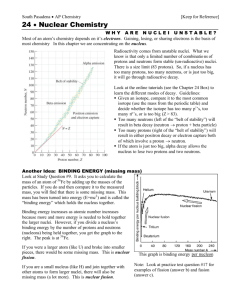
The Nucleus Element Atomic Number An element is a substance all of whose atoms have the same number of protons in their nuclei. This number is the atomic number of the element and equals the number of electrons that surround each nucleus of the element’s atoms. Isotopes The isotopes of an element have different numbers of neutrons in their nuclei. Nucleon A nucleon is a neutron or proton. Mass Number Nuclide Radioactive Decay The mass number of a nucleus is the number of nucleons it contains. Is an atom whose nucleus has particular atomic and mass numbers. Certain atomic nuclei spontaneously emit alpha particles (helium nuclei), beta particles (electrons), , or gamma rays (high-frequency electromagnetic waves). Positron Is a positively charged electron emitted in some beta decays. Half-Life Of a radionuclide is the time needed for half of an original sample to decay. Binding Energy Of a nucleus is the energy equivalent of the missing mass and must be supplied to the nucleus to break it up. The mass of every nucleus is slightly less than the total mass of the same number of free neutrons and protons Binding Energies per nucleon Nuclei of intermediate size have the highest binding energies per nucleon. The fusion of light nuclei to form heavier ones and the fission of heavy nuclei into lighter ones are both processes that liberate energy. Elementary Particle Antiparticle Annihilate Pair Production Four Fundamental Interactions Cannot be separated into other particles. Of an elementary particle has the same mass and general behavior, but it has a charge of opposite sign and differs in certain other respects. Some unchanged elementary particles also have antiparticles in which other properties differ in the particle and antiparticle. A particle and its antiparticle can annihilate each other, with their masses turning entirely into energy. In the opposite process of pair production, a particle-antiparticle pair materializes from energy. In order of decreasing strength, are strong, electromagnetic, weak, and gravitational. Lepton Is an elementary particle that is not affected by the strong interaction and has no internal, structure; the electron is a lepton. Neutrino Is a lepton with no charge and little mass that is emitted during beta decay. Hadron Is an elementary particle that is affected by the strong interaction and is composed of quarks. Quarks Particles with electric charges of ± 1 2 e or ± e that have not been found outside 3 3 hadrons as yet. Protons and neutrons are hadrons. 1. The basic idea of the Rutherford atomic model is that the positive charge in an atom is a. spread uniformly throughout its volume b. concentrated at its center c. readily deflected by an incoming alpha particle d. the same for all atoms 2. Nearly all the volume occupied by matter consists of a. electrons b. protons c. neutrons d. nothing 3. The atomic number of an element is the number of a. protons in its nucleus b. neutrons in its nucleus c. electrons in its nucleus d. protons and neutrons in its nucleus 4. The atoms of the isotopes of an element are different in which one or more of the following? a. number of electrons b. number of protons c. number of neutrons d. atomic mass 5. Which of the following is not an isotope of hydrogen? 1 1 a. 0 H b. 1 H 2 3 c. 1 H d. 1 H 6. The atomic number of an element determines its a. mass b. binding energy c. number of neutrons d. chemical behavior 7. The number of protons in a stable nucleus is always a. less than the number of neutrons b. less than or equal to the number of neutrons c. equal to or more than the number of neutrons d. more than the number of neutrons 8. An electron is emitted by an atomic nucleus in the process of a. alpha decay c. gamma decay b. beta decay d. nuclear fission 9. Gamma rays have the same basic nature as a. alpha particles c. x-rays b. beta particles d. sound waves 10. Radioactive materials do not emit a. electrons c. alpha particles b. protons d. gamma rays 11. Which of these types of radiation has the least ability to penetrate matter? a. alpha particles c. gamma rays b. beta particles d. x-rays 12. Which of these types of radiation has the greatest ability to penetrate matter? a. alpha particles c. gamma rays b. beta particles d. x-rays 13. When a nucleus undergoes radioactive decay, the number of nucleons it contains afterward is a. always less than the original number b. always more than the original number c. never less than the original number d. never more than the original number 14. Which of these particles is radioactive? a. electron c. neutron b. proton d. alpha particle 15. The largest amount of radiation received by an average person in the United States comes from a. medical x-rays b. nuclear reactors c. fallout from past weapons tests d. natural sources 16. The half-life of a radionuclide is a. half the time needed for a sample to decay entirely b. half the time a sample can be kept before it begins to decay c. the time needed for half a sample to decay d. the time needed for the remainder of a sample to decay after half of it has already decayed 17. As a sample of a radionuclide decays, its half-life a. decreases b. remains the same c. increases d. may do any of these, depending upon the nuclide 18. In a stable nucleus other than 11 H the number of neutrons is always a. less than the number of protons b. less than or equal to the number of protons c. equal to or more than the number of protons d. more than the number of protons 19. The electronvolt is a unit of a. charge b. potential difference c. energy d. momentum 20. Relative to the sum of the masses of its constituent particles, the mass of an atom is a. greater b. the same c. smaller d. any of these, depending on the element 21. The binding energy per nucleon is a. the same for all nuclei b. greater for very small nuclei c. greatest for nuclei of intermediate size d. greatest for very large nuclei 22. The splitting of an atomic nucleus, such as that of 235U, into two or more fragments is called a. fusion c. a chain reaction b. fission d. beta decay 23. In a chain reaction a. protons and neutrons join to form atomic nuclei b. light nuclei join to form heavy ones c. neutrons emitted during the fission of heavy nuclei induce fissions in other nuclei d. uranium is burned in a type of furnace called a reactor 24. Enriched uranium is a better fuel for nuclear reactors than natural uranium because enriched uranium has a greater proportion of a. slow neutrons b. deuterium c. plutonium d. 235U 25. In a nuclear power plant the nuclear reactor itself is used as a source of a. neutrons b. heat c. radioactivity d. electricity 26. Fusion reactions on the earth are likely to use as fuel a. ordinary hydrogen b. deuterium c. plutonium d. uranium 27. Of the following particles the one that is not an elementary particle is the a. alpha particle c. neutron b. beta particle d. neutrino 28. An example of a particle-antiparticle pair is a. proton and positron b. proton and neutron c. neutron and neutrino d. electron and positron 29. The interaction responsible for the structures of molecules, liquids, and solids is the a. gravitational interaction b. strong interaction c. weak interaction d. electromagnetic interaction 30. The weakest of the four fundamental interactions is the a. gravitational interaction b. electromagnetic interaction c. strong interaction d. weak interaction 31. The mass of the neutrino is a. equal to that of the neutron b. equal to that of the electron c. equal to that of a quark d. very small 32. Quarks are particles that a. have no mass b. have charges whose magnitudes are less than e c. decay into protons d. decay into neutrinos 33. A particle that is believed to consist of quarks is the a. electron c. neutron b. positron d. neutrino 11 34. The number of protons in a nucleus of the boron isotope 5 B is a. 5 c. 11 b. 6 d. 16 40 35. The number of neutrons in a nucleus of the potassium isotope 19 K is a. 19 c. 40 b. 21 d. 59 13 13 36. When the nitrogen isotope 7 N decays into the carbon isotope 6C it emits a. a gamma ray b. an electron c. a positron d. an alpha particle 37. The product of the alpha decay of the bismuth isotope a. c. 210 79 Au 210 83 Bi b. 210 81 d. 218 85 214 83 Ti At 38. The product of the gamma decay of the aluminum isotope a. c. 27 12 Mg 27 13 Al Bi is b. 26 13 Al d. 27 14 Si 27 13 Al is 39. The half-life of a certain radioactive isotope is 6 h. If we start out with 10 g of the isotope, after 1 day there will be a. none left c. 1.6 g left b. 0.625 g left d. 2.5 g left 40. After 45 h, 1 mg of an 8-mg sample of a certain radioactive isotope of sodium remains undecayed. The half-life of the isotope is a. 5.625 h c. 33.75 h b. 15 h d. 39,375 h