impulse Momentum LEARNING OUTCOMES Momentum At the end of this topic an should be able the is a vector related to the mass and velocity of object. to: p state quantity you law of = mv conservation −1. of linear use of the An momentum law of momentum analysis of after of units shows that the units of momentum are kg m s conservation to find the WORKED velocity the objects before EXAMPLE 1 and impacts. What is the momentum of a minibus of mass 2000 kg when −1 moving at 5.0 m s ? –1 p = mv = 2000 kg × 4 5.0 m s = 1.0 × 10 –1 kg m s Impulse As you saw change the change in force and which it in the previous velocity and momentum this caused impulse is units means resultant the by the a force product forces are momentum of is called the required of an the force to object. impulse and the time The of the for acts: impulse The topic, therefore for that impulse the = are units of Ft = N s. change As this impulse in is and a momentum change of momentum momentum are this identical: –1 kg m s The symbol ≡ means Conservation As A the acts Ft Momentum momentum the size is impulse on both on it then, from object change to’. an object can only be changed when a force an must an interaction Newton’s object by third changes an between equal two law, then and you the can opposite objects A see that momentum and of when the another amount. B the forces acting conserved is the objects. the two objects must be of the same size and last for the same same amount Ft and of the quantities This In gives any This used time. This impulse of the law means the to means on B is momentum interaction law exactly 28 identical momentum of of on as of momentum In 1.13.1 ‘is N s B Ft Figure ≡ as given but in the by total the the of momentum situations where −Ft. of amount amount impulse The opposite conservation the that same analyse of that two objects momentum before A is given gain (Figure by equal 1.13.1). momentum: momentum two object directions linear of on the objects is after conserved. any collision. collide collision This with law each is can other . be Investigating To investigate used (Figure timer T o or carry needs by law 1.13.2). the be of measurements T o is of successfully do this a movement of movement experiment reduced. and conservation Their ultrasonic out to the momentum momentum, monitored their the trolley is trolleys using a can ticker be tape position. effect placed of frictional on a ramp forces and the ACTIVITY ramp is tilted spaced and dots. the trolleys so This friction can that be a ticker shows forces varied that have by tape the trolley been placing attached is to the moving compensated. additional trolley at The blocks has equally constant masses of metal velocity of the onto Use a ramp collect them. data collisions to and for timer Tape Pin of that so results the The 1.13.2 first partway or a The pin trolley down and is a ramp be to Trolley used to compensate allowed The make can B and ramp. data of can pushed the movement WORKED ramp cork conservation In A trolleys them then stick be law of momentum. will to have each analysed to (at rest frictional collide may to for A with demonstrate discuss the is sources exact of error . initially) forces. a Velcro other not relationship experimental Figure the to of Cork Y our Trolley range verify conservation Ticker trolleys a second strips, after confirm the the trolley magnets collision. law of momentum. EXAMPLE 2 experiment a trolley of mass 0.5 kg is pushed at a WORKED EXAMPLE Why running does 3 into a wall −1 velocity mass of 0.3 m s 1.0 kg and . It sticks collides to it. with The a second resulting stationary trolley pair trolley moves of hurt more than walking into it? off The change of momentum −1 with a velocity of 0.1 m s . Has momentum been conserved? will Find the momentum before the collision: = mv = 0.5 kg × 0.3 m s time –1 = the momentum after the collision faster will also remembering that that have a combined mass of –1 = mv = 1.5 kg × shows always that momentum 0.1 m s shorter. This force in momentum = ) time –1 = 0.15 kg m s conserved in the collision (as be much larger. it is). SUMMARY QUESTIONS 1 complete Copy T able was are impact the will This you the 1.5 kg. (F p be the change trolleys when and 0.15 kg m s means Find greater moving –1 p be and T able 1.13.1 by finding the missing values. KEY POINTS 1.13.1 1 −1 Object cricket Mass/kg ball Velocity/m s 0.16 Momentum of the of an mass is the and product the velocity −1 Momentum/kg m s object. (p = mv) 20 2 Momentum is a vector is conserved 6 meteorite 5000 6.5 × 10 quantity. 3 2 A car is pushed using a constant force of 500 N for 5 Momentum all How much momentum does the car in seconds. interactions. gain? 29 1.14 More The LEARNING concept the end should be of this able topic rocket you collide apply of the energy law to of be applied each other to situations and also to where explosions and a collisions range including of collision both objects could be moving. This is often the case collisions a traffic collision. The objects may not stick together in a collision, explosions instead can off conservation in and bounce to: During situations momentum and propulsion. More of momentum OUTCOMES objects At about apply the rocket laws of motion they can rebound and separate after the impact. to systems. WORKED EXAMPLE In of a a game match ball. the billiards/pool, white Initially 1 the cue balls balls ball are of mass 0.20 kg collides head on moving in are with opposite the used. During object directions. The cue −1 ball has velocity of 0.30 m s and the object ball has velocity −1 −0.10 m s . After the collision the cue ball continues to move in −1 its is original the direction velocity of but the at a object reduced ball after velocity the of 0.05 m s . What collision? Before: −1 −1 0.30 m s −0.10 m s After: −1 0.05 m s Figure 1.14.1 Choose the direction ball and Find ? the cue as ball direction negative. object initial as positive, Subscripts t, c and and o the opposite identify the total, total momentum of the balls: ) (0.20 kg –1 p = p t + p c cue ball. = (0.20 kg × 0.3 m s –1 + × –0.1 m s ) o –1 = Find the 0.04 kg m s momentum of the cue ball after –1 Figure 1.14.2 When billiard balls collide p = 0.20 kg × 0.05 m s the collision: –1 = 0.01 kg m s c they don’t stick together. The They often move off total momentum momentum different after the collision is the same as the with before and so the momentum of the object ball velocities. −1 must be Finally 0.04 find − the 0.01 = velocity 0.03 kg m s of the object ball: –1 p 0.03 kg m s o v = –1 = = 0.15 m s o m 0.20 kg o As the answer moving 30 in the is positive, same this direction means as the that cue the ball. object ball is now Explosions The law of conservation momentum following T wo The The track of (Figure held are initial When the gliders the This placed on together released momentum end of and opposite track are at of applies can in a be the even when the demonstrated centre cushion This (explosion) Further experiments masses and the same and means is with they still can measuring a of small will system released the velocities momentum. release are gliders are of the they The momentum zero. in initial the of air a linear and so air track. eliminates friction. when is 1.14.3). gliders floats the of system gliders that way a identical most of they time be is zero will the as move showing therefore that spring forced total their apart that equal between apart them the velocity and they and by opposite momentum is reach have so spring. zero. the equal amounts after the zero. be the carried out velocities – v using after gliders the of different explosion. +v Air track Gliders Figure Similar 1.14.3 Using experiments a linear can air be track to carried investigate out explosions using two trolleys on a track. KEY POINT Recoil 1 When by a gun rapidly the same time momentum. WORKED A rifle is fired the expanding of the It bullet gases. gun must therefore EXAMPLE mass is The propelled bullet gain moves an has equal backwards forwards gained and at high opposite of and a bullet of can of be of applied pairs of explosions. (recoils). SUMMARY fi res conservation collisions objects of 2 4.5 kg to At amount law momentum velocity momentum. The mass 10 g forwards with QUESTIONS a 1 A satellite has a mass of −1 velocity of 300 m s . What is the recoil velocity of the rifle? 500 kg. Use a subscript b to identify the bullet and r for the it rifle. ejects To adjust 50 kg of its speed fuel at −1. 40 m s Find the momentum of the in –1 p = m b v b The rifle = 0.01 kg × 300 m s momentum gain the the change same change in 3.0 kg m s speed also is and –1 = b must What bullet: momentum but in the of the satellite? opposite 2 If the 1 is fuel 1 minute ejected in Question direction. ejected over a period of –1 p –3.0 kg m s r v = –1 = = –0.67 m s what is the average r m 4.5 kg r force acting on the satellite? 31 Section SECTION 1 The 1: 1 Practice Practice exam displacement–time questions graph shows exam questions 2 2 the 4 During an experiment a trolley of mass 4.0 kg −1 height a of Find a skydiver the during velocity of the a dive. skydiver moves with with stationary and Find the of trolley 0.5 m s of . mass It collides 3.0 kg and two trolleys stick together moving off at a 50 s. new b velocity between the 20 s a a velocity of the skydiver velocity. What is the velocity of the trolley between combination? 50 s and 100 s. 5 During a docking manoeuvre a supply pod Height/km of mass 4000 kg International 2.0 accidently Space collides Station (mass with the 100 000 kg) −1 with the 1.5 a relative pod change 6 During to pot velocity attaches in a game the to velocity of final of the of 2.0 m s station the space snooker black. a The . Assuming what is the station? player white attempts ball strikes 1.0 −1 the stationary and the black black ball with is a velocity propelled of 1.2 m s forwards −1 with 0.5 a velocity Assuming calculate 0 Time/s 0 10 20 30 40 50 60 70 80 90 the of that the 1.0 m s both after balls velocity of have the the impact. equal white mass, ball after impact. 100 7 A billiard ball of mass 0.3 kg hits a cushion −1 with 2 A car takes 8 s to −1 15 m s slow down from a speed 5 m s the as it approaches a velocity of 2.5 m s . It rebounds off of −1 to a cushion in the opposite direction with a junction. −1 velocity a What b If is the acceleration of the a the car has a mass of of −2.4 m s car? 800 kg, what is What is billiard braking force required to slow the During take-off 10 000 kg can a jet aeroplane produce a of 40 kN. At the point of maximum take-off What mass momentum of the is the final momentum of the billiard ball? thrust 8 of initial ball? car? b 3 the the the An artillery gun of mass 1500 kg fires a shell plane −1 of experiences a drag force of Calculate the initial 10 kg at a velocity of 400 m s acceleration of the recoil velocity of the gun? the plane. –1 400 m s b Calculate the the of point acceleration of the plane at 10 kg take-off. 1500 kg 32 . 20 kN. is a mass What 9 A group of students conservation X and Y . of Trolley investigated momentum X was the using allowed to law two roll of a trolleys, along and accelerated before moving along adjusted trolley to both then counteract trolleys to collided continue with frictional to trolley move Y acceleration the of trolley X as it ramp? What distance did trolley X travel before a impact with trolley Y? forces. The The the down a the ramp is travels b ramp What mass of trolley X was 0.5 kg. causing c forwards Calculate before separately. d the and Calculate momentum just the after mass the of of trolley X just impact. trolley Y . X The graph trolleys between the two trolleys lasts for 0.25 s. e The impact Y Ramp shows during an the motion of the What Y two is the during average the force acting on trolley impact? investigation. –1 Velocity/m s Trolley X Trolley Y 2.5 2.0 1.5 1.0 0.5 0 0 1 2 3 4 5 6 Time/s 33 1.15 Energy and energy transformations The LEARNING concept due At the end of this topic to a be able define describe energy transfers and transformations calculate (energy acting to energy energy the or is fundamental transformation of to physics. All events occur energy. is defined as the capacity to do work, for example when a to: force energy transfer you Energy should of OUTCOMES work through a by All (and joule often a an object mechanical energy One done transferred) as moves of through a distance. This is sometimes referred work. work energy done) is is quite measured a small a unit called quantity in and so the joule kilojoule (kJ) (J). is used. force distance. The forms Energy is in T able in form T able of sometimes 1.15.1, of to energy 1.15.1 Energy energy referred help is us referred Forms of form to as being describe to as the a in different transfer of forms, energy. as A shown change transformation energy Description thermal This refers to the heat energy stored within an object. light Light carry is an without sound Sound the Kinetic potential energy from need energy vibrating kinetic electromagnetic energy is object for the particles energy Potential place is energy due to its in the is a to wave place associated sound energy the it can quickly medium. energy a and very of energy position with the wave. a moving stored (being off in object. an the ACTIVITY ground), being Think about electrical are so the a range devices useful energy to why they electrical as take you can, in as including energy spring state. an circuits electric transfer energy through wires current. This energy is stored in the nucleus of atoms. radios, transfers involving electric energy see electrical 2.9, 2.10 energy and see 4.5, and for transfers 2.11. motors Energy that energy. you identify transfer and transformation any The ideas explain 34 a bulbs. sure wasted chemical as telephones, microphones, Make or (such many thermal light Electrical in For and stretched) state transformations place televisions, energy Describe nuclear that physical of and us. its of how energy devices transfer and operate. transformation can be used to of Examples: When you chemicals energy When and you stored into walk in in heat kinetic into a light, the the sound, energy as (Figure When as potential transferred catapult elastic. energy the is energy transform heat, potential stairs body stretch the Fireworks up your to fire you a stone, release stone kinetic flies up it in potential the energy outwards energy energy shoot stored gravitational the potential 1.15.1). potential they energy into (Figure stored and into the even air energy is in is transformed 1.15.2). their chemicals gravitational (Figure 1.15.3). Figure Wasted 1.15.1 Chemical potential energy your in gravitational In any energy transformed more useful become transfer into that a force work given by work done This out transformation to energy energy the which has not some of cannot the be disappeared. surroundings or energy used It to has energy is do or W WORKED will and been = moves done force = just dissipated by an the object force. through The a distance amount × distance moved in the of we work say done is direction of the force Figure 1.15.2 Elastic Fd = Fd = a motorcycle flat 80 N road has using a to be force pushed of 80 N. to a garage How much of energy → the kinetic stone 2.5 km work × 2.5 × 10 5 m = 2.0 × 10 J QUESTIONS Figure Describe the energy transformations involved 1.15.3 Chemical using a battery-operated torch charging a mobile c boiling kettle. heat sound + b potential energy in: → a potential elastic done? SUMMARY 1 the energy 3 W energy EXAMPLE along be heat relationship: broken-down away + any in A → potential work acts has the or thermal work. spread Mechanical When cells energy + + light kinetic gravitational + energy potential phone energy a KEY 2 Copy and and complete distances for T able 1.15.2 mechanical comparing work done, work. 1 T able POINTS force Scientists in 1.15.2 terms describe of energy transformation a Work done b c 5.0 kJ 4.3 J 40 N A mechanical energy 2000 N and Distance 60 m and energy transfer. 2 Force events is this transfer called is work found of done using the 20 cm equation W = Fd 35 1.16 Energy sources for electricity Electricity LEARNING over At the end should be of this able describe is an important topic light, you for to: how very large kinetic distances energy electricity or which is to resources generate can be state of some energy as met transform it There by is using a a it is simple into to other transfer constantly wide range forms increasing of energy such as demand sources. energy sources electricity advantages disadvantages alternative heat. to used Non-renewable and alternative Non-renewable energy form OUTCOMES of and and so are in energy limited sources supply or are have fuels that limited cannot be replaced lifespans. using energy resources. Fossil Fossil of fuels fuels plants include and coal, animals oil that and natural died gas, millions of formed years from the remains ago. ACTIVITY These Research and the use renewable Caribbean of fossil energy and write a in fuels large fuels amounts pressure the about what should to reduce the fossil fossil spin energy. turbines producing The heat which waste is drive used gases to and produce electricity high generators. of can produce carbon very dioxide are large amounts released of leading energy to but global dependency Oil this Sulfur is causes the most accounting SUMMARY dioxide acid is also released, especially from burning coal, rain. significant for more energy than 90% source of the used total in the Caribbean, electricity production. QUESTION Nuclear Which energy require a power sources: A a furnace fuels. and 1 a thermal to fuels quantities warming. on of steam in be vast done burnt short Burning report are large amount nuclear power station produces heat using nuclear fuels such as of uranium. These fuels are not burnt but release thermal energy inside a land? reactor b damage c are the core can be used on a near to where electricity power is fairly plants cheap electricity. very to are very operate. However, dangerous a process called nuclear fission. expensive They can to build produce and very dismantle large but quantities and they also requires produce safe radioactive storage for waste thousands which of years. required? Accidents are contaminate No nuclear very large power rare but areas the of stations escaping radioactive material can land. operate in the Caribbean and none are POINTS currently Large-scale production fossil fuel power There station electricity takes place burning or in nuclear had in planned. in been the built. are a range of energy other Renewable can a can started construction abandoned find the additional of project detail a nuclear before about power the reactor nuclear reactions smaller energy sources sources produce energy sources produce electricity without being used electricity up. on You but 5.4. Renewable which Cuba 1980s plants. renewable The sources are either quickly replaceable or have effectively scale. unlimited 36 through the is 2 split small of scale 1 nuclei unreliable? are KEY the atmosphere? Nuclear d when lifespans. Several examples are shown in T able 1.16.1. T able 1.16.1 Energy Solar A source power summary of some Example There are no large-scale cells) power the plants many buildings panels Most solar Caribbean Key No sunlight converted to advantages fuel Plenty in the required. of sunlight Caribbean. Electricity using can produced semiconductor building cells. it is to on in be be Water has is trapped dams a 10% dam used electricity No behind and to as allowed fuel Can flows used Wind turbines The but electricity battery storage required. Quite expensive to build. used. it of required. which amounts electricity Water very quickly. drives on fuel required. Predictable and a energy output. amounts precious small- and systems Caribbean No in large is of land resource islands. Only scale to is estuaries not Floods few trapped are large There islands valleys are estuaries medium- can very in be used as there or few the on are rivers. suitable Caribbean islands. turbines. Alters habitats and wildlife. currently. Wigton farm on wind Jamaica produces of around provide energy so the produce large drive turbines is Caribbean could produce night which escape. the at heaters needs. Tidal Can’t in Dominican providing power disadvantages roofs. Republic Tidal Key have water the the operates electricity installed. twenty it in energy from is sources directly new homes their The of in have Barbados on The solar Caribbean but energy How (photovoltaic Hydroelectricity renewable 20 MW electricity. The wind turbine spins blades mounted on towers. No fuel Plenty required. of around wind island Offshore tourism Some turbines by noise systems. produced Large-scale large wind or could spoiling pollution especially wind affect views. is from farms. farms small-scale local generation possible. Geothermal Beneath energy volcanic of St Kitts Nevis vast the islands and there geothermal Research are are reserves of energy. projects underway develop a to power station. Biofuels Sugar islands could on produce crude is many and be ethanol energy No released by is pollution caused radioactive sources decay reliable. the within Earth used to water Only and are a very few locations are suitable. very is heat into steam and steam drives the turbines. cane grown Thermal some used oils to oil. to or replace Biological New material, be as wood, sugar or such ethanol, is The not add extra carbon to land crops to Does dioxide can fairly quickly. cane, burnt. supplies grown used may grow be food to grow better for fuel used increasing populations. the atmosphere. 37 1.17 Potential kinetic Potential LEARNING energy the end should be of this able topic masses you force to: the calculate and energy can arise because an object is stretched or OUTCOMES compressed. At energy the kinetic energy the change to When the acted you spring through investigated and a the force distance Hooke’s caused and law the therefore in 1.9 spring did to work you added stretch. in This stretching spring. of objects Gravitational calculate gravitational in a range potential of potential energy An object that potential situations. in its can fall energy. gravitational change in due When to it potential potential the pull changes of its gravity height has gravitational then there is a change energy: gravitational gravitational = EXAM energy in mass change × energy × field strength in height TIP or ΔE = mgΔh p T he trans positio n the kine tic equa tion most of This equation is probab ly diffi cult one need strugg le the equation for mechanical work done. the you WORKED will matches energ y to do. then If EXAMPLE 1 you mem orise How much energy is required to 500 kg the sand alter nate show n on lift version s these page 500 kg 20 m (Figure will work be to a height of 1.17.1)? 2 ΔE done equal potential EXAMPLE sand s. The WORKED of lifting to the energy = mgΔh = 500 kg the sand gravitational gained: 20 m p –1 a A fast bowler can bowl × 10 N kg × 20 m a 5 = cricket ball of mass 160 g 1.0 × 10 J at −1 40 m s . How much kinetic sand energy 1 E = does this ball have? 2 mv Figure 1.17.1 Gaining gravitational 2 k potential 1 = –1 × 0.16 kg × (40 m s energy 2 ) 2 = b 128 J What is the speed of powerboat of mass it of kinetic a 600 kg if Kinetic has 40 kJ The The rearrangement of energy energy? kinetic movement. kinetic energy energy of an object is the energy equation The relationship 1 kinetic × E 2 × with its is: gives: energy = 3 2 associated the 40 × 10 1 2 × mass × speed or 2 E = k J 2 mv 2 k v = = √ √ m 600 kg As you more –1 = 38 11.5 m s can see kinetic an object energy than with a a large small, mass slow and moving high speed object. will have Transposition of the equation WORKED To find v if given the mass and the kinetic energy, you need the equation. Here is the coconut 1 Original of mass 0.5 kg falls process. 25 m 3 to A rearrange EXAMPLE equation: E = a tree. How fast will 2 mv it 2 k from be travelling when it reaches 2 Multiply both sides by 2: 2E = mv = v the ground? k 2E Find k Divide both sides by the gravitational potential 2 m: energy m ΔE 2E lost: = mgΔh = 0.5 kg = 125 J p k T ake the square root of both = sides: v –1 m Transfer between E and is common. It of k gravitational happens every 10 N kg × 25 m E p Transformation × Use potential time energy something to falls. kinetic The energy to the find kinetic the equations 2 energy equation speed: × E 2 × 125 J k v for kinetic energy and potential energy can be used to calculate = = the √ m speed of the falling object, assuming there is negligible air 0.5 kg resistance. –1 = Oscillations A pendulum gravitational side there and back is of a pendulum serves as an potential a transfer again. 22.4 m s The example energy. of As of the the gravitational quantities of transfer pendulum potential energy between swings energy involved can kinetic from to be and side kinetic to energy calculated SUMMARY using the equations in the worked example for the falling 1 Calculate mass falling A EXAMPLE pendulum 3 cm above freely. bob its What is of 4 25 m. mass 60 g equilibrium the is displaced position maximum speed and of sideways then the so allowed that to it is 2 An a the gravitational potential energy the bob loses as it of a when you jet loaded It from notice? aircraft mass can 12 km 1.0 kg 2.0 kg ground do A380 fully 550 000 kg. bob? a reach the What height Find will to speeds then Airbus has swing the and coconut WORKED QUESTIONS coconut. of travel with a at a speed swings −1. of to the equilibrium 250 m s position: a –1 ΔE = mgΔh = 0.06 kg × 10 N kg How much does it How much kinetic energy –2 × 0.03 m = 1.8 × 10 J p When the pendulum reaches the central position all of this b potential energy will be lost, matched by a gain in have? kinetic potential and the speed can be calculated (Figure gravitational energy energy does it 1.17.2). have? 2 × E k v = m –2 2 × 1.8 × 10 J = Maximum Maximum gravitational gravitational potential potential energy energy KEY 0.06 kg POINTS –1 = 0.77 m s 1 The change potential ΔE = of gravitational energy is given by mgΔh p 2 Figure 1.17.2 Energy changes Maximum The kinetic energy of an kinetic 1 in a pendulum swing energy object is given by E = k 2 mv 2 39 1.18 Conservation The LEARNING At the should end be most important rule about energy of is the energy law of conservation of OUTCOMES of this able topic energy which states: Energy cannot you be created or destroyed. It can only be transferred or to: transformed. state and apply the law of This conservation provide of rule allows examples the is dissipated surroundings reducing energy the analyse and explain a wide variety of systems. as for energy loss to the surroundings into There heat amount available to where Spotting energy us energy is always a loss transformations, of though future but transfers. no energy longer Dropping If you of energy usually is as heat. disappearing useful for to the This when doing surroundings means in fact that the it in energy often energy is looks still as present work. objects pick up gravitational a wooden potential block energy and will drop it onto transform to the ground kinetic the energy as the Gravitational block falls but transformed sound the Kinetic (Figure surroundings similar kicks and on energy the block thermal will energy 1.18.1). and stop. Even becomes The warming the original up the sound energy block energy has and been some dissipates into heat. energy A Heat then into sound process the ball warm up rest lost is as happens gains some to the and of when loses the you kinetic energy kick a football. energy. turns into Over heat With time within repeated the ball the will ball. The surroundings. impact Pendulums When Figure 1.18.1 Energy is dissipated the the a pendulum swing will decrease. over Some a of period the of energy time, is the leaving height the of system. surroundings. There the two frictional bob. air are The rubs to and Eventually losses. need As force the be time. these is stand. it of this provided masses energy are air the the will This it also will of the loss. the kinetic As forces. pendulum motion of pendulum frictional swinging to a loss keep system downwards to the this frictional force as energy the wire the of is wire the energy. energy provided small energy to to random where reduces stop gradual energy into point a leading experiences kinetic be heat attached move is At into with happening the transformed there pendulum Masses potential reduce energy). stand the Because to will transfers the through energy (heat against system moves kinetic particles attached of transformations pendulum This 40 oscillates into the very due the to swinging which slowly pendulum. these energy pendulums for drives their in long the clocks periods pendulum. gravitational Swinging When even you are though your centre gain kinetic chemical forces on a there of you energy gravity or you your can loss can gravitational potential with swing is energy keep to the drive the potential to drive it moving swing energy. the for a surroundings. swing and so seem you you time adjusting you However, when long By are to using produce muscles. ACTIVITY Figure Investigate ball down the a Measure energy ramp the of losses due different height to friction gradients difference to by rolling (Figure calculate a trolley 1.18.2 or 1.18.3). the loss of EXAM gravitational potential Measure speed to find the the kinetic of the trolley or ball at the end of the ramp If you the losses: do no t know energy. where Explain TIP energy. does a steeper ramp waste more or less the energ y energy? miss ing has gone trans fo rm ation probab ly into KEY Figure 1.18.3 Investigating energy Describe changes trans fo rm ed POINTS 1 Energy is 2 During always energy energy into heat and this longer when a has conserved. losses EXAMPLE the it heat. some WORKED in a heavy ball is dropped onto in transfers is transformed the surroundings energy be can no used. a trampoline. SUMMARY The ball starts transformed with into gravitational kinetic energy potential as it falls. energy When and the this ball hits the 1 trampoline the trampoline stretches, this is because the How is being transformed into elastic potential is energy lost when kinetic you energy QUESTIONS is energy. push a book across your When desk? the be ball has no transferred upwards energy. energy The has eventuallly and ball the into of kinetic continue continue ball but energy the stop the elastic energy and to and the until gravitational be each will moving. all will of heat been will move potential its of When is energy. you and to and place a stretches. pulled released energy until it then up the tranferred 2 spring kinetic potential to have energy ball stretching converted energy strain gravitational upwards during will initial will left energy transferred some all energy kinetic will been will trampoline heat back gaining The cycle kinetic the system down. explain If onto the why and oscillates Describe in a mass downwards transfers eventually mass this the the process mass stops. 41 1.19 Power Some LEARNING devices the should end be transfer energy efficiency at a greater rate than others. For OUTCOMES example, At and of this able topic one you a large second car than a engine small will be moped able to engine, transfer as it is more more energy in powerful. to: Power calculate power calculate the energy efficiency transfer or of an The rate of energy transfer is called power and is defined by the equation: device. work power done (energy transferred) = time taken E or P = t −1 The unit The power done and transfer WORKED EXAMPLE of power the equation so or is can be J s which applies used to for any has the transfer mechanical special of name energy work, or electrical watt any energy heating 1 An electric total This a motor mass of process What is Weight is the = used 600 kg takes Remember 30 work to mass lift the × a an elevator height of and 40 m passengers (Figure with 1.19.1). seconds. done use to through by the weight elevator of gravitational the motor? elevator field and not the strength –1 40 m Elevator = = 600 kg = 6000 N Now calculate W Fd × the 10 N kg weight: with 5 passengers = = 6000 N × 40 m = 2.4 What is the effective power rating 5 E P = 42 Lifting 2.4 × 10 = t 1.19.1 × 10 J 600 kg b Figure (W) work J = 30 s 8.0 kW of the elevator motor? mass: Efficiency During any energy transfer some energy is wasted and lost to the EXAM environment. there is This always wasted some energy heat depends on the specific device produced. If The efficiency transferred of a transfer is a measure transfer As output and there the is input the how the and lift energy is so represents input value some no motor transfer is have try the total the an or power energy output is efficiency or power always of output Y our defini tely a go back ques tion answ er no t is co rrec t. input. smaller greater of an greater then the 100% energy find of 100% agai n. useful wasted can than than 100%. 2 mentioned energy the ever efficie ncy value represents energy EXAMPLE much you and × value always WORKED If much value = input the how than output Here of usefully: efficiency TIP but in wasted Worked when example lifting the 1 is 70% elevator efficient, and passengers? First find out how much output efficiency energy supplied to the motor: value = input was × 100% × 100% value 5 output input value value 2.4 = × 10 J = × efficiency 100% 70% 5 = The energy law of 3.4 × wasted 10 by conservation of J the motor can now be found using the energy: SUMMARY Energy wasted = energy supplied – useful energy 5 = (3.4 – 2.4) × 10 output 1 5 J = 1.0 × 10 QUESTIONS What is the power rating of J an engine which 40 kJ per How much transfers minute? ACTIVITY 2 Use a small (Figure electric 1.19.2). electrical motor Calculate energy supplied attached the to gain the to in a joulemeter potential motor to find to energy its lift objects and is the motor efficiency. 75% Does the efficiency of the motor change when the load by with in power useful provided an an one efficiency hour rating energy electrical of if it of has a 150 W? is changed? Joulemeter Ruler Wire KEY 1 Motor POINTS The power rate at of which (transfers a it device does energy) as is the work given by E P = t 2 Metal Efficiency how useful 1.19.2 Measuring the efficiency of an electric a effective transfer Figure is measure of block is at an energy producing energy. motor 43 1.20 Pressure Pressure LEARNING When At the end of this topic be able you calculate the walk Your on a sandy weight is pressure of your feet causing a a acting you down leave on trail of sand footprints and it acts over the = F or p = surface area a the pressure. force Pressure on beach pushing to: area surfaces you behind. should on OUTCOMES describe how with depth the pressure in a increases A −2 The unit of pressure is N m which is also known as the pascal (Pa). fluid. –2 Pressure can also be measured in N cm 2 . As 2 1 m = 10 000 cm , 2 1 N cm = 10 000 Pa. ACTIVITY Draw to around estimate Calculate your the the feet area onto of pressure your you some feet exert squared and on then the paper . floor A re a: 2 A m 40 0 2 (2 = Figure × 13 .3 N 0 .0 1 5 ) m 2 kP a 1.20.1 WORKED EXAMPLE 1 Figure I F = N = 0.0 15 squares weight. 1.20.2). P 40 0 the your (Figure W eig ht: 150 cm Use measure have a mass of 60 kg and 1.20.2 How much pressure do you produce on the floor? the 2 total area of my feet is 0.04 m . Pressure What is the pressure on Pressure sand? be F p = in liquids the acts throughout demonstrated simply a liquid by and allowing increases water to with flow depth. out of This holes can in a 600 N = = 15 kPa container. The further down the hole is from the surface of the liquid 2 A 0.04 m the further greater the force water or squirts out. This shows that it is experiencing a pressure. ACTIVITY Drill Holes some small with water. holes into a tall, plastic, measuring cylinder and at fill different depths it depth of the Compare water in the the distance container. the What water is happens ejected as the to the water Water beneath level the decreases (Figure 1.20.3)? surface Squeezy Another bottle Jets pressure experiment uses Pascal’s vases. of water Explaining When Figure 1.20.3 The pressure in a a with submarine is in under pressure onto the skin water of the the weight submarine. depth. acting 44 increase of the water acts liquid downwards increases the on the surface of the submarine. This produces pressure Imagine width The the w water and volume given above length of the l the giving water submarine an area directly of l above to be × w the a rectangle (Figure with 1.20.4). submarine would be by: Column volume = length × width × height or V = l × w × liquid The mass of the water above the submarine would be of h given of density by: ρ h mass The force weight = volume acting of the weight × density downwards water = mass W = l weight of the which × or on m the would l × w × submarine be gravitational = given field h × ρ would be the by: strength w or × w × h × ρ × g l The The water acts over the surface of the submarine, l × w. Figure pressure acting on force pressure the l = × submarine w × shows depth At (h) very that as the long great compressed ρ × pressure as the liquid the is so increases is in has pressure a at a liquid × ρ × at a g high is the that particular same the throughout. liquid is slightly. 2 density a h constant liquid 3 Mercury within w of pressure density EXAMPLE a density the the in × Pressure g = l depths and WORKED × = area This h 1.20.4 is: depth of 13.5 × of 30 mm 10 −3 kg m beneath . What the is the surface of increase a pool of SUMMARY QUESTIONS mercury? 3 p = h × ρ × g = 0.030 m × 13.5 × 10 –3 kg m 1 –1 × A nail plank 3 = 4.05 × 10 is knocked into a 10 N kg of wood using a force –2 N m of 60 N. The point on the 2 nail has an area −6 (1.0 Pressure in × gases when The m nail Earth’s atmosphere is a deep layer of gases reaching Water has beyond 100 km above increases in difference the Earth’s depth in in density a as surface. similar you Pressure way travel to the deeper in the greater a is the wood hit? pressure into the than that in liquids and so the sea level referred the to Blue the as pressure ‘one is in a liquid but atmosphere relationship approximately atmosphere’ Mountain Peak in 100 kPa pressure. Jamaica If you you is not as which is climbed would is pressure 40 m above sea level. At this height the air of is the reach surface? Don’t pressure is the forget to far the pressure of the simple. above the water. often to a beneath the the top height of KEY 2.2 km density What atmosphere (0.8 is the atmosphere include of What on −3. 1000 kg m At ). acting the 1.0 mm atmosphere 2 The 10 pressure of 2 only POINTS 80 kPa atmosphere). 1 The pressure acting on a F As water is much denser than air, travelling to a depth of 10 metres surface in water will Deep-sea increase divers experience a can the pressure reach pressure of a 50 depth acting of on nearly atmospheres. you to 500 m two is given by p = A atmospheres. causing them to 2 The pressure given by p = in a liquid is hρg 45 1.21 Floating, sinking and density Archimedes LEARNING has At the end should be was a of this able topic it gold, you that, he when asked discovered explain float why and some calculate acting why some its lived the on an weight a key principle if a in the which crown 3rd was century explains made BCE. why from Legend objects pure float or sink. resultant force object a and principle sink as object, force wholly equal to the or partially weight of immersed the fluid in a fluid, displaced by is buoyed the up by a object. result It of who investigate objects Any scientist to to: Archimedes’ Greek OUTCOMES is important to note that this principle applies to objects which are buoyancy. floating In and addition their own to objects which submerged volume. This are objects fact is submerged will displace often used a to in the fluid. volume calculate of water the equal density to of materials. When you weight of get into that a bath displaced of water water will you be will the notice same the as water the level upthrust rise. you ACTIVITY Investigate called a Archimedes’ Eureka Measure the submerged in the can) weight in the principle (Figure of a displacement can (also 1.21.1). water water. using displaced Compare this when to the an object apparent is reduction weight. Spring balance 2 4 A 6 Weight of steel 8 2 block = 8 N 1 0 4 Weight of steel A 6 block = 5 N 8 1 0 Displacement filled to can overflowing Steel Displaced block water Steel block Beaker Weight 0 of Weight empty beaker 2 2 N 7 displaced water = 4 6 Large on vessels the side Plimsoll line have called or a the line. Figure 46 1.21.1 Investigating 3 7 4 5 marking water 5 N 6 5 1.21.2 2 8 plus 3 Figure 1 9 beaker 8 = 0 of 1 9 empty Beaker Archimedes’ principle The feel. Floating objects WORKED An object will float as long as the upthrust (or buoyancy) is equal weight of the object. Solid objects with a density greater ferry of water will always weigh more than the water they St so will used Lucia to to transport cars Martinique. The displace ferry and is than from that 1 to A the EXAMPLE has a fully loaded mass of sink. 6 5000 Ships are built from steel which has a much higher density than water. tonnes density of 3 They float because their average they density contain to much large lower volumes than the of air and density of this reduces water. 10 of the ship weighs. to float Loading the ship and the ship will it up must the therefore sink the lower displace ship with weight into the the cargo of same will water weight increase of water the displaced. as weight This 10 is kg). 1.02 The × . water What the to is ferry the volume needs remain to floating? it of means × −3 kg m displace For (5.0 seawater that The of water. mass must be the of the the water same as displaced the mass ferry. mass If too the much sea. ensures cargo The is added Plimsoll that line enough to the ship indicates extra then how buoyancy is it will heavily kept sink loaded in reserve too the in low ship case in is Density = volume and waves 6 m cause water to start entering the ship (Figure V 1.21.2). 5.0 = × 10 kg = 3 ρ If the ship is damaged and water enters into the hull then 1.02 × −3 10 kg m the 3 = weight of the eventually, becomes it ship increases sinks. greater The than and average the it sinks density density of lower of the the into ship the as a water 4.9 an object objects is A beneath the surface 3 m whole of a fluid Archimedes’ EXAMPLE hydrogen mass When 10 water. WORKED Submerged × until, of balloon 40.0 kg 2 has and a a total volume principle 3 of tells us there will be an upthrust force acting on the object equal 40 m . If the surrounding air to −3 has the weight of the fluid displaced. This upthrust force will seem a the weight of the object by decreasing the resultant of 1.225 kg m , to how reduce density large is the upwards force force. acting First Submarines on find the the balloon? mass of the air displaced: Submarines can alter their average density by allowing ballast tanks −3 m to fill with water. This increases the weight of the submarine = ρ × V = 1.225 kg m × which 3 40 m makes be it sink pushed the in out weight of the of water. the the To tanks gain using additional buoyancy compressed air and the this water = 49 kg can decreases This means displaced submarine. weight is of the weight 490 N the (W balloon of = is the air mg). The 400 N. Balloons Finally A balloon that the or there weight balloons their airship will of and be air a is also force which airships to submerged acting has been float as in upwards a fluid, on displaced. long as the the the air. This balloon This buoyancy upthrust is means equal to resolve upwards weight the force = = 490 N forces: upthrust − 400 N − = 90 N allows greater than weight. KEY 1 SUMMARY POINTS The density of a material is QUESTIONS m given by ρ = V 1 Draw a diagram showing all of the forces acting on a submarine 2 which is accelerating forwards while sinking in the An a 2 A balloon of mass 10 g is filled with helium of object floating on, or in, ocean. mass 0.4 kg fluid displaces a weight of so fluid equal to the object’s 3 that its volume is 5.0 m . Will the balloon float in the air? weight. 47 Section SECTION 1 A 1: Practice student motion bearing the has of mounted a is is follows fl oor. travelled the compressions the of compression bearing The ball the of the travels bearing compression of and the as spring is the 2 a Describe the energy transformations 3 transfers that take takes off and place when: a a helicopter b a bungee c a tree that jumper dives off a bridge The falls over in a forest. bearing to the 3 distance and range A a of car of force the mass of car 600 kg 500 N. rises by The is pushed slope is up a 50 m hill long using and 4 m. investigates length of times a How much b How much does the work is done in pushing the car? the distance reaching three the spring x. falls for and before fi red it the the spring ball questions ball so distance bearing the a The spring a between from exam 3 investigate table. measures ball relationship ball a trajectory student by into released curved The of to fi red compressed then a edge pushed questions asked bearing the is Practice exam been ball at spring spring 1 the for gravitational potential energy the car gain? fl oor. c each Assuming is spring. counted efficiency 4 x that During a world’s tractor as of gain useful the required and distance of a the 20 m work, to road. tractor in a A is energy calculate the transfer. determine human flat potential energy competition strongest along in the competitor force of moved pulls 4000 N through a is a 70 s. 1 m a What is the work done the effective in moving the tractor? b What d Compression is distance, x/cm 2.0 3.0 4.0 5.0 6.0 Travel distance, d/cm 26.2 38.8 51.0 63.4 76.2 Travel distance, d/cm 26.6 38.8 51.0 63.4 75.8 Travel distance, d/cm 26.6 39.0 51.2 63.4 76.0 a Calculate the average travel distance ball bearing of the for 5 the power competitor? for each State the law of conservation of energy. compression Describe how this law is applied when finding distance. the b Plot a graph distance comparing (x-axis) to the the 6 distance A new business electricity. c Draw straight d Calculate the line of gradient best of They a wind turbine To what the extent hypothesis proportional spring? 48 does that to the the the an electric motor. a paragraph wished are or to generate considering photovoltaic the its own use cells. of Write fit. this data travel for each system describing line. advantages e of compression travel (y-axis). a efficiency support distance compression of is the and disadvantages. the 3 7 A pulley system consisting of three pulleys is 10 A ship is made of 4000 m steel of density −3 used to lift through a a pallet height of of bricks 40 m. weighing The force 600 N 7900 kg m required to a What volume of fresh water (ρ = water lift the pallet at a steady speed is 210 N and the −3 1000 kg m rope is pulled through a distance of Neglecting the mass of the pulleys calculate the useful work done the What is Explain to In the what in order how the displace efficiency of the pulley to for it to float? design this of much the ship enables water. A rectangular form is energy wasted in block of polystyrene foam with system? dimensions c need pallet. 11 b ship in it lifting the and b rope, would 120 m. displace a ) the 2.0 m × 1.0 m × 0.3 m is used to lifting make a raft. process? The density of the polystyrene −3 is −3 50 kg m and What is b What volume when the How raft the that a c foam mass of block much of water of the water 1000 kg m polystyrene will be block? displaced floats? additional support is before weight beginning could to the sink? Pull 12 Explain the 13 why a helium-filled balloon rises in atmosphere. The following from table hydroelectric shows plants Location the on power output Jamaica. Installed Power output/MW Upper White River 1945 3.8 Lower White River 1952 4.9 1949 3.8 1949 2.5 1966 6.3 1989 0.8 1989 0.6 1989 1.1 Roaring River 600 N Rio 8 A tourist submarine has a glass dome Bueno Maggotty allow clear views of aquatic life. The withstand pressure of a maximum 12 000 kPa. The absolute density 3 seawater is 1.02 × kg m Rams of is the maximum safe = 10 N kg ). diving depth of a −3 The density Which float of in of the water is 1000 kg m following cubes of material Find the will b Assuming year of side b output of the system. the (365 turbines days), production of operate estimate the system all the total over a year . Mass c a power that around energy Length total ) water? Material River hydroelectric −3 (1 g cm Bueno the submarine? 9 Horn –1 (g Rio What Spring external −3 10 Falls dome Constant can A to 4.0 cm 10 cm The total electrical energy production 16 60 g in Jamaica was percentage 1.2 kg produced of by 2.0 × 10 Jamaica’s J. What electricity is hydroelectricity? 4 c 3.1 m 2.3 × 10 kg d Suggest why stations have no new been hydroelectric built since power 1989. 49 2 Thermal physics and kinetic theory 2.1 Heat, energy and temperature A LEARNING good understanding behaviours At the should end be outline the of thermal energy is required to explain the OUTCOMES of this able the kinetic topic them. you to: development theory of It of took were hot The end different many and why materials years for objects and the scientists energy to transfers explain why between some objects cooled. of thermal of the caloric theory energy During describe energy the flow from one of heat object was to called another. The the the some. Count heat hot holes cannon Over to the next developed be generated 2.1.1 The mechanical work boring was out this converted to by provided (Figure 2.1.2). would the meant to heat water the always other found the If that heat a was water of Joule This a no to gain involved large amount properties physical to ideas behind the principle of water point. There James Joule, energy the that temperature increase was research of eventually led energy. cannon heat. Paddle-wheels Mass moves at to a energ y from high er an objec t an alwa ys objec t tempe rature at a lower tempe rature. Liquid Insulated Figure 50 2.1.2 Joule’s apparatus for container transferring (no could mechanical TIP T herm al of the temperature conservation in substance under boiling measured in object released. internal the moves. meaning work placed including increase supplied. clearly were heat fluid heat one, cold a caloric. identical being measured the Thermometer EXAM the form energy. and was there heat the of about was when colder and had scientists, was that energy there idea there mechanical cannons of a supply of cannon supply mechanical He to Although This that effect held that objects to caloric limited heating from years object some a idea done the in the lose the between hot have unlimited idea commonly used a transferred. work energy proportional Figure only filings few the would material. an most from cannons. being be the theory transferred flow noted the mechanical seemed was could into released, caloric) This object Rumford original (the that would Objects boring century theory. caloric caloric that the 18th caloric heat in) mechanical energy into heat to The The kinetic thermal theory energy and (heat thermal energy) of an energy object is mainly the result of ACTIVITY the motion motion it of is the called particles the within kinetic the object. theory. Some As the theory potential describes energy is associated with the bonding between, and the arrangement of, Use three water, atoms in means to an this energy that transfers thermal object at a from energy lower a hotter flows object from temperature. an This to a object cooler at process high one. This temperature always occurs one (Figure in Place temperature After coldness are scale of is simply objects. several a way Hotter temperature of expressing objects scales have a the higher relative hotness temperature. including: Celsius freezing 100 °C scale: This point of producing The used Kelvin ideal gases absolute hand is a scale based on the properties of pure a water centigrade temperature scale: and The the zero. kelvin This and is so Kelvin energy At this is set at (100 scale scale of in 0 °C step) is school based particles. temperature Temperate Temperature in SUMMARY What if 2 the Copy to cannot be the conversion 1 they defined makes kelvin in water in in the the hot iced seconds and the scale. boiling Celsius is on The the science the low and place of become same between = as any an the Celsius increase scales temperature degrees colder. have in An zero of fairly degrees one = temperature in Count in which will has feel has feel hands been cold been in and in the the hot. the cold However , are experiencing the temperature. is thermal of water one Hot Room water temperature Celsius. Figure kelvin will of scale degree + which most easy: Celsius lukewarm industry. the increase the point the properties point particles in into 2.1.3 Hot or cold? 273 − 273 QUESTIONS would caloric and hot water. Ice-cold energy one thirty water same commonly hand hands hot both at containing and The water The iced water water. hand The of scales both There one lukewarm water. and of 2.1.3). one water direction. T emperature A one materials. and Thermal bowls, the have happened theory complete had been T able Rumford’s experiments correct? 2.1.1 which shows some important temperatures. KEY T able POINTS 2.1.1 1 T emperature in The thermal object degrees Celsius in is zero point of liquid nitrogen Freezing point of ethanol Freezing point of water Body measure of of an the energy and potential 0 energy Boiling a kelvin kinetic Absolute energy T emperature 77 −117 point of ethanol Boiling point of water 2 310 3 of all make of up Temperature the 0 temperature Boiling that ‘hotness’ the the is a of Thermal energy from object an particles object. measure an is at of object. transferred higher 79 temperature to an object at 100 lower temperature. 51 2.2 Thermometers LEARNING temperature scales A the thermometer the end should be is a device of this able topic substance. describe and the select an of of appropriate for use in will an object with thermometers thermometers expand is proportional A reservoir as to its are based the temperature on the increase in increases principle and temperature that that (Figure this the expansion 2.2.1). a of liquid, mercury or coloured alcohol, is contained at the situations. will is be of the and tube. schools are in a rise up give ice This boiling used liquid to point water the can a The the point. ‘bulb’. tube tube the calibrated level The the 0 °C. of This measurable higher marking represents and 100 °C be a capillary capillary then in give upwards. the and to narrow thermometer melting thermometers thermometer enough further The Most the connected expand 2.2.1 of vary different bottom Figure that thermometer thermometer range temperature properties characteristics liquid on temperature. you Liquid-in-glass of rely to: operation types measure Thermometers Liquid-in-glass to OUTCOMES or At and The distance will of the of the that The which the of there reservoir the bulb liquid liquid in is capillary between so liquid the will liquid rise. placing bulb in expansion. temperature liquid level liquid required through the by is these of liquid the then tube two in capillary placed is in marked points is to then liquid-in-glass. divided into Clinical As to body between cover allows equal divisions giving a centigrade scale. thermometers human vary 100 only the this and narrow to divisions magnify is 42 °C, thermometer temperature designed temperature 35 °C of the approximately a clinical range to be (Figure 2.2.2). designed 0.1 °C. The capillary to be shape tube 37 °C and thermometer of and The very the is will generally designed small range precise, with thermometer make it easier to is read the temperature. Clinical they thermometers can be contracts, breaks the the a most 2.2.2 A clinical follows prevent thread. are now removed inaccurate is built liquid into can from reading the be the patient as the liquid thermometer returned to before the that bulb after thermometer. take display being The the be an constriction flicking and to an a few minutes accurate replaced by to reach temperature. electronic thermal Because of this, thermometers. thermometer the principles by To thermometers equilibrium Figure small liquid reading Clinical read. need same as a basic liquid-in- Thermocouples glass thermometer. A thermocouple potential connected. 52 is an difference The p.d. electrical (p.d.) thermometer produced produced varies when with which two relies different temperature. on the metals are Thermocouples melting p.d. or on they a that There can This display and also can be then making thermocouples very they temperatures recorded be very quite converted them are high respond very used simply and easy to (beyond quickly a by in the changes. data These The loggers temperature read. extensively to shown properties industry. thermometers are Gas measure glass) produce digital More can of computers. mean point several other thermometers types use temperature increases. temperature change. Infra-red ACTIVITY of the This thermometers thermometers: fact that expansion measure a gas is the expands when proportional rate of to emission its your using a dyed the of Make radiation from objects. The intensity of this radiation water the temperature This of the object: measurement can hotter be objects used to find emit the the flask in iced level of the something SUMMARY 1 What the are three without needing to touch tube. water dyed and water infra- 0 °C point. Then place it in depends water to find the level more 100 °C. Divide the range into temperature suitable of containing capillary the for radiation. and mark boiling on thermometer flask Place for red own conical divisions. it. QUESTIONS the ranges, precisions liquid-in-glass and temperature thermometers a b °C shown in readings Figure on 2.2.3? c °C 42 200 100 41 190 40 180 39 170 38 160 37 150 36 35 140 34 130 33 120 32 110 10 1 00 0 KEY 1 Figure What to All thermometers on physical rely changes in substances caused changes temperature. in by 2.2.3 2 2 POINTS a are the advantages liquid-in-glass of using thermometer? a thermocouple compared Thermometers of scales, response have precisions a range and times. 53 2.3 The Most LEARNING states materials the end should be of this able describe the topic of The arrangement the particles use liquids the and particle describe the behaviour and into of solids, three categories liquids and based on their gases. model of matter and materials are composed of tiny particles in different arrangements. in particles are atoms or molecules. The behaviour and physical gases properties placed behaviour: particle These solids, and you to: All behaviour be matter OUTCOMES properties At can of model properties solids, of to these of materials particles and can the be explained forces by between describing the behaviour them. and Properties liquids of solids gases. In solid are materials held in place (Figure 2.3.1a). vibrate about the particles with The are relatively particles closely strong are not packed together inter-molecular free to move and forces around but instead a maintain It is not already the Solid their their easy own to tightly particles strongly fixed positions. compress move means that solid materials shape. packed repulsive This solids. together. slightly force This If closer a is because together. between the compressive them particles force However, and so is this further are applied then produces a compression b becomes volume much more (although Properties of difficult. this can Because change of when this the solids solid is have a fixed heated or cooled). liquids Liquid The c particles molecular will and are are forces 2.3.1b). flow Liquids liquids forces short-range (Figure in very also weaker allow When the are a liquid the difficult than to is fi ll packed those particles liquid will closely placed the for to compress solids. flow into bottom for together a of the past but These each inter- weaker, other container the the the particles container. same reasons as solids. Gas When Figure 2.3.1 The arrangement they in a a solid, b and c a This The use describ ing the move not 54 occur between that liquids also have a fixed volume as the long as is unchanged. gases in gases around have very very quickly weak inter-molecular (Figure 2.3.1c). The forces gas and particles compared term ‘parti cles’ indivi dual mo lec ules. each space. The other and spaces so spread between out gas to fill particles up are all of very the large when matter , to the size of the particles themselves. The space between to particles means atom s pressure or of attract available the to forces TIP do refer means temperature particles they When repulsive gas. Properties EXAM strong a their liquid squashed of particles. particles are to it. that it is possible to compress a gas by applying a Thermal When solid a material this staying expansion in particles (Figures is increase the heated in same slightly 2.3.2 particles causes position. further and the energy This apart gain the increase and as thermal particles a in to energy. vibrate vibration result the will material In a more Ball while force the expands 2.3.3). Ring The forces which produce the expansion are very large and can cause Figure even very strong objects to distort. Long metal bridges may 2.3.2 A metal ball through by several bridges centimetres have on expansion a hot gaps day. at To each allow end. for As this the expansion bridge the warms up the ball expand it longer lengthens prevent As you them have heated. Gases and The in gaps buckling already in hot of a will Similar are used in railway lines ring. pass will and fit Heating cause it will it to no through the ring. to weather. both liquid not gaps a liquids will be and gases increase able to with expand will expand when temperature. and so their pressure instead. SUMMARY QUESTIONS Summarise table T able close. seen, volume containers increases 1 the can expand like the T able properties 2.3.1. of Add solids, liquids additional and gases categories if using you a can. 2.3.1 Shape Does Volume Density it Separation Forces of between particles flow? particles Solid Liquid Figure 2.3.3 Overhead power cables Gas or telephone on 2 Draw gas 3 a as Use diagram it is ideas showing compressed about what inside expansion to a happens to the particles in a to hot days, lines expand causing them sag. syringe. explain why: ACTIVITY a running the lid of a jar under hot water makes it easier to You open of b a house roof sometimes creaks during the can test solids, compressibility and gases. night. Seal the syringe. as end of Place modelling syringe KEY the liquids and a a plastic solid clay push such inside on the the POINTS plunger. 1 In solids the particles vibrate about fixed positions. Repeat water, 2 Liquids and gases are both fluids. Their particles are free with and a a liquid, gas such as (air). to The solid and liquid will be move. incompressible 3 Gases can between be the compressed particles. as there is a large amount of space possible about to half but it compress of its should the original gas be to volume. 55 2.4 Gas The LEARNING gas laws the end should be of this able topic will you or result state the describe gas laws how use the gas changes the behaviour in the altered. change the or can The be gas in of A one, temperature volume laws number laws a is of gases the change or of a both, gas in when of will one the of their these temperature, others. cause an quantities For example, increase in the gas. and they demonstrated volume in increasing to: pressure describe OUTCOMES pressure At laws to of apply to particles a in fixed the mass gas of gas. sample This does means not that the change. describe properties of Charles’ law gases. Charles’ law temperature The volume describes T of of temperature a (in a gas fixed the relationship and is mass kelvin) if usually of the gas is between stated is volume V and as: directly pressure the proportional kept to its constant. V V ∝ T or = = constant T Volume (length of air column/cm) Experimental The graph can be extrapolated data back to zero volume Zero volume WORKED EXAMPLE 1 –273 A sample of gas 0 Temperature/ °C +100 occupies 0 +273 Temperature/K +373 3 600 cm while its temperature Figure is 300 K. 400 K The and gas is allowed heated to expand the pressure remains What volume key would is consequence cooled, the of kinetic this law energy is of the the idea of absolute particles zero. decreases As until a they the stop gas law the gas same. Charles’ so One that 2.4.1 to moving altogether. At this point the gas will have zero volume occupy? and cannot possible Before be cooled temperature: any further absolute (Figure zero, 2.4.1). This is the lowest 0 K. expansion: 3 V 600 cm 3 = = T 2 cm –1 K After expansion: V 3 = 2 cm law law gas its and The –1 = T × deals with pressure volume pressure 3 V Boyle’s of a p fixed 2 cm if the mass temperature –1 relationship the of between temperature gas is is inversely the kept volume V of a constant. 3 = 400 K × = 800 cm 3 2 cm is kept proportional to its constant. 1 K V –1 ∝ or = pV = constant p K An so 56 the while K T So Boyle’s 300 K increase applying in pressure pressure will cause squashes the the volume gas into a to decrease smaller in space. proportion The pressure The pressure The pressure temperature law law of a (in connects fixed temperature mass kelvin) of when gas the is T and directly volume is pressure p. proportional kept to the constant. p p ∝ T or = = constant T This means pressure the that to heating increase. pressure As decreases a gas the to in a closed temperature zero (Figure container decreases will to cause the absolute zero 2.4.2). Pressure, p Zero pressure predicted at SUMMARY absolute zero 1 0 QUESTIONS (0 K) +73 +173 +273 Temperature/K +373 What a happens the volume decreased Figure 2.4.2 The pressure when: of a gas while is the law temperature remains constant? The gas equation b All three gas of the gas equation volume and laws that can be describes temperature of a combined the together relationship gas. As before, to form between the mass the of a the single is pressure, the volume be of while remains a gas the constant? gas c must temperature increased the pressure of a gas constant. is increased while the pV = constant temperature is kept T constant? 3 WORKED EXAMPLE 2 2 A 500 cm sample compressed A weather balloon containing hydrogen gas is released from ground into the balloon at the atmosphere. The volume of the gas of it gas is occupies 3 only the until 100 cm while the in temperature is kept constant. 3 launch is 40 m while the pressure of the gas is The 5 1.0 × 10 final of the gas is −2 Pa (N m ) and the temperature is 35 °C (308 K). found The pressure balloon temperature rises is into the −50 °C upper (223 K) atmosphere and the where pressure is the the to initial be 600 Pa. pressure What of the was gas? only 4 1.0 Let × 10 the Pa. What conditions is the new volume on the ground the upper be of the balloon? represented by V , p 1 and T 1 1 KEY and the conditions in atmosphere by V , p 2 Find the value of the constant at ground 5 p V 1 1.0 × 10 2 2 1 3 × 40 m 4 constant = The be 1 = POINTS T level: –2 N m and = 1.3 × 10 behaviour described gases the can gas laws –1 N m K which T of by link the pressure, 308 K 1 temperature Use the value of the constant to find the new a p and volume of volume: gas. V 2 2 = 2 constant The gas laws lead to the idea T of 2 4 T × constant 223 K × 1.3 × 10 V 3 = = = 2 4 p 1.0 × 10 minimum temperature –1 N m K 2 So a –2 N m called absolute zero (0 K, 290 m −273 °C). 2 57 2.5 Heat Heat LEARNING the end of this topic be able heat capacity, C, of an object is the amount of energy increase the temperature of the object by 1 °C (1 K). to: heat energy E perform calculations capacity = or heat heat capacity capacity describe how capacity of and Specific object and heat capacity of can be heat of 2 kg an object block of temperature water specific required Specific is directly aluminium proportional takes twice to as the mass. much by 1 K as a 1 kg block. This leads energy to heat of specific heat to capacity, increase capacity energy heat the allows required capacities This is a temperature us to are c. to directly increase shown measure of of compare their in 1 kg of a the the materials temperatures. T able amount material by and Some 2.5.1. 1 Specific of a its amount typical EXAMPLE of energy the full capacity capacity increase 1 K. bath (K) measured. of A heat example, concept WORKED Δθ a to material rise the For specific = heat The an ) specific equations the (J K using temperature the C (J) H –1 Heat required you to should capacity OUTCOMES The At capacity has heat capacity is defi ned by the relationship: been heat energy 6 provided with 5.0 × 10 J specific of heat capacity = mass energy of the and the water has (8.0 K). capacity of risen What the change E by –1 or 8.0 °C × in temperature temperature is the c (J kg H –1 K (J) ) = heat m (kg) × Δθ (K) water? Investigating heat capacity and specific E H C = heat capacity Δθ The heat capacity of a metal block can be found using an electrical 6 5.0 C × 10 J heating = element (Figure 2.5.1). The energy provided is measured with 8.0 K a 5 C = 6.3 × 10 joulemeter provided A Some examples of similar to heater rise in insulate method element container −1 air electrical power heat can current the the be block block used is to to × heater p.d. measured reduce find the × with a is placed to in a sample of heat capacity water inside such as a plastic beaker or a polystyrene the 385 gold 129 Thermometer To circuit Metal block Heater 4181 Insulation diamond 509 glass 840 of an cup. capacity 1003 copper time thermometer . loss −1 897 Figure 58 heating heat K aluminium water equation: capacity Specific /J kg the 2.5.1 Measuring the heat capacity of a metal block It is surroundings. water . The specific heating Material = temperature important heat from −1 The 2.5.1 calculated J K energy T able or insulated To find must the also Method We can of heat the the known heat mass The object is heated The object is placed Energy reach is of the same gained mass of energy object. mass of the metal block or water of (or specific specific heat heat capacity capacity) of of water a solid to object the by and in measured its from and recorded. temperature water water gained The is and the the is recorded. water object into is stirred the water thoroughly. until they both temperature. water heat temperature using object transferred the Energy The the 2.5.2). The the value capacity capacity measured. mixtures use measure (Figure specifi c be and by is the of The temperature specific water capacity change. calculated the the equals object specific change using heat heat and the the rise capacity the can energy be in of temperature, water. lost by calculated capacity can be the using its calculated mass. Figure WORKED A to metal of block 140 °C. stirred EXAMPLE until water of The is mass metal the 3 kg is is then and its heated placed temperature 10 kg of the until into a water temperature its temperature container stops increases of is the If the specific specific heat gain by heat capacity rising. from The 15 °C heat capacity capacity of of the water is 4200 J kg and mass KEY POINTS to −1 K 1 what The heat object metal? energy Energy Measuring rises water −1 20 °C. 2.5.2 2 water: E = mcΔθ = 10 kg is capacity, the C, of an amount of heat required to raise its H temperature –1 × 4200 J kg × 5 K = loss from metal = capacity of specific heat capacity, c, metal: c of a of energy material is the amount 210 kJ H heat The 210 kJ E Specific 1 °C. 210 kJ 2 Energy by –1 K = required to raise = metal mΔθ 3 kg –1 = SUMMARY 1 Describe 2 Copy T able 120 K the temperature that –1 material by of 1 kg of 1 °C. K QUESTIONS the and 583 J kg × difference complete between T able heat capacity and specific heat capacity. 2.5.2. 2.5.2 Object Mass T emperature /kg /K change Energy provided Heat capacity −1 water in a 2.0 /J Specific −1 /J K /J kg 15 heat capacity −1 K 4200 beaker gold block copper pan 0.3 1.5 500 120 129 585 59 2.6 Changes of state and evaporation When LEARNING a further At the end should be of this able topic lost you describe changes compare gains of a heating a gas solid. phase). thermal it the liquid energy can These The may change into processes names of also it may change a are the change liquid called changes into into and, if gas. further changes are a a liquid. of shown When With cooling state in energy (or Figure is occurs, changes 2.6.1. All of state the by into to: of solid OUTCOMES evaporation and changes although boiling. during Sublimation During bonds involve is the the changes and this a change change process of state causes the there in the in thermal is which no a particles changes in change solid form the energy in or the substance, temperature. changes directly break properties of of to a gas. inter-molecular the materials. Evaporation Evaporation is a process which occurs at the surface of a liquid. GAS The Vaporisation Sublimation or particles in the liquid have temperature of the liquid is the particles. The higher a range related the to of kinetic the temperature energies average is, the kinetic greater and the energy the of average boiling kinetic energy. Condensation The particles liquid. near However, the the surface more are held energetic in place particles by forces may within escape from the the Melting liquid and form a gas. This process is called evaporation. During LIQUID SOLID evaporation more likely the to particles escape with from the the greatest surface. amount When of these energy are fast-moving Solidifying or particles freezing escape, decreases. This the average decrease in energy the of particles average energy in of the the liquid liquid means KEY that Increase in Decrease internal in the temperature of the liquid decreases (Figure 2.6.2). energy internal energy Examples Figure 2.6.1 Changes of state When you surface the Air perspire, and surface the evaporates of your skin moisture quickly. and on The cools your skin spreads evaporation you takes over its energy from down. in Air and vapour out by ether In a similar way, the contents the thermal The earthenware water will jars in evaporate water and will help remove keep some of sucked a energy from the containers. pump Beaker Refrigerators evaporation Liquid coating cold. and air-conditioning units are also cooled by processes. ether Factors Water affecting the rate of evaporation freezes Sheet of wood (any or There are insulator) temperature The higher Figure 2.6.2 The cooling produced chance the of ether the enough water. of average to they affect the rate of evaporation: the liquid. kinetic The energy of higher the the temperature particles and so the is, the have of escaping the more surface. The surface area of the liquid. A large surface area will allow freeze particles 60 that is the factors effect by evaporation large several plastic to escape at a greater rate and so evaporation will be faster . The humidity saturated with condense and The type others are of as the Evaporation air vapour reduce liquid. weaker. Comparing and the Some forces Ether flow. from holding rate above some of particles more the of liquid the is already vapour will evaporation. evaporate the and are air liquid overall evaporates boiling the liquids evaporation and If the much more together readily than in rapidly the than liquid state water. boiling often confused but they are different processes: Evaporation is a it liquid and a process happens which can whatever only the happen at temperature the of surface the of liquid. A Figure 2.6.3 The flow speed puddle of the of water water When a never liquid temperature. the liquid During will is reaches heated This and evaporate is happens boiling the it the away anywhere will liquid a point. specific turns into though near eventually boiling at even reach Boiling gas a and bubbles of gas can be seen for air will help evaporation. throughout that throughout the ACTIVITY liquid. whole forming within the a little ethanol on the back liquid. of your it. You effect SUMMARY of maximum occurs Pour volume up temperature 100 °C. temperature a the hand and should of blow feel the evaporation over cooling on your QUESTIONS skin. 1 A flask left off floor. 2 3 it why, you Draw ethanol but will Explain Explain day, of a will how after feel take many evaporate this in a hours few to evaporate minutes if it is if the spilt lid on is the happens. getting out of the sea on a sunny but breezy cooler. table stating the differences between boiling and evaporation. KEY 1 POINTS During a change of state a solid can change into a liquid, a Figure liquid to a gas, a gas to a liquid or a liquid to a 2.6.4 The but 2 It is possible called 3 a solid to change to a gas directly in a did has not evaporated boil. change sublimation. Changes a for water solid. of state involve the gain or loss of thermal energy in substance. 61 2.7 Heating, latent When LEARNING water the end should be of this able topic water you more to: to explain how to point measure of a describe of a the cooling substance demonstrates latent and the begins energy its temperature to is the boil, the being state when the rises the temperature provided. of temperature particles the This water. stops increasing additional This is reaches are energy because the 100 °C even is being bonds and though used between the molecules curve how when this concept heated to the are being convert ice molecular it altered. reaches forces to water. A similar 0 °C and and causes The thing happens the the temperature energy ice to taken stops when ice in increasing breaks the inter- melt. of heat. Latent The as EXAM heat energy latent separate used to heat. latent As change there the are state two of a substance changes of state, is referred there are to two heats: TIP Rem embe r , remai ns a and However, substance is heat heated energy. change the melting and OUTCOMES gaining At is cooling chan ge the tempe rature consta nt for for the the change between between solid liquid and and liquid, gas, latent latent heat heat of of fusion vaporisation durin g During of change cooling, the latent heat is released when a gas changes into state. a liquid the or a liquid temperature energy state will that be changes will stay released take place into solid. constant. as the during 100 a C°/erutarepmeT Liquid During Also, amount the exactly of change the energy same taken of state, amount for changes of of heating. phase only A 90 Melting 80 Both liquid or and freezing solid 70 phases point B 60 C 50 Solid phase only 40 D 30 0 5 10 15 20 25 30 35 40 Time/min Figure 2.7.2 Between Figure 2.7.1 The at temperature 100 °C water until has will A and cooling B the curve for cooling paraffin curve wax graph shows that the temperature stay all of the turned to gas. of the liquid (Figure does not latent to heat is decreases Once lose change temperature energy. wax 2.7.2). continues 62 A until all lost the until reaches energy being of it to of by solid the the the wax it its reaches freezing the surroundings. wax wax. has In freezing point (B Note solidified. the decreases final as the to point C) that the the During phase wax (C wax temperature this to phase, D) continues the to lose ACTIVITY: Place EXPLORING some HEATING granulated AND COOLING wax Thermometer (or stearic tube and acid) into suspend a the Temperature boiling tube in a Stirrer Liquid beaker of water (Figure 2.7.3a). Melting point Place a thermometer into the Wax wax and heat the water while Solid stirring it to make temperature is sure even the throughout. Time Watch the thermometer to see Water how as the the temperature wax melting is heated changes past its point. Heat At first will the temperature increase melting as point it wax the temperature reaches the approaches (Figure the of its its 2.7.3b). melting stops wax As Figure 2.7.3 Heating wax and finding the melting point point increasing 0–100°C and to the be wax begins measured temperature Remove cooling of the of to melt accurately. the liquid boiling the wax it After wax tube as instead. all will from This of the begin the solidifies allows wax to water has melting melted, increase and (Figure the the again. monitor the 2.7.4). Boiling Record If you time the plot it a will temperature graph show a of showing cooling the the wax every (Figure of the wax against Wax 2.7.2). Figure 2.7.4 Monitoring of SUMMARY 1 Use the sample 2 A of in Figure 2.7.2 to find the freezing point for cooling wax. sample of beeswax is heated until it melts and then to cool, providing the data in the POINTS table. 1 T able the wax the KEY allowed the QUESTIONS graph small tube minute. temperature curve thermometer point During a change of state the 2.7.1 temperature Time/min 1 2 3 4 5 6 7 8 9 10 11 T emp./°C 85 80 74 69 65 62 62 58 55 52 50 a Plot a cooling b Find c Would the melting using accurate curve a for point larger result? the for does 2 sample beeswax. of beeswax 3 give a more a energy is when required Latent a state heat substance during material change. Additional changes beeswax. the not of is (latent a heat) substance during heating. released changes when state cooling. 63 2.8 Specific To LEARNING compare the end should be of that the take changes this able describe of topic you As energy or produced of state. there energy This of different are required gives two latent heats: specific a to materials, change quantity changes of the we known state, as each state the of need a to unit find specific material has mass The latent two energy during required a change The 1 kg latent of specific specific a heat solid of latent into fusion heat liquid (l ) is of without the a energy vaporisation change required (l ) is in change of 1 kg these of a terms liquid can into be gas without represented by energy a the change energy the same provided in required temperature. equation: (J) E H –1 latent heat (J kg ) = or mass block of lead of its is heated melting until point of it much energy to melt the 4 = 2.3 × 10 the specific latent heat of water latent heat of vaporisation measure the specific latent heat of vaporisation of water , an electrical is element can be used to heat a sample of boiling water so lead? that (I m 327 °C. additional heating required (kg) reaches T o How = mass Specific 300 g l EXAMPLE Measuring small to temperature. v to specific A the heat. f change Both WORKED the of transfers during state the of materials. to: place calculate heats heat OUTCOMES amount At latent latent some of it is converted into steam (Figure 2.8.1). A well-insulated –1 J kg container ) needs to be used to prevent energy loss through the container . f E H l = ACTIVITY f m E = H l × Record Allow the mass of the boiling water and its container. m f 4 = 2.3 × 10 = 6.9 × 10 by –1 J kg × the the water heating to boil for several minutes while being heated element. 0.3 kg Find the energy supplied using a joulemeter or the electrical 3 J power equation: energy After Calculate find supplied heating, the the the current record the change mass Calculate = of in water specific × new mass that latent p.d. × time mass of the of has the water boiled heat of water and and container. container to off. vaporisation using the equation: specific latent energy heat of vaporisation supplied E H or = l = v change Specific As a to electrical into 64 latent sample difficult in of fi nd mass heat ice of melts out element. account Δm how The (Figure fusion naturally much following 2.8.2). at melts room as a temperature, result technique of takes it heating the is more by natural an melting Joulemeter Beaker in insulating Heater jacket ON OFF ON To power IN OUT Top-pan unit balance OFF Figure 2.8.1 Measuring the specific latent heat of steam ACTIVITY Allow the minutes) ice to melt without collected, for any a fixed heating. period Record of time the (such mass of as five water m 1 Use an period electrical of time heater and to melt record the a similar mass of sample water of ice for collected, the same m 2 Find the difference in these two mass measurements (m − m 2 This gives provided The by energy used in the the Calculate mass the of ice has melted as a and calculation the for specific mass the latent of latent heat melted latent of ). 1 the energy heat of fusion ice as a result are heat. of fusion using energy specific result heater . provided the that the equation: ON supplied OFF = change in mass (m – m 2 ) 1 Joulemeter E H or l = f m – m 2 Low-voltage 1 heater SUMMARY QUESTIONS Ice 1 a Calculate oxygen the into energy oxygen 5 (I = 2.1 × 10 required gas at to the change same 250 g of liquid temperature. –1 J kg ) v b Calculate the energy released when 4.5 kg of lead solidifies, 4 without change in temperature. (I = 2.3 × 10 –1 J kg Melted ) ice f 2 In an experiment to find the latent heat of ice as described in Figure the activity above, the following measurements were 2.8.2 Measuring latent when the Mass of Mass Energy of ice ice ice was allowed melted melted provided without when to to melt heater heater heater for = is five = on Calculate the specific heat of specific ice minutes. 5.3 g = KEY 15.7 g POINT 3500 J 1 a the recorded latent heat of fusion for ice (l ) using The amount released or of energy absorbed during ice this data. The agreed a −1 b value for l is 334 kJ kg . Explain why the value change of a of state material is for the 1 kg specific ice obtained by the experiment is higher than this value. latent heat. 65 2.9 Thermal The LEARNING At the end should be in of this topic you to: conduction metals and processes non-metals energy they part of a by process a perform a rod the in an experiment solid are the rates of in called in fixed is rapidly positions. heated, about When the these they energy positions. gain heat passes When through one the object conduction rod flame is will heated become vibrations energy of using hot. the a Bunsen The particles burner, particles will will cause the start nearby part to of the vibrate particles and begin to vibrate more. This process will to continue thermal along conduction object sample The more to gain measure a vibrate solid When rapidly. in OUTCOMES able describe particles conduction the rod until, eventually, all of the particles are vibrating more different rapidly and the whole rod has increased in temperature. The process materials. of T able 2.9.1 Some examples of thermal passing The rate The better energy of along thermal a is called conduction material is at lattice varies transferring vibration from material thermal to energy, material. the higher its conductivity thermal Material Thermal −1 /W m conductivity The rate conductivity of thermal energy 2.9.1). transfer by conduction also depends on −1 K the air 0.024 wood temperature temperature difference difference between will increase the the parts rate of of the object. thermal A large conduction. ~0.06 Conduction brick 0.2 glass 0.8 Metals have metals. cast (T able iron This in metals significantly is because higher there is thermal a conductivities second process than non- transferring the 55 energy aluminium in metals (Figure 2.9.1). This is energy transfer by free 204 electrons. copper ‘sea’ 385 of When diamond Metals free a contain electrons. metal is a The heated, lattice same the of metal electrons electrons ions allow rapidly surrounded electrical gain by a conduction. energy and can 2200 pass along quicker metals the than are metal the good carrying lattice the vibration thermal thermal method energy. This described process above and is far so conductors. ACTIVITY Diamond has a surprisingly high as it is conductivity, a non-metal. + + + thermal especially + + Find + + out why this is and how + + property is used to Ion this detect + Electron fake Atom diamonds. a Figure 2.9.1 b Conduction in a a metal and b a non-metal Insulators Poor thermal thermal particle foams, 66 conductors conductor. to particle. containing It is For lots are very this of called insulators. difficult reason trapped for energy materials, air are Air to such used to is be as a very poor passed from sponges insulate or objects. Measuring To rates demonstrate of different thermal rates of conduction thermal conduction, samples of the Three materials can be heated with a Bunsen burner. There are several rods ways A to monitor or compare the rates of conduction: B C The as rod the can rods be completely heats up. coated Observing in the a thin rate wax at layer which that the melts wax melts Drawing gives an indication of how quickly the thermal energy is stuck transferred. The far to underside rod pin being end of each rod can be coated in petroleum jelly and of with a petroleum drawing heats pin up attached the pin will starting heating thermal conductivity (Figure fall until off. each 2.9.2). When Measuring pin falls off the the can far time be end it of takes used to the rod jelly from compare the Tripod Comparing of the material of each rod. insulators Figure 2.9.2 The to The insulating properties of materials can be investigated by time fall liquids inside identical containers lagged with these materials the and the materials the temperature can be used temperature rise fall to can lag be for each (Figure identical 2.9.3). containers monitored over a of a pin how heat energy is and transferred monitoring for indicates placing quickly hot taken off along the rod. Alternatively cold period of water time. ACTIVITY Carry the out materials 2.9.3 Comparing SUMMARY 1 Explain 2 Why with need to 1 hot some Thermal test cups or material. sure beakers You there are will lids evaporation. insulators drinks frying Metals conduction to are pans sometimes have a sold copper in polystyrene cups. base? 2.9.4 have Materials involves the transfer of energy from particle. conduction 3 to insulating POINTS particle 2 the make prevent Figure KEY of QUESTIONS why do experiment using lined to Figure an properties free electrons much which more which quickly conduct poorly transfer than are is energy possible classed as by in non-metals. insulators. 67 2.10 Convection Liquids LEARNING the should end be describe in fluids of this able topic they you processes explain energy can can explain by how currents coastal both fluids. carry thermal within The energy them. particles from Because they place to the are composed place particles as they can a travel. currents substance contracts. increases During in expansion temperature the particles it in expands. the When substance it cools move them convection produce breezes apart contraction in substance When the air Because As For heated air air A closer currents. a substance together can so transfer These are convection decreases. the heat flows current of in During density of energy by particles air is the within produced used air air the sinks less dense heat as it particles back produces both to is out further that energy rises, the apart become cooler, with loses a downwards continual convection convection are move from each other dense. carrying spreads cools process they particles surrounding air, it. energy closer and cools. together and so the air currents transparent. show convection flow and of the cycle continues. air. currents in air These and two water can simple be quite difficult demonstrations can currents. LIQUID Place a small potassium of a large crystal of potassium permanganate) beaker of water at the manganate( VII) bottom (Figure and (also towards known one as side 2.10.1). Crystal Heat the current The rise and 2.10.1 Demonstrating convection 68 a current in water water will water rises by denser. dense Observing USING the behaviour the less warm Demonstrating be of expands. the hot the The as move This example, upwards becomes This density particles becomes floats The is air The the convection air it so processes: and the substance. these and increases. producing the Figure of move how areas. ACTIVITY: are around be further move Convection it transferred to to: convection and able When thermal gases OUTCOMES are At and currents near upwards with at it. the As directly beneath the crystal and a convection form. the the the other crystal purple water side of expands as colouration reaches the the beaker it is heated from the surface begin to it and as dissolving will sink. the water crystal spread out, will cool, ACTIVITY: Start USING with a 2.10.2). Light candle Close Hold The a air air is front from being rising chamber positioned glass smoking smoke cool hot the CHIMNEY glass-fronted (Figure a A of of cardboard cardboard drawn out one two Hot chimneys air Cold chimney. down the above moves through chimney the other downwards. the above This chimney the chimney. to is because replace the candle. Figure 2.10.2 Demonstrating convection in Air cool air will units be cool denser air using than the a system of surrounding expanding air air and so gases. sink Air warmed land downwards. the room to Air-conditioning produce a cold units flow are of positioned air towards the top by rises of downwards. Sea air Thermal convection in the breeze from During the causes replaced currents day air by the produce land above cooler the air breezes heats land flowing up to in near more coast rapidly warm from the up. than The above (Figure warm rise cooler the warm the sea. sea and Convection air the land flows currents temperatures cools. from within across the cooler flowing air At the This the land causes oceans to the air above rises night the have a to replace warm air and and the is sea Figure 2.10.3 A cool sea breeze the sea flows in during to the and to sea 2.10.3). from remains due the atmosphere in Convection this a current conditioning Air-conditioning The air panel. piece the under with day. sea. significant effect on the globe. ACTIVITY SUMMARY Convection currents responsible for the continents surface Explain why, during a fire in a room, you should crawl 2 floor How KEY can to a make hot-air your of this the Find out works. escape. balloon control its height? POINTS Convection can only occur in fluids as it involves of particles from place to fluids in density caused by rise place. fluids , Changes TIP the Ho t movement 2 Earth. process EXAM 1 the across across how the movement QUESTIONS of 1 the are expansion result in convection Never e.g. say ho t that abov e air co ld rises . heat rises . currents. 69 2.11 Infra-red Thermal LEARNING energy the end should be of this able topic describe affect objects red the the you properties to: factors rate as of they (a is a transferred part longer of of than visible the by infra-red radiation electromagnetic (IR visible light light. including IR the spectrum radiation ability to shares travel with many a of through the empty vacuum). that cooling radiate All of infra- objects their are constantly surfaces. This gives emitting three and absorbing possible IR conditions radiation for an from object: radiation This wavelength space be OUTCOMES radiation). At can radiation describe affect the the objects factors rate as of they that heating absorb at of a up greater sunlight – an rate will object than warm it up will is warm emitting up it. if A it is cold absorbing drink IR radiation placed in direct rapidly. infra red Warming Cooling down – an object cools down when it is emitting more IR radiation. radiation food will and See is absorbing cool temperature are will the stay – same at a to if from room the then steady surroundings. Freshly cooked temperature. rate the its of absorption object will temperature. not and gain Most the or rate lose objects in a of energy room TIP will 3.3 abou t so it gradually Constant emission EXAM than to the spectru m be each find out at ‘room temperature’. They are at the same temperature as other. more electr omag ne tic and absorp tion the and Factors ideas affecting the rate of heating or cooling of emiss ion. T emperature A hot object means that objects. If you see will will very you emit hot far monitor that more objects the the IR lose radiation energy temperature temperature drops at then a of a cool greater a cooling more one. rate drink rapidly at This than cooler then the start of Incident the experiment than later on. Eventually the drink will reach room radiation temperature Reflection stop cooling. and absorption here Properties The an Smooth and of the absorption object and or so surface emission the of IR properties radiation of the happens surface are at very the surface of important: surface The surface energy Incident Scattering The to area of escape colour of or the the be object. A larger absorbed surface. Dark more surface area will allow rapidly. surfaces are much better absorbers and radiation absorption here and emitters into bright The Rough a 2.11.1 A rough larger so is surface overall able infra-red 70 larger to has a surface absorb radiation. area more radiation of 2.11.1). than wearing clothes the surface surface (Figure Figure Your roughness have IR sunlight immediately. of dark start surface. area white to A when surfaces. clothes feel you If will you step notice out this effect hot. rougher surface compared to a will effectively smooth surface ACTIVITY A Leslie’s different poured same cube is colours into the a metal or with container different container and all with the textures. of the surfaces Boiling sides painted water rapidly is reach the temperature. Place a temperature sensor (or the back of your hand) close to EXAM the different being surfaces to measure (or feel) the thermal radiated. T ry You TIP energy should find that the dark or rough surfaces emit to remem ber dark er thermal radiation than the white and shiny colou rs one experiments side painted Investigating To investigate place hot colours, of each for and similar coloured Both cool of a how recorded and the cooling and also the placed up slows room the the are the plates tempe rature with of with 2.11.2). a of patterns can IR The period the water, materials 1 be used radiation. sunlight or near can temperature Stopclock twenty in cooling. to The to you a different radiant monitored. beakers than difference with the in the silvered black surfaces surface. temperature The between rate the decreases. POINTS All 2.11.2 SUMMARY objects are quick ly. different Figure KEY chan ge more shiny. painted over absorb rapidly left been compare cold rises metal cooling (Figure direct that more as in other minute surfaces with surfa ces heating have to very out the black every using confirm heat that and temperature experiments or are and plotted different beakers down water silver is and affecting beakers graph carried black factors into is be cooling experiment, investigate heater matt example beaker minutes A the water can and surfaces. rough er Similar that more constantly absorbing or emitting infra-red Investigating the colour the rate of of the a effect surface of on cooling QUESTIONS (IR) 1 In many tropical countries radiation. buildings light 2 The hotter an object is the more IR radiation it The temperature of dark coloured objects changes rapidly help are good emitters and absorbers of IR The temperature of silver coloured objects changes slowly are poor emitters and absorbers of IR Rough surfaces radiation than are better smooth emitters and to cool warm these keep the during the day during the night? In the Arctic, polar bears are radiation. white. 5 in as 2 they do radiation. and 4 How as building they painted emits. colours 3 are colours. absorbers of them IR How keep does this help warm? surfaces. 3 In Antarctica, huddle numbers. them penguins together How keep in large does this help warm? 71 2.12 Applying energy thermal transfer principles Understanding LEARNING allows At the should end be of this able topic us heating you large to: to describe how transfer of can or be used increase energy of we of objects. thermal need to or thermal energy decrease Sometimes energy prevent into the the loss we the transfer need rates to surroundings or gain of of dispose quickly thermal of and energy. in a flasks to the flow vacuum wide flask is designed to prevent as much thermal energy of as possible. To do this it has features that reduce conduction, range convection, of cause increase energy transfer heat which to the thermal A reduce cooling amounts Vacuum of devices an understanding methods processes design and sometimes the OUTCOMES radiation and evaporation processes (Figure 2.12.1). circumstances. The main with no a particles thermal Plastic chamber vacuum there energy transferred plastic) Plastic constructed can be between the two no of glass layers. conduction the layers. (a As very the or poor conductor) vacuum convection Thermal energy contains to can transfer be cap Double-walled (or is between around the glass layer by conduction but this is a very glass slow container protective The process. inside surfaces of the glass layers are coated in silver to reduce cover radiation. Hot or cold Sponge The surfaces are very poor emitters and absorbers of liquid radiation so there A plastic is very little transfer of energy between them. pad hollow cap is used to prevent evaporation. (protection) Inside surfaces silvered to Solar stop water heaters radiation Vacuum prevents Many houses heat water using solar energy. Cool water is either conduction and pumped or driven by convection currents through pipes in panels convection on Plastic for the 2.12.1 A absorb support vacuum (Figure energy Car The the passing for are heaters used keep the night. 72 pipes are painted black so that sunlight and this energy is transferred they to the them. of for can heat water . washing house large They or warm at cooling radiators engine fuel is in a car burnt. If produces this engine would parts prevent engine. front of the the radiator the air. to the this, conduction amounts the through of the Solar from temperature To 2.12.2 The flask Designs Figure 2.12.2). spring water Figure roof The and car engine soon water water is is and large were would cause 2.12.3). thermal through of As the energy to a car is thermal build up then when the of the up. which thermal radiator moves, energy Expansion seize pipes the through of to rapidly. engine some pumped amount allowed increase the pumped absorbs then (Figure pipes a energy energy through by mounted air transferred pass travels from on the between the water to Cooling The panels back energy Heat panels taken on from refrigerators refrigerators the inside to are be painted radiated black away to allow heat quickly. sinks Microprocessors during their damage sure on the that can produce operation. processor this This if happens, it a very large energy is not heat will amounts reduce transferred sink is of the away mounted heat energy efficiency rapidly. on the top or To of even make the Figure microprocessor (Figure 2.12.4). A highly conductive ‘thermal paste’ 2.12.3 In a car radiator, increase used as the glue between the processor and heat sink. The heat designed to have many fins so that the heat energy can be both by radiated The The convection Earth the a by process, balance However, for more heat of with fans) and area fins and also to be painted increase the black rate of The Earth activity such as to be ocean trapped as light the longer back to at short surface and tends remained and atmosphere. wavelength the dioxide effect, energy to of methane. maintain constant years. burning and gases visible carbon has this through energy as and as reflected such thousand greenhouse and this is warming Sun passes greenhouse deforestation energy atmospheric this the arrives which gases the 2.12.5). human amount as of global by energy re-emits Some several production, radiated Sun’s Earth known and radiation greenhouse (Figure temperature the the radiation. Earth crop The infra-red of effect energy planet. surface infra-red The assisted pipes emission. absorbs wavelength the (sometimes away. greenhouse warms The currents surface carried may away the sink and is fins is fossil keeping in the and fuels, livestock atmosphere. results in an increased is Figure 2.12.4 A This overall heat sink mounted on a microprocessor increasing causes increase in temperatures. SUMMARY 1 Describe QUESTIONS how a vacuum flask Sun can Some back Heat from passes the heat into keep contents cold. escapes space 2 How can warming Sun Heat is back to the be effects of global slowed? reflected through Earth by atmosphere 'greenhouse C0 in gases' air Earth 2 Heat is back from KEY POINT radiated 1 Careful 2.12.5 The increase in selection of materials Earth can Figure its greenhouse gases may lead to catastrophic climate be used to control heat flow. change. 73 Section SECTION 1 2: During 2 Practice an Practice exam investigation exam questions questions into cooling by 5 Compare the behaviour and spacing of the 3 evaporation, into each different and of 20 cm five of circular diameters. ethanol ethanol was was beakers The mass measured of with placed that the a particles had concepts beakers fluidity The ethanol samples were allowed for 30 minutes and of Describe the beakers was recorded 4 Diameter/cm 5.5 explain the then the the three the and gases. Use compressibility states of these and matter. changes in particle behaviour and changes in energy during the following mass changes of to liquids to the evaporate solids, top-pan 6 balance. in of state: again. 6 7 a from solid to b from liquid liquid 10 to gas. 2 Area/cm 7 Start 75.5 mass/g 95.5 121.0 141.0 A metal block strongly End 67.9 mass/g 81.2 104.0 117.9 The 133.3 until block is containing Change mass its 0.8 kg is temperature heated then placed 4.0 kg of into cold reaches a is stirred stops Calculate the surface area of each of rises the until rising. from the water The 10 °C temperature and temperature to 19 °C. of of (The the Calculate the mass loss for each of the of water the is a Calculate b Calculate the c State heat the water the 4200 J kg water heat −1 K temperature water specific −1 capacity beakers. b 200 °C. bucket in mass/g a of 180.5 .) rise of the water . beakers. c Plot the a graph surface comparing area of the the mass loss energy gained by the water. and the energy loss of the metal beakers. block. d Describe the mathematical relationship d between the surface area and the rate Calculate the specific heat capacity of the of metal. evaporation for ethanol. 8 e Use the graph to find the rate T wo metal from evaporation per centimetre plates squared for ethanol during the an electrical Describe can 3 be how a liquid-in-glass a thermometer and period of following calibrated. Describe explain the key features of thermometer as shown in equal distance heater. and the One other plate matt is shiny, black. the of five each plate minutes results is as recorded The shown in over the table. a Time/min clinical an experiment. temperature 2 placed per silver-coloured minute are of 0 1 2 3 4 5 6 7 8 9 10 30 40 48 55 61 66 70 74 77 80 82 30 45 58 65 70 75 78 79 81 82 82 diagram. T emperature of shiny Cross-section plate/°C T emperature 35 36 37 38 39 40 41 42 of black plate/°C 4 Copy and complete temperatures this between table converting degrees Celsius and a kelvin. Plot graph comparing temperature b 0 Celsius/°C 100 −50 Describe 0 of and rise explain the pattern temperatures. Why do the plates temperature? in plates. 234 c 74 the the −127 in Kelvin/K a stop rising in in the rise 9 Liquid nitrogen systems, state from nitrogen a is energy heat of liquid as it 10 in rapid energy to a to it How absorb for cooling when gas. changes vaporisation 5 × used required heat 2.0 is absorbing much 500 kJ state? liquid 13 changes liquid of (The is students evaporation and ether. fume latent nitrogen Some into of much ice with energy a is required starting to convert temperature of students rectangular pour trays out the in a liquids measuring 3.0 kg of steam with a final is the surface area of the liquids in trays? 3.0 kg −5 °C students measure the mass of the liquids temperature and of carried of ethanol 5 cm. What The into is rate water, .) the How The the liquids: experiment cupboard. by investigating different −1 J kg a 10 The identical 4 cm are of trays at the start of the experiment and 105 °C? after −1 Specific heat capacity of ice: Specific heat capacity of water: −1 4181 J kg 2100 J kg 10 minutes. Their results are shown in −1 K the table. −1 Starting K Change Rate in evaporation of mass/g /g min End Liquid mass/g mass/g −1 Specific heat −1 2080 J kg Specific capacity × K latent 10 Specific heat of 11 Describe fusion for water 14.4 14.2 ethanol 13.4 12.4 ether 17.8 12.6 ice: −1 J kg latent heat of 6 water: steam: −1 5 3.3 of 2.3 × how a 10 vaporisation for −1 J kg group of students could b determine the specific latent heat Which liquid has evaporated at the fastest of rate? vaporisation of ethanol. You must include: 2 c the equipment an assessment an explanation What for of the risks of the measurements each the end the of of that of evaporation per cm the liquids? the they experiment did not turn the on students the fume required. cupboard Explain rate and notice 12 the involved At calculations is required following in terms of infra-red extractor fan as they were supposed to. radiation: d a b Some firefi ghters when entering Solar water wear burning heaters on silvered clothing In what way different if would the fan the had results been have turned been on? buildings. roofs are painted black. c Dark-coloured washing line clothing than dries faster light-coloured on a clothing. 75 3 Waves 3.1 Wave Wave LEARNING motion the end should be of this able describe topic you caused of by describe the between give a Wave differences of and the waves examples of oscillation or a series in roun d can will the pulse be demonstrated produce of of spring the is a end the of is coils with vibrations will a until type the of coils along long allow spring generated. the stretched wave The pulse slightly are the Single left, why in us is carry A hand transverse A Vibrations of the describe in spring. wave one part Observing motion. motion one is of the sideways the end in end spring and The (Figure in and the once, out a length 3.1.1a). and spring together back along (Figure pushed produced. closer turn If the rapidly has regions these regions 3.1.1b). movement: movement Fixed end Fixed end pulse hand then 3.1.1 movement: Pulse pull Continuous ACTIVITY continues train a table comparing of transverse longitudinal different Research these them category by of over the a of along produced A the if the continuous spring. The source series of particles of the pulses vibration form oscillate in a a wave a period of time, repeating the same regular pattern. of waves There are two classes of waves: transverse and longitudinal and correct describing the spring range Transverse waves the T ransverse motion a waves waves. types into in waves. produce types are oscillate. moving Classes Earthquakes motion the motion properties Compression waves waves to movement pulse Wave Continuous waves form when the oscillations of the particles are particles. perpendicular transfer). to 76 rest right longitudinal Figure place to move reaches with Rarefaction of a energ y. b producing matter , push, and oscillations springs. the moved coils compressed length then Single Draw the pulse wave fixed T hat’s don’ t of wave springs wave a without single train. Pulse only place a pulses where parti cles in When move wave s to be waves. TIP positio ns. wave behaviour another T he single spring spring oscil late place may transverse longitudinal EXAM a motion the Wave and from There medium transverse longitudinal energy (matter). waves Waves in material progressive to: how propagate transfers OUTCOMES transfer At motion The oscillate to the direction movement and this of of one oscillation is propagation particle passed causes along (direction the the of the energy neighbouring wave (Figure particle 3.1.2). Wave Left movement Crest To fixed end Right Hand Figure Ripples causes Light repeated regularly 3.1.2 A on the the waves surface and Longitudinal In example the waves The forcing oscillations and to propagation. positions water to of a are transverse oscillate from vertically the direction fields in A while disturbance the water source. transverse magnetic waves. wave. which which the Light vibrate wave waves at consist right of angles to propagates. waves longitudinal of of horizontally electric other wave molecules out another oscillating each transverse water spread is Trough movement the their (Figure particles particles oscillate move neighbours back to parallel and follow forth the to the about same direction their pattern rest of 3.1.3). Figure Wave 1 Hand To movement tank ripple fixed A 3.1.3 A longitudinal waves are wave single oscillation a wave wave Progressive motion with ripple (Figure the when The of 3.1.4). water , a A of when the the to motor wave of the waves investigate causes parallel ‘dipper’ propagation see used producing round crests wave SUMMARY Figure waves is a horizontal plane used in transverse waves. place of form wavefronts wave. Observation reflect, change to Circular the and of speed wave beam or motion dip these in waves horizontal the train consist produced of by oscillations. Transverse water and are out produced of oscillate generated to beam. travel in wavefronts change in the waves when are particles perpendicularly direction of wave propagation. the direction allows us 4 Longitudinal to produced direction. oscillate waves when of are particles parallel direction 1 which a tanks 3 are through longitudinal. regular Observing tanks will pulse medium. a Ripple in regularly 2 Sound waves end propagates Figure plane POINTS produce repeated and a Compressions Rarefactions Pull Circular movement KEY Push 3.1.4 to the propagation. QUESTIONS 3.1.5 shows a wave pulse in a rope. a Which type b Redraw of wave is shown? −1 The pulse is traveling at 40 cm s of 2 cm Figure 10 20 30 40 50 60 70 80 90 100 the Sketch the would see centre of the diagram pulse a 0.5 pattern if a of pebble calm showing second wavefronts were the position later. that dropped you into the pond. 3.1.5 77 3.2 Describing Progressive LEARNING At the end should be can of this topic A displacement–position A displacement–time waves use period the in a and amplitude A wave range interpret position time kinds of graph: graph graph displacement–position displacement–position speed of in graphs a wave at a graph fixed represents time the during its shown at position of movement. all The of the positions equation a large number of particles are a single instant, like a contexts snapshot different terms of in two wavelength, particles by to: frequency, speed, represented you Understanding of be OUTCOMES able describe waves waves photograph (Figure 3.2.1). displacement– and graphs displacement– for waves. selcitrap tnemecalpsiD Wavelength λ fo a Position B A C Mean or Wavelength along wavetrain λ a rest position Wavelength Figure The and the metres. and In another (having This the means wavelength EXAM For unit T he is that the no t hertz same is the as the of that it a wave is a is wavelength that is of the is exactly displacement all the distance, troughs fixed distance the the at and a distance the a between wavelength same moving on time in one is between point the transverse in its same wave wave measured a particle motion direction). are also one apart. waves adjacent The amplitude, can have crest the wavelength compressions or is a measure of the distance rarefactions. a distan ce to a from a, the a its of rest a wave is the depth greater of (undisturbed) maximum a trough. distance In from position. displacement high their It is amplitude rest the height a particle waves the of a particles position. um from of or oscillate the parti cle, from Understanding displacement–time graphs a A displacement–time graph shows the behaviour of a single particle troug h. over a period changes particle 78 the particle same of As at the a maxim positio n peak fact, longitudinal between −1 displa cemen t rest λ, next. graph s ampl itude wave displacement–position TIP Rem embe r (Hz) A wavelength, crest in 3.2.1 λ as at a of time. wave point C It shows passes. from how Figure the the 3.2.2 displacement represents displacement–position the of the particle motion graph. of a elcitrap tnemecalpsiD Period WORKED EXAMPLES a the T What is water wavelength wave with a of a frequency fo a of 5.0 Hz and a speed of −1 25 cm s Time Period or ? a Mean T Original rest equation: position v = fλ Transpose Figure 3.2.2 A displacement–time the equation: graph v λ = f The period, T, of a wave is the length of time taken for a particle to Substitute complete one full oscillation. The particle moves from the rest in values: position –1 to maximum positive displacement back through the rest position to 25 cm s the λ maximum The negative frequency, f, displacement of a wave is and the then back number of to the rest = position. oscillations per second. 5.0 Hz Answer: −1 Frequency has the unit hertz (Hz) which is identical to the unit s λ Period and frequency are related by the b 1 period 1 = = 5.0 cm equation: or T What of a is the light frequency wave which = 8 frequency travels f and Therefore the greater the frequency of the wave the shorter at has 3.00 a × 10 −1 m s wavelength of the −7 4.00 period will 10 m? be. v Wave × speed = fλ v (v) f = λ Different waves travel at different speeds. For example, a water wave −1 may travel at 5.00 m s 8 3.00 × 8 whereas a light wave may be travelling at 3.00 f −1 10 m s . The speed of a wave is related to the frequency × by the −1 m s and −7 4.00 wavelength 10 = × 10 m equation: 14 f wave SUMMARY speed = frequency × wavelength or v = A sound this wave has a period of 0.04 s. What is the frequency of 1 wave? Sketch a × The by speed the displacement–time graph for a particle at point B of 3.2.1 if the frequency of the wave is Copy T able 3.2.1. Then calculate the speed in. You must include the missing appropriate values and A can 500 Hz 0.2 m 3 a be A the particles fixed = (v frequency = × fλ) displacement–position the f given used to units. 3.2.1 v is fill represent T able wave 2 Hz. graph them a in 2 3 Hz equation wavelength Figure 10 POINTS wave 2 7.50 QUESTIONS KEY 1 = fλ positions in a wave a time. displacement–time can of at be used to graph represent the −1 b 340 m s c 3.00 3.5 kHz 8 × 10 motion −1 m s −3 5.00 × 10 m (or of point) period of a in single a particle wave over a time. 79 3.3 Electromagnetic Visible LEARNING the end should be the state is of this able topic very the you small properties which on their differentiate by waves of the electromagnetic whole radiation electromagnetic but it only spectrum wavelengths or are how grouped they are spectrum. forms into six Waves major produced in regions (Figure 3.3.1). properties of electromagnetic radiation share electromagnetic their of all Common waves form part electromagnetic based to: electromagnetic a OUTCOMES a At light waves wavelength or All electromagnetic and (EM) radiations share some common properties behaviours: frequency specify sources and uses EM radiation travels as a transverse wave. for 8 each of the regions of EM radiation parts electromagnetic travels at −1 3.00 × 10 m s travel at different in a vacuum. Different the of the spectrum speeds in other materials. spectrum. The wave does The wave motion and no not particles require consists are a of medium (material) oscillating electric to pass and through. magnetic fields required. ACTIVITY The in Produce about a the summary sources, T able detectors and uses of electromagnetic waves are shown 3.3.1. poster properties and Ionisation uses of waves. electromagnetic Make include sure some wavelengths that you examples and of the frequencies involved. Find of out the used how EM for systems different spectrum Some This forms means of EM that radiation electrons charged particles damage living called tissue, carry are ions. enough removed These causing ions mutations are or the to ionise atoms highly atoms. which reactive become and can cancers. parts can be communication including subcategories of SUMMARY the radio QUESTIONS waves. 1 What the 2 is EM Copy relationship 3.3.2. calculate identify T able the spectrum T able given, which and between the Then, the region for of the damage each missing the it of frequency can the frequency EM cause /m a 8.8 × 10 b 1.4 × 10 7 −8 c 2.2 × 10 d 1.3 × 10 −2 a part living wavelength spectrum the Region 15 of to electromagnetic or 3.3.2 f/Hz 80 energy from wave of cells? waves and then belongs in. T able 3.3.1 Sources, detectors and uses of electromagnetic waves Frequency / Sources Uses hertz Radio waves Electron movements Communications 22 10 in transmitter circuits such as radio γ-rays or –13 10 21 of TV and radio television. 10 –12 systems. Microwaves are 10 used = 1 pm 20 to cook food as their 10 –11 X-rays 10 energy is absorbed 19 10 easily by water and –10 10 fat molecules. 18 10 –9 Infra-red Any warm or hot Infra-red 10 cameras = 1 nm 17 10 radiation object. (often used for –8 10 The higher the seeing during night- UV 16 10 temperature of the time). –7 10 object the shorter IR thermometers to Violet 15 light 0.4 μm 10 VISIBLE the wavelength. measure temperature –6 10 = 1 μm 14 without contact. Red 10 light 0.7 μm –5 10 Visible light Very hot objects. Photography. 13 10 Some chemical Lasers. –4 IR reactions. 10 12 Sight. 1 THz =10 –3 Ultraviolet Very hot objects Forensic 10 science. = 1 mm 11 10 light such as the Sun. Chemical lamps Sterilisation analysis. –2 10 Fluorescent Micro of 10 10 containing mercury. waves equipment. –1 10 UHF 9 X-rays X-ray tubes which Forming images of 1 GHz = 10 TV 1 propel fast electrons bones in the = 1 metre body. VHF 8 10 into metal blocks. High-energy radio X-rays Short- can be used to find in other 6 materials. 1 MHz = 10 OIDAR 10 damage 10 7 wave radio 2 10 Med.- wave radio 3 10 Gamma rays Decay of radioactive Cancer = 1 km treatment 5 10 materials. (radiotherapy). Nuclear Gamma Long4 wave explosions. emitters are 4 10 radio 10 Gamma ray bursts used as radioactive 5 10 3 from cosmic events tracers in medicine 1 kHz = 10 Wavelength / such as black formation of or holes. other applications. Sterilisation metre of Figure equipment or 3.3.1 The electromagnetic spectrum some foods. KEY 1 POINTS All electromagnetic vacuum 2 waves travel at the same speed in a space). Electromagnetic (and 3 (empty waves have a wide range of wavelengths frequencies). There is a wide range electromagnetic of uses and dangers associated with waves. 81 3.4 Light A LEARNING light the end should be of this able describe explain the topic be you say how formed situations energy to: path of light describe in a range A of light a lamp energy (taken absorbed boxes operation of ray box in) by and uses produced eclipses radiates is when the being light energy it emitted. in reaches retina at the This all directions. surfaces. back of light For the energy example, can light eye. rays the by lamp. a lamp placing We to a can produce ‘stop’ see light. with these a Narrow single light rays slit beams (or of series because light of some can slits) of the be next light is a off surfaces into our eyes. camera. A laser can look to but spraying the path laser of the the extended pass so a through shadow sprinkling scattering rays. rays talcum and It is are allows harmful not powder us to to usually into see the visible the the path straight shadows straight of line light from then region light light where completely and or light These will will the there be in source. will be a If an opaque region where object the light shadow. produce a very sharp shadow behind an object. regions cannot of the This source narrow beam. beam. a of reach. point An in path very laser aerosol travels in opaque boy laser a causes Light A the some beam at formation cannot Light produce directly The is 3.4.1 also eye of Figure the shadows including the is scattered pinhole as shadows rays to that absorbed Ray are such and OUTCOMES We At source rays blocked all of dark. will be source the The will produce possible regions partially ray where a less paths some sharp are of shadow. blocked the ray will The be paths are shaded. is formed. Eclipses Eclipses only demonstrate source of that visible light light in travels the solar in straight system. lines. The The planets Sun and is the moons ACTIVITY are Construct camera foil your using and some own a box, visible Solar some tracing it to objects view (or darkened brightly light Produce how a dates of guide or and observed some of this sunlight. Sun is very much larger than the Moon but it is much further lit sources) in away. This means when viewed that they appear to be almost exactly the same size a from how safely. the about solar they can Include next the eclipses for be the eclipses. directly Sun regions small in is which not only lie the Earth. between a point some within region total surface 82 reflect A solar eclipse occurs when the Moon room. lunar happen they eclipse passes because paper. The Use only pinhole on the darkness of the the of and light sunlight penumbra Earth’s during Earth Sun source of the the as an the the there is and are blocked (Figure surface lies This within dark rotates. 3.4.2). regions by experience eclipse. Earth Earth the the a on the Moon. partial umbra region Because Earth These eclipse. and moves so across A is the Earth Moon Sun U U P Figure Lunar We 3.4.2 An eclipse of the see Umbra Penumbra Sun SUMMARY eclipse can = = the Moon when sunlight reflects off its surface. A lunar 1 eclipse occurs and the when the Earth blocks sunlight travelling to the The International Moon appears dark (Figure 3.4.3). During a lunar Moon sometimes appears to be dark red, this is because some passes through the Earth’s atmosphere and reaches the of and orbits 100 at 200 km from a the Moon. surface of diagram causes Moon’s approximately long red height light is eclipse metres the Space Moon Station so QUESTIONS the to no Earth. explain obvious Use why a it shadow as orbit it Eclipsed passes between the Earth Moon and 2 the Explain shown Sun. how in the Figure equipment 3.4.5 can be Sun used to show travel in Figure 3.4.5 that straight light rays lines. Earth Earth’s Figure The A 3.4.3 pinhole pinhole image hole An on in eclipse an the Moon camera camera a of umbra is screen opaque a simple (Figure sheet optical 3.4.4). such as A instrument pin is used aluminium used to foil. to make The produce a foil very is an small placed KEY in front of a brightly through the sheet translucent lit pinhole. object. An Rays image of of the light from object can the be object formed on a 1 of POINTS travel Light rays travel in straight paper. lines the Screen, film 2 or and ray are detected reaches Shadows are our when eyes. formed when photographic an opaque object blocks the plate path Pinhole 3 Real, Distant of Solar light rays. eclipses occur when inverted object image 4 Figure 3.4.4 The pinhole the Moon the Earth Lunar passes and eclipses between the Sun. occur when camera the Earth from prevents reaching the sunlight Moon. 83 3.5 Reflection When LEARNING light the end of should be and state strike a surface they light may be absorbed, transmitted or OUTCOMES reflected. At rays of this able topic Describing the all follow specifi c laws. you to: apply Refl ections laws reflection of Reflection occurs when a ray of light reaches a surface or boundary reflection between describe formed how in properties a images mirror of and those strikes are This the a ray mirror . For The reflects off example, ray the that a reflection strikes silvered the surface occurs mirror and is is when called called a an the light ray incident reflected ray. ray images. At a the point normal. surface. Ray materials. where The the normal When incident is a measuring measured relative The of to the line ray touches the perpendicular angles normal, during not to mirror (at right reflection the all surface we can angles) angles draw to a must be itself. box Incident For laws any reflection reflection at a surface two laws apply: ray i = r i the angle of the incident reflection ray, is equal reflected ray to the and angle normal of are incidence all in the same plane. Normal r Figure 3.5.1 shows Experiments show a simple that the reflection angle of in a plane (flat) incidence, i, mirror. and the angle of Reflected ray reflection, Mirror The lie Figure 3.5.1 Investigating the laws second on a using a ray are law plane always of (a equal, reflection flat surface proving states like a that piece the of first two law. rays paper). and This the normal means that all the ray does not twist upwards or downwards during reflection. box Regular Reflections This is so and from because Parallel the rays of diffuse a mirror the object. reflection produce reflective light observed original will size This be and is an surface reflected shape regular of the and the (a will image reflection picture mirror is remain will we very parallel match (Figure can see). smooth. that and of the 3.5.2a). b Parallel a image of a light rays from source Mirror Figure 3.5.2 or polished surface Reflection paper 84 the of reflected reflection r, is b from a Rough mirror diffuse. is a regular but surface, reflection like paper from a sheet of Most surfaces surfaces but in the the is not will roughness different This are rays of in This parallel according surface reflection plane When reflected the directions. diffuse Images smooth. be causes means (Figure that rays to these no of the light laws reflected clear image hit of these reflection rays will to be travel formed. 3.5.2b). mirrors x = y The image we see when we look into a mirror is a virtual image. A Point object Image x virtual no image rays the of same Figure cannot light actually distance 3.5.3 be the projected pass behind through the distances x onto mirror and y a screen the as are or image. the the surface The object is because image in y O I is front. In same. Normals SUMMARY 1 What QUESTIONS are the sizes of angles Q, P, R and S as shown in Mirror Figure 3.5.4? Figure 3.5.3 The image formed in a Mirror plane mirror Normal S Normal ACTIVITY 20° R Design a method to prove that P 30° Q the Mirror image same as Normal in a distance mirror the object is behind is in the the front of mirror it. Mirror Figure 2 A 3.5.4 student face looks exactly a Is the b What directly 30 cm image from real or into the a plane mirror mirror. using a She positions her ruler. EXAM In is the total TIP virtual? distance from her face to the exam ques tions , image? mirr ors may positio ned or KEY verti cally , angl e POINTS as in ques tion 1 2 Angles The all are angle always of measured reflection is from equal to the the normal. angle of sure incidence for to you the be 1. but at no rm an Sum mary If you measu re shou ldn’ t reflections. no t ho riz onta lly al mak e angl es they give you any problem s. 3 The image behind the in a plane mirror as mirror the is virtual object is in and front the of same the distance mirror. 85 3.6 Refraction of light 8 Light LEARNING travels at approximately 8 2.00 At the end should be of this able topic to you × describe at a change to: how light boundary 10 change point describe × 10 −1 m s in air but slows to is m s glass. direction at is This the change boundary always of speed between measured causes the relative two to the the ray of materials. normal light This at the refraction. refracted between in a rectangular block materials how be dispersed by a prism in direction in of −1 Refraction transparent 3.00 OUTCOMES white into or a light When can the causes spectrum (Figure raindrops. and light the ray to 3.6.1). refracts parallel, the originally ray refract, When away ray but it Refraction enters ends is to the from the a ray its travelling it slows path leaves normal. laterally in block change light the up glass towards the glass Because in the down the same and the block two this normal it speeds boundaries direction as it up are was displaced semi-circular block Lateral If the incident ray enters a new material along the normal then displacement Glass the ray cannot the same ‘turn path. The towards ray still the normal’ slows down and or it continues speeds up. along This can be block demonstrated directed with along the a semi-circular normal to the glass block curved where surface the (Figure ray can be 3.6.2). r Refraction i Triangular r < prism from it refracts prism are also the towards used normal. As the the to refract two normal and light. refracting as it As the leaves surfaces are it ray enters refracts the not away parallel the box emergent Figure prisms a i Normal Ray in 3.6.1 Refraction through rectangular glass ray is not parallel to the incident ray. a block Dispersion All electromagnetic but Incident ray will travel different at colours transparent radiation different of light materials. travels speeds travel The size in at the other same speed mediums. at different of the speeds change in In in in a vacuum particular glass direction or other during P Normal i at P refraction depends on boundary (see and the change in the speed of light at the r different degree Refracted Figure 3.6.2 The ray refracts boundaries change but at both does direction ray not is amounts. than called enters is travelling the 86 the Rainbows raindrops. when surface. block as normal it to red For light dispersion produced. it 3.7) are caused The light so different example, as it has (Figure when is a colours violet light greater of light refracts change in will to a refract by greater speed. This effect 3.6.4). light dispersed from and a the Sun passes coloured through spectral pattern is ACTIVITY Normal Use a when filter prism you to to try disperse to produce Refraction in white disperse blue light. blue light Investigate with a what prism. happens (Use a coloured light.) water Apparent When from path rays the of of light normal. the ray travel If will a ray from water travels change into from such that air the the the rays bottom ray of refract a appears pool to away then come Real to actually the is surface. (Figure SUMMARY 1 Copy for causes the pool to look shallower from that 3.6.3 The pool is deeper than it it appears due A can to refraction. 3.6.3). QUESTIONS Figures the This position the Figure closer position 3.6.5 incident and 3.6.6 and then complete the ray path rays. Normal 45° Normal 45° Figure 3.6.5 Figure Figure 3.6.6 3.6.4 prism disperse 2 Which enters 3 colour glass Which of of visible from these light slows by the least amount when The higher statements speed as the it The frequency enters longer speed as the it is/are correct? glass of light from the greater the change wavelength Alwa ys air. leaves glass and of light enters the smaller the change in any will angl e POINTS When a of light travels from one medium to another Y ou it If the the ray point If the ray no rm al refra ction . help you chan ge muc h shou ld to in more slows of down, its path will turn towards the normal at sharp, refraction. speeds up, its path will turn away from the of see that the direc tion boun darie s Avoid 3 the chan ge speed. these 2 the clear ly. ray changes draw diag rams invo lving air. judg e 1 TIP in T his KEY to it on used light. air? EXAM be white sudd en usin g is at a turn . the wo rd normal ‘bend ’. at 4 the White its point light of refraction. will constituent disperse during refraction and separate into colours. 87 3.7 Refractive total Snell’s LEARNING the end should be apply internal and reflection law OUTCOMES Snell’s At index of this able Snell’s refractions topic law states that the ratio of the sines of the angle of incidence you and angle of refraction This constant at a boundary is a constant (Figure 3.7.1). to: law to shows to the is the refractive refractive index of index of different the boundary. T able 3.7.1 materials. calculate sin i change in angle or = speeds refractive index, n sin r find the critical angle for a The same form of relationship also applies to the ratio of the speed boundary of describe total light in the two materials. For a boundary between air and another internal transparent material: reflection speed explain how total is used light in air, v 1 = speed reflection of internal in of light in material, refractive index, n v optical 2 fibres. The two equations can be combined sin i to give: v 1 = = sin r refractive index v 2 Normal WORKED EXAMPLE 1 i Medium 1 Medium 2 A ray of light (refractive is refracted index 1.50). at The the boundary angle of from incidence air is into 30°. glass What is the r angle of refraction? sin i = refractive index sin r Figure 3.7.1 The refractive boundary is index the of ratio the sin i sin r of = refractive sin i to ratio sin r. of the This is speeds also of sin 30° = = index 0.33 1.50 the light –1 r in T able 3.7.1 the Examples indices of for a refractive ray of T otal the vacuum material (or 0.33 = 19.5° Refractive internal index, index light and can 1.50 water 1.33 Perspex 1.50 reaches one be with a boundary lower between reflected by refractive if critical given the angle angle by the of the surface incidence (Figure 3.7.2). is The instead greater critical of of glass high to being air) refractive then refracted. the This than an angle, angle C, for a called the boundary is 1 = refractive index of boundary 2.42 During this material this 88 material (e.g. relationship: sin C diamond a index n occurs glass reflection from air) light Material sin light When entering a = materials. process and process so the there is all of the process no loss light is of is called light reflected total back internal energy. into the first reflection. During ACTIVITY At the critical angle Total internal Incident Incident reflection ray Next time you are swimming ray in P P i C Normal at P Normal at a pool go and look towards Try P to to explain the bottom the the surface. effects that r you see. you are Flash a Refracted ray Reflected a long light fibre 3.7.2 Total internal than the critical reflection WORKED The is (r EXAMPLE refractive the critical index angle reflection = angle occurs C. The if the angle reflected ray of only do this if supervised. into one optic end cable. of You ray should Figure Note: being incidence follows the is the greater laws of see other cable is the end flashing even coiled at when the up. i). 2 of for an air this to Perspex boundary is 1.50. What boundary? Ray 1 sin C 1 = = = 0.66 Normal refractive index of boundary 1.50 –1 C = sin 0.66 = 41.8° Optical fibre Optical signals fibres enters the of glass. the glass use (Figure fibres even The when SUMMARY the process 3.7.3). and ray A ray travels can the of of along travel fibre is total light it for internal (more by being several coiled in refl ection usually to transmit infra-red refl ected kilometres at radiation) the through surface the loops. QUESTIONS Figure 1 Copy rays T able 3.7.2 entering a and then material complete from it using Snell’s law Rays in an optical fibre air. KEY T able 3.7.3 for POINTS 3.7.2 1 Angle of Angle Refractive of index Speed of light The ratio of the sines of in angles of incidence and −1 incidence/ refraction/ degrees degrees of boundary 30 material/m s refraction is called refractive the a constant index. 1.33 2 20 The the 50 refractive index is also 1.45 30 ratio light in of the the speed two of materials. 8 45 1.5 × 10 3 When a along 2 Draw a bottom the diagram of water. a showing pond so Calculate that the a it ray of just critical light travelling escapes angle for from this from the the surface boundary. totally of many is lost ray an of light optical internally times at but these passes fibre it reflects little energy reflections. 89 3.8 Converging T ypes LEARNING the end of this topic be able are (Figure describe the path through a (convex) lens describe the of rays that the converging a images of Lenses glass or usually plastic have which two cause curved refraction surfaces and of each converging are refraction. refraction of the many In order takes to place simplify for each diagrams, ray and this we assume happens in lens. shapes of lens but there are only two main effects: formed by causes single centre There blocks 3.8.1). to: surface curved you light should lens OUTCOMES Lenses At of lenses A converging lens brings rays of light, parallel to the principal axis, lenses. together. A diverging apart. The Converging See lens spreads rays of light, parallel to the principal axis, 3.9. principal axis is an imaginary line which passes through the lenses centre The it of Plano-convex lens principal and the lenses Bi-convex the focus centre cause and lies of more is perpendicular on the the lens is refraction to principal the than it. axis focal weak and the length, lenses f, and distance of so the between lens. have Strong shorter Converging focal lengths. meniscus Diverging Converging lenses Figure These the 3.8.2 rays lens Bi-concave Plano-concave shows are so (marked a set parallel that F). lenses they Each all lens of to rays the meet has passing principal at two a through axis point and called principal a converging they the are lens. refracted principal by focus foci Diverging meniscus Figure 3.8.1 A range of lens shapes Parallel f rays F L Figure The 90 F = principal f = focal L = optical 3.8.2 focal passes through would be (real) centre Parallel plane focus length is a the brought rays passing plane a a converging perpendicular principal to through focus focus. on a to Parallel screen the rays lens principal from placed in a the axis distant focal that object plane. Images We can from the from find an the object. Ray 1 travels Ray 2 so Figure Ray 3 it the it of the 3.8.3 lens image three place through reaches principal parallel passes through takes passes When the to that passes refraction properties In lenses by rays analysing are drawn the ray from paths the top of object. refracted converging the the to the principal axis. through the principal focus. optical there the lens is no centre change principal it is focus refracted of of lens ray and as is no direction. on so the The the that it left of the becomes lens. parallel to axis. Converging lens EXAM TIP 1 2 Draw ing Object requi res Principal F 3 Principal 2F axis axis Real Measu re ray the caref ully use a and sharp care. distan ces image (diminished) diag rams great alwa ys penc il and ruler . Figure The 3.8.3 three should rays of rays be original An meet able object light image to formed and see by form that, in pass converging an image this case, (diminished), really a it through is on the upside the lens the down image right image and is of the (inverted) so it lens. smaller is a You than and real the that image. ACTIVITY Place rays a of converging light principal Draw and focus two lens and more versions between F and 2F b between F and the each case, front what measure a For in describe draw of of a ray happens the focal Figure box to producing the length 3.8.3 but rays. of the place parallel Find the lens. the object: lens. the three rays and describe the KEY image. 1 POINTS A converging rays, axis, SUMMARY parallel so that Define and the key principal terms: focal length, focal plane, principal focal Draw axis of a scale each focal they principal are focus together at the point. axis. 2 2 refracts the QUESTIONS brought 1 lens to diagram 0.5 cm length apart 5 cm. showing as they five pass rays parallel through a to the principal converging lens A diverging rays, axis, lens parallel so come that from to refracts the they the principal appear focal to point. 91 3.9 Diverging the Figure LEARNING the should end be eye 3.9.1 of this able topic rays come you shows describe the through a spread from principal to: focus a path of diverging is one focus the out set of rays (diverge) passing point. and focal As the for in a such a distance between that the diverging lens, diverging the this lens appear lens. point and is the to all called the principal length. rays (concave) images Parallel formed rays lenses. F Figure To find F = principal f = focal 3.9.1 Images Figure a they f describe by through way converging lens and OUTCOMES The At lenses Rays in the focus L (virtual) length passing through diverging properties of a diverging lens lenses an image produced by a diverging lens draw 3.9.2 three Ray rays 1 is appears as for a parallel to converging to come the from lens principal the (Figure axis principal and 3.9.3): is focus refracted (the so dotted that line it shows this). Ray in 2 passes straight through the optical centre without a change direction. Ray 3 is the ray beyond the lens. when passes it which This would ray through is pass through brought the the parallel to principal the focus principal axis lens. f f 1 3 Object ACTIVITY 2 Principal Place a diverging lens in F F axis axis Virtual of a ray box producing parallel image rays of light and happens to principal focus focal length the of describe rays. and the Find what the measure the lens. Figure 92 Principal front 3.9.3 An image formed by a diverging lens As before, formed the image means you Human a is that could Our the eyes the not also rays acts a and and not to come the upright. actually screen there from principal The pass and a point. axis. image You is through see the The image should also that see virtual. point is that This and so image. vision contain at appear point do place two lens muscles objects rays this diminished converging ciliary three between with are a lens a used different as surfaces variable to a the The fixed act focal stretch distances. with which lenses. length lens outer focal as to The (Figure allow layer of lens 3.9.4). us the eye’s to Small focus eye, is the on cornea, Cornea length. Retina Correcting Both converging vision A vision (T able Lens and diverging lenses are used to correct defects of 3.9.1). short-sighted (near-sighted) person suffers from myopia. The Ciliar y lens in their objects. a focus This The in poor diverging of A light the lens focus eye. retina used to power can T able to used reach of blurred this a the vision correct from image problem to object instead of of focus would clearly distant myopia by on be on muscles distant brought its to surface. objects. diverging Figure 3.9.4 The human eye A the rays cornea. person as enough distant retina to (such light thin blurred (far-sighted) a the 3.9.1 be from eye’s leads objects Rays so become light the they correct of of of focusing long-sighted the cannot rays front before cannot to eye the the is by suffers words object formed. from on a would A increasing hyperopia. page) be that focused converging the overall They are lens close behind can be converging eye. Defects not of vision and their correction (refraction by the cornea is shown) Myopia Hyperopia Before After correction SUMMARY 1 Draw axis a scale each focal QUESTIONS diagram 0.5 cm length apart showing as they five pass rays parallel through a to the principal diverging lens of KEY 10 cm. 1 2 ‘The and image smaller Construct POINT formed than some by the ray a diverging object.’ diagrams Is lens this to is always statement test your virtual, true answer. or upright false? A diverging parallel so that from to they the lens the refracts principal appear focal to rays axis come point. 93 3.10 Magnification As LEARNING you size At the end should be have seen, the of this able topic as the object) you or object. The diminished calculate the produced or by a other determine of a an object (smaller be are not than magnified always the (larger the than same the object). and image height magnifying of the images is defi ned by an equation comparing the lens image of may magnification Magnification glass image to: Magnification images OUTCOMES the focal converging height to the object height: length linear lens magnification, m, height of image height of object = experimentally. Magnification and object distance Converging The linear magnification of a converging lens can also be found lens X using the I L object formation LOX O the are (image distance of similar height) an image triangles is and the by and same a distance. converging so as v u image the the ratios ratio of Figure lens. of u OX to image distance, v object distance, u 3.10.1 The shows triangles (object LIY height) and to IY v Y Linear magnification, m, = v A m × A Figure 3.10.1 Linear magnifying magnifying glass uses a single converging to work focal EXAMPLE image of effectively length. This an the object object (Figure needs arrangement will 3.10.2). to be a lens forms candle 10.0 cm. of the an with If lens a the is size image of height of magnification 1.60 of the produce produce a the a to magnifying the lens magnified, than virtual what is virtual i magnifying ii erect ii instrument eyepieces iii magnified spectacles, as iv on of the the lens iii same as O side for glass correction long-sightedness and away candle? F magnification O L = Eye height of image height of object Figure Height of object height linear of 3.10.2 A magnifying glass = image magnification The The lens formula relationship distance, v and between the object the focal = 6.25 cm 1 1 1 1.60 = f 94 length distance, 10.0 cm = image. Uses: i F Linear glass the 1 further actual to For closer Image: A lens magnification magnified WORKED glass u + u v u is of a given lens, by: the image When using the lens formula the distances are positive for real images WORKED but are negative produce a for virtual virtual image images. 0.1 m For from example, the lens a and diverging so v would lens be −0.1 m. A lens has The lens formula can be used to determine the focal length of a illuminating an object and positioning the lens to form a a on a screen (Figure 3.10.3). The distance between used focal produce the lens object is measured as is the distance between the lens the and These two distances can be substituted into the formula the focal length of the of camera 0.10 m. a clear image on a image behind must the be lens. formed How the lens should the far object to be find simple the from screen. a and 0.20 m the in length clear CCD image 2 lens To by EXAMPLE may positioned to produce this lens. clear image? 1 1 1 = v u + f u v White Light box 1 screen 1 1 = + 0.10 m u 0.20 m 1 1 1 = Pearl – lamp Converging u 0.10 m 0.20 m lens Real Front of object light or box: illuminated on 1 image = screen 10 – 5 = 5 u cross-wires 1 Figure 3.10.3 Finding the focal length using the lens formula u = = 0.20 m 5 SUMMARY 1 In which QUESTIONS of these three situations is the magnification the largest? ACTIVITY An object of height An object of 13 cm An object of 3 cm 5.0 cm producing producing producing an an an image image image of of of 20 cm. 1.0 cm. 7 cm. Perform an find focal the range 2 Copy T able 3.10.1. Then use the lens formula and to complete the table for a range of converging lenses. results the lens. 3.10.1 curvature Image Focal distance distance length Magnification focal 2.00 m Use see 0.25 m the a is of the lens the and the glass happens to when positioned the beyond focal length the of image the lens. produced. POINTS The magnification image to the of height an of image the is the original ratio of the height of T ake object. The the The magnification object focal distance is also the ratio of the image distance length of image care a lens can distance be found using the from lens the when fo rm ula, dealin g distance. and to TIP an lens 3 the 0.2 EXAM 2 curvature between magnifying what Describe 1 the 6 cm 14 cm KEY to length. object 15 cm to a Compare Describe relationship Object of lenses. of T able length magnification your equation of investigation with usin g as you the are recipr ocals. object formula. 95 3.11 Diffraction, and theories nature Scientists LEARNING the end should be outline have of this able the topic A particle of describe Young’s and and very brief is behaviour behaving as a of light stream for of centuries particles and or as have waves. history… Huygens the of formulated concept to a theory describe that reflection light and travelled as refraction in a wave terms of evidence Newton believed that light consisted of a stream of particles he wave corpuscles. He used this model to describe the behaviour of light light the this wavefronts. slit the used called behaviour it wave Isaac demonstrating describe light light experiment the the you to: and studied whether Christiaan theories of about OUTCOMES debated At interference interference and his ideas were generally accepted. of Thomas Young devised experiments which demonstrated the wave waves. behaviour explained theory of if was light Albert are Einstein experimental Produce a detailed the history understanding of were results a stream and described used result a of of in more the experiments particles replaced particle called these with detail model of and the so could the wave not be particle theory. These below. light ‘photoelectric to explain effect’. the This could not of using wave theory. our Today light. it is particle accepted behaviours Diffraction When a spreads The water out. amount (Figure a of wave This of gap that in light has different both wavelike properties and situations. waves passes process is diffraction 3.11.1). through that Maximum roughly through a takes same size we and place diffraction the gap diffraction see the place that for depends takes as can occurs on when all the wave types the size the wavelength a of of of wave the waves. the b beam ACTIVITY Use the a ripple tank interference formed by through to Diffracted patterns waves two observe wavefronts passing narrow gaps. Figure Use a laser to interference when light observe patterns passes 3.11.1 Diffraction of water waves a at a wide gap b at a narrow gap the formed through Light diffracts when it passes through a gap. As the wavelength −6 two very narrow slits. of light is very noticeable evidence 96 small when that (around the light is gaps 0.5 are × very behaving like 10 m), narrow. a wave. this This process is diffraction gap passes wave. Vibrating be timeline explained about The abandoned experiments ACTIVITY light. only gives Interference When two and waves superimpose meet (Figure waves are At points where waves cancel each trough). This At points other increase. is This and two-slit pass 3.11.2). separate some the simply other a crest two out will each other, displacements the waves caused by the together. waves destructive is through The added the experiment overlap (for the example, superposition when a crest of the meets a interference meet constructive a crest and the displacements will interference Figure 3.11.3 T wo an a Destructive tnemecalpsiD + position = interference tnemecalpsiD tnemecalpsiD 0 Figure 3.11.4 tnemecalpsiD + = An 1 0 Sketch what of a diagram as waves 2 Why do they pattern (Figure formed by the waves is an interference 3 3.11.3). we in indicates Thomas Young investigated what happened when light clear two very interference through two produce this narrow gaps. patterns gaps (Figure effect. The in He the 3.11.4). patterns found same A that way as stream could only the light water of be as a particles would explained if not light Digital collecting KEY nature however, cameras light packets as POINTS There have been of several energy in of light be explained photovoltaic small energy the behaviour of particle or light. light cannot and about packets, cells by the behave similar to wave as if model they particles. Diffraction are called and interference of experiments provide evidence the are for wave These behaviour small behaves was 2 light. light that wave. particle effects, evidence topic that see waves? passing wave Some the this normally light wave. theories The a produces waves 1 travelling through passed a through showing water pass not of Summarise found to entrance. diffraction resulting pattern QUESTIONS happens waves Superposition interference 0 harbour pattern as superimpose. 0 SUMMARY The waves 0 Constructive 3.11.2 produce pattern tnemecalpsiD tnemecalpsiD 0 Rest Figure waves interference the b water interference of light. photons 97 3.12 Sound Sound LEARNING At the end should produced of this how compare topic objects. Typical sources include: the vibrating the skin moving vibrating a strings on a guitar you on a drum to: sound columns of air in a woodwind instrument waves vocal chords sounds and in terms when you speak drill being used and to to pitch. Sound waves passed to Wires of a sound power bell propagate medium from an remove the to A on placing road. waves air (travel) bell to quieter longitudinal because particle. inside (Figure becomes as necessary particle electric the is a 3.12.1). until This glass it the can waves be jar and the air is cannot be must using a removed, heard at all. a be demonstrated bell As through vibrations by vacuum the pump sound This is of because supply there is Sound removed a up loudness medium. using dig amplitude amplitude frequency to of Propagation relate Air vibrating produced frequency by OUTCOMES able describe are be is waves no longer waves are rarefactions a medium for composed which spread of the a out sound series from of the waves to pass through. compressions source (Figure and 3.12.2). vacuum The compressions are regions of air (or another medium) where the pump particles have rarefactions Bell jar Bell works been are compressed regions where more the closely particles together are while further the apart. but Compressions cannot be heard Loudspeaker Figure 3.12.1 When from the the longer air jar be is removed the bell can Rarefactions no heard. Figure 3.12.2 The Describing T able 3.12.1 Sound travels at Medium in different Approximate and and rarefactions displaying in a sound wave sounds different Just speeds compressions like all other waves, sound waves have amplitude and frequency. media The greater The higher the amplitude of a sound the louder it is. speed the frequency of the sound the higher the pitch. −1 of air sound/m s 340 water 1500 steel 6100 Representing Sound energy on the sound 98 waves connected to into sound can a an be as electrical a on represented microphone. oscilloscope. wave waves The trace The signal an on the the screen microphone and this oscilloscope on oscilloscope screen. signal then of an oscilloscope converts controls displays the the the sound display ‘shape’ of the On the The oscilloscope height wave. The of the period waveform wave. of is show between sound period this waves distance the This of T aller display: the the The related to the amplitude of the sounds. peaks closer sound inversely is louder on the the wave peaks are represents together the the a Loud and high-pitched b Loud and low-pitched c Quiet period shorter wave. proportional to together peaks, the frequency ( frequency = 1 period of ) and the sound Most They so the sound. closer Figure 3.12.3 the shows the higher oscilloscope the traces of frequency simple waves. sounds are are much composed of more waves complex of than different these single frequencies and Figure on in each other. Figure produces more complicated (higher pitch than a) waveforms as 3.12.3 Sound shown on an waves represented oscilloscope screen 3.12.4. SUMMARY 1 This high-pitched frequencies. superimposed Why do QUESTIONS you hear distorted sounds when you are swimming underwater? Figure 2 Sketch a diagram represented a A high b A sound of 3 on Use an with in wave how oscilloscope frequency sound the showing sound half part speed the these two sounds would of wavelength in with low frequency and twice the water c in steel. (The speed T able sound equation for a flute to find the frequency of a sound an oscilloscope to compare 1.0 cm: waveforms in the different media is shown produced musical by instruments or forks. in 3.12.1.) WORKED KEY trace a ACTIVITY tuning of on amplitude a different in played amplitude. air b oscilloscope screen: the a An note Use wave 3.12.4 be POINTS The EXAMPLE period of a sound wave is –3 4.0 1 Sound waves are produced by vibrating objects × 10 s. including What is the frequency of the loudspeakers. wave? 2 Sound waves travel as longitudinal waves through a medium 1 composed of a series of compressions and f rarefactions. = T 1 3 The higher the frequency of a sound the higher the pitch of = –3 the 4 The 4.0 sound. greater the amplitude of a sound the louder the sound. = × 10 s 250 Hz 99 3.13 Experimenting sound The LEARNING At the end should be of this able describe be waves behaviour of sound waves OUTCOMES topic Reflection you to: Sound with how reflected, sound rigid can refracted can surfaces waves and waves are be cause called reflected the just strongest echoes (Figure like any other reflections. wave. The Smooth refl ected and sound 3.13.1). diffracted Refraction describe sound interference waves describe used of in Sound how sound pre-natal materials can scans to be also and will waves as the Warmer Cooler lower of air air air – they the can temperatures when change because increases sound testing. waves another bend higher as they 3.13.2). the speed of of is they The the travel This ground when speed. temperature (Figure near direction change air cooler is from of layers most than the one sound increases. through effect move speed This of air above air means at noticeable air material in that different at night it. sound – speed sound Source of sound Observer Ground Figure 3.13.2 Sound waves cooler can than follow a air on curved a summer path evening through a temperature gradient. Sound Diffraction waves Sound Figure 3.13.1 An echo rigid from a smooth waves so they are as they pass have a much diffracted easily greater by wavelength larger gaps. than Sound light waves waves will and spread and through doorways or around corners as their wavelength surface is similar to the size of these gaps. Interference When two sound waves meet top each they interfere. The waves are ACTIVITY superimposed Investigate waves of follow reflection cardboard sound whether Find the identical laws using tubes sound to direct pattern the waves. out about frequency dolphins. waves also the range of Produce bats how they regions be of used then connected front will of to them. This louder the than this the interfere where other. If effect same two no sources can source pattern and two will detected. will have sound waves be will producing form an regions be constructively interference where heard. an T wo the There interfere will and the normal. and report Using about in are destructively be sound hearing a signal loudspeakers some on sense prey sound waves and If the speed of sound is known then we can measure distances by predators. recording 100 the time taken for sound to reach us. For example, during a storm, light sound takes how far from a away WORKED A the lightning lighting EXAMPLE lightning later. a measureable How strike far reaches of us time almost allowing instantly us to but the calculate struck. 1 hits away strike amount a tree did and the the thunder lightning distance is heard 4 seconds strike? (m) –1 Speed (m s ) = time (s) –1 distance = speed × time = 340 m s × 4 s = 1360 m Figure 3.13.3 A lightning strike Ultrasound High and frequency so are produce Because easily or In ultrasound audible produce care, (Figure echoes (above 20 kHz) ultrasound. pulses to pulses sound, detect can be they are not Animals as bats by or humans dolphins obstacles. directed can detectable such be in used narrow to beams make more measurements images. pre-natal the as ultrasound than foetus sounds classed ultrasound 3.13.4). of these The pulses pulses pulses are are used reflect off processed by to form the a an image different computer of tissues to form the and an Figure 3.13.4 An a image. much An Unlike safer X-rays, to as shoals pulse pipework, of fish does not cause ionisation and so image of is use. ultrasound such ultrasound ultrasound foetus near can to a also be measure boat used the (Figure to ‘see’ depth of into the solid materials sea-bed or to detect 3.13.5). Transmitter WORKED EXAMPLE 2 Sea-bed An ultrasound pulse is used to detect a crack in a metal pipe. The −1 pulse travels at 6000 m s in the metal and the echo is received Figure 4.0 μs after the pulse is sent. How deep within the pipe –1 Distance The = pulse 0.012 m speed has × time travelled within the to = 6000 m s the crack is the Using and 4.0 back × 10 and s so = the crack is pipe. POINTS Sound waves They along a road you hear an ambulance siren can corner but you cannot see the ambulance. Draw explain how this can A dolphin produces a sound pulse which reflects off a fish be hunting. The echo of the pulse second is detected by the is after the 1500 m s . pulse was each and other. detect sent. The speed of can be objects used by echo dolphin sound and to measure in distances. −1 water diffracted with Ultrasound location 0.4 reflected, it to is same waves. happen. 2 2 the other a interfere and as from refracted, the diagram share QUESTIONS walking around to 0.024 m behaviours While pulses depth –6 × 1 1 sound measure KEY SUMMARY 3.13.5 crack? How far apart are the dolphin and the fish? 101 Section SECTION 1 The 3: Practice relationship and the 3 angles Practice exam between of exam questions questions the incidence refractive and index refraction B for Displacement/cm a ray glass crossing is given the boundary between air and by: 3 sin i refractive index = 2 sin r 1 During an experiment to measure the 0 refractive index of a glass block the following –1 results were obtained. –2 Angle of 10 15 20 25 30 35 –3 incidence, Time/s i/degrees 3 Angle of 7.0 10.8 14.3 17.6 21.2 On a calm day you can see your reflection 24.2 when looking into a pond. When the pond’s refraction, surface is disturbed you can no longer see a r/degrees reflection. Why is this? sin i 4 The electromagnetic (EM) spectrum consists of sin r a series a a Copy and complete the table of State waves. two properties electromagnetic calculating the values for sin i and Plot a suitable and sin r graph using the values for allows you to all have in common. Copy obtain a and complete this table showing sin i the which waves sin r b b which by EM spectrum in order of increasing value wavelength. for 2 the Diagram graph a refractive A for shows a water a index for the glass block. wave displacement–time and graph diagram for a B same Visible Radio rays light waves shows particle in c the Gamma distance–displacement Which of the regions electromagnetic a of the wave. spectrum: State: i the wavelength ii the amplitude iii the period of of of the the the i has the lowest ii is a major iii is used frequency? wave cause of skin cancers? wave in radiotherapy to treat wave. cancers? b Calculate the speed of the wave. 5 A An oscilloscope received Displacement/cm different nearby. from a is tuning These used to display microphone forks, signals A and are signals when B, are shown in two struck the 3 diagrams opposite. The oscilloscope has been 2 adjusted so that each horizontal division 1 represents 0 a time period of 2 ms. –1 a Which of louder sound? the tuning Which of higher pitch? forks produced the forks produced the –2 –3 Distance/cm b 102 the tuning c Find the frequency of tuning fork A. 7 During pulse d Without further calculation, state of tuning fork medical used to examination measure the an ultrasound thickness of a the fat frequency a is layer in a patient’s skin. Sound travels at B. −1 630 m s pulse Sine wave is in a How b Why c thick are Why The 0.3 μs echo after 8 A a fat lower the from pulse the is sent. box State the b Copy and and two a scans the used law curved laws of complete reflected frequencies used instead of care? investigated ray layer? frequencies? pre-natal a the the ultrasound in student using is ultrasound of are X-rays wave tissue. A instead Sine this received of reflection mirror as shown. reflection. the diagram showing ray. B c Mark its on the angle of reflection and state value. Normal 70° Mirror 6 During an experiment to measure the speed of 9 sound in metal, two microphones are A group of properties to a long, thin steel bar . The first the stopclock when it detects the bar . The second investigating constructed a optical triangular microphone diamond. Diamond has a prism refractive index sound of in have microphone from starts scientists attached is 2.40. 50 cm 8 further when the a along it the detects stopclock What and sound. is type bar stops The the a stopclock measurement wave b is speed What on 0.2 ms. of The Explain light sound? to prism b What c The is the sound with a speed of source frequency sound in produces of a 1000 Hz. the steel sound in air is The wave speed of 10 What both is wavelength of the ii diamond greater 3 × in 10 −1 m s . diamond? disperses extent white than a glass and a and have index. into a When beaker type a of of block the baby exactly of baby the oil oil the are same glass the is block sound invisible. Why is this? air? What in the much is light glass refractive 340 m s the a of air transparent becomes in why speed in would. Borosilicate placed i the light bar? −1 sound is of the is the metal wavelength of the sound bar? 103 11 A ray of walled shown a light glass in the Calculate to glass passes beaker into the side containing of a water thick- 13 as angle optical with diagram. the An a curved of refraction at the air a as Copy boundary. State the water angle of the incidence at the glass the ray boundary. Calculate glass to the b angle water of refraction at Complete the diagram What the centre of the the the far The a glass fibre is diagram. and entry from 1.60. draw into the the fibre path until of it end. is the critical angle for the glass to boundary? c When bending showing the at the the fibre surface. a small Explain crack why this may ray prevent reaching of the boundary. ray in diagram from at forms d constructed index to air c is shown leaves b fibre refractive light from reaching the end of the beaker. fibre. Normal 50° Air Water, n = 1.33 Glass, 14 A slide projector produces a magnified image n = 1.50 of slides The 12 Rays of light are shone into two prisms, slides B, as shown in the diagram. Prism A of a standard glass with which 1.50. plastic index a with of Prism a B is constructed significantly lower from the critical angles refractive for both of the prisms. b Use the results complete the the the calculations paths until the to rays leave blocks. A 104 of ray 14 cm through from a them. has a focal length of converging 8.0 cm. Draw a ray is diagram showing how the produced. a 1.2. Calculate placed shone refractive image index is is a constructed are light A lens and when B b Describe the properties of c Calculate the distance d Calculate the magnification to this this image. image. of this image. 15 The lens form back the a in a human focussed of lens the eye. and the eye image The eye is on retina can converging the lies form retina 5 cm a and at can a the an object is placed a behind clear minimum three showing on the rays how on the a copy image of this would diagram be formed retina. image b when Draw Describe the properties of the image distance formed. of 20 cm An from object minimum of the eye. height distance 4.0 cm from is the placed lens as at c Calculate the magnification d Calculate the vertical shown simplified ray the image. size of the image on in the the of this retina. diagram. 20 cm 5 cm Retina Object 105 4 Electricity 4.1 and Static Atoms LEARNING the end should be name an of this able the of topic you (Figure three electric 4.1.1). types of Protons particles: and protons, electrons have neutrons a property charge. to: charged particles in atom describe the charged particles explain composed electrons called are electricity OUTCOMES and At magnetism forces charging are nucleus of Neutrons electrical between by Protons Atoms small to be atom. also positively They found charged cannot bound be in and removed the nucleus are bound into the easily. but have no charge. are and are positive Very are Electrons nucleus induction. said the negatively can be neutral charge particles and charged. removed if they because negative They are they charge, are found provided have giving a the outside with same total the some energy. amount charge of of zero. nucleus Separating charges Most are + materials uncharged because they are composed of neutral Proton atoms. When some materials are rubbed together the frictional forces Neutron can cause Because move Electron This electrons the from protons place transfer electrically When to of to be are transferred tightly from bound in one the material nucleus to they another. cannot place. electrons results in both of the materials becoming charged: extra electrons enter an object the object becomes negatively charged. Figure 4.1.1 The structure of an When electrons Rubbing from EXAM is movem ent from place caus e you polythene cloth to rod the with it has extra charged because it has fewer a Perspex leaving the rod rod a polythene because the object becomes to will dry cloth rod. The electrons cause will rod and electrons positively electro ns place to Mak e the than electrons charged cause will cloth positive becom e sure that Electrons by the result losing as move the move become (Figure from cloth to negatively positively 4.1.2). the rod negatively to the charged. Atoms of become electrons gained positive a rod result losing as of electrons pro to ns) gained + movin g + r + by + + + + + + r o + + s r e P cloth Dry Figure d + + x e p P t y l o e h e n objec ts. o of Electrons electro ns Dry 106 will protons to and electrons become + out charged. become that a describ e or positively Atoms of d into object the objec ts charge d. (no t a the Rubbing TIP alwa ys an charged cloth It leave atom 4.1.2 Charging a plastic rod cloth cloth Forces between charged objects + Charged particles produce a force on each other in a similar way – to + – magnets: – + Opposite charges attract each other. (Positive and negative – charges + attract each Similar repel + + + + + + + – other.) + + + charges positive repel each other. (For example, positive – – + charges charges.) a The charged rod is held near the sphere ACTIVITY Suspend rods of different materials from a piece of cotton – so that they can rotate freely. Charge up the rods and then – investigate the the effects suspended of placing other charged rods very close to + + – + + –+ ones. + + + – – Electrons from – Induced charge earth flow onto sphere A neutral placed attracted one part other object nearby by of part a become object the charged 4.1.3). positively the of can (Figure The charged to object when electrons rod become and a in move negatively positively charged the charged. towards charged If object object we will it and touch is b be The sphere is earthed briefly causing leaving the the – positively – charged Earth. area then Removing electrons to electrons the can original distribute move charged themselves on into rod the the will object then object from cause leaving the it – the excess – – charged. – This The process same is referred effect can some uncharged move slightly to be chalk as charging seen when dust. The a by charged electrons in rod the is placed dust are near able to to c one side. and This cause means the that dust the particles dust to particles become become charged attracted on to The rod with an is removed. opposite The sphere charge to the is left rod. the Figure charged – induction 4.1.3 Charging by induction rod. ACTIVITY To investigate small pieces the and effect place of induced them on a charge desk. cut paper Charge up a into KEY very plastic rod 1 using a cloth and place the cloth near the POINTS Atoms charge Describe Explain what why happens the to charged the rod pieces affects of the have no overall pieces. paper. an uncharged pieces equal and of because they number of contain protons electrons. paper. 2 Electrons between can be objects transferred by frictional forces. SUMMARY QUESTIONS 3 1 A a student door an 2 handle. electric Dust walks along A shock. particles equipment are such tiny a nylon spark Explain is television and produced why uncharged as carpet this but then and reaches the out student to attract feels they often Why charged each similarly happens. screens. Oppositely other charged objects while objects repel. stick does to electrical this happen? 4 Charges neutral can be induced in objects. 107 4.2 Electric the fields Van de and Graaff generator Electric LEARNING An At the end fields OUTCOMES of this topic electric field experience should be able describe an electric field effect on field charged explain how generator a Van region of space in which a charged the in direction the field of force (Figures acting 4.2.1 on and a positively 4.2.2). particle experiences how protected buildings from A charged negatively a force in the opposite direction. charges To explain will Graaff Van de Graaff generator + object particles de separates shows placed charged a force. and particle its a to: The is you – are damage Glass dish by lightning. Castor oil sprinkled Conductors with semolina powder a Figure + 4.2.2 Demonstrating an electric field. The particles line up in the direction + + + of the field. Charged + + Lines metal of + + force sphere + The Van de Graaff generator + + + Rubbing charge. a rod To with a produce a cloth can larger only separate build-up of a charge small a amount Van de of Graaff b (VdG) A generator large, hollow, can be metal used dome (Figure is 4.2.3). insulated from the ground by a + plastic tower. Inside the tower passes over As belt two a rubber rollers belt made is of moved by different an electric plastic motor and materials. c the from the negatively Positively comb Figure 4.2.1 Electric fields charged near sphere, b a a the c a point negative charge point the the lower belt roller to the electrons roller, are making transferred the roller charged. are ions trapped surface of are on the attracted the belt belt to as it positively the roller moves from the upwards. metal This leaves charged. At the top of the generator the positively charged belt draws and electrons charge dome As Some The from rubber and belt of effectiveness on through of charged rotates, charge the conditions. water dome positively positive effects weather away the becomes the dome 108 of a positive over part charged but outer passes inner VdG the In more can VdG in by second comb. The induction. electrons build metal up are on removed the surface from of the the dome. generator generator humid molecules the depends conditions the air. the a great charges deal can be on the carried move + + from ACTIVITY + Electrons + to the + dome rubber Perspex + through Metal roller a receives Your and stores comb + + a charge + + + + + teacher may demonstrate positive VdG generator. + metal dome + belt + + + + Place your hand on the dome. + Electrons + + + will be stripped + + + from your body, leaving you + Insulating column positively charged. The hairs + + on your head will repel each + Rotating rubber belt other and apart as try to move as far + + possible. This means + that they will stand on end. + + Once you have been charged + Motor up, touch another student. + + They + + Positive the ions roller are and drawn trapped an towards on the rubber Polythene belt A Van de Graaff may shock. feel Electrons roller their 4.2.3 you) electric from Figure (and the Earth body pass and into through you. This generator movement called a of electrons is current. Lightning Lightning is a very large spark. Clouds can become highly charged Observe the attaching frictional forces acting inside them. Droplets of water and ice against each other and charges are separated. Eventually separation is so large that a lightning bolt is of bolt is often a flow of electrons to earth. A very a close visible current is produced and this heats the air around it a bright white glow. The air is heated so quickly that a thunder, is a to it. Lightning the The in pointed likely up in that have end the Sometimes current two the charges thick are placed of the on tops conductor building, to will lightning cause metal much KEY 1 to of tall buildings, to across but a fire cables leak be will or or allows away induced into the air charge, pass it through strike even bars an the building explosion. that allow the anyway. Lightning have charged objects. electric The to earth safely without current generating from 4.2.4 Very too When an large charge electrical fields around them which affect Graaff generator electric can be used to investigate large electric amounts can in be the of created by atmosphere. QUESTIONS other Why does lightning strike church towers? the charge. 2 3 high large sometimes of in point. conductors SUMMARY Charges effects a less forces de up struck. heat. Van at which making POINTS A curved build protect 1 2 is evenly ways: Figure lightning are produced. building the could may This lightning conductors building builds from You spark. concentrations Protection generator conducting sound surface shockwave, VdG rapidly spread causing the large because electric of to produced. see lightning the bringing object A point the and charge a crystals dome brush effect by charges current. build up they may eventually discharge as Why shouldn’t under a tree in you a stand lightning storm? 109 4.3 Uses and static Dangers LEARNING the end of this topic be have able describe static electricity already a associated seen resulting that in the build-up lightning, for of large example. charges Even can small be sparks or to: currents electricity you dangerous, should of OUTCOMES You At of dangers range with of can sometimes be dangerous or damaging. dangers Refuelling static explosions electricity Large describe static a range of uses for Air electricity. electrical resistance (Figure Car tyres can a explosion. an Aircraft charge can as build they up fly. spikes designed the on to the help build-up of can build frictional a small are in explosion. car up on forces vehicles cause when the they transfer move. of electrons prevent allow charge to can as in even flour the spark can have car’s can ignite been metal be body. If produced. petrol vapour designed to enough When and help cause reduce an this charge. the aircraft’s are to a spark tyres of tanker the car refuelled, the To charge the build-up aircraft this cause tanks build-up connected dissipate moving fuel fuel and of could a charge together by cause spark a a could the build- cause aircraft conducting and cable to safely. explosions when there is dry powder in the air mills. static These Damage thin on refuelling such 4.3.1 car, charge Sparks Figure up Modern dangerous of transfer builds refuelling up other 4.3.1). charge When charges or wing to electronics are reduce Microprocessors and other integrated circuits are extremely sensitive static to static electricity and can be ruined if there is a build-up of charge. electricity. Uses Static of static electricity pollution and electricity can in also spray be useful painting in photocopiers, in reducing air surfaces. Photocopiers Photocopiers on Figure 4.3.2 This is electronic connected through a paper A rotating A bright The The the Earth wristband as a computer. build-up discharge lead the 110 instead delicate is light negative reflected in of static charge to produce images is negatively shone is onto reflected charged an image onto the by a brush. on a sheet charged of paper. drum. can The escape areas from that do the not drum in receive areas light that receive remain the damaging circuitry. charged. This means that the charges on the drum are body through of charge light. Any his arranged will control he negatively static drum light bright careful 4.3.3). engineer to the repairs use (Figure paper. in the same pattern as the image on the original sheet of A positively powder is charged attracted uncharged ‘toner’ to the powder is negatively applied charged to the areas drum. but This not to the areas. Grid of charged A negatively charged piece of paper is pressed against the rotating wires drum. The The toner paper is permanent remaining heated copy of and the on the the drum toner original melts is attracted onto it to the creating paper. a image. Earthed metal plate Original, face Glass down plate High intensity light source Figure Charging Powder 4.3.4 Electrostatic precipitator unit unit Drum Heater Paper feed Paper out Light Charging Powder unit unit Paper out SUMMARY 1 What to a QUESTIONS charge car body negatively Figure 4.3.3 A 2 to printers create the operate image in on a similar the way. drum, a Instead laser of beam using draws reflected a pattern any image can be Why charged phone light will in a What type be stations chimney. to collect and past earthed To produce prevent the the soot soot this grid plates that would pollution particles. metal metal As they on the a the gain side normally charged soot the metal particles positive of escape charge chimney. grid rise can up and The through collected. be the improve it is This the used for are soot process is called electrostatic in a cell station? to the make the electronic Figure 4.3.2? used attracted can to KEY POINTS then Static electricity can damage precipitation components cause explosions. quality of painted surfaces the paint can be Static electricity and The surface is oppositely charged and so the whole surface. This gives a very even is used in charged the paint coating of painting and is reducing to use painting sprayed. attracted evenly? materials photocopying, as it chimney 2 T o you petrol of electronic Improving to the 1 be paint pollution engineer Power given that created. wristband air stick shouldn’t should Reducing be ensure so 3 that to photocopier droplets Laser should pollution. paint. 111 4.4 Electric Current LEARNING the end of this topic be able an terms charge describe metal electric the a of electric potential current in Defining current in a current in an The size in unit a the of a current called the is the and 1 What one of charge coulomb is 1 the current of (A). flow The of flow charge relationship Current Electrical circuit a the a = of charge is caused and is this is normally measured written as: × measured time using or a Q = unit It called the coulomb (A s). a wire if a charge of 360 C is transferred in It = 360 C = = charges in move is circuit around used the to All the of the circuits transfer current components. circuit. 6.0 A 60 s circuits current and energy EXAM in simple wires current ampere second in t In = are = Q The is a as in flow is of pass current. to used the In an electrical electrons not complete they a energy current electrons components up through as whole through electrical devices. it metal travels journey around releasing them. TIP Conventional impo rtant to that curren t positiv e to when circu its. conv entio nal trave ls current alwa ys Conventional from nega tive charge. In charges. moving This in current circuits the means the is described energy that opposite the is in terms actually particles direction of a flow carried carrying to the of positive by a of positive flow charge conventional of are negative actually current. discus sing Conductors Metals ‘sea’ easy are of for Most called insulators electrons charges other carriers and composed free conductors 112 This points. minute? I even two EXAMPLE Q state rate ampere transferred WORKED is current. current Quantities solution. (C) It a wire describe ionic is between transfer charge charge difference to: describe of flow you by should charge OUTCOMES The At and current and of a lattice move can freely move ions about. through surrounded This metals so means they by that are a it is good electricity. solid so to of which it materials is insulators. do difficult not for a have free current to electrons fl ow. or These other charge materials are Semiconductors Some can materials conduct lie between under semiconductors. conduct such one as can be the Silicon altered germanium. direction enough. This Current and in is if a extremes atoms of semiconductors the potential in diodes of conductivity. These semiconductor adding used ionic two circumstances. is by Some only effect the right and other are its They classed ability as to semiconductors made difference and are to conduct across it is in high transistors. solutions ACTIVITY When sodium chloride is dissolved in water an ionic solution is Test a range of materials to + formed. This contains two different types of ions: Na and Cl see If electrodes both of these difference The seen ions and sodium on the two can and flow connected because of to the a battery, potential electrodes. positively When and reach This or if they and the rapidly hydrogen so are attracted electrode reacts gas and they with so the to simple lamp ions and process solution. or can an use a ammeter the obtain water bubbles to can be are negatively When form they chlorine charged reach the and are attracted electrode they to lose the an gas. is called electrolysis and can be carried out on scale to produce Electrolysis of useful other chlorine ionic gas solutions and can sodium produce the material in the circuit an to industrial conductors You 4.4.1). Place This are insulators. (Figure charged they sodium. hydroxide electrode. electron solution particles are form the electrode. chlorine positive in the electrode. electrons The charged sodium form placed between negative are find out if it can conduct electricity hydroxide other useful Figure 4.4.1 A simple conduction test chemicals. SUMMARY QUESTIONS 1 complete Copy and T able 4.4.1 relating current, charge and time. T able 4.4.1 Current/A 2.5 0.05 KEY Charge/C 100 Time/s POINTS 0.2 40 300 1 The current caused 2 A current How 3 of much Molten 150 mA charge electrolytes flows has through been can also a circuit for 4 from minutes. a transferred? conduct electricity. Molten lead 2 a by a in a flow negative positive metal of wire is electrons terminal to terminal. Conventional current is a 2+ bromide when contains electrodes connected to a Pb are and placed battery. Br in ions. Explain molten lead what happens bromide and flow a of positive positive negative charge terminal to from a terminal. 113 4.5 Electrical energy and transformations Potential LEARNING Potential At the end of this topic be able charged calculate the is will the cause move to a of the movement position where it of will charge. have less energy and so will move to a ‘lower’ point in an electric field. potential This difference particle to: potential difference you A should difference OUTCOMES between is a little like a mass in a gravitational field. As the mass falls two through the gravitational field it loses potential energy. The energy points loss calculate the on the distance moved through the field, the strength energy of transferred depends by an the field and the size of the mass. In electricity the energy change electrical depends on the size of the charge and the potential difference it device. moves through. Potential difference is therefore defined energy potential difference by the equation: transferred = E or V = charge WORKED How EXAMPLE much through a energy is potential Q 1 transferred difference when of a charge of 3.2 C passes 5.0 V? E V = Q E In = VQ circuits higher the 5.0 V component measured EXAM is in cal measu red watt way is. 16 J of force by an are battery and and the power force power called the source voltage referred to is across difference simply an at as the electrical (p.d.). Both are voltages. (W) as One powe in in r the the is Just unit same mecha nica l watt is one as device the in is mechanical the rate definitions of at energy which it potential powe r transfer, transfers difference the power energy. and current E V = of This an can as electrical be linked follows: E and Q = It, therefore V = joule It second . E IV = E and, as power is defined t Electrical 114 is power Q per supply electromotive potential often or terminal. electrical (e.m.f.) called and a negative difference usually volts = TIP Electrical Electri the produced electromotive 3.2 C terminal than potential voltage × positive potential Voltage, The = power is measured by P = , t in watts (W). P = IV to WORKED How with EXAMPLE much a P = IV = 0.5 A E = Pt = 3.0 W Electrical Electrical some transferred × 6.0 V × 20 s = = of by a 6.0 V bulb and a in 20 s if current it of operates 0.5 A? 3.0 W 60 J transformations energy Electrical can motors a Electricity wide can fluorescent is difference be transformed in many useful ways. Here are examples: allowing energy potential 2 transform range be of electrical objects converted into to energy into kinetic energy, move. light using filament bulbs, LEDs or tubes. Loudspeakers Conservation convert of electrical energy to sound. energy Figure 4.5.1 Light or Electrical energy is very useful as it can be transferred and emitting fluorescent but most of the production of electricity relies on more so fuels. These reducing resources using their by fuels use using alternative are is only available important. more efficient sources of We in can electrical energy where limited quantities conserve efficient than burning filament fossil are transformed much easily diodes tubes some of bulbs. and these devices (Figure 4.5.1) possible (Figure 4.5.2). or by ACTIVITY Use a voltmeter potential power and difference an ammeter across a to lamp. measure Use your the current readings to in and find its rating. Figure 4.5.2 Electricity using can be generated photovoltaic solar cells. SUMMARY 1 What a 2 is one of charge 4.0 W potential 40 mC 1 12.4 A of an and is and electrical potential transferred operating device which difference when voltage a of of torch operates with 12 V? with 6.0 V a power operates for minute? What KEY power of much rating 3 the current How QUESTIONS with difference 10 J of is required to provide a charge of energy? POINTS The potential difference across a component is defined by E V = Q 2 The power of an electrical device is given by P = IV 115 4.6 Simple circuits and components A LEARNING a At the end should be simple circuit of this able topic battery or draw simple containing power of a source supply), of electromotive connecting leads and force some (a cell, electrical to: circuit a a components. you Circuit consists OUTCOMES range diagrams diagrams of Drawing realistic diagrams of the components is not necessary and components can interpret circuit diagrams them to construct confusing, so a standard set of symbols is used. and use be The same symbols are used around the world allowing all engineers real to understand them. circuits explain why a standard set A circuit diagram components symbols is used for 4.6.1 Component Standard very the important connections between clearly. circuits. The T able shows of electrical Symbol key circuit are shown in T able 4.6.1. symbols Use earth symbols point Component of lowest wires Symbol passing Use indicates potential wires or that are not connected together cell provides an lamp or bulb transforms electromotive electrical energy or force battery in the provides circuit an into voltmeter light measures electromotive the force difference in the circuit potential between two points d.c. power provides supply + an ammeter measures electromotive current force point at the that – in the circuit in the circuit a.c. power supply provides a galvanometer small force in the connects circuit or disconnects semiconductor allows diode only components junction indicates wires 116 that are very currents or electromotive switch measures changing of electrolytic cell current in the direction arrow produces a precise connected electromotive together force Component Symbol Use fuse Component melts off and the cuts circuit Symbol Use loudspeaker transforms if electrical energy or the current is too into sound large fixed resistor reduces the transformer changes current in a of an branch of a current the p.d. alternating or circuit variable resistor a resistor value can manually electric motor whose generator transforms GEN be energy altered kinetic into electrical energy transforms electrical into energy kinetic energy ACTIVITY Draw a SUMMARY circuit diagram for the two circuits in Figure 4.6.1. 1 Draw the diagram shown Figure draw Start Use any correct for in the circuit two Figure circuits 4.6.2. 4.6.1 Figure To QUESTIONS a by a circuit drawing ruler other Work clearly to the draw straight around the follow cell, all of these battery the simple or power connecting 4.6.2 rules: 2 supply. leads and wires A student has drawn diagram poorly Describe and mistakes the a (Figure circuit 4.6.3). and correct the lines. circuit in a logical order. Start from student has the made. positive Draw one terminal If the If any on mark using should at a time working towards the negative battery. has moving circuit There KEY 1 the necessary the component of circuit before terminal. be branches to the the direction small no then complete one branch conventional current next. of the on arrows. gaps in the Figure 4.6.3 circuit. POINTS Circuit diagrams show the connections between components clearly. 2 Standard symbols must be used for circuit diagrams. 117 4.7 Series and parallel circuits There LEARNING are (Figure At the should end be current of this able describe two categories of circuits: series and parallel OUTCOMES the in topic 4.7.1). behaviour you provided to: behaviour series and of to To analyse the the current these in the circuits circuit we need and the to understand changes in the energy components. of parallel Series circuits circuits Series describe the difference parallel potential in series circuits pathway are the connecting simplest the type positive of and circuit. There negative is only one terminals. and Understanding circuits. In of the circuit electrons, current the current moving in is series circuits composed through the of wires. a flow The of charge, electrons in pass the form through a each component They can also cannot enter This only and stay at means in the that components in turn release in a they of cannot the component. same the but some The created they same or carry. destroyed. The number electrons has to leave all of as rate. current series be energy with must each be exactly the same in the other. b Understanding The or source a power used but through. circuit 4.7.1 Circuit bulbs b a in shows series shows parallel a and an this principle passing When they In is transfer in force circuit in the description the through the difference same the electrons some of cell for all gain move the we series will can be assume a a cell, cell is a battery being sources. potential through energy circuits a they energy as component gained they in when pass the passing two resistor with electromotive supply. the Electrons Figure of potential circuit in electric through the electrons started cell. will with, After have the before moving same they through amount passed of all of the components potential through the energy as the they first cell. motor. As energy the as same they gained difference EXAM Appl y TIP the anal ysis stag e you a at will wide force a rules of of be able conserved the around when cell. the This the they means circuit electrons pass must have through that the match sum the to the of lose exactly components the potential electromotive circuits one and to be energy cell. Parallel circuits have branches where the current can divide and re-join. so lve Understanding of of from drops the Parallel rang e always circu it caref ully, time, must amount current in parallel circuits circu it problem s. The of current that junction. junction must junction. 118 into They any junction This be the can’t is must because same stay at as the the the exactly rate rate of junction. match of the electrons electrons current out entering leaving the the If current divides recombine or other to when power potential Electrons as What the difference is circuit current shown each have different the current in to lose the leaving must the before current entering parallel branch path then current cell, it. circuits be same they will the the same. amount of energy re-join. 1 and in match The difference path a EXAMPLE the to branches re-join. across one following WORKED has potential following electrons different branches supply, Understanding The follow the potential Figure difference across the bulbs in 4.7.2? 12.0 V The potential branches difference must electromotive be the force across same of the as the the battery. E 4.0 A This means across there bulbs D is a and The current from The current into p.d. of 12.0 V E. the battery bulb D is 4.0 A. is 3.0 A and E must be 3.0 A D so the current into bulb Figure SUMMARY QUESTIONS 1 of 4.7.2 1.0 A. Sketch all circuits bulbs. More A complex circuit does not combinations to the of circuits have both. appropriate to be The parts and purely rules of the series for or parallel parallel. and Many series are circuits 2 apply Find Z in can Include the Figure equal involve five parallel circuits. current across possible series, combined p.d. circuit. that the the in and the resistors 4.7.4. X and Y Y and are resistances. 8.0 V WORKED The circuit series each The EXAMPLE shown elements. of the This Figure Find the 4.7.3 current contains and both potential parallel and difference across 2.0 A from the means X 0.5 A bulbs. current 2.0 A. in 2 battery that the Y W is 12.0 V Z current 1.5 V through so must bulb the P must current be 2.0 A through and 2.0 A Figure bulb 4.7.4 Q S. The total current Q and bulb R through bulb 1.5 A so the must current be 2.0 A through R and must S P be R KEY 2.0 A The 1.5 A p.d. same is − as = across the 0.5 A. bulb p.d. Q must across bulb be R 4.0 V the so it Figure 1 This total 4.7.3 of p.d. means 12.0 V − The is 4.0 V . The POINTS 5.0 V around that 5.0 V the − the p.d. 4.0 V circuit across = 3.0 V . must bulb S be 12.0 V . must be 2 current equal to into the any junction current out it. The potential around circuit any is difference closed loop in a zero. 119 4.8 Cells Chemical LEARNING the end of this topic be electromotive able describe how cells by force chemical (e.m.f.) required reactions to between drive acids a current and can metals. be Each cell two produce or electrodes: the positive and the negative. are restricted in the size of the current they can provide by their an internal electromotive terminals and Cells batteries cells to: has in you produced should reactions batteries OUTCOMES The At and resistances which are due to their physical and chemical force composition. distinguish between primary A and secondary battery other. draw a diagram to a secondary The simply e.m.f. a of collection the of battery cells will placed be the in sum series of with the each e.m.f.s of the show cells, how is cells cell can assuming all of the cells are placed the same way around. be recharged. Primary In a primary cannot Metal (positive place, cap cell Insulator rod be the cell the reversed. cell chemical This cannot reaction means be that, recharged which once and the produces reaction reused. There the has are e.m.f. taken several contact) types Carbon cells and of primary (Figure cells. One common example is the zinc–carbon dry 4.8.1). seal (+) In Positive Zinc (–) a zinc–carbon cell a metal case of zinc acts as the negative case + electrode and (outer current card metal or electrode a carbon rod acts as the positive electrode. A reaction jacket occurs collector and between the zinc and carbon and an ammonium chloride removed) paste Paste and this reaction produces an e.m.f. of 1.5 V initially. of ammonium chloride Zinc–carbon cannot cells provide have large a relatively currents, high the internal maximum resistance being and around so 1 A. they This solution means that they are not suitable for providing energy to high power (electrolyte) devices. Negative – Paper contact separator Manganese (IV) dioxide (reduces the internal Chemical they 4.8.1 A reactions not zinc–carbon cell become replaced A a but The car a in most battery is a that zinc–carbon the cells cells even discharge when over It can be can also corrode inside time equipment if not In the cell an so cells a the can cell be of e.m.f. have a be reaction 2 V . low forced cell (positive 12 V internal to a is to produce reverse to the by cell. an applying This means repeatedly. is the between Inside of reaction direction recharged e.m.f. very can opposite plates of chemical secondary the oxide total on reaction in type this lead relies lead–acid lead plates electrode) car battery and six cell used recharged. currents required to power a in (negative dilute cells are sulfuric placed produced. resistance and can provide the secondary large cell. time and and Lead–acid car also common produces series They difference batteries. acid cell secondary electrode) 120 inside means cells this potential that A place This useless. secondary e.m.f. 4.8.2 take use. regularly. Secondary Figure in resistance) and Figure are starter motor. They also contain significant sulfuric quantities acid WORKED A car to P = I = lead and so are very heavy and have corrosive EXAMPLE battery required of inside. produces turn a an starter e.m.f. motor of 12 V . with a What power size current rating of is 4.8 kW? IV 3 P 4.8 × Recharging single battery 400 A 12 V secondary lead–acid with W = V A 10 = an cell cells can e.m.f. be recharged greater than using 4.0 V . This a power applied supply e.m.f. is or a greater Cell 2.0 V being recharged than the reaction A car e.m.f. to the happen battery This provides and so the of is a in cell to the reverse recharged reverse battery by be an e.m.f. remains recharged to fully and direction (Figure alternator the will driven battery while force the chemical 4.8.3). by the the car car is engine. in motion charged. Recharging power 4.0 V supply ACTIVITY Figure 4.8.3 Recharging cell. Use a lead–acid condition of equipment cell the to to power plates measure over the a low several current voltage hours and bulb. and e.m.f. Monitor use data provided the electrode logging by the Note cell. supply Recharge the cell the cell behave using in the a power same supply and repeat the test. Does to travel cell SUMMARY 1 Draw a A table be KEY 1 an the the in the to negative cell is negative power the current opposite that when the discharging. summarising the characteristics of primary and cells. zinc–carbon with to of the the QUESTIONS secondary 2 is lead–acid forcing direction way? of connected electrode a how e.m.f. provided battery of by provides 1.5 V . this What is a maximum the current maximum of power 1.2 A that can cell? POINTS Cells produce an electromotive force through a chemical reaction. 2 Primary cells use irreversible reactions and cannot be recharged. 3 Secondary cells use reversible reactions and can be recharged. 121 4.9 Resistance Using LEARNING electrical Electrical At the end of this topic meters be able use an ammeter calculate used to meters measure must be both placed current and carefully in potential order to operate (Figure 4.9.1). An ammeter is used to measure the current at a point in a circuit. correctly The the and voltmeter but to: correctly are you difference should meters OUTCOMES the resistance of current has to pass through the ammeter for its magnitude a to be measured and so the ammeter needs to be placed in series component with state Ohm’s conditions law and under could the which the other reduce components. the current Placing and so the ammeter ammeters need in to this way have very low resistances. it applies. A voltmeter in it the is to measures circuit. It measuring have very through has the high to the be potential placed potential resistance in difference parallel difference so that across. almost between with no the two points components Voltmeters current will need pass them. Resistance In 4.8 it was mentioned restricts the current. Metals high The the current in wires that them. have cells All low have an resistance resistance current of a component through is while defined Resistances an ammeter voltmeter current to are and a have very as the ratio of the p.d. to or R = measured in a I unit called the ohm (Ω). the EXAMPLE 1 bulb. What is current the of resistance 2.5 A V R = Ohmic A was The of a a length p.d. of of metal 3.0 V is wire across if there is a it? 3.0 V = 1.2 Ω 2.5 A conductors simple physical when = I metal wire properties discovered current potential by will and have Georg through difference a a constant conditions Ohm conductor across do and is so ∝ resistance not is change. called directly it I 122 rods a p.d. WORKED across, glass to and measure through, which resistance V = current Using a it: p.d. 4.9.1 resistance have resistance. resistance Figure internal components V as long This Ohm’s as its relationship law: proportional to the The constant which obey of proportionality Ohm’s conductors. For some remain constant classed as Resistor In with a This to resistance see in of resistance) the 4.10) the are wire. called resistance and their Materials ohmic does not behaviour is sometimes difference other resistors EXAMPLE are used to limit components usually small the maximum from being devices size of damaged placed in by series of it is 6.0 V . to 2 current when diode is components. maximum 0.2 A, the the components will prevents The other WORKED The you resistor currents. the other is constant components current. large (as (have ‘non-ohmic’. circuits the law ACTIVITY through operated What ensure that a by resistor the diode a needs battery should current be can to be supplying placed never be restricted Build a variable in potential series greater with a simple brightness than circuit resistor of a so using that bulb a the can be adjusted. 0.2 A? V R = 6.0 V = = I Variable Variable the resistors resistors circuit altered 30 Ω 0.2 A by can (Figure also 4.9.2). turning a dial be The or used to adjust resistance moving a of the the sliding current variable contact. in a branch resistor In can of be laboratories Fixed variable Other A are often called dependent the light level resistor it is (LDR) exposed to. has a LDRs resistance have low which varies resistance to light build when Variable resistor Light-dependent it and light gets Temperature very high sensors and resistance switch on in low light. lighting They systems can Thermistor be 4.9.2 used automatically dark. sensitive resistance changes to heating resistors depending are on called the thermistors temperature. and They their can be used KEY control resistor in Figure bright resistor rheostats. resistors light with resistors or cooling POINTS systems. 1 The resistance component is of a defined by the V equation SUMMARY What = I 2 1 R QUESTIONS current will there be in a 12 V bulb with a resistance Ohmic conductors have a of fixed resistance while the 1.5 Ω? resistance 2 Explain how brightness resistance a of variable a and bulb resistor using current. the can be ideas used of to adjust potential the difference, of conductors to factors non-ohmic changes like light due or temperature. 123 4.10 Investigating and current potential difference characteristics Investigating LEARNING To At the end should be describe the the resistance of components OUTCOMES of this able investigate the behaviour of different components we measure the you current in the component For investigation when different voltages are applied to it. to: how current topic to and this variable measure is the used circuit to shown control the in Figure current 4.10.1 in the is used. The wire. voltage Once characteristics resistor of a range the data is collected a current–voltage (or I V) graph can be of drawn and this reveals the characteristic of the resistance. The shape components of describe metallic lamps, the characteristics conductors, filament semiconductor the ionic is referred to as the I V characteristic of the component. Metal wires diodes As and graph of shown in 4.9 a metal wire is an ohmic conductor. We can see solutions. evidence for produced wire by this in the graph demonstrating finding the (Figure that gradient I ∝ of gradient = to graph the can Test also shows resistance. travel Filament just This as ∴ the is we as in R the is resistance of the gradient direction would either find line = V that freely can straight 1 = Δx The We A graph: I Δy Cell V. this 4.10.2). of the expect current because is not the relevant electrons direction. lamps component Filament gas. does not constant Figure 4.10.1 Finding the voltage characteristics are there is increase just as simply a very small significantly for an thin current ohmic metal in and the so wires kept filament the inside its resistance an of the filament conductor. of the current increasing electrons line to to increases V +I is The the travel curve relationship sufficiently it will cause the wire to heat resistance of through hot below I required ∝ to V that no Semiconductor for for an longer increase characteristic a a the the wire wire. ohmic holds, as as This it is more causes conductor a difficult the (Figure progressively for the characteristic 4.10.3). larger The increase in current. filament lamp is symmetrical. diodes +V V The conduction from that in and the mechanism metals characteristic and graph, currents as is in beyond shown involved semiconductor are in the scope Figure usually diodes of 4.10.4, very small this has so a is very course. an different The unusual recommended When 4.10.2 a for negative the investigation. voltage is applied the diode has a very large Current–voltage resistance characteristic conductor of an and therefore there is a very small current, ohmic microamps. The diode is said to be ‘reverse biased’. I V shape microammeter – I 124 is component up, Figure inert temperature current– When a lamps When a few is In the with forward almost bias no direction current at the all for diode has voltages an up even to higher 0.7 V . resistance Beyond this Current value +I voltage the diode becomes a much better conductor allowing for larger ohmic onductor currents Ionic through. solutions +V V To investigate need to be the I placed V characteristics into the solution of a ionic fixed solutions distance two electrodes Lower value apart. by The resulting ohmic graph conductor. shows that However, the the ionic solution resistance is behaves large when as current caused increasing R an compared – I to metal Instead wires of and it electrons depends there are on the two concentration different charge of the solution. carriers, for Figure 4.10.3 Current–voltage 2+ example, copper ions (Cu ) moving to the cathode and sulfate ions characteristic of a filament 2− (SO ) moving to the anode. lamp 4 ACTIVITY +I /mA Use an the method ionic described solution resistance of the over an to investigate extended solution the period of characteristics time. How of does Near ohmic the Forward change? bias V +V 0.7 V KEY 1 POINTS Ohmic conductors (wires and ionic solutions) have constant – I / μA resistances. 2 The resistance of filament lamps increases at high currents Figure as 4.10.4 Current–voltage characteristic the temperature of the wire of semiconductor 3 Semiconductor threshold diodes voltage and SUMMARY QUESTION 1 A class of students graphs of the T able have low very and high resistance investigated current a increases. a voltage resistance beyond range of below diode a it. components characteristics and collecting identify the the data shown in T able 4.10.1. Plot component. 4.10.1 Component Voltage A / V Current −5 −4 −3 −2 −1 0 1 2 3 4 5 −0.8 −0.76 −0.69 −0.48 −0.24 0 0.24 0.48 0.69 0.76 0.8 −1 −0.8 −0.6 −0.4 −0.2 0 0.2 0.4 0.6 0.8 1 −0.01 −0.01 −0.01 −0.01 −0.01 0 0 0 0.2 2.1 4.2 /A Component Voltage B / V Current /mA 125 4.11 Circuit Most LEARNING the end should be of this able topic you resistive find in the series resistances find the resistor resistance and of to fully in of components analyse series the and usually parallel. We including can groups combine the circuit. combinations resistors parallel power output of need is often to find more the than total one source resistance of of resistance in a circuit and we combinations. combinations. Resistors The R R 1 R 2 3 total their =R S +R 1 would +R 2 series resistance a wire expect of components resistances. with the 5 Ω This is resistance complete wire in series exactly to to a is as wire have a simply you the would with 6 Ω resistance sum of expect. If resistance of you you 11 Ω 3 More 4.11.1 in respective connect R Figure range to: There a components Resistor contain OUTCOMES of At circuits analysis Resistors in formally this is written as: series R = R s Resistors Finding R in the + R 1 + R 2 + … 3 parallel total resistance of resistors in parallel is a little more 1 complicated. R voltage Using acting the across fact that them, this the resistors equation all can have be the same derived: 2 1 1 1 1 R = 3 + R R P 1 1 = R Figure 1 + R P 4.11.2 + … R 2 3 1 Power R 2 Resistors and resistance 3 in parallel To find find the the the power power electrical the The the a each car’s What 1 element with the R a total 120 = a the car The it of resistors resistors is usually current rear of and 1 15 Ω window 120 Ω. strips + 120 8 of 1 120 = and resistance + P However, resistance P R combination individual and much use we then could use easier these two the to find values to 1 battery. 1 = a output. 12 V is of the equation. EXAMPLE heating strips of resistance power WORKED output output power equivalent find 126 R 1 + R 1 + are of the 120 rear heater in window 1 + 120 contains connected 1 + The using heater? 1 + 120 metallic parallel. 1 + 120 eight operates 1 + 120 120 b What Find is the the power current of the through heater? the heating element using the total resistance: V I 12 V = = = 0.8 A SUMMARY R Finally find the power of the heater: 1 Find the three P = QUESTIONS 15 Ω IV = 0.8 A × 12 V = resistance resistor of the arrangements 9.6 W shown in Figure 4.11.4. a 20 Ω Analysing circuits 20 Ω Using the resistance equations we can analyse complex circuits. 20 Ω This b worked example will lead you through one of these analyses. 40 Ω 60 Ω 40 Ω WORKED EXAMPLE 60 Ω 2 c Analyse this circuit, shown 6.0 V in Figure currents 4.11.3, I and to the find the 100 Ω 100 Ω potential 1 differences V and 200 Ω V 1 2 400 Ω First find the total resistance 100 Ω I of the circuit. This finding the parallel section means resistance (R of 1 the Figure 4.11.4 ). 40 Ω P 2 Find the current in each of 5 Ω 1 1 1 = 3 + R 40 the = 20 two parallel resistors in 20 Ω 40 Worked P example 2. V 2 V 40 R 1 = = 13.3 Ω P 3 The total Figure resistance (R ) of 4.11.3 the KEY POINTS T circuit is given by: 1 R = R T + 5 Ω = The resistance components Now of 18.3 Ω P find I , the total current from the battery, using the the and the total series is battery 1 voltage in sum of the individual resistance: resistances V 6 V R = R s I = = = + 1 R + … 2 0.33 A 1 R 18.3 Ω 2 T We can find V because we know the current through the resistor The resistance components of in parallel is 2 and its resistance: V = 2 I R = given 0.33 A × 5 Ω = 1.65 V 1 by 1 we know that the total voltage drop around the circuit R 1 be 6.0 V and so the voltage V can be + R P must equation 1 = Finally the + … R 1 2 found: 1 3 V = 6.0 V – 1.65 V = could resistors power output combination 1 We The continue using their to find of a 4.35 V the resistances current and through the voltage each of across the parallel them. sum the of the of resistors power individual is outputs the of resistors. 127 Section SECTION 1 A plastic they to 4: Practice rod both have a 4 is Practice exam rubbed become positive questions with charged. a dry The is until 7 found What is the What cell are and the a charge on the Describe, how the in terms rod between a primary cell? Why must and of particle cloth a voltmeter have very high cloth? resistance b differences secondary charge. 8 a questions 1 cloth rod exam have movement, low while an ammeter must have very resistance? become 9 A filament lamp operates with a potential charged. difference 2 Sometimes, tumble and when drier, tiny a clothing crackling sparks can be is taken sound seen. out can Why be of a lamp heard does into of 12.0 V transforms heat and across 4000 J light of energy its terminals. electrical in one energy minute. this a What is the power rating b What is the current c What is the resistance of the lamp? happen? 3 Describe how principles accurate 4 Copy of a static copies and electrical photocopier of electricity an complete circuit uses to table reproduce showing symbols. Component Symbol Component lamp semiconductor diode battery 5 The dome charged The to a dome earth of is What What 0.02 6 A 6.0 V the is 128 particles generator charge discharged average by of is 0.01 C. connecting it wire. travel current discharge in the wire if the in the wire process takes What is connected to a resistor of 5 Ω much resistor b total discharge? battery How Graaff a grounding of the the de holds second? resistance a a type during Van it then with during b a until is in charge one the passes though the minute? power output of in the lamp? the image. this the The resistor? Symbol of the lamp? 1 10 A student investigates characteristics of data are gathered Voltage/V Draw this b c a the current–voltage semiconductor shown in the diode. The table. −0.3 −0.2 −0.1 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0 0 0 0 0 0 0 0.01 0.1 0.2 0.3 Current/mA a a diagram of a circuit suitable for 13 investigation. Plot a graph Describe the of Three resistors connected the results. results a shown in the Draw Magnesium chloride dissolves in graph. water to solution containing magnesium (Mg ) a of 2.0 V circuit circuit resistance cell in diagram of the with a showing resistors the 66 Ω variety be ways. the when highest can of they resistance. form b 2+ a each a arrangement form 11 a to Calculate the current the total through one of the and resistors. chloride (Cl ) ions. c a Describe the process which allows Calculate power of the resistor this combination. solution to conduct. d b What is meant by an ohmic Repeat with c Is the solution an ohmic Calculate the resistance of the b and lowest c for the configuration resistance. conductor? 14 12 a, conductor? these three Determine the readings on the four ammeters resistor and two voltmeters in this circuit. combinations: a 8.0 V 50 Ω 20 Ω 40 Ω b 40 Ω A A 1 4 40 Ω 66 Ω A 10 Ω 2 c 66 Ω A 40 Ω 3 33 Ω 33 Ω V 1 V 2 33 Ω 129 4.12 Direct current alternating Direct LEARNING the end of this topic be and able compare direct alternating batteries only in one current identify (d.c.). positive to This is a negative) current and has that a magnitude. peak voltage of an current (a.c.) is used in mains supplies. The direction of the alternating current using current and Alternating current current (from current the frequency direct and Alternating produce direction to: fixed current you travels should current OUTCOMES Cells At and an reverses rapidly, many times each second, and the magnitude oscilloscope varies. trace describe the cathode-ray operation of a Alternating as oscilloscope. a current generator. voltage peak varies is caused These will be sinusoidally by an alternating discussed from a in positive voltage 4.23. peak The source such magnitude value to a of the negative value. 10 V /egatloV ACTIVITY In the Tesla 19th and century, Thomas 8 Direct current 6 4 Nikola 2 Edison 0 disagreed about use or whether to –2 –2 Time/10 direct alternating s current –4 for mains supplies. Research Alternating –6 the background to this current ‘war –8 of the currents’ and find out –10 why alternating eventually current was selected. Figure WORKED EXAMPLE is alternating a.c. power the frequency of the shown in Figure 1 = the From period the of in Figure diagram one to The peak voltage example current 4.12.1 you complete can is for a measure low voltage the maximum the cycle. oscillation. this is is the highest voltage reached over In ±8.0 V . = 25 Hz The period (T) of the alternating current is the time taken for one 0.04 s complete Finding The cycle. the This In the example is of the a.c. related source to the 1 frequency this is 0.04 s. frequency frequency second. = is the period number by: 1 or f = period T −1 The 130 direct 1 = T supply. and shown current 4.12.1? the f voltage alternating a.c. supply Comparing The voltage What 4.12.1 unit of frequency is s or hertz (Hz) of complete cycles per Exploring In 3.12 sound an saw electrical The two volts The The volts per time when and analyse also be be sound be used to to energy displayed used time of when an the voltage investigate was on converted the measure to oscilloscope the properties base if is alternating control alternating the oscilloscope are the per allows you source. division If is to the set measure wave to the occupies 0.5 V/div then 3.0 V . the one set on base: volts is determines example, time used (volts/div) voltage base an then controls the division boxes For the could can important peak-to-peak division. To which could The currents. division vertical the oscilloscope frequency. oscilloscope peak-to-peak six an their signal most per that and alternating The you waves screen. of waveforms time represented complete to 0.2 s/div, source follow wave the this by a horizontal occupies period is four divisions 0.8 s. procedure: Figure Connect Adjust the the source vertical waveform Adjust the the the period can Measure and Measure the the volts can time to per clearly base be a.c. so input division be seen that at terminals until on least the one the of the peak current the period source, and trough of screen. full wave can be seen and peak of the or peak-to-peak source and KEY voltage. calculate the can also which be would used be to measure displayed as the a POINTS frequency. voltage horizontal of a Direct one direct current Alternating direction in only current and switches magnitude. oscilloscopes 3 Cathode-ray oscilloscopes are expensive and can be difficult Oscilloscopes operate. Most waveform analysis is now carried out using which SUMMARY Find the shown be connected directly to a be used properties to of signals. computer. QUESTIONS peak in can the digital a.c. oscilloscopes can to investigate 1 travels direction. line. 2 Digital oscilloscope measured. note oscilloscope An oscilloscope. 1 The 4.12.2 voltage, Figure period and frequency of the a.c. 4.12.3. source 2 Sketch of the appropriate following graphs a.c. waveforms: 20 a an a.c. source with a V / egatloV 15 peak 10 and voltage a of 110 V frequency of 50 Hz 5 b 0 an a.c. peak and –5 source voltage a with of a 20 mV frequency of 2 kHz. –2 Time / 10 s –10 –15 –20 Figure 4.12.3 131 4.13 Mains the Mains LEARNING the end of this topic be supply electricity able and describe the mains in the describe the what would a device the reasons voltage is were wrong voltage mean supplied vary as alternating from island to current. island The in mains the supply Caribbean for (Figure 4.13.1). varying continuously over a cycle the effective is used square to (r.m.s.) describe voltage the as mains shown supply. in T able This is called the 4.13.1. connected Higher to is frequency happen root if frequency Caribbean average and supply As used voltage to: historical Caribbean you voltage should in OUTCOMES Mains At electricity type of voltage supplies can provide the same power using smaller mains currents allowing thinner wires to be used. However, higher voltages supply. are more Mains T able 4.13.1 Mains supply in supplies spikes Cuba Voltage Frequency /V /Hz 115 110 often the slightly supply picks ‘noisy’. up The smooth interference from curves other have electrical 50 60 Some equipment can be sensitive to this noise. 350 V /egatloV Barbados as islands devices. Island are some slight Caribbean dangerous. St Kitts & Nevis, 300 220 V r.m.s. 250 Barbados, 200 115 V Jamaica 110 or 50 220 St Kitts and Nevis 110 or r.m.s. 150 100 50 60 0 230 –50 St Martin 120 or 50 –2 –100 Time/10 230 s –150 Trinidad 115 50 –200 and –250 Tobago –300 –350 –400 Figure Ring An the wires The a live high The 132 are of between wire called domestic used current neutral Many and varies islands in the Caribbean to in the ring several (coloured a ring power main devices brown) main sockets and at they once provides is used (Figure are thick without the as the 4.13.2). basis T wo enough for or to overheating. power and operates at voltage. current supply configuration connection provide a Mains mains electrical three 4.13.1 to wire flow devices yellow). (coloured in are a closed also blue) completes the circuit, allowing the circuit. connected to an earth wire (coloured green L 10 A or 15 A E socket with N earth N E L 2 wires 2.5 mm In a ring each or ‘ring’, and the 2-pin main wire circuit forms which ends at a the consumer loop, fuse 4.13.2 A ring socket earth unit 10 A main with in Double socket Figure Switched begins 2-pin socket with earthed Figure sockets 4.13.3 Make your sure plugs that and you match sockets correctly. Plugs Some and sockets electrical mains supply, disconnected plugs and (Figure Type Type but most easily sockets such are and used as ovens, connected moved. and In these the are are by wired a plug directly so Caribbean referred to that there as into they are type A the can two and be main type B 4.13.3). A plugs neutral devices, B earth have only two pins which connect to the live and wires. plugs have three pins. The additional pin is connected to the SUMMARY 1 The different plugs are matched by different sockets and a type Why should not be inserted into a type B shouldn’t plug type to the wrong there are accidently several connect Connecting to take a a different a device 110 V large device current. mains to the to This a voltages wrong 220 V will most in use it is possible to causing Connecting receiving won’t a too work Connecting 220 V low at a severe a main supply, supply likely will melt cause the the wires to current. the electrical B to a advantages does a ring provide? the POINTS a 110 V The supply device will will not result in operate the device correctly 1 or Mains and designed reverse, is to operate also likely with to a 50 Hz damage supply the to device components will not function electricity supplied at at can different 50 Hz or be voltages 60 Hz. a Always connect a device to as the its type damage. device device a device in all. or from supply. 2 60 Hz change electrical A? What KEY device an supply 2 As on socket. device Connecting you A the plug QUESTIONS wire. correct type of supply correctly. using the correct kind of plug. 133 4.14 Mains Mains LEARNING the end should be of this able topic force describe and you of to: how fuses be able to correct fuse for a enough this, and is dangerous because the voltages are large enough OUTCOMES to At electricity safety current to start safety the through electrical features your fires need body. if to The there be is currents a fault in incorporated can the to also be device. protect large Because the device user. operate identify the Fuses state a the circuit explain device advantages breaker compared a to the a If of circuits when fuse function there of is fault and device will would heat in an connects be small the electrical to the and wires a in device neutral large the where wire current device then will and the the flow. could live wire short resistance This cause large a fire. of the current A fuse an prevents earth a this by being the ‘weakest’ point in the circuit a thin piece and melting wire. before held any other inside Fuses a need operation to An be but WORKED part. glass or selected only if fuse so there consists that is of of wire, often tube. an they will excess not melt in normal current. EXAMPLE air-conditioning power The ceramic rating of unit 1.0 kW. is designed What size to operate fuse should at 220 V be used with with a this unit? First find the normal operating P Figure 4.14.1 Fuses have ratings, 5 A, specific such as P current 1 A, = IV so I current = = = V 3 A, of the unit: 1000 W 4.55 A 220V 13 A. Then It select a fuse that operating current. should noted device. and be A this current small In case, fuses only increase slightly this that of is a in larger a 5 A alone few do fuse not milliamps current than would is the normal appropriate. protect is the enough not cause to a operator kill fuse a to of a human melt. Switch (held closed by spring) Circuit Circuit the breakers excess circuit. having A residual device. Figure 4.14.2 A circuit breaker uses to break too 134 when large. the faster part the manner magnetic and more advantage circuit compares similar a (Figure breaker current of fuses and sensitive being able but they cutting than to a be fuse. reset rely off on the Circuit without 4.14.2). (RCCB) in to switch the is live an even wire and more the sensitive neutral current should be exactly the same. If there is even a very small wire current the leak circuit is the the current a an which electromagnet It have replace in opening process also to operate current This breakers Electromagnet breakers (through the user is the current is cut off. of a device) then the circuit breaker opens and The An earth electrical becomes live wire wire device live (is breaks Some devices made from becomes connected inside the dangerous the live wire). to the This user can if its happen case if the device. automatically insulating very to protect materials the such as user because plastic. Even they if are the live wire Figure becomes feature Figure If the loose are it cannot double electrocute insulated and the user. should Devices carry the with symbol this 4.14.3 safety shown This an indicates is that double insulated. in 4.14.3. case receive a is metal fatal and electric is in contact shock on with touching the it. live A wire, fuse a alone user can would a Safe F use not symbol appliance prevent touches this, the as no additional current would flow until the Heating element user device. L To prevent wire: the these earth accidents wire. This the wire case can provides be a connected low to resistance an additional route for N the current the user. to pass to earth and so the current will not pass through Switch Also, the large current will cause the fuse to melt or circuit E breaker to trip, breaking the circuit (Figure Metal case 4.14.4). Current path ACTIVITY Inspect the plugs and electrical information on a range of b Protected Heating the electrical devices to find out their power rating, operating element metal voltage making and of the fuses yourself type the of mains devices without supply need. expert they Don’t require. try to Work carry out out any touches case, which it live type rewiring L supervision! N SUMMARY QUESTIONS E 1 Why should 2 What you use the correct fuse in an electrical device? Current should you do if the circuit breaker in your house tripping? Figure 4.14.4 The user 3 What T able fuses do the Power washing 1 machine tumble drier electric lamp T able 4.14.1 from wire protects the electrocution. require? rating/W Mains voltage/V 800 110 1200 230 80 120 POINTS Fuses live 2 in earth 4.14.1 Device KEY devices path keeps The an and wire circuit breakers becomes earth wire electrical too break large protects the due user circuits to a if the current in the fault. from electrocution in case of fault. 135 4.15 Conversion to The LEARNING the end of this topic be d.c. need is able much that easier explain why sometimes a.c. need to a to to voltage be of alternating transmitted current efficiently over long in power current is also much easier to produce from the stations. be many to devices work an alternating properly. current However, some will provide devices, the energy including they computers, diagram require how converted the can d.c. circuit showing increase energy Alternating generators sources need draw current to: For converted direct to electrical distances. for you so should a.c. OUTCOMES It At of a.c. can direct current at very steady voltage levels to operate correctly. be d.c. Rectification The process called shown so if in one current Figure is one half a.c. of diode the alternating will to only an relies allow current on the current alternating to in supply a direct properties one there current of diodes direction will only is as and be a time. rectification from shows an the a circuit alternating direction represents power A an Rectification connected 4.15.1 current in converting 4.10. Half-wave Diode of rectification. and so the resistance of which supply. output the can The be current device used diode the is to only produce allows direct. power The a direct current load supply is resistor connected to. supply To Resistor oscilloscope Figure 4.15.1 Simple If we an can see The current that half-wave rectification use it from oscilloscope what is happens provided only in maximum all. to in by measure the half-wave one direction. zero current at This current required is by very rectification However value and inefficient electronic Improving rectification One improve voltage across the load, we circuit. to a the for the direct voltage periods and is does of not is in still time the varying there provide sense is the no type of devices. + way egatloV 4.15.2). a period short produces a A of the profile capacitor time. smoothed is a Adding d.c. of the simple it on output to as current device the is to simple shown use which by a capacitor stores charge half-wave the blue for rectifier line. 0 Time When the charge When Figure 4.15.2 Smoothed half-wave rectification plates voltage will the current than 136 to (Figure in is rising stored voltage the and the be is load. provide current on and the decreasing However a there current. provided is a current in the load some capacitor. by there the This the would capacitor current a.c. normally will will source. be release decrease a decreasing charge more from slowly its During the continue If the sections to where discharge frequency of and the there is no provide source a were voltage the capacitor will current. low, the capacitor would + discharge has to almost discharge completely. slightly and At the high frequency, current the provided is capacitor only therefore fairly egatloV constant. Full-wave In is Time rectification half-wave current 0 rectification actually complicated you producing circuit can a involving see useful four that only direct diodes half of current. can be the A used alternating more to produce Figure a positive potential producing supplies a a negative much SUMMARY 1 Sketch difference even voltage. steadier d.c. This when gives supply the alternating full-wave (Figure supply rectification 4.15.3 which 4.15.3). QUESTIONS the a.c. full-wave rectification oscilloscope trace you would expect to see when EXAM the Smoothed is source a half-wave b full-wave in Figure 4.15.4 TIP is: rectified Alth ough rectified. have to you know capa citor s fullwave do no t how wo rk or rectifi catio n 110 o pera tes, V / egatloV a.c. can into 0 a remem ber be steady contin uous 0 0.01 0.02 0.03 and d.c. 0.04 fairl y that trans fo rm ed supp ly easily . Time / s –110 Figure 2 Redraw been KEY 1 4.15.4 the traces smoothed as by they a would appear if the output had capacitor. POINTS Alternating not all, current electrical can provide electrical energy for most, but devices. 2 Rectification is used 3 Rectification relies to on convert the a.c. into properties of d.c. diodes. 137 4.16 Logic circuits 1 Binary LEARNING OUTCOMES Computer At the end of this topic binary should be able describe are how binary represented in numbers computer find 0 and the output from a number gate unlike are 4.16.1 using digital system only information contains two represented digits as a represented our decimal but only decimal as 1s system a string and 0s numbers which of are contain digits used. just T able represented as ten as in digits. any 4.16.1 four other shows binary digits. simple numbers can be represented physically in a number of ways. In system. Examples numbers four Decimal binary represented system electronics T able operate The example Binary logic 1 Numbers some systems system. to: by systems you of binary Binary high low The decimal represented as which which exact what they are represents represents voltage type of represented the the levels as digit digit vary is 1 0 from semiconductor two voltage and and is is system being levels: called called to logic logic system 1 0. depending on used. digits Decimal Binary 0 0000 6 0110 1 0001 7 0111 2 0010 8 1000 3 0011 9 1001 4 0100 10 1010 5 0101 11 1011 Logic A gates logic input gate and is an electronic produces a component binary output which based on processes a set of a binary rules. Examples: A NOT input. 1 gate This output As the An is The AND Y ou of a are to expect ed find logic the gate to a in three any the the there logic is 1 opposite a logic input of 0 the input there the same is input as there a is logic these the a 0 logic output. gates are a logic 1 output if one or other of its inputs gate produces are list both of the a logic logic five 1 output if its first input and its 1. gates . logic gates you are required to know is you 4.16.1. Truth tables system tables show the output the gate. states of a logic gate for all of the maxim um T his can combi natio n three Figure be output possible be a not inverters. produces input complete Truth contai ning of is is TIP shown able when which 1. second EXAM output there always called gate logic is an that when output OR An means but sometimes produces see on of these the inputs output When in consult doubt the to would be about truth These when the what table. For a allow gates logic us are gates example the to easily determine connected does, truth you table to input should shows what sources. always that an OR page s. gate logic 138 will 1. still produce a logic 1 output even if both of the inputs are Symbol Gate low voltage voltage OUTPUT A Truth Function (high = = = 0) WORKED table INPUTS 1, A B 0 0 0 0 1 1 1 0 1 1 1 1 0 0 0 0 1 0 1 0 0 1 1 1 0 0 1 What system if B A OR B = OUTPUT A are required the for input the conditions logic gate 1 OUTPUT OR EXAMPLE OUTPUT 1 = to shown produce in an Figure output 4.16.2 of 0? A 1 OUTPUT if AND B A AND B = 1 Output B OUTPUT OUTPUT A = C 0 0 1 0 1 0 0 1 1 0 0 0 1 0 1 1 1 0 1 1 1 0 if NOR B A OR B = OUTPUT Figure 1 = 4.16.2 Draw up the truth table for the 0 OUTPUT A logic if NAND B A AND B = 1 a sensible sure OUTPUT OUTPUT if INPUT = 1 = 0 0 1 1 0 gate all of system order the combinations working and making possible are in input covered NOT OUTPUT = 0 = 1 (T able 4.16.1). INPUT if INPUT T able Figure 4.16.1 Combining Logic gates together to be To work table of all have they and can their truth 4.16.1 tables Input Input Input out the in be use used output turn output. the all of work Keep possible A B C individually to process but when information they and are 0 0 0 1 1 0 0 0 0 1 0 1 1 1 0 1 0 0 1 1 1 0 1 1 0 1 1 1 1 1 1 1 combined cause actions for the your on input any collection possible way going of input through until you gates draw up combinations. the logic have a truth For each combinations found the set and outputs for combinations. From SUMMARY that gate Draw up the a truth table for the logic gate systems shown 4.16.3 and the table we output of is 0 system can logic the logic only in when Figures truth QUESTIONS see 1 Output gates limited representing the of logic gates taken. inputs find Logic A is 1, B is 0 and C is 0. 4.16.4. a b A KEY POINTS A 1 Binary digits can be Output B B Output represented by high and low C voltage Figure 4.16.3 Figure 2 Logic gates inputs 2 Design a logic gate system to match levels. 4.16.4 the and process produce binary binary following: outputs. a Three inputs when any Three inputs A, one B of and the C which inputs is produce an output of 1 3 1. Simple be b D, E and F which produce an output of logic combined all of the inputs are to can provide 1 additional when gates processing. 0. 139 4.17 Logic Logic LEARNING gates are the end should be of this able topic you are then devices to: output find the output combined from combination to of conditions processed are and through triggered. device with digital The activates a produce sensors sequence logic only logical which gates when of are the signals. logic are logic gates designed required and so that simple produce Logic gates when logic the gates desired sensors are devices triggered level outputs are process circuits by results signals of produce input met. gates logic involving the environmental Output design signals output sensors Sensors detect These a conditions attached often 2 OUTCOMES environmental At circuits processing which output activated. Sensors The sensors symbols as produced T able connected shown by in these to the T able sensors logic 4.17.1. in system The different can table be also represented shows environmental the by outputs conditions. 4.17.1 Sensor Symbol Produces switch logic 0 Produces when when open closed logic 1 S temperature temperature is low temperature is high T light in darkness in light L pressure not under pressure under pressure P Figure 4.17.1 A data logging connected to a device computer In reality these thermistors supplies. output A 0 sensors light point to a temperature the pressure is applied other infra-red are at which logical could it can such sensors sensors, be 1 level as mechanical (LDRs) be from finely to switches, connected switches can designed a to voltage logical tuned. switch from 0 to 1 40 °C. output somebody possible sound sensor of above switch as are rises such resistors the output sensor sensor to devices dependent temperature A Many 140 The of when or from 0 stepping such sensors as tilt and to on 1 a when switches, even a large force mat. pH sensors, radioactivity sensors. WORKED An alarm and a The alarm will check open the must check NOT if gate is is if the dark, a dark a is dark gate of its door be light it only system be inputs and inverted when be using are a door logic is the a is dark is required switch thus it implemented? and will mechanical the to this AND both AND so triggered when open, 0 be could an when needs into the that logic input it in a to How triggered door goes zero be alarm the this need indicates the if input may opened. produce so logic is This trigger would To signal door opened. To EXAMPLE and 1. needed. switch NOT this This were gate before gate. sensor this is input needed. needs to This be produces inverted a with a too. S Output L ACTIVITY Complete Figure devices This gives would This on a a survey of the 4.17.2 be the logic logic could be 1 circuit only shown when connected to the an in Figure door alarm is 4.17.2. opened system or The and be it rely output is used a dark. to on short would switch you use every electronics report be day and about different that write how life without this technology. light. Computers Digital They computers process ‘clock’ which electronics contain binary stops and millions operations the signals at of logic very getting microelectronics have gates high a integrated speeds, mixed had in up. controlled The massive circuits. by a developments impact on in society SUMMARY over the phones past fifty around years. the For world example, are linked computer together systems through and the internet 1 providing almost instant access to information for billions of A bank POINTS alarm Digital can be designed to produce logic level when somebody or floor on environmental Logic the circuits required can process signals from sensors and to produce Computer undertake only the sound vault. happen is steps on This when the has been Design primed accomplish a logic by a this system task. outputs. 2 3 of a conditions. switch. 2 have signals alarm depending to which heard the sensors needs system activates should 1 vault people. an KEY QUESTIONS mobile systems very contain complex millions of processing. logic gates to Design could a logic activate raining at circuit an which alarm if it is night. 141 Section SECTION 1 Describe current 2 4: An Practice the and time supply base Practice exam differences direct oscilloscope power 4 has is questions between exam used to alternating 7 been set to analyse an trace 0.05 s a.c. 8 shown. per The the volts/div has been set to is the function of a fuse and how does operate? What are when compared the advantages to a of a circuit breaker fuse? division 9 and What it the 2 2 current. producing questions Draw a circuit diagram showing how an 2 V/div. alternating to current produce a rectification 10 Sketch a a b an a direct system d.c. source a.c. source and half-wave period of of be be How voltage with rectified can this improved? graphs peak a showing: 10 V period voltage rectified 0.2 half-wave current. voltage–time seconds c can a.c seconds of of 0.2 8 V source and with peak a voltage of 8 V d a What is the peak voltage of the a half-wave rectified a.c source with a power period of 0.2 seconds and peak voltage supply? of b What is the period of the c What is the frequency power 8 V large of the a.c. Explain sockets why in a ring main circuit is used for wall Copy Select output smoothed using a capacitor. and complete describing their this table of logic gates functions. houses. Logic 4 the source? 11 3 with supply? the appropriate fuse for each of Symbol Description the gate following and devices from the range 1 A, 3 A, 5 A Inverts 12 A. the symbol Device Operating logic Power vice voltage/V filament lamp 110 80 W 110 500 W AND air- Produces and conditioning to logic a 1 and versa) logic portable 0 input (changes 1 an output when input B input are logic 220 electric drill 220 electric grill 110 1 2 kW 300 W 1.5 kW NAND 5 How does faulty an earth electrical wire protect the user of a device? NOR 6 Explain why the core of an electrical cable Produces from copper but the outer sheathing 142 from flexible plastic. 1 when both is inputs made logic is output made a are logic A both unit kettle of 0 12 Copy the and complete possible logic inputs this for truth this table for all arrangement of 13 of How can NOR gate an AND and gate two be NOT constructed from a gates? gates. 14 Design only the logic sounds circuit during for an daylight alarm and which when it is A raining. B Y C D Input A Input B Input C Input 0 0 0 0 1 0 0 0 0 1 0 0 1 1 0 0 0 0 1 0 1 0 1 0 0 1 1 0 1 1 1 0 0 0 0 1 1 0 0 1 0 1 0 1 1 1 0 1 0 0 1 1 1 0 1 1 0 1 1 1 1 1 1 1 D Output Y 143 4.18 Permanent Magnetic LEARNING the end should be of this able describe the topic Only you to: effect a on other magnetic describe a can be materials magnadur. be metals (unless All magnets have seeking) when placed these as steel, will is a poles south poles Opposite Like near a magnet. not magnetic include that iron, contain attract materials. nickel, these cobalt metals non-magnetic may materials, current in them as we will see later). which (or are south referred seeking) to as poles. the In a north simple (or bar are at either end but magnets can be made in The Magnetic of magnets coloured: and blue red for are attracted the between attract repel poles closer so poles may be found in other locations. interactions poles poles between poles and interactions north there two and shapes north often such These same made. The The magnetic. these permanent Magnetic 4.18.1 are Magnets from and several Figure made Alloys, magnetic. magnet magnet forces are magnets magnets materials how few also north themselves and even have experience OUTCOMES Magnets At materials magnets each repel the the magnets each other: north magnets can other: south be summarised south poles poles repel by attract south these north poles, rules: poles. and poles. are to each other, the stronger the force magnets. materials towards placed near either pole of a magnet bar magnet will be it. for south. Magnetic When an induction iron nail is placed on the end of a it can be ACTIVITY used to attract induced Suspend a magnet as shown 4.18.2 and use a the iron iron nail. nails. The We nail say is that magnetism temporarily acting has as a been magnet in but Figure in other will stop acting this way when it is removed from the permanent second magnet. magnet identify prove with its that between the the the closer known poles. polarity Can size you of the magnets they are to also If force increases we try magnet has been Iron process also removed and used do S Iron same may with retain from the a steel some of nail its permanent the nail magnetic will act as properties a after it magnet. together? Force the but N of to not Steel is mumetal make retain classed magnetism classed magnetise bar are temporary but as it a hard will as soft magnets. magnetic They are easy materials to and magnetise are but well. magnetic retain its material. magnetism for It is more longer. It difficult is used to to attraction make Figure 4.18.2 Testing the between 144 forces magnets permanent magnets. Magnadur magnets are also permanent. Making A a new magnets permanent permanent permanent magnet magnet magnet can along must its only ...and be made length be as from moved repeat many a shown in one steel in bar by Figure rubbing 4.18.3. The direction. times ACTIVITY Make your using the Figure be for Figure 4.18.3 Making Breaking magnets A will magnet dropping a lose a permanent some permanent of its Can stronger longer shown the if it in magnet is rubbed time? magnet strength magnet magnet technique 4.18.3. made a own onto if the it is struck. floor will Repeatedly weaken EXAM its TIP magnetism. Only a few mag ne tic . the SUMMARY QUESTIONS all 1 Copy and between T able complete the to North north Why bar KEY 1 is to south steel two showing the me ta ls of mak e thinki ng are. interactions magnets. rather South to south than iron South to north used to make small permanent magnets? POINTS Magnets produce magnetic 2 of 4.18.1 mista ke are 4.18.1 North 2 poles T able materi als Don’ t Permanent as forces which act on other magnets or materials. magnets are made from magnetic materials such steel. 3 Like 4 The poles size distance repel of the and force unlike poles between attract. magnets decreases with their apart. 145 4.19 Magnetic Magnetic LEARNING the end of this topic be able have already produced draw and plot the around a bar are describe the magnetic magnetic by the explain how a shape The an as of the of Iron objects between by magnetic fields. is a region of space in which a magnetic material will magnetic field is not as simple as that of an electric are always two poles involved. magnetic bar represented north the lines as pole point south pole a series would away of be which pushed from (Figure lines the by show the north the field. pole of direction This the means magnet and 4.19.1). magnet field the shape two of a common magnetic ways of field finding the shape of a magnetic field magnets. filings filings align with can the be sprinkled field lines around and the the shape magnet. of the These fi eld filings will be will seen clearly around (Figure shape To A A 4.19.2). of the prevent placed the plotting plotting the The will field technique when filings a piece more sticking of is particularly than to paper one the or useful magnet magnet wrapped is the in for looking at the involved. magnet plastic can be film. compass compass The line can needle and compass curve This field under magnet. is back a of dot moved to the be the placed against compass can be placed repeatedly south will pole where until of the the north point a the trail pole along of the of compass dots magnet. The is a bar direction is pointing. formed. dots of can This be 4.19.2 joined with starting the The is is Earth to curve several to form different a field points line. near The the whole poles process until the is repeated shape of revealed. magnetic produces the whole south a Earth’s due the at field The a rotation planet pole geographic 146 a field are around S Iron Figure charged acting force There N a forces force. imaginary Finding The field a is towards Lines caused there field that 4.19.1 between the compass works. Figure acting Earth field forces Similarly, field The produced the fields. magnet experience that electric magnetic A field by to: magnets seen you are should fields OUTCOMES You At fields of this north as if field magnetic of its there imaginary pole. field metallic were bar a around core. bar itself The magnet magnet lies (Figure magnetic within near, but 4.19.4). field the not This envelops Earth. at, the The The Earth’s magnetic field has been used for navigation for ACTIVITY thousands thin will slice of of align years. Lodestone lodestone itself to the can be Earth’s is a naturally made to float magnetic magnetic material on The water. and a lodestone field. Use on the shapes Magnetised steel pins are used in compasses and follow the techniques page 146 of the but these are suspended inside a case so that they freely. The ‘north seeking’ pole will point towards the of the Earth because it is attracted to the as it magnetic fields two permanent arranged as shown in north Figure pole the magnetic may magnets rotate described find same surrounding principle to south 4.19.3. pole. a The Earth’s from the harmful magnetic Sun and field is protects also the useful Earth’s defl ects surface from charged particles potentially S N S N N N S effects. b Magnetic Axis of rotation S axis ~1 1° c S N S N d S N N Figure Figure 4.19.4 The Earth’s magnetic field 4.19.3 EXAM When SUMMARY S TIP draw ing field lines remem ber 1 How can iron filings be used to reveal the shape of of fields? go An iron nail whether is this seen to proves attract that the a paper bar is a clip. Comment into they no rth the po le south come and po le. on T he magnet. the a KEY the alwa ys that magnetic out 2 mag ne tic QUESTIONS term ino log y Earth ’s bit sure POINTS po les confus ing. you abou t can be Mak e unde rstand it clear ly. 1 Magnets have magnetic 2 The magnetic fields around them which affect materials. shape of the fields can be found with a compass or iron filings. ACTIVITY 3 Field lines come out of the north pole and enter the south Find out about the aurora pole. borealis 4 The and Earth has protects a us magnetic from field some which cosmic assists rays. with navigation and and what aurora causes australis these phenomena. 147 4.20 Electromagnetism Currents LEARNING The At the end of this topic be charges able sketch the magnetic a shape field of the formed current-carrying until around wire and apply to This right-hand the grip rule direction of field state a was first compass there point in a To was a demonstrate filings with some in a wire produce demonstrated uses of near a wire. current different Demonstrating determine the the moving a magnetic field around the by in The Hans Christian the compass wire. This would Ørsted caused point when he the northwards compass to direction. a solenoid fields to: placed magnetic you wire. should and OUTCOMES or bar the the plotting shape shape of of the the compasses in field magnetic a similar field way to we can the use iron techniques used magnets. electroThe current-carrying wire is made to pass perpendicularly through a magnets. sheet is of horizontal placed show be on that the there sprinkled confirming card is onto the card a (Figure and the circular the card shape of 4.20.1). current field and the is A of turned around these set will the plotting on. wire. form a The Iron compasses compasses fi lings pattern of can will also circles field. Card Field Figure To determine hand Figure 4.20.2 Using your imagine fingers while out the The right curling around sticking in the the your thumb of current. direction fingers The of magnetic wire field 148 way shows loops. the as way shown the in of Ørsted’s field lines Figure experiment loop you can use the ‘right- 4.20.2. called solenoid is field around solenoids similar in a are shape single used. to wire The that of is not strong magnetic a bar field magnet and so coils outside but there the is a field of the coil too. This internal field is almost uniform. The fi eld lines you are which which rule’ version Solenoids inside your updated wire direction curved grip An hand, your conventional 4.20.1 all parallel to each other. One end of the coil acts magnetic and the other acts as a south pole (Figure 4.20.3). as a north pole Increasing The the strength using increasing placing of more strength the field loops of the magnetic produced (turns) of by a field solenoid is increased by: wire Field the an current iron concentrating core the in the inside field (north) wire the lines solenoid creating a (this has strong the fi eld effect at the of end of the Current core). Powerful electromagnets can be produced by using all of these Figure 4.20.3 measures. To find field of the use your own electromagnet using a large iron nail, a power supply and a length of insulated wire. Wrap around the nail in a tight coil and test the power of Be careful not to use large currents. You again. fingers of the around in the thumb will direction your of electromagnet. can direction Your the the point wire we hand curving coil. of direction your the current low- the voltage right time follow Build direction the current, our This ACTIVITY the from can the magnetic field add (northwards). a variable Using resistor their They are medical control the current. electromagnets Electromagnets and to Scrap can strength used for be can a very be wide useful. They controlled range of by can be turned adjusting applications the from on and current heavy off in them. industry to diagnosis. yards objects. use powerful T urning off the electromagnets electromagnet to lift allows up cars them to and be other put down again. An electromagnetic remotely. the A small, current where large a small current the a can from starter fields are be current switch. Examples current to safe, pulls circuit. Electromagnetic in and electromagnet high relay of the used to enters The operate the switch these relays ignition switch electromagnet is actually can circuit a is be part found used to and of in turn a cars on Figure 4.20.4 A very electro- motor. also vital in electric motors as you will see SUMMARY 4.23. 1 What QUESTIONS advantages electromagnets KEY strong magnet a do have when POINTS compared to permanent magnets? 1 A current plane at in a right wire produces angles to the a circular magnetic field in a wire. 2 Imagine there was current-carrying 2 A solenoid is a coil of wire which produces a magnetic field coming similar to that of a bar The strength of the magnetic field of a solenoid is using more loops of wire, increasing the Sketch current an iron out of this magnetic pattern around this and imaginary adding the increased field by vertically magnet. book. 3 a wire wire. The current is core. flowing upwards. 149 4.21 Forces from magnetic fields When LEARNING a wire surrounding At the end should be of this able topic from you with to: a describe field the has on effect a a the a current wire. permanent each magnet. carries there will be a magnetic field OUTCOMES other. This is If the magnet A force the wire then will motor is placed the act on two the inside the magnetic wire and magnetic fields the will field interact permanent effect magnetic current-carrying Demonstrating the motor effect wire sketch the resultant surrounding carrying two use a wire permanent find the left-hand direction acting on Figure The magnets be demonstrated simply using the apparatus 4.21.1. T wo produce an almost uniform magnetic field between them. of a thick wires hold another loose wire, so that a complete circuit rule made. the When the circuit is switched on the current in the loose wire will current- produce carrying can magnets force in effect between is to motor shown current- placed Fleming’s The field a magnetic field which will interact with the field from the wire. permanent The If the loose magnets. wire direction reversed then will of the experience the current loose wire a or force the will making direction move to the it of jump the to the right. magnetic field is left. S-pole Plastic ACTIVITY If you have shown motor in the copper brass wires or or support a wooden held in stand apparatus, Figure effect Thick 4.21.1, using the test the method described. Iron yoke Magnadur (unlike Figure The The 4.21.1 size size The of of the The current The be 150 in strength of of greater acting wire. around field field length a the field the motor V, high I, d.c. supply effect force force magnetic stronger the Low facing) Demonstrating the magnetic magnets poles will the wire larger will the and a the wire field the following: stronger the the interaction with magnets. the permanent magnets. A force. permanent is on the greater from greater depend current the permanent magnetic inside if the wire the produce wire force The the from the on longer. magnetic field. There will Fleming’s left-hand rule EXAM It can wire be in motor when a difficult magnetic rule) we Using to lets know your left work field. us find the out the direction Fleming’s the left-hand direction direction of of the of the the rule (also force current and force acting known acting the on on as the magnetic the When wire in field. Separate feel thumb, first and second fi ngers so that they all silly to right angles to each other (see Figure Point pole your to Point your positive your First south finger the direction terminal finger to about angles Your thuMb (or you of of the magnetic Field find does the movem ent. 4.21.2). wo rk It . (north the to do in the direction negative this but of terminal). always the You keep Current may the (from have three to the twist fingers at First right lef t-h and help pole). seCond hand in don’ t usin g point really sitting hall, abou t direc tion at are exam Flem ing’s hand: your you the rule TIP a to each will finger = Field other. show the direction of the force acting on the wire Movement). Left hand SeCond = Understanding The force magnetic field on the fields (Figure the wire so it field arises is finger Current because important to of the know interaction the shape of of the the two resultant ThuMb = Movement 4.21.3). Figure 4.21.2 Fleming’s SUMMARY 1 How a The rule QUESTIONS the force current-carrying acting wire in on a field between two can left-hand magnetic the field be increased? permanent magnets almost 2 is uniform Figure 4.21.4 carrying field In is acting which acting A into the field a current- magnetic the direction on Magnetic shows wire. is paper . the force wire? into the paper Current Neutral Flat magnets point Figure 4.21.4 alone The fields combine Direction like KEY this of 1 The field POINTS force A current-carrying placed by the wire is magnetic 2 wire 4.21.3 alone Interaction The direction acting of the magnetic experience found Figure a field field will in in circular Combined Current wire produced magnetic on the using a of force. the wire force can Fleming’s be left- fields hand rule. 151 4.22 Electromagnetic induction When LEARNING a conductor through At the should end be of this able topic you to: describe current how in a to coil induce of the field force which force (e.m.f.). e.m.f. is moved will lines, attempts If the produce explain the the factors induced the to electrons make them conductor an a magnetic is induced in the field so conductor that it cuts move. This connected current in to the is a experience the a electromotive circuit then the circuit. a wire Demonstrating through OUTCOMES induction affecting To current. demonstrate through circuit must be moving wire a field will is be the as shown very used. effect small Note through in can simple Figure and that the you so the a 4.22.1. The sensitive current magnetic move field. only a wire current ammeter exists There is rapidly when no induced or in the galvanometer the current wire when is the stationary. Movement of wire Cutting field lines Ammeter To understand imagine the places occurs 4.22.1 the reason magnetic conductor which Figure the as a when it on is a the lines moves. force there for field It the is induced around the cutting electrons. relative current the you magnet of these need being lines Electromagnetic movement between a to cut of by force induction magnet and electromagnetic induction conductor so that the Investigating It is to usually a several is times Pushing the in Pulling Reversing will The also faster the The the This field more lines magnet are of than induction connected field lines increase (north lines magnet direction the pole pass being the to you to cut cut. a move can use through into the the sensitive induced first) through that loops of is that is of induces the direction magnet shows lines the therefore reverse current. field the a wire bar and magnet ammeter. the A conductors current. solenoid loops of wire so that induces the wire a magnet of the moving it is the cause in current The stronger Leaving the There no is conductor the magnet magnet relative so a the the opposite (pushing in the direction. south pole) current. relative rate of in the to that the the increased solenoid the coil wire the greater moves current. greater the no the inside larger the movement field lines are coil the does between being induced induced not the cut. current produce magnet a is. current. and the through current. so wire. the the the wire) that field the out of magnet magnetic current so and the move properties (coil used magnetic induction to the solenoid solenoid easier investigate and 152 a Demonstrating the The The direction current of induced the in the current coil will change direction as the magnet is ACTIVITY moved able To to do The in we current This coil means that This the that end of the the the the rule direction in a that work as current as the We need magnet to be moves. rule: always the a field out current). Use of acts in such a direction as a magnet, wire to solenoid investigate and coil induction. to it. push acts the solenoid solenoid we coil alternating of causes magnetic to (an following when the coil north north of the end pole the coil direction of but the reverses. of magnet when the the We can current into the magnet in use the coil 4.22.2). means coil that and mechanical of use change solenoid (Figure of induced the withdrawn the out determine this oppose is and a force will mechanical work conservation is of be work required must transformed energy is be into to push done in electrical the magnet doing energy so. into This and so the law observed. Motion Like poles repel 0 Motion Unlike 0 Figure 4.22.2 The solenoid rule allows us determine the poles attract direction of the induced current. SUMMARY QUESTIONS 1 How can 2 How does a KEY 1 you increase rotating a the induced magnet at current the end of in a a solenoid? solenoid induce current? POINT Electromagnetic movement current depends magnetic induction between field on and a occurs magnet the the strength speed when and of a and the there is conductor. direction relative relative The of induced the movement. 153 4.23 The d.c. a.c. generator The LEARNING the end of this topic be able direct force describe d.c. motor the current on a motor operation of a is not designed very to useful cause explain a.c. the motor wire. effect Making as a seen wire in 4.21 move in a to exert straight a in most coil of circumstances wire to and so the motor is rotate. motor Understanding uses current-carrying to: line the you a should and OUTCOMES A At d.c. motor the operation of generator sketch the produced an a.c. A coil The waveform by the d.c. motor an of other generator . wire magnets and is mounted are on arranged produce a an so strong, axle that between the uniform, a pair of opposite poles magnetic field magnets. face each between them. The coil ring the is connected commutator . direction of to This the a circuit allows current by the in a pair coil the to coil of connectors rotate to freely switch called but every also a split- allows half-turn. Force A current north hand pole Split-ring is pole rule will The two When passed will to through experience check this) experience forces will a the an coil. while the side downwards cause The upwards the coil side force of of the (use the coil coil near Fleming’s near the the left- south force. to rotate on the axle. commutator the coil has rotated a half-turn the commutator will reverse + Force Metal or brush contacts graphite the current the coil right Figure 4.23.1 A d.c. motor As spin, will side the in the still is cause pushed force Applications of motors a This the means left that side to the be direction pushed of the upwards force while on the downwards. remains producing Electrical coil. in the turning same direction moment which the can coil be continues used to do to work. motors are used in a wide range of devices from a small ACTIVITY battery-powered Use an make electrical your own motor model kit of fan to a motor which can drive cars. to a The a.c. generator motor. Alternating as seen energy into in 4.22. generators Physically transformation electrical Operation 154 current of the construction A coil The coil wire is they the are on electromagnetic similar reverse. to Kinetic the d.c. energy induction motor is but the transformed energy. The of is rely is of a.c. a generator simple mounted rotated. a.c. on generator an axle is inside shown a in strong Figure 4.23.2. magnetic field. Steady rate of rotation Meter pointer Coil swings from to side side Commutator Brushes Alternating rings Figure As 4.23.2 the coil An a.c. rotates voltage generator it cuts through the magnetic field lines and a ACTIVITY current is induced. Use As the coil rotates through the field lines the rate at which it to them changes as its alignment changes. This means that the a hand produce in the coil changes with the position of the When the coil is horizontal it cuts field lines at a generator alternating Describe what happens coil. when an e.m.f. current. induced powered cuts high rate and the generator is spun a faster. large e.m.f. When is the is produced. coil is vertical it does not cut field lines and so no e.m.f. induced. During shown a full in rotation Figure the coil will produce an alternating e.m.f. as SUMMARY 4.23.3. 1 Describe QUESTIONS the operation of the + motor in battery e.m.f. 2 1 Figure is 4.23.1 if the reversed. Comparing the a.c. generator cycle to 0 the d.c. factors motor, beside what the other rotational Time will e.m.f. produced? KEY X X speed increase the peak POINTS Y Y X Y N S 1 A d.c. motor electrical Y transfers energy into kinetic X energy using the motor effect. Figure 4.23.3 The e.m.f. produced by an a.c. generator 2 The d.c. motor uses current-carrying which Changing the rotation the speed of rotation of the coil will cause it to produce peak e.m.f. It will also increase the frequency of the a.c. a magnetic power station An a.c. generators are spun 50 times each a frequency of 50 Hz while others are spun 60 times field. generator to uses produce in a wire by a rotating each a second wire second current providing of rotate supply. induction Some coil to a 3 larger made speed inside Increasing is a coil inside a magnetic field. (60 Hz). 155 4.24 Transformers The LEARNING voltage called At the end should be of this able topic describe the you best to: operation of a an alternating transformer. current. to cable of current can be changed with a device OUTCOMES When do will so The electrical at high cause less transformer energy voltage heating is and also changes transmitted low effect over current. and so less A the long lower energy size of the distances current will be in it is the wasted. a In power lines the voltages may be several thousand volts. transformer state the difference between Transformer a step-up and a A transformer transformer arms use the operation step-down transformer of a consists laminated of iron two coils core as of wire shown wrapped in Figure around 4.24.1. opposite These coils equation. are the The primary (input) alternating magnetic field coil current in the and in the the iron secondary input coil (output) generates a coil. varying core. Laminated iron Primar y core Secondar y coil I The magnetic The varying output I p field is focused in the iron core. coil magnetic field induces an alternating current in the coil. s The iron induced Lamp core in it. is laminated These to ‘eddy’ reduce currents currents would which heat the would core be and waste a.c. energy. generator There is no direct transformer Figure 4.24.1 The construction of a are said to and be electrical so no connection current can between pass the between two them. coils The in the coils decoupled transformer Note that, magnetic The because fields, it the transformer There is a transformer cannot work operates with direct by using changes in current. equation relationship between the voltage on the primary and the number coil (V ) p and the voltage on the secondary coil (V ) of turns of s wire on the respective coils (N and N p V N s turns and see the that I p there currents in s p N p also s is the relationship coils (I and p If the on If number the output there voltage Figure 156 4.24.2 is a will of turns coil both on then difference the there in the change. by: = V can given I s = You ) input will be I the number of ). s coil number between no of matches change turns the in then number voltage the or of turns current. current and Step-up Step-up transformers transformers have more turns on the secondary coil than on EXAM the primary The The coil. voltage is current factor as is This has two T ake increased. decreased voltage is TIP effects: (compared stepped with primary current) by same and up. care check answ ers WORKED EXAMPLE A transformer step-up the output What are coil. the The usin g the equa tion that mak e your sens e. 1 has input output when trans fo rm er 50 turns voltage on is the 5.0 V input with coil a and current 400 of on 2.0 A. characteristics? ACTIVITY Find the output V voltage N s N s = first: × V s ; V 400 × 5.0 V p Construct = = = a simple transformer 40 V s V N p N p 50 from find the output efficiency N p I s = and × N p ; I of investigate the device. current: Measure I cores p the Now iron 2 A × the input and output 50 p = = = currents 0.25 A and voltages and see if s I N s Step-down A 400 s the power the same. ratings transformer secondary coil. This has also more has turns two on the primary coil than The voltage is decreased. The current is increased the by the same factor as the voltage 1 is Why are down transformers 110 V used in are homes needed or even to reduce lower for transmission step-up some How EXAMPLE many turns transformer the primary V in of electrical devices. 2 What order coil has V s = are N the on the voltage secondary from 200 V coil to of 6.0 V a if × N × in is the output is 40 V if voltage the and input there are 400 turns on the primary and 2500 on the secondary Is down 6.0 V step- used distribution? transformer coil? turns? s ; required lower 500 N s to a voltage 2 wire and transformers voltages of WORKED are QUESTIONS electricity to IV) effects: down. Step-down = on SUMMARY stepped (P transformers step-down the N p this a step-up or coil step- transformer? 500 p = = = 15 turns s V N p V p 200 V p KEY Transformer power 1 The relationship secondary output between voltage power POINTS of a and the primary current can transformer as voltage be used and to find current the and input the transformer increase and voltage ; V I s = V s I The p V p N a.c. supply. equation IV P p = N p as we have the = out I p s relationship: 3 Transformers are very P in efficient and so P = out above equation transformer and so the is is actually equation is: s V The to the I s = I p defined an transformer s V is used p = power of is decrease I s As or follows: 2 V A used a can for highly be an ‘ideal’ efficient used to transformer. device find with results However, low close to power true a loss provides results close P in to true values. values. 157 Section SECTION 1 The 4: Practice resistance given by 4 the of a Practice exam set of questions resistors in exam parallel is 4 relationship: 1 1 = A + R P R 1 student strength it by + … group of resistors in by using the relationship electromagnet the magnet to between and lift a the the current chain of steel paper clips from a desk. The results are 3 students relationship an R 2 shown A tested of 1 + R 3 3 in 1 questions were placing parallel a with asked to number each verify of in the table. this Current/A 0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 Paper 1 3 4 5 7 8 11 13 15 60 Ω other. They clips then lifted measured potential the current difference through across the set and the them. a Plot a graph paper Number Theoretical Current Potential Measured of resistance /A difference resistance resistors /Ω /V /Ω 0.19 12.0 2 0.38 12.0 3 0.57 12.0 4 0.76 12.0 5 A Describe group factor of The factors: Sketch the five arrangements of Calculate set the of Calculate set d of Plot theoretical resistance the e graph measured resistance of to the difference results comparing the the measured theoretical resistance the magnets Mass their effect how large on electromagnet. electromagnet investigated the a was mass two current of loops of wire. in the three it could different and They tables causes the of the effect to of each of loops of wire lifted/g Current possible and next of increasing recorded shown. 0.5 A 10 15 20 25 1.1 1.3 1.5 1.7 the 10 15 20 25 2.2 2.6 3.0 3.4 10 15 20 25 3.3 3.9 4.5 5.1 1.0 A Number of loops of wire measured theoretical lifted/g resistance. Current Describe greatest which of Mass 2 the testing students Number between resistance of by the investigating each combinations. Describe of the for resistors. a in resistors. the resistance number shown. was strength increasing Current c pattern have the number their each the current resistors. the b the 12.0 lift. a to students strength measured 0.95 the would increasing The 5 comparing lifted electromagnet. b 1 clips placing other two in all 1.5 A bar six possible Number of loops of wire orientations. Mass 3 Which of these will increase the strength of lifted/g an a electromagnet? Why should during a Increasing b Reversing c Replacing the current in the direction of the iron core with the the students use an iron core experiments? What conclusions can be made about the current. effect the of coil. b the all an of increasing the current? aluminium c What conclusions can be made about the core. effect d Increasing the number of turns of wire in wire? the e coil. Switching rapidly. 158 the electromagnet on and off of increasing the number of loops of 6 A bar magnet shown there in to the be is inserted diagram. a current into An in a solenoid ammeter the wire as as a Which b What way will the motor rotate? shows is the function of the split-ring the commutator? magnet moves. c How can the power of the motor be increased? Hollow cardboard tube 8 Y X What are increase the the factors e.m.f. that of an can a.c. be used to generator? N S 9 An a.c. generator revolves with a frequency Magnet of 20 Hz voltage a a What is causes the a name current of in the the process What will happen to of produces an e.m.f. with a peak voltage–time graph showing 50 V . Sketch a supply provided the which by this generator. wire? The b and the reading on frequency of the generator is increased to the 40 Hz. meter when the magnet is pulled out of b the Sketch how c What will magnet happen is to inserted the more reading if current in the solenoid causes it 10 An ideal being d e of a magnetic will the What field when the magnet transformer a.c. mains is graph showing change. used voltage resistance to to convert power a a 12 V lamp 5 Ω a What is the current be the magnetic polarity of b What is the power c What is the current in the lamp? solenoid? will be solenoid the direction (clockwise or of the motor is constructed as the lamp? current in the primary coil? in A transformer has 500 turns on the primary coil anticlockwise)? 6000 current d.c. of end and A would is 11 the 7 voltage–time inserted. What X new supply to with produce the the quickly? 110 V The a solenoid? shown in on the when the output coil. primary What current is is the output 0.2 A. the diagram. 12 The diagram using current of a the shows step-up in the and 132 kV transformers calculate an electrical step-down cable are is distribution transformers. 0.2 A. grid The Assuming all ideal: the current from the current that the power station b calculate can be provided + to light industry. + To power source Transformer Transformers 132 kV 25 kV 33 kV 11 kV 230 V Light industr y Factories 159 5 The physics 5.1 of Models Initial LEARNING the end of this topic be able summarise of models civilisations, substances describe the of the describe the the until such the the substances smallest development as the Greeks, simplest used components the idea were of cutting reached. These parts were possible: ‘elemental’ and were built from the atoms. atom ‘plum pudding’ model Geiger–Marsden experiment atom to: The the ideas simplest of you up should atom OUTCOMES Ancient At the At modern view the end discovered of of (electrons). atom. from inside possible As Sphere a positive 19th century heating Thomson the atoms metals the proposed and British could that therefore physicist release the the J. small electrons atom J. Thomson particles were was not released the smallest particle. beam fields, of the that of electrons Thomson original atom could realised was be that deflected the uncharged, by electrons he magnetic were proposed a and charged. model electric As where the the charge electrons in ‘sponge’. (Figure an This atom was were the trapped ‘plum inside pudding’ a positively model of the charged atom 5.1.1). Electrons Rutherford Figure 5.1.1 The ‘plum pudding’ and the nuclear model model A few years students, structure charged later Hans of Ernest Geiger an atom, particles) most passed some were some bounced Rutherford and Ernest using (Figure a asked beam 5.1.2). two Marsden, of alpha When straight through the foil deflected through large of to his research investigate particles fired at with a a thin very the (positively gold small foil: deflection Incident α-particles Atoms in the metal angles foil + straight back. + These results could not be explained by the plum pudding model and + so Rutherford proposed a new model ‘the nuclear model’. + + As most of the alpha particles pass straight through, most of the Nucleus + atom α-particle must be composed of empty space. tracks + As some alpha particles are deflected they must be repelled by + a dense positively nucleus) Figure 5.1.2 The deflection paths which charged contained part of the protons atom and (later most of named the the mass of the of atom. alpha gold particles foil fired at The apparent size of was a 160 the radius nucleus. suggested way similar that to the of As atoms the they was much electrons occupied planets the orbiting greater were known space the than to around Sun. the be measured very the small, nucleus it in The The of Bohr model previous the models electrons and electromagnetic Bohr suggested (shells were or of of atom the why atom they could did not not fall explain into the the behaviour nucleus due to forces. that energy possible the the levels). and this electrons Only could certain prevented only occupy transitions the electrons certain between from regions these falling shells into the Nucleus nucleus. Discovery In 1932 James neutron), (Figure Our of of neutrons Chadwick similar discovered mass to the that proton, an uncharged existed within particle the (the nucleus 5.1.3). understanding of the structure of the atom continues to develop Electrons through the standard model, where the forces binding the around together of are explained, subatomic particles and are quantum described mechanics, as complex where in the the Figure 5.1.3 2 the pudding’ a what charge b what is c what force a the the why do electrons remainder holds the of did most of the an atom electrons Geiger–Marsden without KEY 1 why were in composed of? place? ACTIVITY experiment: alpha particles pass through the gold an online simulator to deflection? some The concept Scientists that in of the The developed modern containing ‘shells’ or matter ancient experimentation 3 the alpha particles directed straight back? scattering the Rutherford experiment. POINTS developed 2 of have? reproduce b model model: Use foil nuclear QUESTIONS ‘plum During The atom SUMMARY In nucleus positions mathematical probabilities. 1 orbit nucleus view of energy composed models and protons is of atoms was initially Greece. of analysis the and the of atom atom through evidence. has neutrons, a central nucleus surrounded by electrons in levels. 161 5.2 Electrons, and The LEARNING At the should end be of this able compare of nucleus periodic components of table atoms OUTCOMES topic Experiments have particle make the electrons, now shown that there are three different types of you that up all atoms (T able 5.2.1). to: T able the the 5.2.1 Properties of subatomic particles properties protons and Particle Location Mass proton in 1 Electric charge neutrons identify the constituents the nucleus unit +1 e of −27 (1.67 × 10 × 10 −19 kg) (1.60 × 10 C) isotopes neutron describe the in the nucleus 1 unit −27 (1.67 between atomic structure periodic kg) 0 C and electron the 0 relationship outside the 1 −1 e table. −19 nucleus in ~ energy unit (−1.60 × 10 C) 2000 levels or ‘shells’ −31 9.11 Neutral atoms protons and have the neutrons same are × number found in 10 of the kg electrons nucleus and they protons. are referred As to as nucleons Nuclear A EXAM nucleus Very specifi c used in phys ics. unde key can be described using the number of protons and TIP neutrons is notation Mak e rstand , sure all The The nucleus. of protons, number) total is number the proton represented of nucleons by number the letter (protons and (also called the Z neutrons) is called the you nucleon of the number atomic nucle ar wo rd s, nucle us lang uage in number (or sometimes mass number) and is represented the by the letter A especia lly nucli de and iso to pe. The the number letter of neutrons, the neutron number, is represented by N A X Z The neutron found number simply number of by is not normally recorded because it can be remembering: neutrons = mass number − atomic number (N = A − Z) Examples ACTIVITY Use a periodic table to A nucleus of magnesium with an atomic number of 12 and 24 study number of 24 is represented by Mg. 12 the patterns in electron arrangement and the A nucleus of patterns chemical elements in behaviour the same by protons and 5 neutrons is B. A nucleus of iron containing 56 represented by Fe. 26 162 5 5 of group. containing 10 represented of boron 26 protons and 30 neutrons is a mass Isotopes WORKED Nuclei of a single element always have the same number of protons Sketch and so have the same atomic number. However, the nuclei may different number of neutrons. For example, most carbon six protons and six neutrons but some carbon isotopes structure of for boron B 5 nuclei nuclei atomic 10 two 11 and contain the have the a EXAMPLE B. 5 contain 5 seven neutrons or even eight. × Nucleus Atoms are with called the same proton number but different nucleon numbers × 1 0 isotopes B Three isotopes of carbon are: 13 C 5 neutrons 14 C 6 protons × 5 12 with: 5 C 6 × 6 × As the proton number identifies the name of the element it is often Both more convenient carbon-13 Nuclei or that to write isotopes in a simpler format: carbon-12, 5 carbon-14. share the same number of protons and neutrons isotopes have electrons: 2 in the first 3 in the second shell shell are 5 referred to as nuclides. × Nucleus × with: 5 protons 6 neutrons 1 1 Patterns in electron shells and the periodic × B table 5 × The periodic table is arranged in order of proton number from left × to right in shells the and (or working energy behaviour outermost of down levels) through and elements the with the periods. periodic the same table Electrons shows number of a are found pattern electrons in in Figure shell. KEY Each for complete an 5.2.1 the energy period in the periodic table represents the filling POINTS up level. 1 Atoms are because The are first 2 The shell can elements second in shell contain the can first a maximum of 2 electrons and so The third shell can equal number of positive charged contain up to 8 protons to negatively electrons. contain up to 8 electrons and the fourth up electrons. to Mass number (A) = atomic electrons. For example, arrangement a silicon is atom has 14 electrons and the number (Z) number (N) Isotopes in of same the elements number nucleus numbers of but of have protons different neutrons. QUESTIONS Fluorine has number of a proton 2 Copy T able 5.2.2. Then calculate and fill in the missing values. 9. T able a neutron electron the SUMMARY + 2,8,4. 3 1 particles an period. 2 18 are there charged neutral there How many does a 5.2.2 electrons neutral Number fluorine of Nucleon Number of Number of Atom atom b Sketch of c In protons, have? the these what periodic placed? arrangement a carbon b sodium c gold d osmium 6 Z number , A neutrons, N electrons 12 electrons. group table of is 11 12 the 197 79 fluorine 76 114 163 5.3 Radioactive A LEARNING short the end of this topic 1896 Henri be able in describe three the properties nuclear of the which Marie radiations in know the sources background a drawer noticed near some that, after leaving photographic a film, sample the film containing became to: exposed. Becquerel you uranium should history OUTCOMES In At emissions of He theorised caused and a Pierre sufficient that chemical Curie amounts the uranium reaction in investigated to allow the was the producing film. other study Over minerals of its the ‘invisible next and few isolated rays’ years radium properties. some radiation. Background There us is always from Nuclear a natural radiation radiation varies small and amount artificial from place of background sources to place. (Figure Some radiation 5.3.1). locations The around level have of more this than weapons twenty times the level of radiation as others. 0.4% Food Air and travel Cosmic 0.3% drink Investigating 13.0% 15.6% nuclear radiation Nuclear reactors Three different types of nuclear radiation have been discovered: Ground 0.1% 16.0% Air alpha (α), beta As nuclear (β) and gamma (γ). 33.6% Medical 21.0% the material it stripped away Alpha Figure 5.3.1 Typical origins cause from particles are interacts with ionisation, their the where atoms some of or molecules the electrons most pass A They are atoms. strongly charged absorbed penetrating and this makes them interact easily By of absorbed by all, as they less materials. easily interact and Beta particles least gamma with the rays counter can particle electrical placing counter, pulse used which different the be passing is penetrating to detect through logged materials a by can G–M ionising matter radiation. Geiger–Müller a between power Source be counter the tube attached source and investigated A to the radioactive safety (Figure tube rules handling sources. Explain why Support these rules are needed when or Plasticine Set handling the different types range to give a large of Fixed distance enough deflection sources. Figure 164 5.3.2 Measuring the penetrating powers of nuclear the tube. Geiger ACTIVITY the the they produces Ratemeter when are are through. Geiger an and penetrating radioactive required a radiation more Investigate of are of strongly. background radiation can radiations 5.3.2). Effect of magnetic and electric fields a Beam Charged particles experience a force when they move a magnetic or electric fields (Figure 5.3.3). The size and direction of depends upon the charge of the particle. Alpha particles mass than beta particles so do not deflect as magnetic easily. Also alpha will curve in the opposite direction to beta particles have opposite charge. Gamma rays are not affected by right to plane because of they at angles the particles enters field have field more radiation magnetic the A force of through the the fields paper as they T able are not 5.3.1 charged. Properties of alpha, beta and gamma β-radiation radiation α-radiation deflected deflected Alpha particles Beta particles Gamma in the by opposite rays the is is direction magnetic to α-radiation field Symbol α β γ-radiation γ by Nature A particle A fast moving the is undeflected magnetic field Electromagnetic b consisting of electron ejected radiation Electric two protons and from the emitted by field positive two neutrons ejected from nucleus nucleus. the energy A between the and negative high α metal plate Beam of photon. γ nucleus radiation β Electric +2 e (charge of −1 e (charge of None + charge two protons) one electron) Penetrating Can travel Can travel power only Note that electric a few metres centimetres in in Blocked a few Can air. travel distance by α- field and are β-particles deflected in passing in air. Figure 5.3.3 Radioactive passing Intensity metal foil. reduced by paper by or Ionising Strongly power ionising is such Ionising as massive highly metal skin. of alpha ionisation and the Deflects particle beta the No electric the particle fields in Why would to alpha chamber we they cannot make see when α or water β particles droplets or γ-rays, condense we on can ions see the formed in careers you than average having a annual of radiation? Radiographer Airline Miner in a hospital pilot a 2 cloud to particle dose tracks lead an higher Although these opposite direction cloud QUESTIONS effect possibly The an it 1 and b field charged Deflects magnetic a lead. SUMMARY Effect a and plating as Some field thick electric Blocked an directions emissions through magnetic air. through opposite long What type(s) of nuclear chamber. radiation: KEY 1 a can penetrate b causes There are three types of nuclear radiation: alpha, beta skin? the c are deflected magnetic There and is always artificial some sources. background radiation from both most ionisation? and gamma. 2 your POINTS by a field? natural d can travel farthest in air? 165 5.4 Nuclear and reactions Unstable LEARNING the end of this topic nuclides be able describe a the nucleus changes decay or time. a nuclear construct nuclear not. them For have example, a emits an nucleus not break has the apart. correct However, many neutrons causing carbon-12 nuclei will or the nucleus is stable a decay over too to many decay nuclide time protons (break but and apart) carbon-14 changing into this over is nitrogen-14 decay occurs new it can be described in these simple terms: nucleus → daughter nucleus + α, β, or γ alpha decay an unstable parent nucleus releases two protons α-particle and a the does decay During forms that and changes. Alpha and means equations nuclear parent nucleus This neutrons too unstable Carbon-14 When describing The and nuclei. reaction stable. protons nuclides makes in undergoing radioactive of to: some are you balance should isotopes OUTCOMES Many At decays two neutrons bound together forming an alpha particle (Figure nucleus 5.4.1). a The daughter nucleus alpha particle nucleus the with resulting is ejected less mass. daughter is a from As the two parent protons different nucleus have element left than leaving the the parent. α-particle The general form of an alpha A X → α Z Proton 5.4.1 Alpha particle beta was EXAMPLE particle not proton 1 is the alpha decay decay equation isotope a is a fast proton originally the present. nucleus from the moving inside and → 84 of After so general form of a beta α + A β-particle created in is nucleus or beta energy. may cause nucleus decay equation is electron additional a different is: → A β + –1 Y Z + 1 be any and decay The released change so may nucleus the by in the to daughter be in an a gamma decay. the number of product of the Gamma protons decay is nucleus excited the or nuclide. general form of a gamma A X Z the changes proton emission decay 0 → γ 0 equation A + X Z is: with state. decay some This does neutrons same emitted The particle said and instantly Beta leave is the parent 166 nuclide an decay alpha not 5.4.2 is neutron Pb energy Figure there a The 82 excess a daughter 0 X An into the decay when 5.4.2). parent. lead? Gamma nucleus the produced (Figure 206 2 in nucleus polonium-210 4 neutron 2 electron the Z 210 A – Y 2 A Po Z + is: for The of to in element What an 4 decay converts into – emission A WORKED equation A Neutron Beta Figure decay 4 in element the as the Other nuclear reactions WORKED Nuclear nuclei process is induced can split usually fission. becomes so into caused The two by smaller hitting neutron unstable that it is nuclei, the nucleus absorbed splits up. releasing and During with then a energy. neutron the fission This in beta nucleus several a decay caesium-137 of neutrons decay reactors chain alongside these the neutrons two are new used to nuclei cause (Figure further 5.4.3). fissions In form for undergoes an isotope barium. 137 0 Cs released equation which to → 137 β 55 are 2 fission Write Large EXAMPLE + Ba –1 56 nuclear and form a reaction 235 1 U 141 + n 92 → 92 Ba 0 + 1 Kr 56 + 3 36 n 0 Fission Target 2 product 1 H H 1 1 nucleus Neutron Fission product γ 3 He 2 Figure 5.4.3 Nuclear Nuclear Figure fission 5.4.4 Nuclear fusion fusion ACTIVITY Stars this produce process (Figure their small 5.4.4). processes, isotopes. An nuclei The mostly energy Sun are 2 the of all isotopes a fusion 1 process merged produces fusing example using of of together of its to form energy hydrogen reaction nuclear is to by larger nuclear form shown fusion. In ones fusion helium below. Most nuclear place in there have formed H + H → 1 SUMMARY Write He + KEY decay equations for the The alpha reactors. Find but naturally and how they out about worked. decay of americium-241 POINTS Alpha decay involves the following. 241 a some γ QUESTIONS complete been 2 1 1 takes reactors 3 these 1 fission nuclear ( release Am) into an of an alpha particle isotope 95 (helium of neptunium parent 137 b The beta nucleus) from a (Np). decay of caesium-137 ( Cs) into an isotope nucleus. of 55 2 Beta decay involves the barium. release 2 Copy and complete these nuclear fission and fusion of adding the missing values for proton number beta particle equations (electron) by a and when a neutron nucleon converts to a proton in a number: nucleus. 235 a 1 U + 92 2 b → + + → ? He ? + 1 Sr ? 4 H 1 90 Xe 54 ? H 1 ? n 0 + 2 n 0 3 Gamma changes decay in the involves energy nucleus. n ? 167 5.5 A model of radioactive decay Radioactive LEARNING Radioactive At the end of this topic be able of describe a simple simulate of a the random an number of discuss the are process but, involved, because the decay a very process large can be fairly simply. effective model of the decay process all of the particles need to be identical so that they each have exactly the objects same random decay involved large a nuclei experiment For to is identical to: modelled decay you number should decay OUTCOMES limitations of chance of decaying. In the model all of the particles (dice) are the identical (Figure 5.5.1). They all have the same chance of decay as model. each other Follow decay when the they procedure and the are rolled. described patterns in the Activity to gather data about produced. ACTIVITY 1 Put 60 2 Make dice sure 5.5.1 A large can be number used radioactive to of If you have If you are 3 Roll all 4 Remove simulate decay. Results table Roll Dice Dice Dice Dice number left left left left 0 60 Try 1 Try These side but Record 2 Try 3 6 Roll 7 Repeat 60 all have or is box so that marked cubes normal you clearly then don’t on colour numbered one in a dice lose them. face. spot then on the one face. number six spot. dice, a handful at a time. do the of dice dice not lose number in a the dice end of dice and like upwards Put the (or showing decayed dice to a the (not the T able number of dice that 5.5.1. dice. recording or spot left table, remaining left up ‘decayed’. them. results rolling no that have you the have number rolled 20 of dice left until you times. T otal 8 60 simple the any six). decayed) 5.5.1 dice using as the tray dice 5 T able a each counts Figure in Repeat the whole process from stage 3 two times to fill in the 180 results table. 1 The more sets of rolls you complete the better the final 2 graph will be. If you do not have time for three sets then 3 you may share results or just use one or two sets. … 9 Work out the total number of dice left for each roll 19 by 20 168 adding the three sets of results across the table. number T able Analysing the You see should 5.5.2 Example results data that the three sets of data are not identical although Roll Dice remaining number the pattern enough of Producing Plot a a label of A should axes graph remain This shows process that, follows when using predictable large patterns. the a is called plot the number the smooth this to roll on y-axis. curve an on the Add a following exponential graph x-axis line but you of the total fit pattern decay must and best of curve. make to the You sure used certain You Eight- T en- sided sided sided dice dice dice 0 6000 6000 6000 1 5000 5250 5400 2 4167 4594 4860 3 3472 4020 4374 4 2894 3517 3937 5 2411 3077 3543 6 2009 2693 3189 7 1674 2356 2870 8 1395 2062 2583 9 1163 1804 2325 10 969 1578 2092 11 808 1381 1883 12 673 1209 1695 13 561 1057 1525 14 467 925 1373 15 389 810 1235 16 325 708 1112 17 270 620 1001 18 225 542 901 19 188 475 811 20 157 415 729 can that you graph be a Six- the clearly. can questions. with be like the after similar. decay remaining curve Analysing The graph spreadsheet the is the graph dice This points. use a scatter number graph. decay numbers, may to determine number use of what rolls. fractions of a fraction Use the of the graph to original answer dice these roll. 1 How many rolls did it take to get down to the original number of 2 dice? 1 How many more rolls to get down to of the original? of the original? 4 1 How many more rolls to get down to 8 You should remaining see dice Limitations Because results do entirely for If the you a a to of halve the limited not dice to The is always number and number of rolls required for the 3.8. model follow smooth use pattern. this you halve or dice pattern may each thousands of were used exactly. find that it you The did may decay not find curve take that may exactly your not 3.8 be rolls time. millions of dice the curve would be much SUMMARY smoother and monitoring the the relationship decay of would radioactive be more nuclei precise. there are generally 1 billions of nuclei. Even though the decay of an individual nucleus a Plot there is a consistent pattern to the number of a dice after a certain types b results the 5.5.2 shows the Do example (idealised) results produced the experiment with 6000 dice. You may use this data graphs if you don’t all of have enough dice. The table also that would be obtained when using eight- and shows the ten-sided How may the rolls dice pattern does number How dice of it take eight- to many halve? rolls for the POINTS number The random model the ‘decay’ decay of of large numbers of dice can be used to to Although pattern in the the of ten-sided dice halve? nuclei. 2 2 same of dice. d 1 types the sided KEY the decay? for results all to c plot for shown by of completing dice table. follow T able of time. in Example showing remaining nuclei three remaining graph is the unpredictable QUESTIONS When decay decay of of a single large dice cannot numbers can. be predicted the Why are after twenty sided the there dice more rolls when six-sided for dice left the ten- compared to dice? 169 5.6 Nuclear decay and half-life Rate LEARNING of Although At the end of this topic be able define the half-life radioisotope activity or isotope in of a find a terms amount of of the model to predict of decay time when nuclide will a specific decay nucleus decays exponentially in is the random, same way a large as the described the is in 5.5. behaviour not affected of by This a regular sample external of decay pattern radioactive conditions can be material. such as used The rate temperature or pressure. remaining Decay of to: dice the you sample should decay OUTCOMES the half-life radioisotope of from constant a a graph. The decay constant decay in in model. the one likelihood nuclei exactly have second. of have the Some very same the a nuclide This is nuclei decaying a exactly of decay same similar are and small is so to very a All constant. chance of likelihood the chance unstable have chance. the large nuclei For and of dice a same all each a will ‘decaying’ high constant the example, decaying nucleus have decay of a while nuclide carbon-14 other have nuclei second. Activity The activity This of is the nuclei left, the Decay A of is of showing gniniamer over a a nuclei sample that is the decay proportional sample of ielcuN graph the period remaining falls but remaining time remaining never showing because number of to rate each the over the the a of decay second. number period (Figure radioactive called zero a but of of As of the the nuclei nuclei. number that are time. activity is nuclei decay the decreases. reaches activity of is approaches nuclei approaches shape radioactive decaying activity graph number A a curves sample nuclei of number rate This remaining curve. of leads The decay to a in a number slows curve as of the which zero. of the sample proportional will to have the exactly number the same of nuclei of the 5.6.1). Time Half-life Figure 5.6.1 A decay curve showing The the number of half-life nuclei remaining of time. over a to decay. a nuclide The is the half-life of time a it takes particular for one nuclide half is a active constant. perod As the the activity half-life activity 170 of nuclei of a is can proportional also sample be to to defined fall to the as half number the of its of length nuclei of original time value. remaining, taken for the T able 5.6.1 Number of half-lives Fraction of sample 0 remaining 1 1 1 2 EXAM TIP 1 2 4 Y ou can find the 1 halflife 3 of 8 an an Figure 45 5.6.2 minutes. measuring means same is the that as shows It time the the the decay important for time time half taken taken curve for note that to of for for it the the to a sample the remaining activity fall from with half-life sample fall 300 from to a half-life can to 600 be decay. to of found 300 or by iso to pe activ ity a decay num ber remai ning usin g of eithe r cur ve nucle i decay cur ve . This is the 150. Activity 45 minutes 45 minutes 45 minutes 600 300 150 75 0 50 100 150 Time/min Figure 5.6.2 The 45 decrease minutes. every Some nuclides indicates that example, Some For 45 have the nuclides very nuclei have activity for activity a radioactive (and number sample of nuclei with a half-life remaining) of halves minutes. dubnium-261 example, in The short are has very a of half-lives, has a fractions unstable half-life long uranium-238 half-lives, very and 27 of a second. very This quickly. For seconds millions half-life of decay 4.5 or billions billion of years. years. SUMMARY 1 KEY The The for half-life the of activity a sample of that of a radioisotope sample to fall to is half the of time the taken Half-lives original The the 4 fermium-253 The half-life fifteen can vary from milliseconds to billions of years. 2 If the longer the half-life the lower the activity (rate of decay) of fraction sample falls 3 a What activity. 2 isotope POINTS has 1 QUESTIONS will three of the remain days. original after days? activity from of 2000 an isotope counts per of second to second in 500 counts per radioisotope. decay process is independent of external conditions. half-life one of the day what is the sample? 171 5.7 Applications of radioisotopes Although LEARNING also At the should end be explain radioactive of this able how topic very useful can medicine and Medical uses be used are potentially how to very dangerous they are archaeology. of radioisotopes containing radioisotopes can be used as tracers inside industry the age They materials can injected gathering into in the target body and organs. move The through tracers emit the gamma be radiation carbon are of bloodstream, using medicine tracers patients. found from in Compounds organic fields radioactive materials describe in you to: Medical materials OUTCOMES which is detected outside the body with a gamma camera. A dating. computer as produces blockages gamma in trace blood being Technecium-99m diagnostic radiation has a long images which short vessels used to (Tc-99m) medicine. is This easily half-life from and the can be detect is information the identified. a blocked most isotope detectable therefore by Figure kidney common decays by not problems 5.7.1 (Chart tracer releasing gamma does and in shows a B). used in gamma cameras. stay such the Tc-99m patient also for a time. Radiotherapy Radioactive cancer rays normal than radiation. radiation Chart recorder A Chart recorder emissions However, the by 10 Figure 5.7.1 A A Chart gamma detected recorder trace from can B be a gas Geiger at a a be destroyed high tumour. The intensity cancer cause by the beam cells cancers. from gamma gamma of gamma receive a high dose and, hopefully, die off. The gamma rays may be and civilian out the applications leaks from a pipeline it can be difficult to fi nd exact pipe body during diagnosis. is of the leak. counter A can radioactive be used to tracer detect can the be added radiation to the leak gas even if and the underground. outside The the thickness of aluminium foil can be measured with a beam of beta medical radiation. too thin Smoke source If the the foil count detectors produces the air fire absorbs current is in too rate rely from a is thick the on gap the detected. then increases the radiation ionises in 172 can can damage Min location recorder so and to 20 When Chart and ionising susceptible cobalt-60. Industrial Min highly radiotherapy directed radiation produced 20 be more B of 10 are cells During is can cells and This (Figure ionising which ions the the off an of drops. radiation. through resulting by rate If the foil is 5.7.2). effect passes created sets count the a current alpha alarm. small is An air alpha gap, detected. particles and Smoke the fall Radiocarbon dating Radioactive Rollers Any organism during but a its contains lifetime. small carbon Most proportion of is the atoms absorbed carbon carbon-14, is a from carbon-12, beta emitter. the a source atmosphere stable When isotope, carbon-14 Metal decays it forms a stable isotope, 14 nitrogen-14. 14 C → 0 N 6 + β 7 –1 Geiger The proportions of carbon-12 and carbon-14 in the atmosphere are tube constant, rays at as the new same carbon-14 rate that is produced carbon-14 by interaction decays. This with means cosmic that all living Machinery organisms the maintain organism dies a it constant no longer ratio takes of in carbon-14 new to carbon. carbon-12. The Once control carbon-12 Figure nuclei are stable but the carbon-14 nuclei continue to decay and 5.7.2 A beta ratio of carbon-14 to carbon-12 decreases over control the can activity ratio can measure of a then the sample be used of to ratio of carbon find the two taken out how isotopes from long by the remains. organism has a half-life of 5700 years and so This the which died 5700 years ago would have remains half of as an organism that died today. For out some as example, 2000 that is an years the important old. plants techniques However, used can religious be to artefact radiocarbon make used with years ago. it died originally dating only uranium to on the the find the to Shroud years billions of ages using T c-99m. of Why a 2 A would sample an alpha particle emitter not be suitable taken from an arrow is found by safety in place? for use as radiocarbon EXAMPLE ancient found to piece have to carbon-14 of a modern Estimate piece wood be rocks tracer? of What to Similar QUESTIONS medical need showed ago. is 1 diagnostic carried be An SUMMARY the are T urin WORKED formed of foil. much thought 700–800 of that precautions Shroud aluminium an out carbon-14 or ACTIVITY procedures organism used died. Find Carbon-14 be thickness measuring biological ago can the time. paper Scientists source so to the to rollers The of the of an cotton activity, decay, ¼ cotton age of cloth of due that sample. the ancient cloth. cloth has decayed over 1 dating to have an activity of that for a modern piece of 2 the half-lives and so is 2 × 5700 8 same KEY 1 In medicine wood. Estimate radioactive cancers Radioactive a 3 of the age of the =11 400 arrow. years old. POINTS treat 2 type material Radioactive organic materials or materials are used as tracers and to (radiotherapy). detect dating can flaws be in used to measure the thickness of it. techniques can be used to find the age of materials. 173 5.8 The release nuclear The LEARNING law the should end be energy conservation of energy states that energy cannot be OUTCOMES created At of of of this able topic or realised you given to: destroyed. that mass Energy was must equivalent be to conserved. energy and Albert this Einstein relationship was by: 2 energy calculate the energy = mass × speed of light released 2 when mass nuclear changes in or a describe the advantages disadvantages nuclear associated with power. Whenever in mass. In nuclear to energy mass and this a change means (splitting) of these mass in that a large smaller change EXAMPLE energy mass there can be nucleus nuclei leads to is a is an associated transformed is split less into than large two the release change into energy. small original of energy. 1 TIP How as is also fission WORKED give mass there This The nucleus Mak e mc and nuclei. EXAM = reaction Converting E sure adva that ntag you es can this much energy well 1 U + 92 disadv anta ges released during the nuclear fission shown in equation? 235 as is 144 n → 90 Ba 0 + 1 Kr 56 + 2 36 n 0 of −25 nucle ar powe r . mass of uranium-235 = 3.902996 × 10 kg −25 mass of barium-144 = 2.389897 × 10 kg −25 mass of krypton-90 = 1.493157 × 10 kg −27 mass of Calculate a neutron the = 1.674927 difference between × 10 the kg starting mass and the end mass. Δm = mass before fission − mass after + 1.674927 fission −25 Δm = (3.902996 × 10 −27 × 10 ) kg −25 − (2.389897 + 2 × 10 −25 + 1.493157 × 10 −27 × 1.674927 × 10 ) kg −28 Δm = Now 3.1927 convert × 10 this kg loss of 2 E = mc mass to energy. −28 = 3.1927 × 10 8 kg × (3.00 × 10 −1 m s 2 ) −11 = Although there very large Once how the are this to Figure 5.8.1 A nuclear energy vast of 10 of mass bomb. enough into The energy than any seen by one nuclei energy understood convert J released number amount was × in could provide before. decay gram began The conversion to nuclear every potentially scientists energy. explosion greater 174 the a nuclear release 2.8734 of an first a to of be is very matter small so a released. develop application amount explosion small, and ideas of of about the idea matter thousands of was would times Nuclear fission power stations WORKED Nuclear fission power stations use the thermal energy released water into steam which drives turbines. These are then used much generators which produce During fission this is caused process by several a neutron other colliding neutrons are with a large released and nucleus. these used to neutrons. split This other nuclei, process is releasing called 1 would gram a more chain energy reaction and (Figure yet completely of be matter converted into energy? can 2 E be if electricity. were Induced energy to released drive 2 to How heat EXAMPLE = mc = 0.001 kg more × (3.00 × 5.8.2). 8 −1 10 m s 2 ) 13 Key components Fuel: Uranium Moderators: or of a fission plutonium is = reactor used inside canisters called fuel rods can be This 9.00 is slow down the neutrons so that they by chemical absorbed Coolant: and by This carries There are national T able the fuel removes it to a for governments cause the heat arguments 5.8.1 and energy 20 000 than that tonnes of explosives. fission. thermal and energy for against the consider and against For from the reactor core use (T able the use of nuclear power, which 5.8.1). of nuclear power Against Running Only a costs small are low. amount of Commissioning waste and produced. J exchanger. must Arguments 10 more released These × Large decommissioning (dismantling) supplies of nuclear fuel Nuclear available. (building) costs waste radioactive are Figure high. 5.8.2 A chain reaction contains isotopes. ACTIVITY No so carbon no dioxide is contribution produced to Waste Accidents global products nuclear can be used in weapons. warming. such as occurred Research the advantages disadvantages Chernobyl and Fukushima large a fusion in form energy larger possible been at is also ones. very This high reproduced released process when very occurs temperatures QUESTIONS 1 What nuclear 2 How much 2 H + chain energy 3 1 is → in and small the nuclei cores pressures, of combine stars which and have is how only not Find arguing construction station out what up yet long the on your went and of island. it wrong Fukushima will take to contaminated in and clear land. in this fusion 1 process? Mass can in be converted nuclear into reactions n 0 according = 3.344494 × 10 to the relationship 2 −25 hydrogen-2 POINTS energy + 2 of against Chernobyl to 1 He 1 (mass report reaction? released 4 H or power KEY a a sustainably. SUMMARY is Write stars Nuclear nuclear areas. for Nuclear of can energy. contaminate and in kg, mass E of = mc −25 hydrogen-3 = 5.008267 × 10 kg, mass of helium-4 = 2 Nuclear power can provide −25 6.646476 × 10 kg) large 3 A sugar cube equivalent to has a this mass? mass of 12 g. How much energy is amounts energy and but of there useful are risks disadvantages. 175 Section Section 1 The 5: Practice activity measured the a 5 of a over Practice exam sample a of radon-220 time as is 3 shown in a During to table. Plot of activity against time for the sample. b Identify Most the anomalous result. A Use the graph to find the half-life Geiger–Marsden the structure results of the through small were c the explore following graph questions questions of period exam were alpha the gold by experiment the atom, the noted: particles passed directly foil. percentage deflected of of the angles alpha particles between 0 and of 90 degrees. radon-220. d Predict the time it would take for A few alpha angles activity to fall to 10 counts per particles were deflected by the greater than 90 degrees. second. a For each describe the conclusions about −1 Time/s Activity/s the 0 500 20 389 structure made b Why of based on was gold was the the atom this used that can be evidence. as the material for the foil? 40 302 60 235 c Why evacuated 80 182 100 150 4 Electrons or 120 110 140 86 160 67 180 200 What of 40 iii Calcium iv Neon table different Number hydrogen-2 showing Z hydrogen-3 Which two of similar the elements chemical 176 nitrogen-14 d chlorine-35 will Nucleon A Number of neutrons, an levels N Number of electrons of have properties? Electron configuration 1 3 (tritium) c energy the number, in the elements? (deuterium) b in nuclides. of out configuration following Sodium this found electron the ii protons, a the of 52 the Nuclide are Magnesium complete components atoms i most and is atoms b Copy carried shells. a 2 in experiment chamber? 7 2,8,7 the 5 Which of the isotopes listed in the table: 8 This graph argon-39 a are isotopes of the same b contain the same number of nucleons? c contain the same number of neutrons? d contain the same number of protons? shows and a the activity sample of of a sample of silver-108. element? a Determine the half-life of argon-39. b Determine the half-life of silver-108. c Which sample has the higher initial activity? 35 40 36 Cl Ar 17 Ar 18 38 18 36 Ar S 18 Activity/Bq 37 5000 Cl 16 17 4500 Silver 35 36 S Cl 16 4000 40 Argon Ar 17 3500 18 3000 2500 223 6 The nuclide thorium-223 ( Th) 2000 undergoes 90 1500 nuclear decay isotope of by alpha emission to form an 1000 radium 500 (Ra). 0 a Which nucleons are present in an 0 alpha 100 200 300 400 particle? b 500 600 700 800 900 1000 Time/year Write a alpha decay. decay equation representing the 9 Write decay equations for the following: 107 a The alpha decay of ( tellurium-107 Te) 52 7 This graph shows the relationship between into the proton number (atomic number) and an isotope of tin (Sn). of potassium-40 the 40 neutron number for all of the stable b nuclides. The Describe the relationship for nuclides of decay ( K) into 19 an a beta isotope of calcium (Ca). a 90 low b proton Describe higher N number the of < relationship proton (Number (Z number (Z c 20). for > The gamma decay of ( strontium-90 Sr). 38 nuclides of a 10 20). A single all of a neutrons) nuclear the Write idea on b a of few reactor could requirements paragraphs building a for provide Trinidad. supporting nuclear power the station Trinidad. Write of fission electricity a few building paragraphs the power opposing the idea station. 126 11 Nuclear the radiation thickness of manufacture. can be used materials Describe to during how the measure their thickness 82 Protons = neutrons of a metal radiation foil so can that a be measured constant using thickness beta can be 50 maintained during the manufacturing process. 28 14 6 6 14 28 50 Z 82 (Number of protons) 177 Glossary A C Absolute zero The temperature (0 K temperature all lowest or of possible −273 °C). the At thermal D Celsius this scale temperature energy has of water of water. A centigrade with and 0 °C 100 °C at at scale the the for Decay freezing boiling point constant decaying per removed from a material. a Absorb an To take increase in in radiation. This results in temperature. Centre of the of gravity weight of The an point object at which seems to all velocity. The The unit rate of of change acceleration of is Chain metres reaction neutrons are During released. nuclear second squared transfer a = Δv/t cause further fissions These of fission neutrons nuclei and The material. Accuracy An accurate measurement set which of is close to the measurements of true a value. quantity neutrons causing an energy if their mean is close to do not but share which them. mass per = unit mass volume ÷ of a volume. in variable response A to variable an that independent during an experiment or release. A investigation. can the that current between Density be Charles’ law For a fixed mass of gas the Destructive accurate a increasing variable of nucleus can is rate one more a release changes even circuits through energy Dependent ). (m s of act. −2 per T wo connection Density Acceleration chance point Decoupled been The second. true volume is proportional to the two value. temperature if the pressure interference When absolute is waves meet out of phase (e.g. a kept crest meeting a trough), the waves constant. Activity sample (the The is activity the number rate of of of a radioactive decay nuclei of which the superimpose nuclei decay each Compression pressure in a An area sound of higher air and displacement partly out at cancel that each other’s point. wave. Diffraction The spreading of a wave second). Conduction Alpha particle consisting neutrons of (a A two radioactive protons helium emission and energy two pass nucleus). also through energy radiation The emission of transfer a via energy of material though conduct transfer Alpha The thermal when vibrations. free electrons more when atoms an Metals which The and two emitted neutrons particles from form a a nucleus. helium Conductor electricity through depends can quickly. meter used to greatest current (in ampere, measure A) in the A (or material thermal that The maximum a circuit. waves to flow Diffuse surface distance of meet crest), interference from its rest position in a of incidence The angle incident ray and the When phase waves the of reflection reflected ray and distance Average speed The the The measured speed = gap; when the the these are (e.g. size. a crest superimpose reflection which travel in is Reflection not smooth. different from The directions a reflected can be and so no seen. meeting and amplitude at that Diminished point. when An image reduced to original compared the in size object. force objects A are force in which direct acts contact when with each Dispersion frequencies The of spreading light of caused different by angle difference White between variable A variable that in light their can speeds be in a the dispersed medium. by a prism. must normal. rate over occurs between the normal. be Average of between Control the size two other. Angle the past diffraction relationship and or vibration. two an of gap a Contact Angle in the increase particle the a allows energy) it. Constructive a Amplitude amount diffraction same image electric on wavelength rays A The through two nucleus. Ammeter passes edge. the protons it of a change period distance of travelled of time. ÷ time or kept constant investigation relationship during in an order between independent variable Convection current the to experiment to allow dependent be Displacement the origin and determined. (starting direction. The distance point) in a Displacement is from an particular measured in metres. taken. of B particles transfer fluid. Background radiation The A of the surroundings. This rocks, radon gas and radiation A of a from cosmic radioactive fast-moving the A a gas whole body temperature of a in law is For a fixed inversely a 178 if the a particular of a fluid as it heats graph instant. graph A graph up the changes in displacement The line on a temperature– an object gradient of over a a period section of of the time. graph The is that shows how a substance speed of the object at that the time. down. angle The angle of incidence at To spread surroundings. out Thermal into the energy an incident ray will be refracted into the surroundings where dissipates it cannot be throughout at a to mass Current of gas proportional is the boundary. used to do any additional work. specific point). temperature at A particles liquid to kept The an rate of object. flow The of charge current is Distance measured the amperes where 1 A the of volume wave the electron in pressure density curve graph through Boyle’s a of emission from liquid boiling in graph positions the Dissipate state happens of (the the by of parallel the caused nucleus. change which is the contracts. which into throughout current representing rays. Critical Boiling Displacement–position the showing cools ejected energy by Displacement–time time consisting movement caused radiation Cooling Beta thermal The fluid radioactivity includes or from a convection changes in within constant. charge per second. is the transfer of 1 C point. The How SI far unit an of object distance moves is the from a metre. Glossary Focal E length where Echo The reflection of a sound The parallel distance rays from parallel to a the lens to I optical wave. Impulse axis will be brought together caused Efficiency A measure of how effective a lens) or seem to come from by is energy at transferring usefully. or Efficiency useful energy ÷ total energy in. Efficiency can as a decimal or as a plane The plane, perpendicular to 0.4 or 40% surface axis, were the principal focus ray such Free efficient). electrons which limit The point beyond which will no longer return the Metals to its outer form when the force on the electrons a structure object. The as a ray which strikes a mirror. These are free free to variable A variable which is in in an experiment to see how that move change atoms. electrons affects the dependent variable. are original responsible length an lies. a between spring on percentage changed Elastic momentum Ft Independent (e.g. in acting a be lens expressed = Incident Focal out change force lens). transforming = a (diverging Impulse device The (converging spring for electrical conduction and Induce To transfer magnetic or electrical is most thermal conduction in metals. properties without physical contact. removed. Frequency Electromagnetic Related to electric pass magnetic a point complete second. The waves unit Induction of The electromotive production force (a of an voltage) in a wire by −1 Electromagnetic an many each fields. frequency by How and relay Switch operated is s which is also called hertz a magnetic field moving relative to it. (Hz). Inertia The change its reluctance of an object to electromagnet. Friction Electromagnetic spectrum waves properties. with similar A set two of amount of coulomb battery. of A Electrons = as it to The which each passes found The releasing through acting opposes between movement. planets, joining energy. releases of small Fusion energy is in the process charged energy particle. levels that of radiation electromagnetic High to Large objects, amounts speed up released thermometer measures the the infra-red of or to from a object determine at a A intensity radiation Instantaneous frequency radiation large difficult object Gamma or have Infra-red Sun. G in are motion. such inertia slow as and down. nuclei the a E/Q negatively are which force so (e.m.f.) provided charge e.m.f. Electron force energy contact surfaces Fusion Electromotive The the speed particular thermometer and frequency emitted by an temperature. The speed moment. of an Measured −1 shells can around be the ejected nucleus from the of an atom nucleus or Gas beta in nucleus. metres per second equation A relationship Insulator between A poor Electrostatic precipitation Using a of pressure, a fixed volume amount and metal grid to induce charge onto pV/T = heat or of an ideal gas. smoke from particles the Emit To air . that Used give radiation so or in out they can power be station radiation, e.g. Gas thermometer A uses the a Evaporation infra-red pressure from to do work. energy A process the where surface of a energetic the particles average escape interference of gas to indicate patterns formed energy of potential associated a are experiment with gravitational energy an object field. The same liquid. This reduces change it is in Inverted it is as An upside potential energy can be Clear destructive interference the in Young’s overlapping two-slit waves have image is said to be inverted if down. found Ionisation and the relationship ΔE = so particles temperature from Gravity two The objects force due of to attraction their The removal of an electron mgΔh an atom creating an ion. between Atoms of the same element mass. (having the waves. The when The first the the pattern and frequency. Isotopes the The constructive particles liquid. from reduce of temperature. gravitational most which the capacity the chimneys. inside escape thermometer light. The by removed Gravitational Energy pattern constant. formed the same number of protons) that liquid. Greenhouse Extension spring (or applied The other to it. spring is increase object) For proportional the of electricity. temperature Interference charged of conductor decay. the in ). (m s during to a in spring the size extended length when the of a of a force thermal is extension the beyond force the to is effect energy greenhouse radiation by the gases emitted The by capture atmosphere reflecting the have of numbers of neutrons. due infra-red K Earth. Kelvin unless limit different on of the scale A temperature properties of ideal scale gases based and the H energy proportionality. Half-life The time taken for the activity of of Kinetic a radioactive sample to fall to half of with initial value. Hard magnetic energy the movement 1 E The splitting accompanied by a of a release nucleus of material which be induced using mv 2 magnetic properties theory Heat A liquid or gas. The particles are capacity move to flow. past each other allowing the The amount of energy The of kinetic solids, particle theory is liquids model used and of the to gases. explain the able which to The well. neutrons. behaviour Fluid associated object. retains Kinetic can an energy. its Fission energy of 2 = k A The its F Fission particles. can be stored in a body per degree changes of state and pressures within fluid kelvin (or Celsius). gases and liquids. 179 Glossary Momentum L an Laser A device which produces a object as intense (light with beam a of single monochromatic heat change the The the property of required P relationship Parallel = mass × effect inside A a current-carrying magnetic field wire are the parallel Pascal a force. that This is the current have can junctions diverge or at merge circuits. will to experience Circuits velocity. which Motor wavelength). energy by light placed Latent movement narrow momentum and The defined motor (Pa) A unit of pressure. The pascal effect. −2 to a liquid state or of an liquid to object a (from a solid is heat of fusion The Penumbra energy to melt a substance. The of energy is released when electric to of vaporisation vaporise a liquid The a during an This carry charge field which will affect causes Period The oscillation other time of a it takes system. for It is one complete measured in liquid energy is to a gas). released The when same the An back into a uncharged particle found in Photon A packet of energy carried by it nucleus of an atom. light. The higher more energy the frequency of light the amount each photon carries. vapour Neutron condenses seconds. energy (change the of Electrons charge. objects. Neutron from shadow solidifies. heat required charged electric charged Latent partial the an substance The same negative quantity N m eclipse. Negatively required to solid). N Latent equivalent number The number of liquid. neutrons within a particular Pitch nucleus. A high frequency sound has high pitch. Laterally displaced When a ray passes Newton through a rectangular block of are material it is refracted on (N) entry vector the it ray but leaves the travelling shifted block. in the (displaced) This unit quantities. of force. This Forces means that the and direction when The transparent results original which they act is significant. Non-renewable from will its eventually An run energy out or source become which path. Lattice vibration positive Potential volts. cause particles in a solid The vibrations which cause of Normal the A line at right angles to surface. thermal Angles refraction are of all incidence, measured reflection relative to dependent resistor which depending to. In but on bright in dark resistor changes the light light, conditions it have they carries difference A by a unit potential 1 (p.d.) charge, difference coulomb of The of charge energy measured to 1 volt in will joule of transfer energy. Power The measured rate in of watts transfer (W). P of = energy E/t (LDR) Nuclear resistance level LDRs proton and this normal. A A the 1 conduction. Light charged charge. transferred very scarce. original Positively in direction sideways in is exposed low have resistance very Related atom. Nuclear nuclei are to the energy split nucleus is (fission) of released or Precision an when merged (fusion). The degree to measurements agree. A measurements will be all which set of very repeated precise similar. high Nucleon A particle found in the nucleus. Pressure law For a fixed mass of gas the resistances. The Limit of which a proportionality spring can be The limit stretched possible remaining proportional to acting on the spring (still protons and pressure is absolute directly temperature if the to the volume is kept number The total number of the and neutrons in a Primary nucleus. cell A cell which cannot be obeying recharged. Hooke’s proportional constant. protons force are the Nucleon extension nucleons neutrons. to with two law). Nucleus The central part of an atom. Zinc–carbon cells are primary The cells. nucleus Linear magnification A comparison size of the image to the size of image Linear ÷ magnification height of = the volume height all A object. wave Nuclide motion where of the particles are small amount of of the the atom but Principal contains mass. centre the lens. A particular carbon-14 type is a of nucleus. different direction of wave motion. parallel Sound Principal rays nitrogen-14. that is M A ray centre which focus enter principal For a nearby magnetic magnetic axis The passes central through point the of a lens. the rays point seem exerts a materials force by does not change a Origin or turning measured in N m. is as the defined perpendicular the The starting central point point in an of a movement in (pl. foci) The converging are through point lens brought lens The effect of moment force distance a of multiplied to the force a by pivot. force the Oscillation a point. Most sinusoidal. the A regular to at which parallel together to by the principal the focus pattern of object a sine the original parallel through. of moments to the or the For an clockwise object to anticlockwise moment is moment. oscillation. movement oscillations The which pass equilibrium, equal studied particle wave. around a are direction moves in wave. ripples light a 180 runs perpendicular optical be field. The which and path. on producing from to Propagation Moment a diverging Principle centre magnet line O wave. is A A lens a Optical Magnet a to lens. longitudinal of For nuclide the the axis the the to oscillations very of example, Longitudinal a the nearly object. only of of the occupies The of on rays lamp. a The wave spreading is said to propagation. pond For propagate propagate in all of energy travel in example, in a circle directions by the the and from Glossary Proton in the A positively nucleus of charged an particle found atom. Specific energy of Proton number The number of the nucleus of an capacity one to kilogram raise of a The the amount of Umbra temperature specific material during The an in complete darkness Unit A system of measurements of kelvin. atom. quantities. Specific to region eclipse. by protons one in heat required latent change the heat state The of energy 1 kg of a required is material. the For metre. example, Scientists International (SI) of the use unit the for length Système Units. R The specific latent heat of fusion is the Unstable Rarefaction An area of lower pressure in energy required to change the state of sound wave. of the The Real image An image which can specific onto a screen. Rays of from latent a solid heat of into a unstable equilibrium An if, object when it is is liquid. vaporisation disturbed, it falls Upthrust The over. is be the projected material equilibrium 1 kg in a light energy required to change the state force acting on an object pass −1 from through a real liquid to a gas. Unit is J Kg (or submerged or floating on the surface of image. −1 J g °C Rectification The conversion of ). an current into direct liquid. weight Spring alternating a constant The force required If the of the upthrust object it is less will than the sink. to current. −1 extend Reflected ray The ray which leaves a or other reflecting index The of light in a vacuum of light in a material. ratio (or of air) the to speed the position The larger the is N m An object is in stable slower light if it after returns being to the Van original disturbed de device (pushed). form A numbering format A travels large or small numbers by produce In standard (VdG) generator generate VdG charges generator can A by differences large generate enough to scientists in engineers. to used potential form large sparks. numbers material. b are Regular reflection The reflection from a represented where a is a in the number form a × between 10 1 Vector , and flat produces a surface clear Renewable An such as a mirror which and is image. energy source b is used an to integer. For represent example, 3.4 × 10 an inexhaustible inexhaustible) which (or force, displacement graph the velocity a Thermal current. R = Resolution opposition to an conductivity material The rate at can conduction. A conductivity will The smallest increment can detect. the The be resolution of a ruler resolution of a referred heat energy or internal with energy) the is particles The sum of the or an object. As forces within direction of the directions (e.g. forces must forces must be acting be subtracted in a thermistor each other). or the area distance below the line The the image rays of of light an object appear A virtual image cannot be to projected a screen pass as the rays through of the light never image. A meter difference used (in to measure volts, V) the between points in a circuit. The voltmeter must temperature. be Transformation of the resistor. two form by changes taken the gradient) image potential of opposite from the (shown Voltmeter Temperature-sensitive resistance with account The acceleration material. vectors The the an forces are find object x-axis). and onto the to the the movement thermometer of used (on displaying an graph). actually force by which come. associated time of to be Thermistor on the from may be graph thermal Virtual (sometimes of A y-axis) quickly. a object into high energy 0.5 °C. Resultant acting with transfer For arrangement may material energy velocity. which of instrument and include by V/I energy 1 mm. energy travelled as example, thermal electric Thermal measuring transfers (on period (shown The magnitude vectors and the graph a Resistance with example, Velocity–time over effectively supply. quantity For 34 000. T has A direction. 10 4 smooth, the Graaff used friction. speed and the Unit the for index metre. equilibrium Standard refractive 1 surface. equilibrium Refractive by V Stable mirror spring a energy energy can energy into be A change into from another. transformed For one placed have very in parallel high in the circuit and must resistance. example, from electrical W S Scalar A quantity magnitude. include For mass which example, and only Transformer has scalar quantities to change supply. energy. the Secondary cell A cell which can A A lead–acid cell is a the voltage while the oscillations A material which certain a lamp. which of an is alternating transformer a pulse Wave train other in an electrical placed after each A wave are magnetic magnetic A that loses its of wave The propagates. For on of points on motion where perpendicular to the the crest example, a a ripple all wave on a of as the it points pond. the The peaks distance U the symbol is λ (or between troughs) measured in in a wave. metres and has (lambda). circuit. material properties series propagation. Ultrasound Soft continuous Wavefront successive one oscillation. transformer conditions. Components A Wavelength of single voltage. Wavelength Series A pulses. increases step-down Wave used will direction in voltage step-up Transverse conduct device by secondary cell. Semiconductor A energy be reduces recharged. light easily. the range used in of High-frequency human depth sound hearing. measurement above Ultrasound and is Work The measured transfer in joules of energy. Work is (J). pre-natal scanning. 181 Index Key terms listed in are the in bold and are also causal cells glossary relationships 120–1, photovoltaic A Celsius absolute a.c. see zero alternating acceleration in second accuracy acid rain 170, 100, see 24–5 171 in also 68, 160, to d.c. of transformers 122 ampere 112 gates 132 78, 98, 99 of incidence of reflection results principle atmosphere 45, 84, 88 84, 88 47 69 163 160–2 speed 17, 167, 41 175 114, circuit diagrams motor 164, 165 125 current of energy 35, 41 of circuits d.c. decay decay 138–41 in in thermal 124–5 emission circuit 70, 71 45, 76, 138, diagrams, diffuse diodes 141 direct 60 112, 66–7, 6, 9 interference 96, current 97, 100 116–17 100 84–5 images 91, 92–3 136–7 (d.c.) motors 130, 136–7 154 86, displacement 72 37 3 reflection dispersion 113 2, 124–5, d.c. 168–71 energy 46–7 circuit diminished 98 170 166, variables units diffraction 116–17 54 170 geothermal 3, 173 current radioactive destructive 86 radiation electrical 149 150–1 characteristics 169, dependent 28–30 conduction 22 direct derived condensation 134 148, effect constant and 123 momentum waves motor curves decay, 122–7 116–17, compression fuses wires radiocarbon see density light 148–9 150–1 of current-voltage dating, 36 and 152–3 from D 116–17 112–13 resistance effect ratings the 124–5 123 134 126–7 in current 127 122 current-carrying 116–19 analysis field measuring and potential limit current magnetic 118 118–19 circuits characteristics induced 56 breakers 112–13, resistors 60–5 112, law circuits I-V 110–11 circuit and 162, in 109 carriers computers 106, 16, radiation components, 46, air number average lightning circuits 115 88–9 112–13 complex 112, nuclear 97, colours 8 Archimedes’ 54, in collisions, 139 angle atoms circuits coal angle also in logic 156–7 state symbols 138, anomalous 166 130–1 154–5 change ammeters amplitude 164–5, 136–7 153, electricity (A) of 106–7, chemical (a.c.) 37, gravity charge angle current reactions Charles’ 69 current generation mains 69 critical 51 change on atmosphere particles atomic 27 scale of charge converting see chain 111 alternating AND current 26, conditioning alpha centre 5 currents air 23, law 56 36 activity air 51, 6 130 87 10, 22 B conductors background radiation 47, 112 122–3, units of 120–1, particles equivalence 164–5, systems waves dissipation model 174 of momentum 28–9 interference 97, distance 100 diverging forces lens 12 waves 10, energy 22, 35, 25 lenses formula 90, 92–3 95 76 161 E control boiling 60, Boyle’s law variables 6 61 Earth convection currents 68–9, 45, 73, eclipses current earth lenses 90–1, wires vision 132–3, 135 94–5 echoes in 82–3 112 47 converging 146–7 72 56 conventional buoyancy 100 93 C eclipses cooling callipers 82–3 70–3 4 eddy convection cameras 83, 95, currents in currents and evaporation and dioxide 36, latent heat 73, 182 19 curves potential energy 41 62–3 149 coulomb batteries limit 62–3 elastic 72, 157 60 73 cooling cars 43, 136–7 elastic carbon 156 68–9 97 efficiency capacitors 78 22–3 78–9 of 37 continuous Bohr for 115 138 contact biofuels 40–1, 166 constructive binary energy 130 conservation beta 125 graphs 2 mass batteries 124, displacement–time 57 conservation base graphs 164 ohmic balloons displacement–position for (C) electric charge electric current see charge 112 120–1 see current 40 Index electric in fields waves electrical 108–9, 77, energy 34, transmission meters electrical power also electricity mains 122 energy sources energy transfer forces electromagnetic induction generators electromagnetic gamma see infra-red 70–1, see speed radiation gamma 73, eyes of 80, 118 electromagnetic spectrum 16, 17 gas 72 gas 31, 110, decay in 80, waves 174 curves lines force (e.m.f.) 36, induction in beta 46–7, plane 152, 160–1, particles charged circuits in electrons induction and 90, 92, de 57 of state in 68, in 118, 45, 66, 9, 62, charged between magnets 107 fields curves 73 24 169, characteristics transfers 144 170 gravity 106–7 12, 40, 106 78–9 124–5 strength potential 12, 38 energy 38 35 39, 40, 41 12 greenhouse 119 law of 78 22–3, 24–5 field transformations 108 160 37 36, velocity–time objects 64 55 experiment 22–3, gravitational between Hooke’s 60–1, 69 energy gravitational 12–13 electric 53 56–7 effect 73 18–19 112 and immersed and impulse objects 46–7 H 152 ionisation Van 94–5 90 half-life 28 170–1 80 inter-molecular in I-V 163 in free liquids 166 objects 112, 36 displacement–time 155 162, 166 displacement–position gases; length 164–5, 6–9 decay 68 focal friction in graphs 151 46–7 focal in and 175 rule 106 atoms as 174, 118 forces electrons 167, left-hand also 64 175 172 warming gradient 120 116, 54, global 114 see in 152 26 80–1 fluids circuits 148, motion 148–9 floating in 146, 70 34, 62, thermometers currents 18 of of 56–7 change 169 149 produce laws pressure Fleming’s cells natural gases 81 law 137 167, 81, emit geothermal fission electromotive rays equation gas 68 149 first electromagnets heat nuclear tracers 93 field radiation 175 135 latent gamma F relays electromagnetism 36, rectification G 86 electromagnetic 37 Geiger-Marsden electromagnetic infra-red fusion, 106 115 fuels 134, fusion, 166 55, extension 81 114, 43 60–1, states exponential light fuses 42 76 15, explosions 80–1 39, gas, expansion rays full-wave 4–5 excited 152–3 154–5 nuclear 36–7 40, 73, biofuels 163 66–73 34, evaporation 12 12, 36, 114–15 waves errors fuels 43 161, movement 55, equilibrium 113 electromagnetic a.c. efficiency by 106–11 34–5, charge friction energy 40–1, (fuels) and thermal see of electron and electrical 115 132–5 static light levels, 175 36–7, 35, energy 114–15 sources electrolysis in 153 power 155, energy wasted/lost 42 115, 156 electrical of transformation transformations see transformation 165 80 Graaff generators half-wave 54 rectification 136 108, in laws of motion hard 26–7 magnetic materials 144 109 electrostatic e.m.f. see precipitation electromotive and magnetic and moments 34, of pressure state 62, of 40–1, equivalence decay thermal capacity kinetic fuels energy 82, 34, 14 internal energy 36 50 35, transfers 38 of 35, 43, 36, 73, transformations 68 115 40, 66, see 112 also thermal 41 energy heating 60 42, 55, point 62 and change 68 rate of of state 62–3 79, 98 70–1 174–5 see of as 97 potential see energy alternating current 130, 155 hertz (Hz) 130 energy of transfer of 34, electrons of potential 34 generation 58 frequency nuclear 55 electricity energy freezing light effect work freezing see expansion in 166 free heat 44–5 174 fossil gamma kinetic 73 energy 115 and and sinks heat/heat 64 turning conservation and heat 14–15 42 change mass 150–1 58–9 force and and 146, capacity 111 and energy fields heat mains electricity 132, 133 homes, ring mains in 132–3 transfer on oscilloscopes 99 Hooke’s law 18–19, 38 183 Index human reaction human vision humidity times and L M lamps, evaporation hydroelectricity hyperopia 4 93 61 filament lasers 37 125 magnetic 82 latent 93 124, in heat 62–5 forces latent heat of fusion latent heat of vaporisation 62, 64 62, 64 fields 146–7 electromagnetic from radiation 80 150–1 in induction in light 152–3, waves 154–5 77 I lateral I-V characteristics displacement lattice images vibration and 66 nuclear radiation transformers 165 use 156–7 induction 144–5 85 law in 86 124–5 lenses 91, of conservation momentum magnification immersed impulse of objects magnets 144–5 94 laws of motion laws of reflection 26–7 electromagnets 149 46–7 LDRs rays independent 84 magnification 94 (light variables 6, lead-acid cells current 123 magnifying left-hand rule, mains glasses electricity 94 132–5 9 Fleming’s measuring 151 mass 4 4 and energy and kinetic conservation and momentum 174 152–3 lens charging by formula 94–5 energy 38–9 107 lenses induction, resistors) 120–1 107 length, induction, dependent 84 charge induced magnetic 28–9 28 incident induced of 92–3 electromagnetic 90–5 28 152–3, light 82–3, 96–7 in second law of motion 26 154–5 diffraction induction, magnetic 96 infra-red 86 radiation 70–1, energy 34, measurements 35, 82 the infra-red greenhouse effect thermometers instantaneous speed 66, 67, on melting 97, in lenses melting lenses see 84–5 internal heat thermal resistance a 86, as wave of 122 mixtures micrometers 59 4 microprocessors resistors microwaves 73, 110 81 123 rays mirrors, 82 reflection moderators at 84, 85 175 lenses 90–1, formula 92–3 molecules 94–5 54 moments 14–15 125 164, magnifying glasses 94 momentum 28–31 165 164, of with 84–5 Moon 82–3 165 refraction conduction 81 91 113, radiation 77, 50 reflection 112, of 86, 87, 88 motion, Newton’s laws of 26–7 113 total isotopes electrical method energy in ionising meters, 90 88 dependent lens 80, 63 124 97 in ionisation 62, 112, 120 images solutions 88–9 86–7, of (LDRs) inverted point 66, 100 light internal metals 9 patterns energy, 172 54 as also 101, 60 53 light internal work applications 112 forces graphs interference ions, 4–5 see 22 speed interference medical 97 refraction intercepts 115 interference total inter-molecular ionic 3, work 73 reflection insulators mechanical 81 transformations see 162 26 as and number 144–5 dispersion inertia mass internal reflection of 89 motor effect 150–1, 154 163 lightning radioisotopes 166, 101, 109 motors, electrical myopia 93 154 172–3 limit line of of proportionality best fit 18 8 J N linear joule (unit) 34, magnification 94 114 NAND linear relationships gates natural liquids 54, 55, 139 8–9 gas 36 68 K negatively change Kelvin scale of state 60–1, 62–3, neutron pressure kinetic energy 34, in number circuits logic of gates 138, chain 162 175 139 and long-sight 39, 40, in electrical energy isotopes waves 77, nuclear fission 166 167 98 115 newton loudness theory 163, 93 41 longitudinal 98, (N) 12, 26 99 51 Newton’s lunar eclipses laws of motion 83 non-contact 184 161, reactions 35 transformations from 106, 138–41 60 in kinetic 107 44–5 neutrons evaporation transfers 106, 162 38–9 logic in charged 64 51 forces 12 26–7 Index non-ohmic conductors non-renewable NOR gates normals NOT 84, gates 86, 138, energy nuclear explosions nuclear fission nuclear forces nuclear fuels nuclear fusion nuclear model 160 nuclear power 36, 174–5 plum fission 36, in 175 167, power nuclear reactors in fission in fusion in radioactive in see 175 162, 166, 144, 163 170–1 radioactive emissions radioactive waste ray 111 rays 106, 107 (p.d.) 114 130 118, 119 and charge transfers of 52–3 of 51 114, 118 39, 40, 41 84 waves internal 90 waves index refrigerators regular 100 88–9 86–7, sound refuelling transformations 121 84–5 refractive 35 cells 136–7 rays sound total 38 95 secondary 31 refraction 34, movement of 91, reflection voltage bonding rays recharging 122 and light reflected 124–5 98 82 rectification 127 173 172–3 4–5 76, images recoil circuits energy see real 164–75 172 errors boxes decay 36 166, rarefactions 148 difference also 147 see dating radiotherapy random 146, thermocouples potential 168–70 133 160 charged circuits and 162 163, devices reducing measuring 160–3 decay number magnetic complex 174 115 81 decay radiocarbon model components 106, 167, electrical alternating 167 167, on potential stations 97, waves radioactive radioisotopes positively 175 37, 83 solenoids 164–5 nuclei/nucleus cells camera pollution, 175 radio 97 pudding poles, 12 144–5 110–11 98 plugs 174 see radiation nuclides pitch decay 34, nuclear nucleons pinhole 139 see magnets photocopiers photovoltaic 87 nuclear nucleon 36 photons decay also sources 139 nuclear see permanent 123 energy 100 88–9 73 explosions reflection 110 84 O power ohmic conductors 122–3, 124, and Ohm’s law 114–15 resistance 126–7 renewable energy resistance 122–7 sources 36–7 122–3 of oil 42, 125 transformers 157 internal resistance 120 36 power optical axis optical centre stations nuclear OR of fibres gates origin 123, 126 lenses power 36, 175 resolution 4 91 care 101 resultant force precision 5 prefixes 3 and first law 12, of 13, 15 motion 26 139 2, resultant moment resultant vectors 14 22 pressure oscillations 39, from 44–5, 76–7, gas laws 98–9, 56–7 rheostats 10–11 123 79 pressure oscilloscopes 55 40 in waves resistors 89 138, 9, 155 92 pre-natal optical 111, law 57 right-hand 120 ring grip rule 148, 149 131 primary cells mains principal axis 90, 91, 92 ripple principal foci 90, 91, 92 r.m.s. principle of 132–3 tanks 77, 96 P parallel components resistors parallelogram 118–19 moments rule refraction nature of 15 86, light 96, 76–7, rockets 87 root 27 mean 98 rough square surfaces, voltage emission 132 from 70, 71 97 proportionality, 54, in 10–11 propagation particles 132 126 prisms, particle voltage limit of 18 56 S proton in atoms number 162 106 safety protons and conduction 66, 106, 160, and scalars and in evaporation isotopes 163, 166 scale waves theorem diagrams 78–9 (Pa) voltage 130, 7, 39, penetrating power penumbra 82–3 semiconductor 164, radiation 26 99, 164 see also diodes 124–5 diodes see semiconductors radiation sensors 113 140–1 130 in table 120–1 165 electromagnetic 79, cells 131 electromagnetic periodic motion 40 background period of secondary radiation pendulums law 44 R peak 10–11 11 second pascal 134–5 10 60 Pythagoras’ in electricity 162 68 heat transfer 70–1, 72, 73 series components 118, 119 163 radioactive emissions 164–75 resistors in 126 185 Index shadows shells, 82 electron short-sight 161, 163 for logic for sensors systematic 93 gates 139 uranium 140 errors 164, fission 4, 36, 171, 167, 173 174–5 5 V SI units 2–3 T significant figures temperature sinking Snell’s law and change eclipses 72 of state and evaporation 62–3 vaporisation generators 108–9 60 materials solenoids the gas laws 60 latent 148–9, variable heat of 62, resistors 64 123 144 and heat and infra-red capacity radiation 37, 58 variables and speed sound 70 vectors 6, 8–9 10–11, 12 72 of 100 velocity 22–3, 24 152–3 temperature solidifying 56–7 82–3 power/energy scales 51 and momentum 28 60 thermal solids Graaff de 88 magnetic solar flasks Van 133 in solar 50–3 47 sockets soft vacuum 5 conductivity 66 velocity–time graphs 24–5 54 thermal change of state 60, 62–3, in 66, 34, virtual 50–1 images 85 64 in conduction energy change of state 60 in lenses 92–3, 94, 95 112 and geothermal and nuclear and particles and thermal energy 37 vision 93 sound as energy 34, transformations speed of 98, 40, 55, voltage of 114 mains 66 and electricity rectification 132, 133 136–7 100–1 51, 66–73 transformers change 156–7 101 nature of 77, heat specific latent capacity see also potential difference wasted energy 35 voltmeters (p.d.) 122 58–9 see heat 40 98–101 as specific also heat/heat energy volume 3, 56–7 64–5 thermistors speed 175 conductivity transformations wave 36, 68 115 transfers ultrasound power 35 123 22 W thermocouples of electromagnetic radiation 52–3 80, wasted/lost thermometers energy 35, 40–1, 52–3 86 water, third light 86, law of motion energy power watt of sound 98, of waves 79 waves 77, measuring (W) 42, wave base nature commutators of internal reflection pulses wavefronts 18, 81, 77, wavelength 76, 96 172 19 transformation 18–19, 96–7 76 88–9 154 tracers constant light 131 wave total springs 97 114 4 100–1 time spring 96, 37 38–9 time, split-ring 87 27 water kinetic in 88 tidal and refraction of energy 78, 79 34–5, 77 waves 76–81 39 stability 16–17 a.c. electrical stable equilibrium waveforms diffraction in standard form induction 153, energy fusion in 96 154 2 loss stars, 167, to surroundings transfer interference of matter sound of state waves 76–7, truth electricity tables also effect wind two-slit experiment objects 46, 35, 38 heating 73, effect of 50 47 ultrasound in 101 induction 153 167 ultraviolet eclipses 34, 47 U submerged 37 97 60 submarines light 81 and power 42 82–3 umbra surface 16 turbines work 37, 12, 14 18 sublimation area 60, 82–3 70 X uncertainty 4–5 surfaces X-rays units pressure on radiation emission 186 116–17, 123 equilibrium 70–1 17 Z upthrust symbols 81 2–3 44 unstable and 76 98–101 138–9 weight Sun 77, light 106–11 turning stiffness waves 80 60–5 see static 34, 97 156–7 54–5 transverse change by 40–1 175 transformers states 130–2 115 17 12, 47 zinc-carbon dry cells 120 43 Acknowledgements The author permission Alamy: Aurora A.T . Getty Lunardi: Images: 4.24.2, Works 5.5.1; & 1.2.1, Corbis: Bert 1.20.1, f. effort the 2.6.3; has necessary Library: thank the following Researchers, made 4.5.1, 3.4.1, to 2.2.1; for 3.13.4, Inc: Vidéo 1.9.1, trace arrangements the the at du NASA: 4.20.4, Staples: CSG: Hart 1.8.4; Davis: 1.12.3, 1.5.1; SuperStock: 2.12.4. copyright first 4.13.3; 2.6.4; Transport, Adrienne publisher the of Darren Library: Commons: overlooked Ocean: 4.5.2, Rendle Metta 4.12.2, Fotolia: Ministry Clive John 2.12.3, 3.9.2, 1.2.2, 1.8.2; 2.1.1, 4.2.4, 4.14.1, Lyroky: 2.7.1, Reuters: Optique Images: Rout: Green: Inspirestock: Photo / Arcaid Chris J. Hoferichter: 1.1.1; Wikimedia been 1.18.2, 2.9.4, Science inadvertently to 4.19.2, David Chillmaid: Jamaica: 1.12.1; like sciencephotos: ESA-CNES-ARIANESPACE been make Picture 5.8.1, 1.15.3, Housing, Stockbroker: have 2.12.2, 1.14.2; GIPhotoStock/Photo Every Images: Martyn Features: 2.2.2, Blend Garland AFP: also Alchemy: Lyons: Rachko: iStockphoto: Rex 2.3.3, would material: 4.17.1, David Leslie 4.18.1, Simone publisher Willett: 4.8.2, 1.21.2, Stock: the reproduce Photos: Streeter: NZ: and to holders will be but if any pleased to opportunity. 187 Physics for CSEC® Achieve your potential Developed guide in will CSEC® exclusively provide by Physics an syllabus information key ● ● to Engaging the with additional experienced in and an learning designed ● the Caribbean Examinations support to Council®, maximise your this study performance Physics. Written the you with team examination, easy-to-use outcomes enhance activities of your that teachers this study double-page from study help the of you and guide and subject , develop the in covers format . syllabus the experts Each all a CSEC® the topic contains such the essential begins range of with features as: analytical skills required for examination Examination tips with essential advice on succeeding in your assessments Did You Know? boxes to expand your knowledge and encourage further study This study choice also questions examiner Physics The guide and feedback, includes sample to build a fully interactive examination skills and answers confidence Caribbean Examinations Thornes subjects at to CSEC® produce and a Council series (CXC®) of with in O x f o rd has Study multiple- accompanying preparation for the CSEC® worked Guides exclusively across a wide with range of CAPE®. How of incorporating examination. Nelson Pa r t CD, University P re s s to get in touch: web www.oup.com/caribbean email schools.enquiries.uk@oup.com tel +44 (0)1536 452620 fax +44 (0)1865 313472