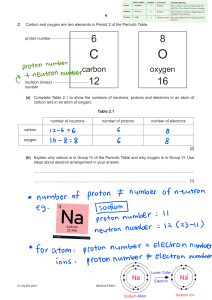
Modelling atoms of elements By: Reyam Mohamed Grade 8 A Introduction A model is a representation of a system in the real world. Models help us to understand systems and their properties. For example, an atomic model represents what the structure of an atom could look like. Atom is smallest unit into which matter can be divided without the release of electrically charged particles. Atom has neutrons, electrons, protons, and shells. The nucleus contains the neutrons and protons. Protons have a positive charge and neutrons have no charge. The shells are having the electrons and they are negatively charged. Rational: Significance of Project in Daily Life Atoms are extremely important structures that make up all of the materials on earth. Atoms are in our bodies and they bond together to form molecules, which make up matter. Everything living is made up of atoms. Research Question: How are electrons, protons and neutrons arranged in atoms of elements? Hypothesis: Electrons, protons and nutrons has a specific arrangement in an atom Aim/Objective: To construct an atomic model of helium using metalic rod Materials: 1. Metal rod 2. Balloons 3. Tape Balloons Tape Metal rod Procedure Bend the metallic rod in to a circular shape Blow the balloons Procedure Stick the balloons that represents the nucleus (Neutrons+Protons) together and label them (N= neutrons, P= Protons) Procedure Stick the balloons that represents the electrons to the metallic rod Procedure Put the balloons that represent the nucleus in the middle of the metallic rod Result Helium atom is constructed It has 2 neutrons, 2 protons, and 2 electrons Interpretation of results: The atomic model of helium was constructed showing that it has 2 protons, 2 neutrons and 2 electrons. The middle part is the nucleus and the metallic rod is the shell having the electrons. It has only one shell which means that helium is in period 1. Although it has 2 valence electrons only, but it is a group 18 element. Conclusion: Atoms are every where around and are very important. Each element has different atom. Protons, neutrons and electrons arrangement differs from one element to another. There is a specific arrangement of there molecule. Protons and neutrons are in the neucleus of the atom, whereas electrons are on the shell. References: https://www.siyavula.com/read/science/grade-10/theatom/04-the-atom-02 https://www.britannica.com/science/atom https://sites.google.com/site/theatomcom/atoms-ineveryday-life Thank You!