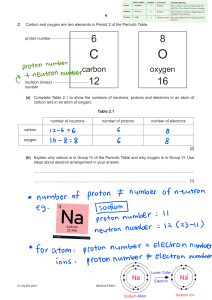
Answers to end-of-chapter questions Chapter 2 a atoms of the same element with different nucleon numbers / mass numbers / different numbers of neutrons b i protons = 5 ii neutrons = 6 iii electrons = 5 1 1 1836 charge = −1 ii mass = 1 charge = zero iii mass = 1 charge = +1 c i mass = [1] [1] [1] [1] [1] [1] [1] [1] [1] [1] Total = 10 [1] Zr ii 51 [1] b 70 [1] c i It is deflected / bends away from the anode, or towards cathode, or downwards. [1] Proton has positive charge since like charges repel / or opposite charges attract. [1] ii no deflection / go straight through [1] neutrons have no charge [1] Total = 7 2 a i 91 40 positively charged nucleus; containing protons and neutrons; electrons outside the nucleus in energy levels; protons are positively charged with relative mass of 1; neutrons are neutral with relative charge of zero and relative mass of 1; electrons are negative with relative charge 3 a of −1 and relative mass of [allow [1] [1] [1] b atomic number = number of protons (in the nucleus); [1] nucleon number = number of protons + number of neutrons [1] c Mg = 12 protons, 12 electrons, 12 neutrons [1] Al = 13 protons, 13 electrons, 14 neutrons [1] d number of positively charged protons = number of negatively charged electrons [1] e it would not be oxygen / it would be another element / oxygen can only have 8 protons [1] f The mass of an electron is negligible. [1] Total = 13 a atoms of the same element with different nucleon numbers / mass numbers / different numbers of neutrons [1] b i both have 92 protons; [1] both have 92 electrons [1] ii uranium-235 has 143 neutrons; [1] uranium-238 has 146 neutrons [1] c 90 [1] Total = 6 4 5 a number of protons = 17 and 17 number of electrons = 17 and 17 number of neutrons in chlorine-35 = 18 and in chlorine-37 = 20 b the chlorine-35 isotope is more abundant; the weighted average is nearer 35 than 37 c i it has more protons than electrons ii it has 17 protons and 18 electrons / it has 1 more electron than protons [1] [1] [1] [1] [1] [1] [2] [it has more electrons than protons for 1 mark] Total = 8 [1] [1] 1 1836 1 or negligible] 2000 AS and A Level Chemistry © Cambridge University Press [1] Answers to end-of-chapter questions: Chapter 2 1