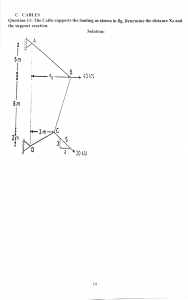
Everyday Physics Unusual Insights into Familiar Things (Jo Hermans) (z-lib.org)
advertisement

Everyday Physics: Unusual insights into familiar things Jo Hermans To Hanneke, Reinoud and Francine, whose questions inspired me to write this book PREFACE Some people think that science and technology are only for people with a formal scientific education, but that’s a misconception. Our society is more and more dependent on technology, and anyone without a basic knowledge of science is at a disadvantage. Apart from that, it’s often just nice to understand a bit more about the world around us and to see how things work. This book gives the inquiring reader scientific insights into everyday things. It contains a collection of interesting – sometimes surprising – examples, many based on questions from family, friends and students, often just motivated by curiosity. The choice of topics is therefore slightly arbitrary. You don’t have to read this book all in one go, or even from front to back! Chapters are self-contained and can be read individually, although related topics are loosely grouped together. Some more complex topics and calculations are shown in boxes in the text, for advanced readers. You can do almost all the experiments in the book with simple materials found at home. Some are just for fun, while others are illustrative and enlightening, but they all show that science doesn’t have to be boring! Suggestions and errata: you can suggest new topics to include in future editions, and obtain the latest list of changes, corrections (and interesting additions) via the book’s webpage at www.uit.co.uk/everyday-physics Continue the conversation at #EVPHYS on Twitter. Published by UIT Cambridge Ltd www.uit.co.uk PO Box 145, Cambridge CB4 1GQ, England +44 (0) 1223 302 041 Copyright © 2021 UIT Cambridge Ltd All rights reserved. Subject to statutory exception and to the provisions of relevant collective licensing agreements, no part of this book may be reproduced in any manner without the prior written permission of the publisher. First published in 2021, in England. The author has asserted his moral rights under the Copyright, Designs and Patents Act 1988. Diagrams prepared by Hayley Wells hayleywellsillustration@gmail.com Front cover photographs sourced via Adobe Stock Photograph credits are on page 301 Design by Mad-i-Creative biscuits79@icloud.com The publishers have endeavoured to identify all copyright holders but will be glad to correct in future editions any omissions brought to their notice. ISBN: 9781906860806 (paperback) ISBN: 9781906860820 (ePub) ISBN: 9781906860813 (PDF) Also available as Kindle Disclaimer: the advice herein is believed to be correct at the time of printing, but the author(s) and publisher accept no liability for errors or for actions inspired by this book. ep-1-1 CONTENTS PART A I OUTDOOR LIFE Chapter 01 I 0 Chapter 02 How does GPS navigation work? (With a nod to Albert Einstein) Why are some mountain winds so warm? Chapter 03 What is wind chill? Chapter 04 Why is ice so slippery? Chapter 05 Waves at the beach Chapter 06 How fast do raindrops fall? Chapter 07 Why don’t fog drops fall? Chapter 08 Skydiving: how fast can you fall? Chapter 09 How high will the sun rise today? Chapter 10 How hot does the sun feel? Chapter 11 Parallel light beams from the sun Chapter 12 Summer and winter, why such a big difference? Chapter 13 Why do you walk the way you do? PART B I BICYCLE AND CAR I Chapter 14 The human engine Chapter 15 The human energy equivalent of a vacuum cleaner Chapter 16 How do you keep your temperature constant? Chapter 17 How efficient is cycling? Chapter 18 What forces affect a cyclist? Chapter 19 Can you cycle at 100 km/h? Chapter 20 How fast can you cycle on the moon? Chapter 21 Is cycling really harder with a side wind? Chapter 22 Minimizing your journey time Chapter 23 The cyclist’s soggy back 1 5 7 11 13 15 19 21 25 29 31 33 37 42 43 47 49 55 59 63 67 69 75 79 Chapter 24 Chapter 25 Chapter 26 Can you get less wet by cycling faster? Rolling resistance, air resistance and fuel consumption How many cars per hour can a road take? PART C I LIGHT AND COLOUR Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter Chapter 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 I Chapter Chapter Chapter Chapter Chapter 46 47 48 49 50 Chapter 51 Chapter 52 98 Cosy candlelight Why are incandescent bulbs so inefficient? Luminous ideas: fluorescent lights and LEDs Why is the sky blue and the setting sun red? Two kinds of smoke from the same cigarette? Swimming pools are deeper than they look Sunlight filtering through the leaves of trees How sharply can you see? Your eye is more sensitive than a camera Puddles on a dry road Seeing the sun after sunset Transparent windowpanes & opaque lace curtains Seeing clearly underwater What makes rainbows? Why are soap bubbles so colourful? Why are CDs so colourful? How do holograms work? Why does the sea look so blue? What’s special about Polaroid glasses? PART D I SOUND AND HEARING 83 87 93 I What do your ears hear? Why isn’t there more noise pollution? The energy cost of talking How can you tell where a sound is coming from? Discriminating between different voices: the cocktail party effect Do you hear better at night? Can the wind blow sound to you? 99 101 105 107 113 115 119 123 129 133 135 139 143 147 155 159 165 169 173 180 181 185 187 191 195 199 203 Chapter Chapter Chapter Chapter 53 54 55 56 Do noise barriers work? Can you hear whether the curtains are closed? Doh-re-mi: the physics of musical scales Why orchestras go out of tune PART E I IN AND AROUND THE HOUSE I Chapter Chapter Chapter Chapter 57 58 59 60 Why do eggs explode in the microwave? Can you cool your home with your fridge? Curve balls, backspin and topspin How much power can you get from solar energy? Chapter Chapter Chapter Chapter Chapter Chapter 61 62 63 64 65 66 Chapter Chapter Chapter Chapter Chapter Chapter Chapter 67 68 69 70 71 72 73 The mystery of the wandering carpets Common misconceptions about the radiometer Is thick glass a better insulator than thin glass? Is there a vacuum inside double glazed windows? Can you feel energy consumption? Is a black central heating radiator better than a white one? Does black paint get hotter than white? Does baby really need a hot water bottle? The lid on the saucepan Why does the air get so dry in winter? Why don’t you die of heat in the sauna? The wine-mixing problem The wandering tea leaves I ACKNOWLEDGEMENTS I HANDY REFERENCE DATA II I I I APPENDIX: THE WINE-MIXING PROBLEM – CALCULATION TWO MORE EXPERIMENTS RESOURCES CREDITS INDEX 205 207 209 217 220 221 226 229 239 245 247 251 255 261 265 267 269 271 273 277 281 283 286 287 296 298 300 301 302 0 8 Everyday Physics: Unusual insights into familiar things 01 HOW DOES GPS NAVIGATION WORK? (WITH A NOD TO ALBERT EINSTEIN) I’m riding on the bike paths along the southern California coast and I’ve lost my way. I do have a detailed map of the region with me, though. Fortunately there is a crossing with a sign showing that I am 14 kilometres from Santa Monica and 12 kilometres from Malibu. I am therefore on a circle around Santa Monica with a radius of 14 kilometres and at the same time on a circle around Malibu with a radius of 12 kilometres. The two circles intersect in two points. I am at one of these points. Which one? In this case I’m lucky: one of the two points lies in the Pacific, and I am not standing in the ocean, so I must be at the other point. I can now easily find the crossing on the map. If I also use the distances to the other towns on the sign in addition to Malibu and Santa Monica, my location becomes even more reliable, and the location in the ocean is eliminated. A GPS (Global Positioning System) receiver determines position in an analogous way but it does it in three dimensions: not just the location on the map but the height as well, and with astounding precision. So how does GPS (or its European counterpart, Galileo) work? Broadly speaking it works like this: GPS has 24 satellites orbiting at a distance of 20,000 km above the Earth (Fig. 1) and making two revolutions about the Earth per day. (The satellites are not geosynchronous/geostationary, by the way.) Each sends a radio Everyday Physics: Unusual insights into familiar things 1 signal that can be picked up by the GPS receiver in your car, on your bike or in your phone. Fig. 1: GPS uses a network of 24 satellites. Credit: After commons. wikimedia.org/wiki/ User:Yuma You need two things to determine your position: (1) the position of each satellite that is visible to your GPS receiver and (2) the distance to each. (1) The position of each satellite is accurately monitored by a number of ground stations spread over the Earth and coordinated from the “mother station” in the US, where the system was originally developed for military purposes. These data are transmitted from Earth to the satellites. In this way each satellite knows its own position and reports this to your GPS receiver. (2) Now you need to know the distance to each satellite. Your receiver finds this by measuring the time it takes for a radio signal from the satellite to reach the receiver; this is the difference between the time when the satellite sent the signal and the time your GPS received it. Multiply the time by the speed of the radio signal (i.e. by the speed of light) to give the distance. If you know the distance for (at least) three satellites, then you can calculate your position, just as in the road-sign story: 2 Everyday Physics: Unusual insights into familiar things (a) The distance to the first satellite tells you that you are on an imaginary sphere around that satellite, with a radius equal to the measured distance. (b) Similarly, the second satellite yields another imaginary sphere – around the second satellite (Fig. 2). So you must be on both spheres simultaneously, that is, on the intersection of these two spheres. This is a circle, just like two adhering soap bubbles form a circle. (c) The third satellite yields yet another sphere, which intersects the circle in two points. Fortunately one of those drops out: it lies deep in the Earth or far above it. The remaining point is your position. Of course there is always a margin of error in the measurement; if your GPS receiver can see more satellites and takes them into account, the error margin will be smaller. Fig. 2: The intersection of two spheres is a circle. It all comes down to measuring time. That has to be very accurate because the radio signals travel at the speed of light (300,000 km per second). A timing error of one millionth of a second would mean an error of 300 metres in your position. To achieve the required accuracy all satellites and ground stations have precision clocks (but note – not the GPS receivers) that are based on an extremely precise and stable transition within a caesium atom. Clocks of this type attain fabulous accuracies: the best ones on Earth run so precisely that in 1,000 years from now they will be out by less than one tenth of one millisecond! The satellite clocks Everyday Physics: Unusual insights into familiar things 3 need not be that good, but they must be synchronized to within one billionth of a second. The clock in your GPS receiver isn’t an atomic clock like the ones in the satellites and ground stations, and in fact it doesn’t need to be. By using an extra satellite (i.e. four instead of three), the GPS receiver can precisely synchronize its (cheap, quartz) clock with the atomic clocks in the satellites, and so measure the times accurately, and therefore the distances. See the Resources appendix for an animation of GPS satellites orbiting the Earth. GPS and relativity We’ve seen that GPS depends on precise time measurement. Now something unexpected happens: we need Einstein and his theory of relativity. Einstein himself would never have guessed that his theory – such an elusive piece of theoretical physics – would ever play such an important role in something as practical as GPS. It’s a lovely example of an unexpected and unintended application of cuttingedge fundamental science. Here’s how it works: according to relativity theory, the clocks in the satellites don’t run at the rate they would when sitting on Earth, for two reasons: (1) because they are moving at high speed, they run somewhat slower, and (2) because they are so high up they feel less gravity (only a quarter of what they would feel on Earth), so they run somewhat faster. The second effect is the greater, and the satellite clocks get ahead of the clocks in the ground stations by 38 millionths of a second per day. That does not sound like much, but it means that after one day the error in calculating a position would be more than 11 km, which would render your GPS useless. To compensate for this relativistic effect, before launch the clocks in the satellites are set to run at a slightly slow rate – exactly slow enough so that after launch and once they are in orbit, they are precisely synchronous with the clocks in the monitoring stations on Earth. Problem solved. Destination reached. With a nod to Einstein. 4 Everyday Physics: Unusual insights into familiar things 02 WHY ARE SOME ALPINE WINDS SO WARM? In the Alps, the Föhn is the warm wind that blows from the mountains into the valleys, especially in winter. Curiously, even though it comes down from the mountains it’s warm and dry. Why? To see what’s happening, let’s first look what happens to dry air that comes over the mountains (Fig. 1). The air pressure at the top of the mountain is lower than in the valley below, because there is less air pressing down, to cause the pressure. So air that streams up a mountain expands and therefore cools down. (This is the opposite of what happens in a bicycle pump: in the pump, air is compressed and therefore warms up.) By rising and expanding, air cools about 1°C per 100 metres, which explains why it gets colder when you hike up into the mountains. When air rises, say, 2,000 metres, Everyday Physics: Unusual insights into familiar things 5 it cools by about 20°C. When it comes down the other side of the mountains it warms at the same rate. The net effect is therefore zero: no Föhn (Fig. 1). Fig. 1: In this model example the dry air starts out in the valley at 20°C, cools to 0°C at the top of the mountain, and warms again to 20°C in the valley. With moist air (as in the Föhn) things are different (Fig. 2). As the air rises up the mountain it cools, just as the dry air did. But something else happens too: as the air cools, the water vapour in the air condenses and forms rain, just like morning mist can form above a meadow as the temperature goes down, or like the water vapour in your breath condenses if you blow on a cold windowpane. Heat is released when vapour condenses – the opposite of evaporation which absorbs heat. Because of the heat that is released at condensation the rising moist air doesn’t cool as much as the dry air did. The net result is that the temperature rises to 27°C as it comes down the other side of the mountain. And the air is not only warm: it is also dry because it dropped most of its moisture as rain on the slope while rising. The relative humidity (the amount of water vapour the air contains divided by the amount that it can contain) can easily get below 20% this way. Warm and dry air, that’s what you get. Fig. 2: The rising moist air cools down by only 13°C rather than 20°C. But as it descends the other side of the mountain, it warms up by the full 20°C, just as the dry air did in Fig. 1. 6 Everyday Physics: Unusual insights into familiar things 03 WHAT IS WIND CHILL? The weatherman reports that the temperature is 0°C, but that the “wind chill” factor is minus 17. What does this mean? When it is cold outside you feel particularly chilly if there is a strong wind blowing. The moving wind cools your body more than stationary air does. This is often called “wind chill”, i.e. subjective temperature. It is important whenever you skate, ride your bike, sail or ski; if you ignore it, you could end up with frostbite. There is a lot of misunderstanding about wind chill. One newspaper said that if there is enough wind the water in a car radiator can freeze, even when the temperatures is above 0°C. Everyday Physics: Unusual insights into familiar things 7 That’s wrong. The radiator temperature will drop until it is the same temperature as the air and it can’t become colder than that. What is true, however, is that in a wind the radiator will cool down more quickly than in stationary air. And what’s true for the radiator is true for any object suspended in the wind: it will reach the temperature of the air, period. (The only exception is where something is wet; then evaporation takes away heat and can lead to excess cooling, but that’s not what we are talking about here.) However, for a human being who has to generate heat constantly to maintain their body temperature of about 37°C the rate of heat loss is important. The environment is normally cooler than your body, so your body loses heat to the environment. In still air, the heat loss is relatively small: even exposed skin is still enveloped by an insulating air layer a few millimetres thick, which you carry with you as though it were a sweater (see How do you keep your temperature constant? p49). That air layer limits the heat losses and keeps your skin relatively warm. In contrast, when there’s a wind, it blows away the insulating air layer, so that the cold gets right up to your skin, which becomes colder quicker. It becomes just as cold as it would be at a lower air temperature without wind. That lower temperature is the subjective temperature or “wind chill”. In other words: it is the temperature that still air would be to cause the same heat loss as the real temperature does in the wind. 8 Everyday Physics: Unusual insights into familiar things Fig. 1 shows the wind chill for various temperatures and wind speeds in average conditions. The left-hand column shows the real air temperature; the other columns give the subjective temperatures in degrees Celsius for various wind speeds. Note that the wind speed is relative: if you are standing still, and the wind is blowing at 25 km/h, the effective wind speed is 25 km/h. But if the air is stationary, and you are cycling through the air at 25 km/h, the effective wind speed is again 25 km/h, so a moving cyclist will cool down a lot more than someone standing still. Fig. 1: Wind chill values under different conditions. Wind speeds of 13 and 20 m/s roughly correspond to the speed of a professional cycling time triallist (47 km/h) and a skier on a fast slope (72 km/h). Wind speed 0 m/s = 0 km/h Real air temperature 7 m/s = 25 km/h 13 m/s = 47 km/h 20 m/s = 72 km/h Subjective temperature (“Wind chill”) 0°C 0°C −11°C −17°C −20°C −2°C −2°C −14°C −20°C −23°C −4°C −4°C −17°C −23°C −27°C −6°C −6°C −20°C −26°C −29°C −8°C −8°C −23°C −29°C −32°C −10°C −10°C −25°C −32°C −36°C −12°C −12°C −28°C −36°C −39°C We see that it makes a lot of difference whether or not there is a wind blowing. At 0°C with a 72 km/h wind (or when skiing down the mountain at 72 km/h), it feels as cold as −20°C would be without wind. And if the temperature is −12°C at that speed, it feels like −39°C. It’s like being in Siberia! If the air temperature is 37°C (i.e. at body temperature), there is no wind chill. However, you will probably find moving air more comfortable than still air, because the moving air evaporates some of your perspiration, and cools you a bit. Everyday Physics: Unusual insights into familiar things 9 A different way of looking at all this is to realize that while wind chill doesn’t reduce the temperature, it does increases the rate of heat loss and this matters to humans and animals. For example, say you are sitting at rest in still air at 0°C. Depending on body mass and clothing, you use about 120 joules/second, i.e. 120 watts. However, when there’s a wind of 20 m/s, you will need 185 watts to keep your body at constant temperature. If your internal heat production doesn’t increase fast enough, your temperature will fall and that’s how you get hypothermia. 10 Everyday Physics: Unusual insights into familiar things 04 WHY IS ICE SO SLIPPERY? Why is ice so slippery? It sounds obvious: of course ice is slippery – that’s why you skate on it. But what exactly makes it so slippery? It’s not just because it is smooth: glass is very smooth, but it’s not a good surface to skate on. “As smooth as glass” isn’t the same as “slippery as ice”. To slide on a surface you need more than just smoothness – a layer of water, for example: a surface that is smooth becomes slippery only when it is wet. You slip on a wet floor, not on a dry one, so a bit of water is what you need. And indeed, that is precisely what makes ice slippery: a thin layer of water between skate and solid ice acts as a lubricant. The next question is: why would there be water under the skate when the ice is below freezing? An old misconception is that this is due to the pressure that the skater puts on the ice: the weight Everyday Physics: Unusual insights into familiar things 11 of the skater is concentrated on a tiny area – on the few square centimetres of the skate that touch the ice – and that produces a lot of pressure. It’s true that, when under pressure, ice melts below the normal freezing point. That is just plain logic: ice has a larger volume than liquid water (which is why ice floats). So if you compress ice, it tends to become liquid, which means that the melting point has dropped a bit. However, if we calculate that change in melting temperature, we find it’s very little in the case of the skater – a few tenths of a degree at most. So the fun of skating would be over as soon as the temperature is a few degrees below the freezing point. (Incidentally, this pressure argument would also fail to explain why a light object like an ice hockey puck slides so well on ice.) The real answer is that ice has a layer of water at its surface by nature. Ice is made up of a lattice of water molecules, tightly bound to their neighbours all around. But at the outermost layer of molecules things are different. The molecules at the surface have no neighbours on the outside. Therefore, they are less tightly bound, have a high mobility and behave like fluid water even at freezing temperatures. At freezing point, this layer of water on ice has a thickness of some 200 molecules – about 70 nanometres (roughly one thousandth of the thickness of a human hair.) That’s not exactly thick, but it is enough to act as a lubricant. In addition, the friction between skate and ice releases heat which, in turn, melts some extra ice, making sliding even easier. However, as the temperature goes down, the water layer becomes thinner and the lubrication reduces. If it gets sufficiently cold, far below −40°C, even the outermost molecules freeze, and there is no water layer left. You might now be tempted to conclude that skating is best at temperatures just below freezing point, but this is not the case. The ice gets a little soft and loses mechanical strength since it’s closer to the melting point, and the skate cuts into it, causing some extra resistance. To skate really fast you therefore must go down a bit in temperature. The optimum – depending on the type of skate – is around −7°C or −8°C. That is cold enough to keep the skate from cutting into the ice too much, but not so cold that the water layer becomes too thin. So the recipe for beating the world speed skating record is no wind and −7°C. 12 Everyday Physics: Unusual insights into familiar things 05 WAVES AT THE BEACH If you’ve walked along a beach you may have noticed that the waves always roll in parallel to the beach. For a north–south beach like the Cape Cod National Seashore this is pretty logical in the case of an easterly wind. But if the wind is from the north, isn’t it strange that the waves don’t change direction: they still keep coming in parallel to the beach? There is a bit of physics behind this. The indirect cause is that the beach rises gradually. Towards the beach the water becomes gradually less deep, and that depth influences the speed of the waves. As the water becomes shallower, the speed goes down (see box). Now take those lovely broad waves far out to sea, driven by a north-easterly wind, so they are travelling at 45° to the coast (Fig. 1a). As the waves approach the coast, the side of a wavefront that is further from the shore will be in deeper water than the side nearby (Fig. 1b). Now remember that the speed at the deep side is higher than at the shallow side. So, the waves gradually turn in a curve towards the shore, with the deep part automatically on the outer side of the curve because it is travelling faster. The wave therefore turns towards the beach (Fig. 1c) until the speeds of the wave ends become equal, which only happens when it is parallel to the coast, where the depths are equal (Fig. 1d). After that there is no more reas on to depart from the straight course and the waves remain neatly parallel, provided of course that the gradient of the ocean floor is more or less equal everywhere. So: wave-surfers should thank heaven for nicely sloping beaches. And for the laws of physics, of course. Everyday Physics: Unusual insights into familiar things 13 Speed of water waves The speed at which water waves propagate depends on several variables. Two forces that want to restore a surface to smoothness are surface tension (which makes raindrops round, for example) and gravity. As soon as a wave crest is formed, both forces want to suppress it. Therefore, both influence the speed. The surface tension is important only for small ripples on the water with wavelengths of a few centimetres, so we can neglect it. For the larger waves, like at the beach, gravity prevails. In this case the ratio of wavelength to the depth of the water is important. For a shallow beach we can assume that the wavelength is large compared to the depth, and then the expression for the speed (v) is simply: v = √ (g D), where g is the acceleration due to gravity and D is the depth. So the speed of the wave decreases as the depth decreases. For a depth of two metres and g = 10 m/s2, we find that the speed is about 4.5 m/s. If the depth decreases to one metre, the speed is about 3 m/s – now only two or three times walking pace. Fig. 1: Waves approaching the coast at an angle turn gradually as the water becomes shallower. 14 Everyday Physics: Unusual insights into familiar things 06 HOW FAST DO RAIN DROPS FALL? In a heavy rain shower it is quite striking that the big drops come first and as the shower wanes the drops are smaller. So it seems that big drops fall faster than small ones, which is strange, because in a vacuum everything falls equally rapidly, as we remember from school. For example, a feather in a vacuum tube falls as fast as a marble or a coin. The explanation is that raindrops aren’t falling in a vacuum; they’re falling in air, so friction (or “air resistance”) plays a role, acting against gravity. To understand the difference between large and small drops we have to look at the process of falling. That process starts as gravity makes the drop accelerate. But as the speed increases, so does the friction, directed upward, against the force of gravity. So the net force downward becomes smaller, and therefore the speed increases less rapidly – according to Newton’s “force is mass times acceleration”. The speed continues to increase until the net force becomes zero, at which point the speed remains constant: there is equilibrium between the downward gravity force and the upward friction. The size of the drops is critical. For a falling drop bigger than about 1 mm, air resistance is proportional to area, i.e. to the square of the diameter (D2). So a drop 2 times the diameter of another will feel a resistance 4 times as large at the same speed, and a drop 3 times the diameter will feel 9 times as much resistance. It is the area Everyday Physics: Unusual insights into familiar things 15 that counts because the air stream becomes turbulent, just as it does for a cyclist (as we will see on p59) and for a car (p87). Now suppose a drop falls at a certain speed and suddenly grows to twice the diameter (because it merges with several other drops, say). The friction force becomes 4 times as big. But since mass is proportional to the cube of the diameter (D3 ), the mass has becomes 8 times as big and thus the gravity force becomes 8 times as big. So there is now suddenly a greater net downward force, and immediately the drop starts to accelerate. This continues until the speed has increased so much that the forces are in balance again. That’s why large raindrops fall faster than small ones. 16 Everyday Physics: Unusual insights into familiar things How large do raindrops actually become? Could we have raindrops that are 10 cm in diameter? To answer that question we first have to look at how drops behave. Just like a balloon, water drops like to be spherical, because this gives the smallest possible surface area for a given volume. It is the attractive forces among molecules that minimize a drop’s surface area. These forces are the basis of surface tension, which acts as though an elastic membrane (like a balloon’s) were wrapped around the drop. Everyday Physics: Unusual insights into familiar things 17 As a drop falls, it feels the air resistance. For larger and faster drops the increasing air resistance causes the drops to flatten on the downward side (Fig. 1). As the drop reaches a diameter of 5 mm it becomes so unstable that it breaks into smaller drops, which is why we don’t get raindrops the size of footballs. (This can be nicely observed in a vertical wind tunnel, in an air stream at just the right speed to make the drops hang stationary in the air.) Raindrops stay nice and round up to a diameter of about 3 mm. If they become larger, they flatten on the downward side, the resistance increases and the drops no longer gain speed (Fig. 1). Fig. 1: As raindrops get bigger they tend to flatten on the underside. How high are those speeds? For drops 1 mm in diameter the speed turns out to be 16 km/h and for 3 mm drops it’s 28 km/h, which is nearly the maximum. For larger drops any further increase of speed is resisted by the flattening. The fastest drops are those that approach 5 mm in diameter, which achieve about 29 km/h – very little faster than 3 mm drops. So if you cycle fast, you can travel at the same speed as the fastest raindrops. Of course, they fall vertically and you go horizontally, so the drops strike you at exactly 45°, from the front, which is good to know if you want to use an umbrella on your bike. 18 Everyday Physics: Unusual insights into familiar things 07 WHY DON’T FOG DROPS FALL? Rain and fog both consist of water drops, but rain falls whereas fog just hangs in the air. The only difference is the size of the drops. Large drops fall much faster than small ones (as we just saw in the preceding chapter, which dealt with drops bigger than about 1 mm diameter). Fog drops fall very slowly if at all. The reason is that the gravity force on such tiny drops is very small – because it’s proportional to the mass of the drop, i.e. to the cube of the diameter. Everyday Physics: Unusual insights into familiar things 19 So far this looks just like the raindrops story. However, the fog drops are so much smaller and fall so much more slowly that a different law of friction applies. The air that flows around the little fog drops is well-behaved, forming nice streamlines that run more or less parallel; this is called laminar flow, and is less turbulent than with larger and faster objects. For droplets smaller than 0.1 mm the friction is directly proportional to the diameter. So when a droplet becomes half the diameter, it will also feel half the air resistance at the same speed, but the force of gravity is only one eighth (because that’s proportional to D3). Does this mean the fog droplets fall eventually? Probably, but only very slowly. Take fog droplets of 0.002 mm diameter. That’s about four times the wavelength of light and therefore large enough to be seen with your eyes. (You see the drops as a white cloud.) If you do the fairly complex calculation, you get a speed of 0.1 mm/s (or 36 cm per hour – a bit more than a foot per hour). That is not much – less than 10 m in a whole day. A breath of air will overwhelm the small force of gravity and keep such droplets afloat. However, some fog droplets really will continue floating even if the air is perfectly still, provided they are small enough! This isn’t so strange – after all, the molecules of air and water don’t fall to the ground either; instead they form the Earth’s atmosphere. Because of thermal motion, gas molecules and other small particles continue floating, so that the atmosphere does not fall on your head, but stretches above you for many kilometres. You can calculate how small the droplets must be in order to continue floating in the Earth’s atmosphere, like gas molecules do. Because fog drops are much heavier than individual molecules we cannot expect that they will float just as high and form as thick a layer as the atmosphere. To form a “droplet atmosphere” of modest thickness (say one thousandth of the thickness of the real atmosphere, i.e. a layer of fog having an effective thickness of 8 m instead of 8 km) the water droplets will contain some 1,600 water molecules. The diameter of such drops is 4.5 nanometres – about one ten thousandth of the thickness of a human hair. These are tiny droplets, so small that individual drops are invisible to the naked eye. But they don’t fall. 20 Everyday Physics: Unusual insights into familiar things 08 SKYDIVING: HOW FAST CAN YOU FALL? Suppose you bail out of a plane without a parachute. How fast will you be falling when you reach the ground? Does it matter at what altitude you bail out? Everyday Physics: Unusual insights into familiar things 21 As long as the plane doesn’t fly too low (not below about 1,000 metres) the altitude doesn’t matter. Once you start falling, your speed increases and so does the air resistance. However, the gravitational force remains constant, and at some point the downward-acting gravity is balanced by the upward-acting air resistance. Then, just as with raindrops (see How fast do rain drops fall? p15), the net force is zero, so your speed remains constant; this speed is called the terminal velocity, and that’s what we want to determine. The air resistance of a skydiver – just as for a car or a large raindrop – is proportional to the frontal area, to the square of the speed, and to a constant factor Cd, the drag coefficient. The value of the drag coefficient depends on the aerodynamic characteristics of the body: the more streamlined the shape, the less drag there is, and the smaller Cd is. As every skydiver knows, the frontal area and the drag depend on the shape you adopt when you fall. The area and the drag are largest if you spread out your arms and legs, and the terminal speed will be almost exactly 200 km/h, or 124 mph. That’s bad news: you have to be really lucky with your landing spot to survive. (Hope for a hay stack!) If you roll yourself up like a ball, the terminal speed will be even larger, for two reasons: first, your frontal area decreases, and second, your aerodynamics improve – you are more streamlined. The result is that you hit the ground at a frightening 320 km/h. (Hope for a very big hay stack!) Can you go any faster? Sure – if you start really high, in thinner air. That gives a lot less air resistance, since the resistance is proportional to the air density. Obviously, the nicest place to start would be at the edge of the atmosphere. But it is freezing cold up there, and you’d need an oxygen bottle for breathing. Even so, on 16 August, 1960, US Air Force pilot Joseph Kittinger jumped out of a balloon at 31,300 metres (about 100,000 feet, approximately three times the cruising altitude of a commercial airliner). The air pressure at that height is only about 2% of the sea-level value, so Kittinger started his fall practically in vacuum, with almost zero air resistance. His “free fall” lasted 4.5 minutes in total. He reached a speed of 980 km/h. Some people claim that he broke the sound barrier (about 1,050 km/h at that altitude), but that is most probably not the case. 22 Everyday Physics: Unusual insights into familiar things This was an irresistible challenge for Felix Baumgartner from Austria. On 14 October 2012, dressed in a kind of space suit, he jumped from a balloon at an altitude of 38,969 metres. During his free fall of 4 minutes 19 seconds he reached a maximum speed of 1,357.6 km/h, well above the speed of sound. So he became the first person who managed to break the sound barrier without using any sort of engine. But it wasn’t exactly a pleasant trip: sometimes it takes a bit of discomfort to end up in the Guinness Book of Records. Now let’s see how a parachute changes things. It increases your frontal area and thus decreases your speed dramatically. Using the traditional round parachute (with a surface area of about 60 m2 and Cd of about 0.8) the landing speed is about 18 km/h, which is the same as jumping off a 1.5 m high wall. Calculating the terminal velocity Let’s first look at a skydiver who jumps out of a plane in relatively dense air. After a few seconds she reaches a constant speed where her weight (mass m × acceleration due to gravity, g = 9.8 m/s2) is balanced by the air resistance Cd A × (½ ρ v2), where Cd is the drag coefficient, A the frontal area, ρ the density of the air (= 1.3 kg/m3 at sea level) and v the speed. This yields v = √ (2 m g / Cd A ρ). For a fully equipped skydiver we take m = 100 kg, and with arms and legs fully stretched we estimate A = 1 m2 and Cd = 0.8 for such a poorly streamlined body. Let’s assume that the skydiver jumps from an altitude of 3,000 metres (10,000 feet). There the air density ρ is only about 70% of what it is at sea level, or about 0.9 kg/m3. This gives v = 52 m/s or 188 km/h – in fair agreement with the actual value of 200 km/h. And now for Joseph Kittinger’s and Felix Baumgartner’s record jumps. In free fall in the absence of air resistance (i.e. very little or no atmosphere) the speed of any object increases with the acceleration due to gravity, g = 9.8 m/s2. In other words each second the speed increases by 9.8 m/s. After 30 seconds of free fall this yields a speed of 294 m/s or 1,058 km/h, practically the speed of sound. In practice the increasing air resistance will keep the speed somewhat lower. Even so, if the jump is continued some more, the sound barrier can be broken, as shown by Baumgartner. Will you hear a sonic boom? No, because there is hardly any air up there and therefore little to propagate sound, so you don’t hear anything. Anyway, there can’t be a boom because a boom requires a shock wave, and you can’t have a shock wave without air. Everyday Physics: Unusual insights into familiar things 23 Home experiment: Magic trick – the slow fall If you drop a pebble into a vertical pipe one metre long, you can calculate exactly how long it will take to reach the bottom: 0.45 seconds. Whether it’s a pebble, or a metal sphere or a coin, and whether the pipe is made of PVC, steel or copper, it makes no difference: it always takes about half a second, as long as the falling object easily fits the tube and the air resistance is negligible. Now you repeat the experiment, but using a small metal sphere instead of a pebble, and guess what? In the PVC tube it takes half a second to fall as expected, but it takes nearly ten seconds in the copper pipe. Your audience is flabbergasted. The explanation is simple: the metal sphere is a strong little magnet. Since the tube is made of a good electrical conductor (copper), the falling magnet induces electric currents in the pipe, just as happens in the copper windings of the generator of your car, or in your bicycle dynamo. The experiment is a simple and dramatic illustration of Lenz’s law which says that an electrical conductor resists a change of magnetic field by generating currents that induce an opposing magnetic field. The currents can circulate nicely in the tube provided this is conductive, and copper is a very good conductor. (The induced currents in the copper tube actually warm it up, but not enough for your hand to feel.) (For this trick, if you use a length of 22 mm copper pipe, your magnet should be about 19 mm diameter.) We can also view the experiment in the context of the law of conservation of energy. Part of the kinetic energy of the falling sphere is converted into heat which warms up the tube by means of electric currents. The reduction in the kinetic energy of the object emerging from the tube means it comes out of the tube at a lower speed. 24 Everyday Physics: Unusual insights into familiar things 09 HOW HIGH WILL THE SUN RISE TODAY? The Earth’s axis is inclined at 23.5° to the ecliptic – the plane in which the Earth orbits the sun (Fig. 1). Fig. 1: The Earth’s axis is inclined at 23.5°. Cambridge (C) is 52° above the equator. Working out the height of the sun at the equator is easy. At noon on 21 March or 21 September (the two equinoxes) the sun is directly over your head, i.e. at 90° above the horizon (E in Fig. 2). But for people in the US, Canada and Western Europe, it is a bit more complicated. Take Cambridge, England, or Kiska Island, in Alaska US, or Quebec in Canada, all at about 52° north (C in Fig. 2). When the sun is directly overhead at the equator, it is 52° lower here. Measured from the horizon, it is only 90 − 52 = 38° high. Everyday Physics: Unusual insights into familiar things 25 Fig. 2: At noon on an equinox, the sun is directly (90°) above the equator. For Cambridge (C) it is 38 degrees above the horizon. However, at midsummer (the summer solstice, 21 June), in the northern hemisphere the Earth’s axis is slanted 23.5° towards the sun, so we add 23.5° to the height of the sun for places in the northern hemisphere. Now the sun is directly above the northern tropic, the Tropic of Cancer (T in Fig. 3). So in Cambridge (C in Fig. 3), the height is 38 + 23.5 = 61.5°. For the winter solstice, 21 December, we subtract the same amount: 38 − 23.5 = 14.5°. Fig. 3: At noon at the summer solstice, the sun is directly above the Tropic of Cancer (T). For Cambridge (C) it is now 38 + 23.5 = 61.5 degrees above the horizon. What about dates in between? Here we can use the lucky coincidence that there are about as many days in a year (365) as there are degrees in a circle (360). The Earth proceeds by roughly 26 Everyday Physics: Unusual insights into familiar things 1° each day in its orbit around the sun. It is even simpler in terms of months: on average the Earth proceeds 360° / 12 = 30° per month. Now things become straightforward if we remember a bit of math from high-school. Continuing with the example of Cambridge, the height that the sun reaches is to a good approximation a sine curve (Fig. 4) around the average of 38°, with maximum and minimum amplitude of 23.5°. Now remember that a sine curve reaches half its maximum at 30° (one month in this case) and needs another 60° (2 months) to reach its maximum. In everyday terms, that means that at the equinoxes the day length changes most rapidly from one day to the next (the slope of the graph is at its maximum – points E1 and E2), and at the solstices it changes most slowly (the slope of the graph is zero – points S1 and S2). Fig. 4: The angular height of the sun at different dates. The slope of the curve is at its minimum at the two solstices (S1, S2) and at its maximum at the equinoxes (E1, E2). Everyday Physics: Unusual insights into familiar things 27 So, starting at 21 March, by 21 April the sun has already made half its gain reaching about 38 + 12 = 50° above the horizon (Fig. 4, M). It takes two more months for the sun to reach its full maximum of 38 + 23.5 = 61.5° on 21 June. Two months later, around 21 August, it passes 50° again, and one month later it is back at 38°. We have simplified things a little. For example, we assumed the orbit of the Earth around the sun is a circle, which is not quite right: the orbit is really an ellipse and the Earth travels a bit faster in the northern hemisphere’s winter, when the distance to the sun is a little smaller, and a bit slower in its summer (when the sun is a bit further away). Therefore, that “21st” date isn’t 100% accurate. And of course, not all twelve months of the year are equally long. Even the year is not exactly 365 days, but 365.25 days, so that every four years a leap year has to be introduced to remain in step. For that reason these “21st” dates shift a bit more now and then. But in spite of all these effects, our results are fairly accurate. 28 Everyday Physics: Unusual insights into familiar things 10 HOW HOT DOES THE SUN FEEL? Why do you sun-burn so easily while skiing or swimming? There are two obvious causes. First, you are outdoors all day, which most people aren’t used to. Second, the sunshine comes not only from above but it’s also reflected by the snow- or water-surface, unhindered by trees or buildings as happens in town, nearly doubling the intensity. In addition, the contribution of the blue sky under those circumstances is substantial: the great open sky not only contains indirect blue and violet sunlight, but ultraviolet too. Two other factors may be involved. The first is geographic latitude. Suppose you live in northern Europe and for your ski vacation you travel south to the Alps. For every 100 km southward the sun rises higher by roughly 1°. (The circumference of the Earth is 40,000 kilometres, so the distance from pole to equator is 10,000 km. That corresponds to 90°, and 10,000 / 90 gives 111 km per degree, to be exact.) So if you travel to the southern French Alps from London, say, then the sun is already 10° higher. Therefore, in the winter, at midday the sun is about 25° above the horizon instead of 15°. The distance the sun rays travel through the atmosphere is then much shorter, and that makes a big difference (Fig. 1). This is because the sun’s radiation is attenuated by the atmosphere, and when the sun is near the horizon, it has to radiate through a much thicker layer of atmosphere than when it is high in the sky. A very low angle of incidence therefore makes the trajectory through the atmosphere very much longer, so more of the radiation’s energy is absorbed. This is especially true for the ultraviolet part of the solar spectrum that tans or burns your skin. Everyday Physics: Unusual insights into familiar things 29 The second factor that can make a lot of difference is the height above sea level. The fact that you get closer to the sun is irrelevant because the change is at most a 2 km in a distance of 150 million km! What does count, though, is that you leave a lot of atmosphere below you, so that the amount of atmosphere that the sun’s rays have to travel through is less. At a height of 1,000 m the atmospheric pressure is 12% less, at 2,000 m 22% less. And at 3,000 m it’s 31% less (see box). Those are big changes, and they apply everywhere in the world. On the other hand, the damaging effect of UV on your skin is not a simple function of height. For example ozone, an important attenuator of UV radiation, is not evenly mixed throughout the atmosphere; it is found mostly at heights between 15 and 35 km. So it does not matter greatly if we skip the lowest few kilometres. Still, using sun screen in the mountains is a must, even when there’s no snow. Fig. 1: When the sun is high in the summer, the distance the rays travel through the atmosphere (AB) is much shorter than the distance in the winter when the sun is lower (CD). Air pressure and height The atmospheric pressure initially goes down rapidly with height, about 12% for the first 1,000 metres. After that, the pressure drops more slowly with height. In fact, it goes down roughly as an exponential according to e−h/8 km, where h is the height in kilometres. At a height of 1 km, the pressure is then only e−1/8, that is 0.88, of the pressure at sea level – a reduction of 12% as we saw above. On Mont Blanc, or Mt Vancouver, Alaska (both 4,800 m high), the air pressure is e−4.8/8 or 55% of that at sea level. At 5,000 m (5 km) the pressure is halved, which gives a nice rule of thumb: atmospheric pressure halves for every 5 km you go up. From 5 km to 10 km (the cruising height of aircraft) the air pressure is halved once more, and is only a quarter of what it was at sea level. 30 Everyday Physics: Unusual insights into familiar things 11 PARALLEL LIGHT BEAMS FROM THE SUN On days with lovely thunder clouds in the sky, you sometimes see several broad beams of sunshine emerging from holes in the cloud cover (Fig. 1). The beams don’t look parallel but seem to come from a point not very far above the clouds, making it seem like the sun isn’t very far away. However, the idea that the sun is so close is purely an optical illusion – or rather a perspective distortion. The beams are actually parallel. The perspective distortion you see here is the same as you see on a train line: the tracks seem to come from a single point far in the distance, although in reality they are parallel. Fig. 1: The sun’s rays seem to come from a point not very far above the clouds. Everyday Physics: Unusual insights into familiar things 31 (The comparison isn’t perfect: the sun’s rays do in the end come from something like a point (the sun) while the tracks do not – they remain truly parallel for the whole length of the track. But in practice this makes little difference.) How parallel are the light beams exactly? The sun is 150,000,000 km from the Earth. The distance between the holes in the clouds letting the light beams through might be 15 km, which is 1/10,000,000th of the Earth–sun distance. Then the angle between these two beams is the same as the angle between two strings that you hold 1 m apart and that are fixed to a nail 10,000,000 m (10,000 km) away. The curvature of the Earth prevents you from really holding those strings straight over such a distance, but if you could, you would see that, for distances of a few kilometres, to all intents and purposes the strings are parallel. Conclusion: if we say that sun rays are parallel beams, we are not far out. 32 Everyday Physics: Unusual insights into familiar things 12 SUMMER AND WINTER, WHY SUCH A BIG DIFFERENCE? In countries like England or the Netherlands the average temperature in the summer is about 17°C. In the winter it’s not much above freezing; 1 or 2°C. That sounds like the summer temperature Everyday Physics: Unusual insights into familiar things 33 is about 10 times more than winter. Is the sun so much further away in the northern hemisphere’s winter? No; in fact the sun is closer in January than in July. Actually, the question is rather misleading. The difference between summer and winter is not large at all: it only looks that way because our Celsius temperature scale has a somewhat arbitrarily chosen zero point. For a true comparison we should look at the temperature values with respect to the absolute zero point, in other words compare the summer and winter temperatures in kelvin rather than in degrees C. Then the difference is much smaller: about 290 kelvin compared to 275 kelvin, i.e. the summer temperature is 290 / 275 = 1.05 times the winter temperature, not 17°C / 2°C = 8.5 times. When we look at it this way, we should be surprised that the difference is so small in England or the Netherlands, for two reasons: 1. Winter days are much shorter than the summer’s. In midwinter the sun is above the horizon for only half the time that it is in midsummer (Fig. 1). 2. The sun is much lower, which makes a particularly big difference. In midwinter the sun hardly rises 15° above the horizon. So a sunbeam of 1 m2 cross-section is distributed over 4 m2 on the ground (Fig. 2) whereas in the summer, with the sun at its highest point, the sunbeam is distributed over just 1.1 m2. Conclusion: the winter sunshine on the ground is many times more “dilute” than the summer sun. Fig. 1: The length of the day in summer (ABC, left) is much longer than in winter (ADC, right). (Point D is Vermont, USA, at 45° north.) 34 Everyday Physics: Unusual insights into familiar things Fig. 2: When the sun is high the same amount of solar energy is spread over a relatively small area and is spread over a bigger area in winter. This combination means that, in the south of England or in the Netherlands for example, during the shortest winter day (around 21 December) only about 1⁄7 of the solar energy is received per unit area compared to the longest summer day. On top of this, at very low elevation the sun is also weakened by the much thicker air layer it has to travel through (see Fig. 1, How hot does the sun feel? p30), so overall a unit area on 21 December receives hardly 1⁄10 of the solar energy compared to 21 June. This prompts the question: why is the temperature difference between summer and winter not much bigger? Part of the answer in western Europe is the equalizing effects of the warm Gulf Stream, and of the atmospheric currents and the entire “weather machine”. However, the most important reason is that the seasons aren’t long enough for the land to reach equilibrium with its surroundings (atmosphere and sea), and this is especially true for maritime regions. The sea acts as a heat buffer: it has a huge heat capacity, and this attenuates temperature fluctuations. As a result, the differences between summer and winter are much smaller than in mid-Siberia (or Mongolia), for example, which are actually at the same latitude. The dry Siberian soil heats up and cools down much more quickly than the sea. In addition, the water in the sea is constantly being mixed, so the deeper layers contribute to the buffering effect, something that can’t happen on land. The slow coming to equilibrium also shows up in the lag of the seasons. The warmest weeks in the summer on average do not occur around the summer solstice (approx. 21 June in the northern hemisphere, 21 December in the southern hemisphere), when the sun reaches its maximum height, but more than a month later. Everyday Physics: Unusual insights into familiar things 35 Similarly, in winter the coldest time isn’t the winter solstice, but quite a bit later. All in all, we can be glad that the Earth orbits the sun so rapidly. If it took five times or so longer, the seasons would be five times longer. The land would have time to cool down fully in winter before starting to warm up again in spring, and we would have truly Siberian seasons even in western Europe. 36 Everyday Physics: Unusual insights into familiar things 13 WHY DO WE WALK THE WAY WE DO? The way you walk seems normal because everybody does it. You swing your arms and legs in opposite directions; when your left leg goes forward, your left arm goes backward. Same thing on the right. Why do you do that? Because you unconsciously take account of a law of physics – that the angular momentum (the amount of rotation) of a body remains constant in the absence of external forces. Your arms and legs are not attached to the middle (vertical axis) of your torso, but to the side. When your right leg moves forward it must push itself off from the right side of your body, so the right side of the body is pressed backward (just as there is recoil if you shoot a gun from your right shoulder). The consequence is that you tend to turn around your vertical axis. You prevent that by moving your right arm backward at the same time, in compensation. Everyday Physics: Unusual insights into familiar things 37 You could move your left arm and left leg forward or backward at the same time, but then the movement of the arm wouldn’t compensate for the movement of the leg; instead it would make the rotation worse. This would happen at every step, and your feet would have to absorb this rotation in their contact with the ground. So, walking this way not only looks funny, it’s inefficient too. The same consideration applies when you walk up the stairs with a cup of tea, to surprise your partner in the morning. As you walk up the stairs with the tray in your hands, you inadvertently move the tray to and fro, because you miss the natural swing of the arms. Thus, a piece of applied science prevents you from spilling tea all over the place. Ten-pin bowlers are especially troubled by this problem when throwing their heavy bowling ball. However, experienced bowlers have found a solution: just as they release the ball, they automatically make an elegant sweep with their free leg to compensate for the torsion. The law of physics provides an unintended bit of sporty elegance! Home experiment: A law with an unexpected twist With an office chair or a bar stool that swivels smoothly, you can give two nice demonstrations of the law of conservation of angular momentum. (The chair must swivel very smoothly – lubricate it to make sure.) Demonstration 1. This illustrates the pirouette principle: the more mass we move inwards towards the axis of rotation, the more the angular speed increases. (You can see this very clearly with a rotating figure skater. When their arms and one leg are outstretched, they rotate slowly, but as they draw in their limbs, they rotate faster and faster.) Raise the seat of your swivel chair to its highest position (to keep your feet off the floor). Sit on the chair holding a full bottle of water in each hand. Spread your hands horizontally and get someone to spin you and the chair (Fig. 1a). Now bring the two bottles towards your chest or – better still – above your head, because that is as close as possible to the axis of rotation (Fig. 1b): your rotational speed increases. If it goes too fast you can slow down by extending your arms again. 38 Everyday Physics: Unusual insights into familiar things Fig. 1a: Spread your hands containing bottles of water, and get someone to spin your chair. Fig. 1b: Raise the bottles above your head and your rotation speed increases. This is the law of conservation of angular momentum in action. During these operations, there is no external force, so the amount of rotation – the angular momentum – remains constant. However, it depends not only on the rotational speed, but also on the distribution of mass around the axis of rotation. As a consequence, if the mass gets closer to the axis, the rotational speed must go up to compensate. We see the same phenomenon in tornados: as the moving air gets closer to the “eye” of the tornado, its rotational speed increases. Demonstration 2. For this demonstration you need a bicycle wheel that spins freely on its axle. If there’s a tyre on the wheel, so much the better: what you want is a lot of mass on the outside, so a tyre on a heavy rim is best. Everyday Physics: Unusual insights into familiar things 39 Attach a handle to each side of the axle. Make each handle from a metal clothes hanger from the dry cleaners. Put one side of the axle in one narrow end of a clothes hanger, and tighten the nut securely, just as if the wheel were in the fork of a bike. Now bend the clothes hanger at a right angle out from the wheel, and fold the protruding part of the hanger in two, to make a nice grip. Do the same with the second coat hanger on the other side of the axle, so you can hold the wheel with both hands (Fig. 2a). Now you’re ready to go. We will now use the fact the amount of rotation – the angular momentum – is characterized not only by its numerical value but also by the direction of the axis of rotation, and this is included in the law of conservation of angular momentum. (In other words, angular momentum has magnitude and direction: it is a vector quantity.) Sit on your swivel chair and using both hands hold the wheel firmly with the axle horizontal (Fig. 2a). Get someone to spin the bike wheel rapidly. Now tip the rotating wheel so that the axle is vertical (Fig. 2b). Behold: the whole set-up – you, the chair, and the bike wheel – starts to rotate! And if you tip the wheel by 180° (i.e. turn it upside down, Fig. 2c) you start to rotate in the opposite direction! Here’s why. When the wheel started to rotate with the axle horizontal, there was no angular momentum in the vertical direction (i.e. no spinning around a vertical axis), and since there are no external forces the conservation law says that must remain so. When you tip the wheel axle over vertically, there is now a rotation around a vertical axis, which must be compensated for by a rotation of the whole configuration (you and the chair included) in the opposite direction. a) b) c) Fig. 2: A surprising experiment with a bicycle wheel. 40 Everyday Physics: Unusual insights into familiar things Home experiment: The reckless wine glass party trick Take a piece of string about 120 cm long. Tie one end to an ordinary wine glass, and tie the other end to a wine bottle cork. Take a wooden stick about 40 cm long; the handle of a wooden spoon or ladle will do fine. Hold the stick horizontally in you right hand. Take the cork in your left hand and pull the string to the left, so that the glass dangles just below the stick (Fig. 3a). Now tell your audience that you’re going to let the cork go. Everyone will think the glass will crash to the floor! When you let the cork go (Fig. 3b) the laws of physics come into play: the cork end of the string wraps itself around the stick, and the glass doesn’t fall to the floor! (Practise with a big soft cushion under the glass until you are confident – and don’t use your best glasses!) Hold the stick at a slight angle towards you, to prevent the cork from hitting the string and instead of wrapping round the stick bouncing back and ruining your demonstration (and the glass). What’s happening is a beautiful illustration of the law of conservation of angular momentum. When you let go (Fig. 3b), the cork is pulled towards the stick, and simultaneously it falls downward and starts to wind around the stick, getting shorter as it does so. Now the conservation law kicks in: as the string gets shorter, it revolves more rapidly (Fig. 3c), just like the pirouetting ice-skater. Before the glass hits the ground, the cork has safely tied the string around the stick (Fig. 3d) and the glass is saved. Once again the laws of physics yield another astounding experiment. a) b) c) d) Fig. 3: Because of conservation of angular momentum, the cork wraps the string around the stick and stops the glass falling to the ground. Everyday Physics: Unusual insights into familiar things 41 42 Everyday Physics: Unusual insights into familiar things 14 THE HUMAN ENGINE We’re going to see how much energy the average person uses when performing various activities. However, before we do that, we’ll define a few terms so that we can be precise in our analysis. Energy, work, heat When you hit a squash ball you do work, and transfer energy to the ball. With that energy the ball in turn can do work – break a window, compress a spring, or move another ball. So work and energy are interchangeable and therefore have the same unit: the joule (rhymes with cool), symbol J, in the international MKS (metre, kilogram, second) system of units (also known as SI, from the French Système International). One joule of work is equal to one unit of force (one newton, N) times one unit of distance (metre, m). In other words: 1 J = 1 Nm (one newton metre). Work and energy finally convert to heat. (The squash ball becomes noticeably warmer after playing for a few minutes.) The unit of heat is also the joule. One calorie (an old unit of heat) equals about 4.2 joules, so one kilocalorie or kcal (often wrongly called a calorie when talking about food and diets) is 4.2 kJ. Power Power is the rate at which work is done, or energy is used, or heat is produced. So the unit of power is joules per second. Because this is such a widely used unit, it is also given the specific name watt, Everyday Physics: Unusual insights into familiar things 43 abbreviated W: 1 W = 1 J/s. Conversely: one joule is one watt for one second, or 1 watt second (in the same way that a “person-hour” is one person working for one hour): 1 J = 1 Ws. Most people are familiar with watts in the use of electricity. The speed of rotation of the electricity meter at home is a measure of the number of watts that are “on” (i.e. are being used) in the house. If 1,000 W is on for one hour, the energy used is 1,000 watt hours, that is 1 kWh, which is the unit you see on your electricity bill. You can convert the kilowatt-hour into “proper” energy units (joules) as follows: 1 kWh = 1,000 W for 3,600 seconds, that is 3,600,000 watt seconds (Ws) = 3.6 million joules or 3.6 MJ (megajoules). The same goes for heat. The wood in a matchstick has a total energy content of about 2,000 J. If the match burns for 20 seconds, it’s acting as a heater with a power of 2000 J/20 s = 100 J/s, that is 100 W. 44 Everyday Physics: Unusual insights into familiar things People at rest When a lot of people are in an enclosed space like a classroom or theatre or concert hall, they warm the place up. Each of us produces heat, because in a sense each of us is a small engine. When we sit still the engine idles, and all it does is produce heat. The cat knows this and is glad to crawl into our lap (although we prefer to think it’s because she loves us). How much heat do you produce? You can work this out easily, by considering your food intake. For an adult this is about 2–3,000 kcal per day. (Kilo)calories are dieters’ measures; the physics units are joules. One kcal = 4.2 kJ, so your food intake is equivalent to about 10,000 kJ or 10,000,000 J per day. When you don’t do much physical work, and if your metabolism is functioning properly and your weight remains constant, all that food is converted into heat. That happens over the full 24 hours of the day, i.e. over 24 × 60 × 60 = 86,400 seconds. So your average heat production per second is 10,000,000 J/86,400 s = 115 J/s, which is 115 W. So apart from top athletes or people doing heavy physical labour, a human generates about as much heat as a 100 W incandescent light bulb. That’s your resting energy expenditure. Everyday Physics: Unusual insights into familiar things 45 It’s amusing to see what’s equivalent to an adult’s 10,000 kJ daily energy consumption. Burning oil or petrol generates 40,000 kJ per kg, so energy-wise a quarter litre (one cup) of oil supplies an adult’s energy needs for a whole day. A car travelling at 120 km/h on the highway uses a quarter litre in about two minutes, compared to which a human’s energy use is pretty economic. Here’s another way of looking at it. Of the food constituents that you eat (carbohydrates, proteins and fats), fat has the highest combustion value. Because fat is not so different from fuel oil, its combustion value is about the same as that of oil. This means that the energy in a quarter kilogram of fat is enough to live on for a day. Conversely, if you ate nothing, your energy for one day would come from burning a quarter kilogram of body fat. So be wary of any slimming advertisements that suggest you can lose more weight that that every day, especially if you continue to eat a little. (When you start dieting, you often lose weight rapidly through loss of water, which is great for morale – “see how many kilos I’ve lost!” – but makes no long-term difference.) The polar explorer’s diet People often expect that solo Antarctic explorers, who have to carry all their food and fuel themselves, bring only dried “astronaut” foods, but they don’t. They use about 8,000 kilocalories (or 34,000 kJ) a day, so they need a very high calorie diet with minimum weight that requires minimum cooking – so they take a lot of butter. (“It’s horrible for the first few days, but then you crave the fat content and eat it like slabs of cheese.”) However, it’s difficult (and boring!) to live on nothing but butter, so they also take cereals/cereal bars, nuts, dried fruit, chocolate and salami – and maybe some freeze-dried meals as treats. 46 Everyday Physics: Unusual insights into familiar things 15 THE HUMAN ENERGYEQUIVALENT OF A VACUUM CLEANER How many watts of mechanical power can a human generate? Take a guess. A human is nowhere near as powerful as a car (which is obvious as soon as you try to push a car). The power of a car engine is about 40 kW, so a human is far less powerful that that. But how much less? Is the power of a human 10 watts, or 100 watts – or even 1 kW, which is the power of a vacuum cleaner? You can estimate the power of a human by looking at the simple but efficient movement of walking up the stairs. The movement involved looks a lot like cycling and uses the same muscles. Most of the effort expended is in raising your body the height of the stairs. (You can ignore the energy for the horizontal component of your travel without significantly affecting your calculation.) It’s easy to calculate the energy needed to gain height. You just need to know the gravitational force you have to overcome (your weight), and remember that work done equals force times (vertical) distance. Everyday Physics: Unusual insights into familiar things 47 A reasonable value for how fast you walk up a stairway for a sustained period is one step per second. Compare that with hiking in the mountains: one 15 cm (6”) high step per second corresponds to gaining 500 metres height in one hour, which is fairly strenuous – so one step per second is a reasonable estimate for going up stairs. The calculation (see box) shows that you generate 100 W going up the stairs, well short of the power of a car. Note that here we are talking of mechanical energy only; the total energy used is a lot higher, because the efficiency of your muscles is well short of 100% and a multiple of that 100 W is released as heat (see How do you keep your temperature constant? p49). How much – or how little – 100 W is becomes clear if you generate electricity with a home trainer. Converting mechanical to electrical energy can be about 90% efficient, but for simplicity let’s say it is 100%. So, if you spend 10 hours generating electricity on the home trainer, cycling all the time, then at the end of the day the total work done = power × time = 100 W × 10 h = 1,000 Wh or 1 kWh altogether. You get that out of the wall socket for about 15 pence or 25 cents. Conclusion: even if electricity is expensive, pedalling on a home trainer is not a good alternative. The energy you use going up stairs The potential energy you gain by rising a height h in the gravitational field with an acceleration due to gravity g of ≈10 m/s2 is mgh, where m is the mass (and therefore mg is the weight in newtons). For a step height of 15 cm and a person weighing 70 kg, the work done for one step is: mgh = 70 kg × 10 m/s2 × 0.15 m = 105 J If you do this at a rate of one step per second, then the power is 105 J/s, or roughly 100 W. 48 Everyday Physics: Unusual insights into familiar things 16 HOW DO YOU KEEP YOUR TEMPERATURE CONSTANT? When you exert yourself physically, e.g. by running to catch a train, or biking into town, you produce not just mechanical energy, but also a good deal of heat. Your muscles don’t convert nutrients into mechanical work at 100% efficiency. (In that respect people are like car engines: a large fraction of the energy used is wasted in the form of heat, which has to be removed.) The efficiency of your muscles depends on the kind of work that you do: for cycling or climbing stairs the efficiency is about 25%. That means that the rest of the expended energy (75%) comes off as heat, which is three times as much as the useful work done. And that is on top of your resting energy expenditure that just keeps your body systems going. Everyday Physics: Unusual insights into familiar things 49 To maintain a constant temperature, the heat production and heat removal have to be equal all the time. If you exert yourself more, you produce more heat, so the rate of heat removal has to increase. You could adapt your clothing to your exertion (which is why athletes wear shorts and singlets) but that’s not suitable for everyday life. In fact you cool your human engine – remove heat – in three ways: (1) conduction to the ambient air, (2) radiation and (3) evaporation. Does the wind not contribute? We tend to think of it as a separate mechanism, but the only thing the wind does is bring the cool air closer to your body. It doesn’t penetrate your body to bring the coolness in, and if you were to look closely enough you would find that it streams parallel to your skin. Therefore, its only effect is a steeper temperature gradient close to your skin, which causes more rapid heat removal by conduction. In short: conduction, radiation and evaporation are all you’ve got. The rate of conduction is completely determined by the temperature difference between the outside of your body and the environment. (This is also true for radiation, although it’s not obvious – see the Radiating body box below.) The amount of heat removed by conduction and radiation remains practically constant, 50 Everyday Physics: Unusual insights into familiar things as long as the ambient temperature and your body temperature do not change. (During exercise, the temperature of your skin increases a little, and gets closer to your core temperature of 37°C due to increased blood circulation, but it never exceeds your core temperature.) Conclusion: heat removal by radiation and conduction hardly increase even when you exercise hard. Evaporation is the only heat removal method that you have more control over, so it’s your main control mechanism for achieving heat balance. And it’s very effective, because evaporating water requires a lot of heat. (You know that from your kitchen: if you put a kettle on the stove and it takes, say, 5 minutes to come to the boil, it will take another 20-30 minutes for the kettle to boil dry. This shows that evaporating the water takes about five times as much energy as heating the water from room temperature, about 15°C, to 100°C.) The importance of evaporation is illustrated schematically in Fig. 1, which shows how evaporation affects someone in normal dress on a home trainer at ambient temperature. The horizontal Everyday Physics: Unusual insights into familiar things 51 axis shows the muscular work done per second; the vertical axis is the energy production per second. ► The green line shows the total energy produced per second, including both heat and mechanical work. ► The blue line shows how much energy is produced as heat, which needs to be removed. ► The height between the blue and the red lines is the amount of mechanical/useful work done. This is zero, (i.e. the green and blue lines coincide) when there is no muscular work done, representing the resting energy expenditure. Fig. 1: Where the energy goes when you exercise on a home trainer. 52 Everyday Physics: Unusual insights into familiar things For a cyclist to generate 100 W of useful muscular power (AB, Fig. 1) at an efficiency of 25%, the power required is 400 W. On top of that there is the 100 W resting energy expenditure, so the cyclist has to generate a total of 500 W (AC in Fig. 1). Only 100 W is useful, and the remaining 400 W is heat that has to be removed – BD being the amount lost through evaporation, and DC the amount radiated or convected. When little or no muscular work is done (bottom left of Fig. 1), the heat removal is largely by radiation and conduction. As the exertion increases, evaporation becomes progressively more important, and you sweat more when you work harder. Because radiation and conduction can’t increase appreciably, evaporation has to remove practically all the extra heat. (If you can’t increase the evaporation – as in the simple example of a cyclist on a home trainer where there’s no rush of passing air to aid evaporation – you may overheat.) There’s a lesson here if you swim in water that is too warm. If the water is cooler than body temperature (37°C), your body loses some heat to the surrounding water. But if the water is above 37°C, the only cooling available is from your head that is above water, and evaporation from such a small area can’t help much. If a swimmer tries to establish an Olympic record in the 400 metres under those conditions, they could well come to grief from overheating. Everyday Physics: Unusual insights into familiar things 53 Radiating body The amount of heat radiated by a body is proportional to the temperature raised to the fourth power. It is equal to σ T 4 times the area of radiating surface where σ is the Stefan– Boltzmann constant and T the absolute temperature in kelvin, K. However, to find the net radiation transfer to the environment, we must also factor in the radiation coming back from the environment to you, which in terms of absolute temperature is not much cooler than you are. Then the net amount of heat you lose is proportional to the temperature difference. If Tb is your body temperature, and Ts is the temperature of your surroundings, then the net radiation that you emit is proportional to: Tb4 − Ts4 Using standard mathematics – remember, a2 − b2 = (a+b)(a−b) – we can rewrite that as (Tb2 + Ts2) (Tb2 − Ts2) and by rewriting the second factor again, we get: (Tb2 + Ts2) (Tb + Ts) (Tb − Ts) Now the difference in temperature, Tb − Ts, which we’ll call ΔT, is small. So, in the factors with the plus signs, we can forget the difference between the two, i.e. Tb ≈ Ts ≈ T. That gives us: (T 2 + T 2 ) (T + T) (ΔT) which is 4 T 3 ΔT. We can also view that graphically: if on the T4-curve two points are close together, we can regard the line joining the two points as a straight line – the tangent to the curve. In the language of differentials: Δ(T4) = 4 T 3 ΔT 54 Everyday Physics: Unusual insights into familiar things 17 HOW EFFICIENT IS CYCLING? While walking is fairly efficient compared to flying and other ways of locomotion in the animal world, humanity improved transport performance by a factor of four to five with the invention of the bicycle. Cycling beats everything in the transport category for the amount of energy per kilogram moved over a given distance. For a bike this is about 0.6 kJ/(kg⋅km) (i.e. it uses 0.6 kJ to transport 1 kg a distance of 1 km) although this depends on the kind of bike and the speed, as we’ll see in the next chapter. Everyday Physics: Unusual insights into familiar things 55 Fig. 1 shows how efficient the bike is. In the figure, several modes of transport are arranged by mass – light ones on the left, heavy ones on the right. The vertical axis shows the energy used per displaced kilogram per km. You can see that a human cyclist is about 100 times more efficient than a rat, and about 6 times more efficient than a human walker. Fig. 1: Comparison of the energy different animals use, per km per kg transported mass, arranged from light (left) to heavy (right) (Data from SS Wilson, Scientific American March 1973, p90). 56 Everyday Physics: Unusual insights into familiar things Why is cycling so much more efficient than walking? This is partly because the centre of gravity of a cyclist doesn’t move up or down: when one leg rises the other goes down, so overall there’s no change. That’s not the case with walking: your centre of gravity moves up and down about 3 cm with every step, and that wastes energy. How much energy does cycling take? The muscular power it needs is comparable to that of mounting stairs, because cycling involves much the same movement as climbing stairs. As we saw in The human energy equivalent of a vacuum cleaner, p47, that requires about 100 W. From that we can deduce a cyclist’s energy use by means of a thought experiment. We saw that 100 W means a gross energy production of 400 W, given a muscle efficiency of 25%, and that a continuous power output of 400 W is the equivalent of burning 1 litre of oil per day (see The human engine: People at rest, p43, where we stated that 100 W = ¼ litre/day). So 1 litre of oil is the Everyday Physics: Unusual insights into familiar things 57 energy equivalent of a 24 hour bike ride. In those 24 hours you cover a distance of 24 × 20 km, approximately 500 km. Conclusion: a cyclist uses roughly 1 litre per 500 km, which is enormously efficient (especially compared to an SUV at about 50 litres per 500 km). But there’s a catch: you don’t bike on oil or petrol, but on bread, milk, steak, cheese. Producing all that food in the first place needed much more energy than the energy (calorie) value of the food itself. Take milk, for example. The cows have to eat, they have to be milked, and the milk has to be cooled, transported, heated to be pasteurized, cooled again, and transported again. If you add up all these steps, a litre of milk turns out to cost half a litre of oil before it comes on the table. It is much the same with the rest of your food. So while biking is wonderful, and great for staying in shape, to save energy you might be better off buying a light motorcycle. 58 Everyday Physics: Unusual insights into familiar things 18 WHAT FORCES AFFECT A CYCLIST? In places like the Netherlands it is easy to see the forces involved in cycling: the terrain is flat, so you don’t have to go up hills against gravity. The bike itself has little resistance if it’s in good condition: the ball bearings only cause an energy loss of about 1%, a well running chain about 1.5%, and a derailleur gear at most 5%. The real forces that resist you, once you are up to speed, are the rolling resistance and the air resistance. Since these behave quite differently with increasing speed, we’ll look at them separately. Everyday Physics: Unusual insights into familiar things 59 The rolling resistance is largely caused by the tyres. They convert some energy into heat as they are deformed by contact with the road. The resistance is independent of the speed, and is proportional to the weight and to the “rolling resistance coefficient” Cr (see box). The magnitude of Cr relates to the deformation process of the tyre, and is strongly influenced by the tyre pressure. It may seem strange that the tyres cause resistance. After all the elastic rubber returns neatly to its original state after deformation, so no energy is consumed in permanent deformation. But the whole dent-and-bulge cycle does consume energy, because the tyre has to be dented with more force than it delivers when bouncing back. Therefore, it warms up, something you can clearly feel easily with car tyres after a long drive. The air resistance increases with the square of the speed. So at 20 km/h it’s 4 times greater than at 10 km/h. It is the air resistance that troubles you most when you want to go fast. Also, air resistance is proportional to the surface area seen from the front (the “frontal area”). The proportionality coefficient is the “Cd value”, drag coefficient or air resistance coefficient, which is determined by how 60 Everyday Physics: Unusual insights into familiar things streamlined the shape of the body is. For a cyclist sitting upright, Cd is about 1, or about 0.88 for a bike racer (and in this case the frontal area is also smaller, leading to a double gain). For cigar-shaped super bikes – the HPVs (Human Powered Vehicles) – it is 0.1. Fig. 1 shows the resistance for an ordinary town bike at different speeds. At very low speeds, there’s almost no air resistance so it’s mostly the rolling resistance you have to overcome. At 10 km/h, the air resistance (AB in Fig. 1) is about half the rolling resistance (AC). At about 15 km/h they are equal (DE = DF). Thereafter the air resistance dominates. Therefore, for high speeds streamlining is essential to reduce air resistance. HPVs can exceed 100 km/h, something an ordinary racing cyclist cannot even dream of (see Can you cycle at 100 km/hr? p67). Fig. 1: How resistance increases with speed, for a town bike. An interesting feature here is that the total resistance that the cyclist meets also represents the energy consumption per km. This is because energy = force times distance, so force = energy divided by the distance, or energy per unit distance. Take the case where the total resistance force is 20 newtons; 20 newtons = 20 joule per metre = 20 kJ per km. Everyday Physics: Unusual insights into familiar things 61 Since the total resistance increases with increasing speed, the energy used per kilometre also goes up. Biking fast not only costs more energy per hour (which is obvious, because you’re working much harder), but also costs more per kilometre, although it does save time and keeps you fit. (At a steady 20 km/h you burn about 2 grams of fat per kilometre, see box. If you want to lose weight by cycling, then you know what it takes.) However, if you have a particular distance to cover and want to save energy, cycle slowly. Rolling resistance and air resistance The rolling resistance can be written as Cr × m g, where Cr is the rolling resistance coefficient, m the mass and g the acceleration due to gravity. For a town bike Cr is about 0.006, and for a racing bike 0.003. The losses are mainly due to the fact that the force with which the tyre is dented is greater than the one with which it bounces back; this is a form of hysteresis (the closed loop integral F·dS is not equal to zero). Another part comes from “Reynolds-slip” (the friction of interfaces due to the deformation). The air resistance is directly proportional to the air density, ρ, and proportional to the square of the speed (unlike that for small raindrops) because the flow pattern is not laminar but turbulent. From Bernoulli’s law (p + ½ ρ v 2 = constant, where p is the pressure) we can derive right away that the dynamic pressure in front of a flat surface perpendicular to the air stream is equal to ½ ρ v 2 . For an object with an arbitrary shape and a frontal area A, this yields a force Cd × A × ½ ρ v 2 . The air resistance or “drag coefficient” Cd is about 1 for the ordinary bike and 0.1 for super bikes. The limiting value is about 0.05 for the ideal streamline, more or less a fish shape. For the relevant speed range Cd can be considered a constant. The total resistance for an ordinary town bike (Cr = 0.006; m = 90 kg; Cd = 1.1; A = 0.51 m2) at 20 km/h turns out to be 16 newtons (see Fig. 1). The mechanical energy use is therefore 16 kJ/km. With a muscle efficiency of 25% this means a total energy use of 64 kJ/km, which is equivalent to burning 1.6 gram of oil per km (assuming a combustion value of 40 MJ/kg), or 1.6 × 500 g per 500 km, = 800 g/500 km. As the density of oil is about 900 g/litre, this agrees well with our earlier rough estimate (see How efficient is cycling? p55) for the oil-equivalent of a cyclist’s energy use of 1 litre per 500 km or 2 grams per km. 62 Everyday Physics: Unusual insights into familiar things 19 CAN YOU CYCLE AT 100 KM/H? If you want to cycle fast, it’s clear from the figure on p61 (What forces affect a cyclist?) that you have to minimize the air resistance. Racing cyclists do this by crouching down over the handlebars and wearing streamlined helmets. Even better is a reclining “super bike” with a small frontal area and a perfect streamline. Everyday Physics: Unusual insights into familiar things 63 Not surprisingly, the top speed you can reach increases with the power – energy per second – that you can develop for the duration of the speed test. Official bike-speed records are normally measured over a 200 m course, with a flying start. We can read off the power directly from the necessary force in Fig. 1, p61, if we remember that power is force times distance per second, that is, force times speed. If we know the force, then we know the power at every speed. A surprising point is that a cyclist starting from stationary – or a car or train starting from stationary – is expending zero power while stationary. This is logical: even when there is a force, as long as there is no movement, no work is done. However, as every cyclist knows, getting your bike moving from rest requires a lot of effort, so is there a contradiction here? No, because how the force is generated differs between a human and a concrete block, say. Think of a concrete block placed on the pedals when the bike is stationary: it’s clear that this expends no energy. However, for a human to press hard against a wall, or to press down on the pedals, or for a train to start moving, energy is expended, because of the chemical and/or biological actions (in your body or in the engine) needed to exert the force. Fig. 1: Power (in watts) required to propel a town bike (red line) and a human powered vehicle (HPV) (blue line) at different speeds. Fig. 1 shows the power for two different cases – an ordinary town bike, and an HPV with Cr = 0.0045, m = 90 kg, Cd = 0.10, and frontal area A = 0.44 m2. The difference is striking. For example, we see that using the power needed to go 20 km/h on a town bike, 64 Everyday Physics: Unusual insights into familiar things we could reach about 50 km/h on an HPV. The maximum power that can be developed by a professional cyclist for a short interval is around 750 watts (one horsepower!). On an ordinary town bike you soon run up against that limit – at about 40 km/h (X, Fig. 1) – but an HPV could do nearly 100 km/h (Y, Fig. 1). And many HPVs have done so. In the 1980s and 1990s a number of HPVs exceeded the then speed limits in the US of 55 mph (88 km/h). Some speed freaks got themselves fined on purpose, and became the proud holders of an “honorary speeding ticket” from the California Highway Patrol. Everyday Physics: Unusual insights into familiar things 65 The 100 km/h limit was exceeded in 1998, and in October 2002 the Canadian Sam Whittingham rode 130.36 km/h (not at sea level, but at 407 metres altitude on Battle Mountain, Nevada, where the air pressure is lower). Meanwhile this record has been broken a few times. In 2016 it was raised to 144.17 km/h by the Canadian Todd Reichert, also at elevation in Nevada. (The International Human Powered Vehicle Association registers these records.) To cycle faster still, you can almost completely eliminate the air resistance by riding very closely behind a racing car with a large flat vertical windshield attached to the back. That’s exactly what Fred Rompelberg from Maastricht, the Netherlands, did on the Bonneville Salt Flats, near Salt Lake City, Utah. On 3 October 1995 with a breath-taking 269 km/h he became the fastest cyclist of all time, even without using a super bike! 66 Everyday Physics: Unusual insights into familiar things 20 HOW FAST CAN YOU CYCLE ON THE MOON? Nobody has ever cycled on the moon (yet!). There are no roads and no oxygen. But let’s just suppose. The moon would be perfect for a record attempt: there’s no atmosphere (so no air resistance), and gravity is only 1⁄6 of that on Earth, so the rolling resistance is only 1⁄6 too. Of course you’d need an oxygen tank, which means extra weight, but that should be manageable. Everyday Physics: Unusual insights into familiar things 67 So how fast could you go? The calculation is straightforward. Since the rolling resistance is the only counterforce, we need just a reasonable assumption for the mass of the “moon racer” and for the rolling resistance coefficient Cr ; let’s assume m = 100 kg (in view of the oxygen tank and the space suit) and Cr = 0.0045, the value for a super bike on Earth and about the average between the value for a town bike and a racing bike. If you develop 750 watts, as on Earth, your top speed is 3,700 km/h. Isn’t that faster than sound? Well, it would be three times the speed of sound on Earth in air at normal temperatures. But on the moon you have no problem with the sound barrier because there is no atmosphere in which sound can propagate. You do have to look out for lunar hills, though: 3,700 km/h is half the escape velocity, so if you shoot up a hill, you might launch off into space! 68 Everyday Physics: Unusual insights into familiar things 21 IS CYCLING REALLY HARDER WITH A SIDE WIND? When you’re cycling, a tail wind is an advantage, and a head wind definitely makes pedalling harder. But does a side wind coming at you from right angles give you any trouble? At first thought you might think that this cannot make any difference, but in fact you’d be wrong. Strange as it may seem, pedalling is definitely harder in a side wind. Everyday Physics: Unusual insights into familiar things 69 The explanation is that the air resistance on a cyclist increases disproportionally with the speed (see What forces affect a cyclist? p59). On a completely windless day, if you double your speed the air resistance is four times greater. That’s easy enough: if you imagine the air as a sea of small ping-pong balls you have to cross, then doubling your speed means you hit those balls at twice the speed (the momentum is doubled). But in addition you meet twice as many ping-pong balls every second, so that gives a total of four times as large a force (the momentum transfer per second is quadrupled). How does the side wind affect things? Let’s take an example: you cycle at velocity V with no wind at all (Fig. 1a). You feel an effective wind speed of V in the opposite direction, and the consequent air resistance, D, say (Fig. 1b). Fig. 1: How drag increases with a side wind. 70 Everyday Physics: Unusual insights into familiar things Now what if there’s a side wind blowing exactly from the right (Fig. 1c)? Because you are moving forward, effectively the wind is coming from your front right (Fig. 1d) because of the parallelogram of forces. Assume that the wind blows so hard that the air speed with respect to you is doubled, i.e. is 2V. The resulting drag is now four times as large as when there was no wind, i.e. it’s 4D. What matters is its component in the direction of motion. That is also much bigger than you’d have had without wind: twice as much air resistance, i.e. 2D (Fig. 1e). To take a concrete example, if you’re cycling at 20 km/h and there’s a cross wind of about 35 km/h (force 5 on the Beaufort scale), you’ll have twice as much wind resistance. Two other factors make things even worse. With the effective wind coming from the side, your “frontal” surface area, i.e. the area facing into the (now sideways) wind, increases quite a lot, which has a big effect on the air resistance. Second, your shape is no longer the front profile of a streamlined cyclist, but practically the (completely unstreamlined!) side surface of cyclist plus bike. And a croucheddown position over the handlebars doesn’t help much, since it hardly reduces the side-area facing the wind at all. Everyday Physics: Unusual insights into familiar things 71 If a side wind is to benefit you rather than impeded you, it has to blow from some angle from behind you. How much of an angle? That depends on the speed of the wind relative to your speed. In our scenario of Fig. 1 you feel no net effect if the wind blows at an angle of 104° (where 0° is straight ahead); if it goes further behind you than that, you get an overall benefit. A strong side wind has a big effect on bike road-races like the Tour de France. Consider a large group of riders. The apparent wind comes diagonally from the side, so riders benefit by staying diagonally behind riders in front (much like a group of migratory birds) in a formation that cyclists call an echelon (Fig. 2). However, given the limited width of the road, only a small number riders can do this and get good shelter from the wind, so a large group breaks into several smaller echelons. Because of the increased resistance caused by the side wind, groups of less-strong riders can’t keep up with the faster groups, and the result is often large time differences between the groups by the end of the race. Fig. 2: In a side wind, racing cyclists travel in a diagonal formation (an “echelon”). 72 Everyday Physics: Unusual insights into familiar things Home experiment: Healthy blowpipe Occasionally you come across them at a funfair: a kind of plastic blowpipe with two exit channels, one above and one below, like a “T” turned on its side. A ring of woolly fluffy thread is run through it. If you blow into the pipe, the thread starts to rotate: it comes out the top of the pipe, and goes back in at the bottom – a sight to see (Fig. 3). Fig. 3: The “rotating string” blowpipe. Fig. 4: The airflow through the blowpipe. But how does it work? The secret is in the diameter of the exit channels (Fig. 4). The diameter of the top channel is a bit larger than that of the lower channel. Therefore, the air speed going up is larger than the air speed going down. The air stream thus pulls the fluffy thread harder upwards than downwards. The net result is the rotating thread. With simple tools you can make a pipe like this yourself. Take a 3 × 6 cm rectangular piece of Plexiglas (Fig. 5), about 1 cm thick. (You can use wood instead, but it’s not as easy to see that the holes are in the correct place.) Drill a hole about 4.5 mm diameter in the top, and one about 3.5 mm in the bottom, to take the thread. Drill a bigger third hole in the side and attach a robust hollow handle to blow into – some plastic tube or part of an old ball-point pen will do. Make the fluffy thread-ring with a circumference Everyday Physics: Unusual insights into familiar things 73 of about one metre. To make it easy to see the thread moving, make the ring from two pieces of different colours, each about 50 cm long, or use ink to mark noticeable spots on the thread. Make the knots very small so the thread can easily pass through the drilled block. Fig. 5: Making the “rotating string” blowpipe. (Not to scale.) 74 The experiment illustrates a phenomenon that is important for your health. What goes for air also goes for liquid, and – within limits – for your bloodstream. The experiment shows that the stream velocity is larger in wider tubes than in narrow ones (given the same pressure difference, of course). To be exact, for a tube 2 times as wide, the stream velocity is 4 times as large. But that is not all: the streaming takes place over a larger cross-sectional area, which is also 4 times as large, so the amount of fluid moved per second is 16 times as much. This is Poiseuille’s law, which says that the throughput per second is proportional to the diameter to the fourth power. This means also that a 1% smaller diameter implies a 4% smaller throughput (because (1 − 0.01)4 ≈ 0.96). So it is good for your health to keep your blood vessels wide and elastic. Everyday Physics: Unusual insights into familiar things 22 MINIMIZING YOUR JOURNEY TIME Some things you automatically do right, because you have an intuitive understanding of some of the laws of physics without even realizing it. Take walking: when your left leg goes forward, your left arm goes backward; it’s the same for the right. That way the net amount of rotation of your body is zero and you don’t suffer any unnecessary strain by trying keep your body straight. Cycling is a bit more difficult, but once you know how to do it, your “automatic pilot” does exactly what’s needed so you don’t fall off, even when going round corners. (If you wanted to teach a robot how to do it, you’d need a huge amount of physics and control software.) Everyday Physics: Unusual insights into familiar things 75 Here’s an easier problem. Suppose you are an express cycle courier and every second matters. You have to deliver to someone at the far corner of a campsite. You have to cross a car park of very rough gravel, where you can cycle (at 6 km/hour, say), and then an expanse of grass, where you walk at 4 km/hour. (“No cycling on the grass.”). What route do you take to minimize your journey time? Remarkably enough, you intuitively take a nearly optimal route. What is the fastest route? Of course, you don’t take a straight line path (X in Fig. 1). It’s the shortest path, but not the quickest, because you lose the advantage of being faster on the gravel than on the grass. You cycle a bit further on, but how far? You don’t continue until the tent is directly in front of you (Y, Fig. 1); you feel somehow that dismounting a bit earlier is better (Z, Fig. 1), but how do you work this out exactly? Fig. 1: Which is the cycle courier’s fastest route from the entrance to the tent? Path X is the shortest, and path Y involves the least walking, but is there another path Z that’s quicker than X and Y? You can see why path Y (A-B-C) isn’t the fastest, by comparing it with path Z (A-D-C) in Fig. 2. In Z you walk further but cycle less. To 76 Everyday Physics: Unusual insights into familiar things see how much further you walk, draw an arc through B with centre C, and you see that the red line DE is the extra walking distance. This increases your journey time by (DE ÷ 4 km/h). To see how much less you cycle, draw an arc through D with centre A, and you see the cycling distance is reduced by green line FB, which decreases your journey time by (FB ÷ 6 km/h). So as long as (FB ÷ 6) > (DE ÷ 4), your journey is quicker. Fig. 2: The effect of walking a little more (red line DE) may save more time by reducing the distance on the bike a lot (green line FB). So more generally, it is the relative speed of cycling compared to walking that matters. Clearly, if walking and cycling were equally fast, the straight line (X in Fig. 1) would always be best. At the other extreme, if you walk as slowly as a snail, it’s best to cycle to almost exactly in front of the tent. So where exactly is the best place to dismount? Nature points the way with an analogous case – the path a light beam takes going from air into water. Fermat’s principle says that light always follows the path which takes the least time. The light kinks at the transition to the water because light propagates more slowly in water, whereas in air it travels almost as fast as in vacuum. (The speed of light in Everyday Physics: Unusual insights into familiar things 77 water is about three-quarters of that in air.) In glass the speed of light is about two-thirds its speed in air. Therefore, a light beam entering glass is just like our cycle courier it travels through air a bit longer to profit from the higher speed, and enters the glass quite late on, with a kink, i.e. it is refracted. Fig. 3: Snell’s law: v1 and v2 are the respective velocities of light in air and water. Credit: After nl.wikipedia. org/wiki/User:Sawims 78 The best place to get off the bike is given by Snell’s law which describes the refraction of light at a boundary between two media. Snell’s law states that the ratio of the sines of the angles of incidence and refraction is equal to the ratio of the velocities in the two media (Fig. 3). The cycle courier has to make the same kink as a light beam when it enters glass where the speed is two-thirds of that in air. (This is why we chose the slightly unrealistic speeds for cycling and walking). Doing the calculation shows that your path over the grass is not perpendicular, but has to make an angle of about 65°. Next time you do it, notice what you do: in practice, you intuitively get it almost exactly right. Long live our intuition for the laws of physics! Everyday Physics: Unusual insights into familiar things 23 THE CYCLIST’S SOGGY BACK Racing bikes don’t have mudguards, so when the road is wet you get a nice straight muddy stripe down your back. This is caused by water drops that are thrown in a forward direction from the rear tyre (Fig. 1). The drops are thrown from the wheel by centrifugal force, which increases as the speed increases, so fast cyclists have soggier backs than slowcoaches. Everyday Physics: Unusual insights into familiar things 79 Fig. 1: Water spraying from a bicycle wheel. Credit: After pixabay.com/ vectors/silhouette-wheelcyclist-bike-3067346/ pixabay.com/ users/mohamed_ hassan-5229782 You might think that most of the water leaves the tyre at the highest point, because this is where it attains its maximum forward speed (combining the speed of rotation and the forward speed of the bike). However, it is not the speed with respect to the road that determines the centrifugal force but the speed of rotation around the axis, which is the same all around the tyre. Gravity is involved too: because it always acts downward, the drops near the lowest part will be easiest pulled off, which promotes splashing near the spot where the tyre touches the ground. Higher up, gravity becomes less and less of a help. At the highest point it even works exactly against drops flicking off the wheel. Even so, water does splash off, because the centrifugal force overwhelms the gravitational force. But which drops wet your back? These are not the drops leaving the tyre exactly at the highest point. After all, the drops leave 80 Everyday Physics: Unusual insights into familiar things the tyre along an imaginary tangent and then, just like a ball, they proceed along a ballistic curve or “bullet trajectory” (ignoring any effect from the wind). When they leave the tyre at the highest point, they start out horizontally, and they can’t get any higher, so they never reach your back (Fig. 2). Fig. 2: Drops thrown from the top of the wheel can never reach your back. So the bad guys have to let go earlier if they are going to make you soggy. Just where? That is hard to say. How high above the tyre is your saddle? How upright is your back? What’s your speed? We’ll do a detailed calculation in a moment, but a reasonable guess is that droplets which let go about 45° before the highest point can just touch the saddle. If they want to make it to your back, they must follow a steeper trajectory and therefore must let go earlier, at least about 60° before the highest point. At what bike speed does the back-splashing begin? To start with, the centrifugal force has to overcome gravity. At the highest point that happens above 7 km/h (see box), and then droplets will let go. At 60° earlier, where the bad guys come from, that happens already at a lower speed because gravity does not yet fully counter the centrifugal force. As well as overcoming the gravitational force, the droplets also need some speed to travel up to your back, and they manage that only at a higher bike speed than the 7 km/h above; this turns out to be about 12 km/h. Below this speed the droplets at best reach the saddle. However, a racing cyclist travels much faster than 12 km/h, and the price is a wet back! Everyday Physics: Unusual insights into familiar things 81 Water drops thrown off a wheel First we’ll work out at what bike speed droplets let go of the wheel at the highest point. The centrifugal acceleration is ω2 R, where ω is the angular velocity in radians per second and R is the radius of the wheel. ω = v/R where v is the speed of the outside of the wheel with respect to the axis (or wheel hub in this case). So we can rewrite the centrifugal acceleration as v2/R. At what value of v does v2/R = g? In other words, at what forward speed does the centrifugal force just overcome the force of gravity? That’s a straightforward calculation: v2/R = g so v2 = R g so v = √(R g) If R = 0.4 m and g = 10 m/s2, this yields v = 2 m/s or just over 7 km/h. By the way, we see from the equation that for a folding bike with small wheels this speed is lower, so the droplets are released earlier. For R = 0.2 m, for example, we find v = 1.4 m/s or 5.1 km/h. And when do you get a wet back with small wheels? We consider droplets that will let go at 60°. These must be fast enough to gain at least 40 cm in height on their way to the back. That turns out to be at a bike speed of 3.3 m/s or 12 km/h. Because this is faster than the 7 km/h that was necessary for the droplets to leave the tyre at the highest point, this is automatically also enough to release them from a lower point of the tyre. From a precise calculation with a computer model it turns out that most of the droplets that hit you are released earlier still. The model also shows that up to 12 km/h you will remain dry. 82 Everyday Physics: Unusual insights into familiar things 24 CAN YOU GET LESS WET BY CYCLING FASTER? When you cycle in the rain, it’s easy to work out how wet you get per second. Your topside area is smaller than your front side, so you catch the least rain when you are stationary. As you start to move, your frontal area becomes increasingly important, because you bump into some of the raindrops in front of you, so you get wetter as you go faster. However, you’re probably interested in figuring out how wet you’ll get covering a certain distance – say from home into town. If you cycle fast you take less time (which keeps you dryer) but you get the rain slanting from the front (which means wetter). Which of the two effects wins? The question is complicated by the wind. If there’s a tail wind, you can adjust your speed to be the same as the wind’s. That’s surely optimal because practically only your upper side gets wet. But what if there’s no wind? To work this out, consider your top area and your frontal area separately. The top area is easy. On a horizontal surface a certain amount of rain falls per second, whether the surface moves or not. That amount is independent of the speed. The less time you’re in the rain, the less wet your top area gets. Everyday Physics: Unusual insights into familiar things 83 It’s different for the frontal area. Think of this as a thin vertical plate that can move horizontally. When stationary it catches no rain. When it moves, consider it to be moving through a shower of stationary droplets. (This is because in a constant shower every droplet is immediately replaced by another one, so that the number of droplets in a cubic metre remains constant.) The amount of rain caught by the moving plate is equal to the volume swept out, which is the area of the plate times the distance travelled. Therefore, the wetness of the plate does not depend on the speed but only on the distance. 84 Everyday Physics: Unusual insights into familiar things Another way of looking at this is to imagine the rain droplets are suspended motionless in the air and instead give the plate an equal and opposite vertical upward speed. For the process of wetting that makes no difference. Because the plate also moves forward (since you are cycling), it sweeps up a volume of air containing suspended raindrops. All the droplets in that volume are caught. Looked at from the side, that volume looks like a skewed rectangle (Fig. 1) – a parallelogram on its side. The parallel sides are formed by the plate in the initial and in the final state. The horizontal distance between them is the distance cycled. How much the parallelogram is skewed depends on the speed. Now the nice thing about a parallelogram is that its area depends only on the length of the parallel sides and their distance apart; the skewness makes no difference. For the cycling plate this means that the volume swept up is independent of the speed, dependent only on the start and end points. Fig. 1: The volume swept out by a cyclist’s frontal area depends only on the distance travelled, D, not the speed V. Everyday Physics: Unusual insights into familiar things 85 Conclusion: you gain a bit by cycling faster. It makes no difference to your front side how fast you go, but your upper side stays a bit dryer, so you gain overall. All of this goes for the ideal case where raindrops neatly fall vertically. So the answer to the question is yes, provided there is no wind, cycling faster keeps you dryer. 86 Everyday Physics: Unusual insights into familiar things 25 ROLLING RESISTANCE, AIR RESISTANCE AND FUEL CONSUMPTION You waste fuel if your car tyres are soft, because soft tyres have a large rolling resistance. And if you spoil the streamline of your car by driving with the windows open or by stowing stuff on top, again you use extra fuel, because the air resistance increases. How exactly do those resistances influence the fuel consumption? Everyday Physics: Unusual insights into familiar things 87 For the rolling resistance of a car – the friction to be overcome to get the car moving – the wheel bearings don’t really play a role, as we saw for a bike (p59). Rolling resistance is pretty much entirely caused by the deformation of the tyres on the road. That may seem odd. After all, isn’t rubber elastic, and doesn’t it spring back nicely into its original shape after being compressed? While that is true, some energy is lost in the process of compression and expansion. The reason is that the force needed to compress rubber is greater than the push-back force when it expands (see box Rolling resistance and air resistance, in What forces affect a cyclist? p62) and the energy difference is converted into heat. ( Just feel how warm the car tyres get after driving on the highway for a while!) The resistance that results from all this – the rolling resistance, or rolling friction – is proportional to the weight of the car, just as for a bike. It is, in fact, a fixed percentage of that weight, for a given type of tyre at a certain pressure; this percentage is called Cr , the “rolling resistance coefficient”. 88 Everyday Physics: Unusual insights into familiar things (The fact that the rolling resistance is proportional to the weight explains why it’s easier to push a broken-down car if everybody gets out of it: you’ve reduced the weight so you have reduced the rolling resistance.) Let’s try to guess what the value of Cr might be. Would it be around 1 or 0.1? We can make an educated guess by thinking of it in a different way based on everyday experience. Suppose your car is parked on a slope, and you release the brakes. For the car to start rolling, a slope of, say, 1% would probably do. On a slope of 1%, if you decompose the gravitational force on the car (i.e. its weight, W) into a component parallel to the road and one perpendicular, you find that 1%, i.e. W/100, is parallel to the road (Fig. 1). That force pushes the car forward. If that is enough to beat the rolling resistance, the value of Cr must be around 0.01. And sure enough, it is – at least if the pressure isn’t too low: since all the rolling resistance is down to deformation of the tyre, it is obvious that Cr will increase as the pressure goes down. Fig. 1: On a 1% slope, 1% of the car’s weight W acts parallel to the road. (Angles not to scale.) It is important to note that speed is not involved here. The rolling resistance is, for all practical purposes, independent of speed. The air resistance, or drag, is different. It goes up with the square of the speed; as is the case for a cyclist (see What forces affect a cyclist? p59), if you double your speed, the air resistance is four times as high. You might guess that from experience: if you stick your hand out the window at 100 km/h, the force you feel is much greater than at 50 km/h. Furthermore, the air resistance is proportional to the density of the air, so air resistance goes up with barometric pressure. It also means that in the heat of the summer, the air resistance is lower than in the winter, given a certain atmospheric pressure: warm air expands and therefore gets “thinner”. Another consequence is that the thinner air at higher altitudes offers less air Everyday Physics: Unusual insights into familiar things 89 resistance. That why it’s easier for cyclists and speed skaters to break speed records at higher altitudes. Another factor determining the drag is the size of the car, or more precisely its frontal area. Low and narrow cars therefore have an advantage over high and wide ones when it comes to air resistance. Finally, the air resistance is proportional to the famous drag coefficient Cd. While the value of Cd can be as high as 1 for awkwardly shaped objects that aren’t streamlined at all, it can be around 0.3 for modern cars. Why are rolling resistance and air resistance so important? They directly determine the energy consumption as we saw with cycling (see What forces affect a cyclist? p59). Their sum is the total resistance that a car experiences when driving on a horizontal road, and the engine must continuously produce an exactly matching force to maintain a constant speed. The interesting point is that 90 Everyday Physics: Unusual insights into familiar things this force is equivalent to the work done – or the energy produced – per unit distance. And that can be directly translated into the energy consumption of the car (see box Resistance and fuel consumption overleaf ). In order to assess the influence of the two resistances on the fuel consumption, we have to know their relative magnitude. For a typical midsize car this is shown in Fig. 2. Fig. 2: Resistance increases with speed (midsize car). When driving through residential areas at low speed (below, say, 50 km/h), Fig. 2 shows that the rolling resistance dominates. Clearly, it is advantageous here to have a light car (and a light car also uses less energy to accelerate up to speed after the frequent stops in urban driving). Everyday Physics: Unusual insights into familiar things 91 On the open highway things are different. Here air resistance dominates by far, so it pays to have a car with a small frontal area (low and narrow), and streamlining of the car body is of great importance (driving with open windows or stowing stuff on top can increase the fuel consumption dramatically). Moreover, since air resistance increases with the square of the speed, driving fast does use more fuel. Fortunately, things are not as bad as the steeply rising curve in Fig. 2 suggests, because the efficiency of the engine improves at higher speeds, partly compensating for the steep increase in air resistance. But driving fast remains inefficient. Therefore, a reasonable speed limit may be a good idea after all. Resistance and fuel consumption Fig. 2 shows the behaviour of rolling resistance and air resistance (or drag) with increasing speed, for a car that is not particularly streamlined. The equations for the two kinds of resistance are given in the box about rolling resistance and air resistance (p62). The values used in Fig. 2 are: mass = 1,000 kg; rolling resistance coefficient Cr = 0.01; drag coefficient Cd = 0.4; frontal area A = 2 m2. Here the figure shows that above 50 km/h the air resistance dominates (whereas for a cyclist that point was at about 15 km/h.) The unit for this resistance is the units of force: newton (N). The resistance is a direct measure of the energy consumption, so as resistance increases with speed, so does fuel consumption. Fig. 2 shows that at 100 km/h the resistance is about 500 newtons for this car. Work is defined as force × distance, so force = work/ distance, i.e. force = work per unit distance, i.e. 1 N = 1 J/m, or 1 kJ/km. So our 500 N resistance is equal to 500 kJ/km. At an estimated efficiency of 20% that comes to a total energy use of 2,500 kJ/km. Oil and petrol yield about 40,000 kJ per kg, so this works out at about 6.25 kg per 100 km, or a fuel consumption of about 7 litres per 100 km. 92 Everyday Physics: Unusual insights into familiar things 26 HOW MANY CARS PER HOUR CAN A ROAD TAKE? To avoid traffic jams without building new roads everywhere, we must use the capacity of existing roads effectively. But how many cars can a highway actually cope with in an hour – what is its capacity? Can we increase it? Everyday Physics: Unusual insights into familiar things 93 At first you might think that driving faster increases the capacity, because the higher the speed, the greater the throughput. But this doesn’t work in practice. Imagine a single lane on the highway, 1 km in length, with all cars travelling at the same speed. The number of cars the lane can handle per hour depends on the car density (number of cars per km) as well as the speed. If this string of cars drives at 100 km/h with 15 cars in each kilometre stretch, this lane can handle 1,500 cars per hour: the capacity is speed times car density. 94 Everyday Physics: Unusual insights into familiar things The density must not be too high or it becomes unsafe. What is a safe density? Traffic rules differ from country to country, but a typical requirement is to “keep far enough back from the car in front that you can brake and stop safely”. However, on a motorway this is too strict, because it is not reasonable to expect that the car in front of you will stop abruptly, or that a concrete block will suddenly appear on the road. If you took the above law literally on the highway, then at 100 km/h you would have to maintain 100 m separation between cars, reducing the capacity of the lane at high speed. (This distance is large because the braking distance increases with the square of the speed: driving twice as fast require four times the braking distance.) Fig. 1 shows how capacity changes with speed. The red curve is for the strict separation rule, and shows that we reach maximum capacity (of 2,000 cars per hour) at 25 km/h. At higher speed each car makes more progress, but the space between cars is disproportionally larger, resulting in a capacity reduction. Everyday Physics: Unusual insights into familiar things Fig 1: Two scenarios for the capacity of a lane: maintaining distance according to the book (red curve) and maintaining distance according to the two-second rule (blue curve). 95 Several countries have a rule of thumb for driving on a dry straight highway with good visibility, which allows a smaller car-tocar separation. The French, for example, have the two-stripe rule (“deux traits = sécurité”): keep two stripes visible between you and the car in front. The British have a similar two-chevrons rule. The Netherlands applies a two-second rule: keep at least two seconds back from the car in front. What does that mean for the capacity of the lane? One car every two seconds means 30 cars in a minute, that is 1,800 cars per hour. (In reality it is a bit less, because those two seconds are calculated from the rear bumper of one car to the front bumper of the next car. We have ignored the length of the car; taking that into account reduces the capacity. That especially adds up at lower speeds, because then the in-between space and the car length are comparable. At higher speeds the in-between space is so large that the length of the car is negligible.) The blue curve in Fig. 1 shows how this rule affects capacity. At speeds of around 50 km/h and above, the two-second rule yields a greater capacity than the strict braking-distance rule, because the two-second rule allows cars to drive closer. So the twosecond rule entails more risk than the strict rule, but the risk is pretty low in good visibility on a dry road, so for the open road it’s a reasonable compromise. While the two-second rule is OK for 120 km/h on a clear road, if there’s congestion (or if it’s likely), 120 km/h is no longer safe so we have to drop the speed. The red line capacity at 50 km/h is the same 96 Everyday Physics: Unusual insights into familiar things as the blue line at 120 km/h, i.e. 1,700 cars/h. So when the traffic authorities say “drive at 50 km/h in congestion” it’s reasonable, because it’s safe, and we haven’t reduced the capacity of the road. The capacity of a road Assume that all cars in a lane have the same speed and the same separation from the car in front. Since the number of cars per hour is equal to the number of cars per kilometre times the number of kilometres that the row of cars moves per hour, we can write the capacity C of one lane as C = v n, where v is the speed in m/s and n is the number of cars per metre. (The result is number of cars per second, which we must later convert to cars per hour). Now n is equal to the inverse of the amount of space occupied by one car; the space occupied by one car is equal to the length L of the car itself (say 4.5 m), plus the space between cars. For the two-second rule the space between cars is the distance driven in two seconds, which is τ v where time interval τ is equal to 2 seconds. We then find C = v/(L + τ v). At very low speeds (bumper to bumper) only L remains. Then the car density is constant, and C = v/L: the capacity is proportional to the speed. At very high speeds we can neglect L and that yields C = v/(τ v) = 1/τ. This is ½ per second, or 1,800 per hour, and is independent of speed (which is why the blue line in Fig. 1 flattens out towards the right). The strict-rule case is more difficult. The space between cars consists of two parts: 1. The reaction-space t v – the distance you cover in the time t which you take to react and shift your foot from the accelerator to the brake pedal, where v is the speed in m/s 2. The actual braking distance v2/2a, where a is the braking deceleration. (For instance, Dutch law specifies a = 5.2 m/s2). That gives C = v/(L + t v + v2/2a). For very low speeds both the braking distance and the reaction distance are negligible, which makes the in-between space negligible. All that is left is the length L of the car, and we get C = v/L, exactly as with the two-second rule. For the other limiting case – very high speeds – only the last term (the braking distance) is of importance. Neglecting the other terms we see that C is proportional to 1/v. That is why the right part of the red curve decreases. In between lies the maximum capacity, at about 25 km/h. Everyday Physics: Unusual insights into familiar things 97 98 Everyday Physics: Unusual insights into familiar things 27 COZY CANDLELIGHT Candles are really cosy, adding an air of intimacy and romance to a relaxed dinner. But originally they were practical light sources, much handier than oil lamps. In fact, compared to an oil lamp, a candle does give a lot of light, and some people even say that it is a super energy-efficient light source. Is that true? How much energy does a burning candle use? A candle consumes a surprising amount of energy. This becomes obvious when you feel the heat produced, which is considerable. A reasonable guess is that a candle produces as much heat as a 100 watt incandescent light bulb – both of them can burn your hand. However, you don’t have to guess: you can easily measure the energy used by the candle by finding out how much wax it burns in a measured time. Combine this with the heat of combustion of wax (see box Candle heat), and you find that the heat generated per second is roughly 100 joules per second which is 100 W. However, the light produced is much less than that of a 100 W incandescent light bulb, let alone a cluster of compact fluorescent bulbs or LED (light emitting diode) lamps whose wattage adds up to 100 W. Everyday Physics: Unusual insights into familiar things 99 The wattage of an item is a measure of the energy required to run it, not the energy it emits in the form of light: an electric radiator consumes 1,000 W but nobody would call it a good light source. It radiates primarily in the infrared which your eyes can’t see, but when we talk about “light”, we mean radiation that can be detected by your eyes, specifically by the rod and cone photoreceptors in your retinas. This type of visible radiation is not measured in watts, but in lumens. The efficiency of a light source is determined by the amount of light it gives (in lumens) per unit of energy consumed (in watts), so efficiency is expressed as “lumens per watt”. From that perspective, a candle performs poorly, producing about 0.15 lm/W, whereas an incandescent lamp gives about 12 lm/W – 80 times as much. LED lamps do even better: at 80 lm/W they are more than 500 times as efficient as the candle! However, that’s not the whole picture, because most electricity is generated from heat in a power station, which isn’t very efficient. The electricity that you use in your home represents only about ⅓ of the energy that the power station needs to produce it, so perhaps the LED lamp is only 500/3 = 167 times better than the candle, but that’s still pretty good. So while candles may produce a romantic ambience and even help to heat your home with 100 watts each, if you want them to produce a lot of light, take them to the power station, throw them in the furnace, and then rush home and use the electricity they produce to power an LED lamp. Candle heat To find out how much wax a candle uses, weigh it before and after burning it for an hour or so (making sure you measure the time interval carefully). Not all candles are equal, and not all flames are the same, but a typical candle burns around 8 grams of wax per hour. Look up the heat of combustion for candle wax: it’s about 48 kJ per gram, so 8 grams represents 8 × 48 kJ = about 380 kJ of heat, produced in 1 hour, or 380,000 J in 3,600 seconds = 105 J in one second, which by definition is 105 W. As we saw, 105 W is about the energy a person uses when not doing heavy exercise. You can sanity-check this. Using 100 W for one day, i.e. for 24 hours × 3,600 seconds/hour, gives 100 × 24 × 3,600 J – or 8,640 kJ. That is a perfectly normal diet for an adult (see The human engine, p43). 100 Everyday Physics: Unusual insights into familiar things 28 WHY ARE INCANDESCENT BULBS SO INEFFICIENT? Moving from candle to incandescent lamp was a big improvement: no fumbling to find matches, no hot wax spilled on the furniture, no danger of fire, and a lot more light. Even so, the old-fashioned incandescent bulb isn’t very efficient: it uses a lot of electricity for the amount of light it gives. Everyday Physics: Unusual insights into familiar things 101 Why is that? The efficiency is determined by how much of the emitted radiation can be sensed by your eye. Comparing the emission spectrum of the bulb with the eye’s sensitivity will tell us a lot about the bulb’s efficiency. Fig. 1 shows the emission spectrum. Fig. 1: The emission spectrum of an incandescent bulb compared to the eye’s sensitivity. ► ► The tall thin curve on the left shows that your eye is sensitive to wavelengths between about 400 and 700 nanometres, which are the colours of the rainbow. (One nanometre is one millionth of a millimetre, or – for traditional readers – 10 angstrom units.) The peak lies about 550 nm, in the yellow/green; that’s where your eye is most sensitive. The red curve is the spectrum emitted by the incandescent wire in the bulb. Its peak lies near 1,000 nm, which is in the infrared. The two curves don’t match well: most of the bulb’s emission falls outside the range of the eye, so its remains invisible – useless as far as lighting is concerned. An incandescent bulb with an emission spectrum like this is always inefficient. 102 Everyday Physics: Unusual insights into familiar things Can we get a better match for the two curves? One way is to make the incandescent filament hotter than the usual temperature of about 2,700°C, because at a higher temperature not only is more radiation emitted, but also the colour changes. Think of an oldfashioned poker in the fire: its colour goes from red to orange to yellow as it heats up. Or think of a dimmed incandescent bulb: as you turn up the dimmer to make the lamp brighter, it gives more light but the light is also “whiter”. In other words, as the filament gets hotter, the emission spectrum contains more shorter wavelengths, and the peak shifts to the left. Twice as hot a filament (in terms of its absolute temperature) would be perfect: the peak would be at half its normal wavelength, right where the eye is most sensitive. This would require a temperature of about 5,700°C. That’s approximately the temperature of the surface of the sun. (So the solar radiation peaks where the eye is most sensitive. That is not a coincidence, of course. Why might that be?) Unfortunately, when we heat a tungsten filament beyond 2,700°C we fairly soon run into its melting point, ~ 3,400°C. Even Everyday Physics: Unusual insights into familiar things 103 approaching the melting point too closely reduces the lifetime of the incandescent bulb dramatically. Filling the bulb with a halogen gas helps a bit, but the melting point is a hard limit. And other metals aren’t the solution: their melting points are even lower. So using incandescent filaments is just not a good way to generate light. For photographers, another disadvantage of incandescent bulbs is that the light is not white. Fig. 1 shows that the emission in the orange/red is two to three times stronger than in the blue, giving a warm light, which makes it difficult to see the true colour of blue objects. You may have noticed this while shopping for clothes in artificial lighting: sometimes you have to step outside to be sure that something really is blue, not black. 104 Everyday Physics: Unusual insights into familiar things 29 LUMINOUS IDEAS FLUORESCENT LIGHTS AND LEDS Lighting based on a glowing filament cannot be efficient, as explained in the preceding chapter. What we’d like is something that mimics sunlight more closely. The temperature of the surface of the sun (where sunlight comes from) is so high that the molecules and atoms get into a higher energy state – an “excited” state – by colliding with each other. As the molecules fall back into their unexcited (“ground”) state, they lose that extra energy largely in the form of visible light – our sunlight. Clever light sources emulate this by a combination of electronics and choice of materials. In fluorescent tubes and compact fluorescent lamps (CFLs), it’s achieved by a smart choice of the filler gas. In Light Emitting Diodes (LEDs) it’s achieved by the choice of the solid material that makes up the diode. The result is that the input energy is converted directly into radiation and not – as in the incandescent bulb – indirectly by heating the filament. They therefore give more light for a given amount of energy – more lumens per watt (lm/W). Fig. 1 shows how much better these “new” lamps are. All of them are more efficient than the traditional incandescent bulb, they Everyday Physics: Unusual insights into familiar things 105 have a longer life, and there is no glowing filament (which slowly but surely evaporates, which you notice by the blackening of the inside surface of the glass). Candle Incandescent bulb Compact fluorescent lamp LED lamp Fluorescent tube Efficiency (lm/W) 0.15 12 50 80-100 100 Life span (hours) 10 1,000 > 5,000 ~ 50,000 10,000 Fig. 1: Efficiency of various light sources compared. These values potentially understate the efficiency of LEDs. LEDs have the useful property that they can produce light in a single colour: red, blue or green, which is useful if you want a specific colour – for example for traffic signals or for a car’s brake lights. In such cases it’s more efficient to produce the proper colour directly instead of producing the entire spectrum of colours and filtering out the unwanted ones with the attendant loss of useful (visible) energy. If you want to make white light with LEDs, you can do it by a clever combination of the three colours, or you can use an intermediate step, where the light of the LED first irradiates a fluorescent layer, as also happens in fluorescent tubes. And the LED offers possibilities unthinkable for old-fashioned lamps: a specific type of LED lamp is already available that can produce any colour required, by combining a red, green and blue LED and regulating the current in each LED separately to produce the desired colour. If the colour of the lighting is not so important, the efficiency can be extremely high. Sodium street lights, (which give a pure orange light) deliver about 200 lm/W – about 1,300 times the efficiency of an old-fashioned candle. What is the highest lighting efficiency we could ever achieve? That would be when all the light is radiated at the peak of the eye’s sensitivity curve (around 550 nm, Fig. 1, Why are incandescent bulbs so inefficient? p101), producing a yellow/green light, and (from a calculation we haven’t shown) the efficiency would be 680 lm/W. 106 Everyday Physics: Unusual insights into familiar things 30 WHY IS THE SKY BLUE AND THE SETTING SUN RED? When there are no clouds, the sky is blue. We take it for granted because we’re so used to it, but why is it blue? Where does the blue come from? Indeed, why is the sky bright at all, and not just black, which is how astronauts in space see the sky? After all, light travels in straight lines, so unless you’re looking directly at the sun or a star, you should see no light. Everyday Physics: Unusual insights into familiar things 107 We must conclude that the atmosphere bends the light rays from their straight paths. It scatters the light, or part of it. Dust particles can cause scattering: just think of the glowing dust particles you sometimes see when you open the curtains a little in the morning and the sun shines in. But scattering also happens in completely clean air, which may seem a little strange, because air is transparent. How does this scattering come about? Air molecules consist of charged sub-atomic particles – positive nuclei and negative electrons. Because light waves consist of an alternating electromagnetic field, the positive and negative charges are set in motion by the electric component of the field. (This also happens in the antenna of a TV 108 Everyday Physics: Unusual insights into familiar things or phone.) The charges in an air molecule shift in the electric field: the positives ones go one way, the negative ones the other way – the molecules are “polarized” to the rhythm of the alternating field. Because of the moving charges, the molecules now act as transmitters themselves: they emit light at precisely the same frequency (and therefore of the same colour) as the original incoming light. However, the important difference is that the emitted light travels in all directions. A small part of the original light is thus removed from its straight path and scattered by the air molecules, although that scattered light is very faint. This explains why the sky is bright, and not black. But why is it blue? That’s because some colours are scattered more than others. The scattering is much stronger for violet and blue light than for orange and red light (Fig. 1). The scattering intensity is inversely proportional to the 4th power of the wavelength: a wavelength 2 times longer means scattering is 16 times less intense. The wavelength of blue light is only half that of red, so its scattering is therefore about 16 times stronger than red’s. This type of “Rayleigh scattering” (named after the British physicist Lord Rayleigh) occurs as long as the scattering particles are much smaller than the wavelength of the light – and they are, because the particles here are air molecules. (The light’s wavelength is about 500 nm and the diameter of an oxygen molecule is about 0.33 nm – about 1,500 times smaller.) Fig. 1: Air molecules scatter different colours by different amounts; blue is scattered much more than red. In summary: the sun’s rays contain all the colours of the rainbow. Most of the rays pass through the atmosphere in their original direction, but some are scattered, and some of this scattered light Everyday Physics: Unusual insights into familiar things 109 reaches your eyes. And because blue light is scattered more than red, overall the sky seems blue. (The surprisingly simple experiment on p111 illustrates this nicely.) The same mechanism also explains why the setting sun is so red. At low solar elevation, i.e. when the sun is low in the sky, the layer of air that the sun rays have to pass through is very thick (line CD in Fig. 2) so the scattering is very large. When you look at the setting sun, the light that reaches your eyes comes in a straight line from the sun with little or no scattering (because the scattered light won’t reach you). Not much blue is left because it has been scattered in all directions. The red part of the spectrum is scattered far less and simply propagates straight to your eyes, which you perceive as the beautiful red colour of the setting sun. Fig. 2: Proportionally, the Earth’s atmosphere is thinner than the skin on an apple. When the sun is directly overhead, its rays only have to pass the short distance AB through the atmosphere, whereas at dusk they have to travel the much longer distance CD. We’ve said that at low elevation the sun has to shine through a much thicker layer of air than at high elevation, but this is true only because the thickness of the atmosphere is small compared to the diameter of the Earth (Fig. 2). If the atmosphere were very deep compared to the diameter of the Earth, the sun’s elevation wouldn’t make much difference. However, if in a thought experiment we swept up the rarefied upper layers of the atmosphere into a single layer that had normal sea-level air pressure throughout, we’d get a layer about 8 km thick. So the “effective thickness” of the atmosphere is about 8 km. Comparing this to the diameter of the Earth of nearly 13,000 km, then proportionally the thickness of the atmosphere around the Earth is less than the thickness of the skin on an apple. This is why light has so much further to travel through the atmosphere as the sun lowers, and the scattering becomes very large. 110 Everyday Physics: Unusual insights into familiar things Home experiment: The blue sky and the red setting sun You can demonstrate the selective scattering of the colours in a light beam with a simple experiment. You need a glass with a flat bottom, a few drops of milk, a piece of white card and an ordinary flashlight. (An incandescent flashlight is best because it emits all the colours of the rainbow. How well an LED torch will work here depends on its emitted colour spectrum; a single-colour LED torch won’t work.) The experiment works best when the lamp has a beam that is more or less parallel and is narrower than the base of the glass. (In this particular case the glass was supported by the (relatively large) torch.) First, pour water into the glass, and shine the torch through the glass from the bottom up. The white light of the torch exits the glass at the top and virtually no light is scattered sideways, and the beam striking the white card above the glass is white. (Fig. 3a). Next add a few drops of milk, and stir (Fig. 3b). You have now made a medium that scatters light much more strongly than air. It is no longer transparent but whitish (Fig. 3c). The scattered light (as viewed from the side of the glass) is a little blue, even though it is lit by ordinary white light, which is the principle of the blue sky we explained earlier. (In Fig. 3c, the blue at the edge of the glass is just visible.) a) b) c) Fig. 3: The plain water lets the white light beam through (a). Add a few drops of milk to the water (b) and now the transmitted beam only contains orange/red (c). Everyday Physics: Unusual insights into familiar things 111 If your torch is wide enough, you can support the glass of water on the torch itself (Fig. 3b). Otherwise, hold the torch in one hand and the glass in the other, and get someone else to hold the card above the glass. The convincing proof comes only when you look at the transmitted light. If the flashlight has a strong, largely parallel beam and the room is dark, you will see an orange-red spot on the ceiling. It illustrates the setting sun. Alternatively, you can hold a white sheet of paper just above the glass to view the transmitted light; it’s clearly redder than the scattered light although it comes from the same light source. It shows the difference between the light that is scattered and the light that is transmitted without scattering. (If the transmitted light is too faint, there is too much milk in the water. A few drops are usually sufficient.) 112 Everyday Physics: Unusual insights into familiar things 31 TWO KINDS OF SMOKE FROM THE SAME CIGARETTE? Curiously, if you look closely at a burning cigarette you see two kinds of smoke. Blue smoke comes from the burning end, whereas the smoke that comes out the filter end is white or grey. The explanation is Rayleigh light scattering, as with the blue sky. In fact, light scattering is the only reason you see smoke at all because smoke itself gives no light. Smoke scatters incident light in all directions. You see smoke best when a bright light, like the sun, shines on it. The scattered light then contrasts nicely with the dark background. This is seen most clearly in photos taken into the light. Everyday Physics: Unusual insights into familiar things 113 What colour will the scattered light be? The reason the smoke at the burning end of the cigarette is blue is the same as why the sky is blue. The smoke particles are much smaller than the wavelength of light – as are the air molecules that produce the blue sky. The degree of scattering is not the same for all colours (p107): blue dominates in the scattered light. Even a small puff of cigarette smoke scatters the light a lot, because the smoke particles consist of quite a large number of molecules in a clump, and these scatter disproportionally more light than a single molecule. (The intensity increases with the square of the number of particles in the clump.) So even a small quantity of smoke produces enough scattering to be clearly visible. Light scattering and photography You can use your understanding of light scattering to take better photos in smog, or when there’s a blue haze surrounding mountains in the distance. Use a lens filter that selectively blocks the short wavelengths, which scatter the most (blue, violet and particularly UV). An extreme example of this is infrared photography, which only makes use of long wavelengths, resulting in minimal scattering. What about the smoke at the filter end of the cigarette? Why isn’t that blue? The explanation above is valid only for particles that are small with respect to the wavelength (and at the burning end of the cigarette, the particles are small). However, as the smoke travels through the cigarette towards the filter, the particles clump together and can grow as large as the wavelength of light or even larger. For such particles all light is scattered more or less equally, or in other words, the intensity of the scattered light is approximately independent of the wavelength (whereas for small particles it’s inversely proportional to the 4th power of the wavelength). So if the incident light is white, the scattered light is also white – or maybe a bit grey because the intensity is decreased. For the same reason clouds are white or grey. They consist of small water droplets that scatter light just as the big smoke particles do: white light in, white light out. 114 Everyday Physics: Unusual insights into familiar things 32 SWIMMING POOLS ARE DEEPER THAN THEY LOOK A problem, especially for children, is that swimming pools are deeper than they seem to be. You stand at the edge of the swimming pool, look across to the other side, and think: “It’s not deep, I can jump in safely.” But you are wrong: the water is deep – you’ve been tricked by the refraction of the light. You’re looking at point D on Everyday Physics: Unusual insights into familiar things 115 Fig. 1: Why a swimming pool looks shallow: the bottom appears to be located at the extension (B) of the ray that enters your eye, and not where it really is (D). the bottom of the pool. Light leaves D in all directions, and one ray reaches your eye E. But this ray is not straight: it bends at the transition X from water to air, because light rays always follow the fastest path. By making that bend at X and entering the air sooner than a straight line from D to E, the ray profits from the greater speed in air and travels a shorter distance (line DX) in the water where the speed is lower. (If the light travelled in a straight line from D to E, it would spend a much longer distance DY in the water.) This is exactly similar to what the hurrying cycle courier does (see Minimizing your journey time p75). Fig. 2: Why a swimming pool looks shallower to a child than to an adult. Fig. 2 explains why the problem is more serious for a child than an adult. First, the child has less experience, and may not know that the apparent depth of water is misleading. But physics also plays a part. The ray seen by the child (Fig. 2, red ray) comes at a glancing angle over the water. In contrast, an adult looks at the pool bottom more nearly perpendicularly, and sees a different ray (Fig. 2, green ray). For the adult the pool looks somewhat deeper, although still not as deep as it really is. (You can see this for yourself: look into a pool when standing; now squat down – the bottom of the pool will appear to rise!). 116 Everyday Physics: Unusual insights into familiar things If you look down perpendicularly to a point on the bottom of the pool directly beneath you, will you see no distortion at all? You might think so because the light ray entering the water precisely perpendicularly doesn’t bend at all (after all, in which direction would the bend have to go?). Everyday Physics: Unusual insights into familiar things 117 But now the fact that you have two eyes comes into play. Each eye looks at the bottom from a different position. This means that the rays you use to see the bottom are slightly bent (Fig. 3). Again the bottom looks closer than it is, although the difference is small: the real depth is 4⁄3 times the apparent depth. (This 4⁄3 factor arises because the ratio of the speeds of light in air and in water is 4⁄3.) Granted, the difference is only 30%, but it can make a world of difference to a child. Fig. 3: When you look straight down into water, the actual depth is 4/3 times the apparent depth. 118 Everyday Physics: Unusual insights into familiar things 33 SUNLIGHT FILTERING THROUGH THE LEAVES OF TREES When the sun is high in the sky and shines through the leaves in a forest, you get a playful pattern of small and large light spots on the ground. But there is something special about those little spots: they are always round. How come? Do the leaves form circular holes? Everyday Physics: Unusual insights into familiar things 119 That would be unlikely, and indeed isn’t true. The roundness of the light spots has nothing whatsoever to do with the shape of the holes that the light passed through. The spots are in fact small pictures of the sun. Each hole between the leaves acts as a “camera obscura” (see box). The only thing the holes have in common is that they are small. Fig. 1 shows how it works. Consider a single point S on the sun. That point emits light rays in all directions. A single ray just passes through the hole between the leaves, and hits the ground at a specific point G, whereas all the other rays are blocked by the foliage. This happens with each point on the sun: each produces a bright point on the ground. The result is a picture of the sun, although it’s a mirror image and upside down. Fig. 1: An image of the sun is projected on to the ground through a small gap in the leaves of the trees. You can use the size of the spots to determine the height of the trees (or more precisely, the height of the hole between the leaves). For this you need to know the angular diameter of the sun (Fig. 2) which is almost exactly half a degree, which is also the angular diameter of a 1 cm coin at a distance of 100 cm = 1 metre. So, if the spot on the ground is 15 cm in diameter, the hole the light passed through is 15 cm × 100 = 15 metres above the ground. It doesn’t matter much if the hole isn’t perfectly round, as long as it is small compared to the spot on the ground, and anyway, who can complain about a camera that cheap? You can use the small-gap idea as a simple and safe way to observe a solar eclipse (Fig. 3): when the moon starts to obscure the sun, you will see many sun crescents on the ground! An easier way, instead of having to find a convenient forest, is to use a piece of 120 Everyday Physics: Unusual insights into familiar things cardboard with a pinhole in the middle (Fig. 4), and project the sun’s image on to a piece of white paper on the ground. You can even use an unmodified kitchen colander to project the sun’s image on to the ground – try it! Fig. 2: The angular diameter of the sun, and of a 1 cm coin 1 m away from your eye. Fig. 3: Images of partial solar eclipse projected through the leaves of a tree. Credit: commons. wikimedia.org/wiki/ User:Ellywa Creative Commons AttributionShare Alike 3.0 Unported. Unmodified. Everyday Physics: Unusual insights into familiar things 121 Fig. 4: Using a piece of cardboard with a pinhole in the middle to project the sun’s image. Camera obscura “Camera obscura” is Latin for “dark chamber”. The name refers to the phenomenon by which an image is produced in a dark room from a bright scene or object outside, by using a small hole in the wall instead of a lens. The “pinhole image”, as it is often called, is inverted – upside down and right to left (Fig. 5). Leonardo da Vinci wrote about the camera obscura back in the 16th century. Many tourist destinations have camera obscuras open to the public (see Resources appendix). By the way, a normal camera also produces an inverted image. However, the camera lens is much bigger than a pinhole, and it allows many more light rays to contribute to the image without the image getting blurred, thus making it much brighter. Nowadays many camera obscuras use a lens instead of pinhole, to produce a better image. Fig. 5: How a camera obscura creates an image. The pinhole is at C. Credit: commons. wikimedia.org/wiki/ File:001_a01_camera_ obscura_abrazolas.jpg You can easily make a small camera obscura yourself, with a shoebox. Make a pinhole in the middle of one of the short sides. In the opposite short side cut a rectangular hole, and over it stick a piece of tracing paper or kitchen parchment, and put the lid on the box. That’s it! Indoors at night you can get a reasonable image of a switched-on light bulb. During the day, if you point your “camera” out the window, you can see the sun’s image – but don’t look at the sun directly! – and you can probably see (upside-down) images of buildings too. (If your image is too dim for comfort, try making the pinhole bigger.) 122 Everyday Physics: Unusual insights into familiar things 34 HOW SHARPLY CAN YOU SEE? Why do you see things? Because the light coming from them reaches your eyes, of course. Some objects – lamps, candles, the sun – emit light of their own, but you see most things because they reflect light from an external source. How do you see an image of an object? The lens of your eye casts an image of the object on your retina, where the light is detected by the photoreceptors (the rods and cones that convert the light falling on them into nerve impulses that are then processed by your brain). The image needs to be sharp, i.e. in focus, independent of the distance from the object to your eye, but the position where the image is formed behind the lens depends on how far the object is in front of the lens (Fig. 1). Everyday Physics: Unusual insights into familiar things 123 Fig. 1: The distance between the lens and the image formed depends on how close the object being viewed is to the front of the lens. Credit: teddy bear: oNline Web Fonts – www. onlinewebfonts.com In a photographic camera, you adjust the focus by changing the distance between the lens and the film (or digital image sensor). You can’t do that with your eye, because the diameter of the eyeball would continually be growing larger or smaller. So instead, your eye adjusts the power of the lens in your eye, a process called accommodation. When the muscle is in its relaxed state, the lens is stretched and becomes less curved (Fig. 2 top), suitable for viewing distant objects. In contrast, if the object comes closer, your eye dynamically changes the shape of the lens using the ciliary muscle that surrounds the lens. When this muscle contracts around the lens, it exerts less tension on the lens, and the lens, which is elastic, reverts to its natural convex (more powerful) shape (Fig. 2 bottom). This is what you need to view something that is close-up. Contracting the muscle becomes more difficult as you age because the eye lens becomes less elastic, which is why many older people need reading glasses. Fig. 2: The muscle around the lens of your eye controls how powerful the lens is, so the image is always focused on your retina. (Not to scale.) 124 Everyday Physics: Unusual insights into familiar things Who needs reading glasses? Just as with a camera, you want your eyes to provide a sharp image of an object, whether it’s close or far away. That doesn’t happen automatically: the power of the lens has to change according to the distance of the object. For both a camera and your eye, the formula specifying the lens we need is: 1/o + 1/i = 1/f where o is the distance from the object to the lens, i the distance from the lens to the image (or to the retina, in the case of the eye) and f the focal length (Fig. 2). The figure 1/f is called the strength of the lens, and its unit is dioptres, or inverse metres (m-1). Fig. 3: How the object and image distances (o, i) and the focal length (f) of the lens are related. Because the distance, o, of the object varies, to get a sharp image, either i or f must vary accordingly. In a camera we change i by moving the lens closer to (Fig. 1 top) or further from (Fig. 1 bottom) the film or digital image sensor. In contrast, in your eye, i (the diameter of your eyeball) remains constant, so your eye must accommodate by changing the focal length f, which it does by contracting or relaxing the muscles controlling the shape of the lens. Young people can see objects sharply between infinity and about 0.2 metres. At infinity, o = ∞, so 1/o = 0, and our formula becomes 1/i = 1/f . At 0.2 m 1/o = 5 m-1, so our formula is 5 + 1/i = 1/f. As i remains constant, only f can change, so that 1/f, the lens strength, must vary by 5 m-1; in other words, the accommodation range is 5 dioptres. As you get older, your accommodation range gradually reduces to practically zero, and adjustments for distance must be made artificially – with glasses. For an eye that sees sharply at infinity, with reading glasses of 3 dioptres the region in focus will be ⅓ m = 33 cm away, even when the eye does not accommodate at all, and 33 cm is a comfortable reading distance. Everyday Physics: Unusual insights into familiar things 125 If you can’t see an object sharply enough, it may help to squint. This makes your pupil narrower, at least in one direction, and blocks most of the light rays that come through the outer part of the lens. As a result, the rays entering through the central part of the lens dominate the image formation, which reduces the effect of any defects in the shape of the lens near the edges. Moreover, a narrower light beam sharpens a poorly focused image – which photographers will recognize is the same effect as increasing the depth of field by reducing the aperture (reducing the size of the opening that lets the light into the camera, by narrowing the camera’s diaphragm). Can you make the eye exceed its performance by making the pupil artificially smaller than it is by nature? To some extent you can, by reducing the aperture of your eye to look through very small holes. Because this reduces the amount of light entering your eye from the object, the object might need to be more brightly illuminated for you to see it properly. (Occasionally this can be useful. If you want to read the wattage printed on an incandescent bulb without turning it off, you can’t normally do it because the light is too bright. However, if you clench your fist so there’s only a tiny hole through it, and view the bulb through this, with one eye, you can read the writing on the bulb clearly.) But you can go too far with this approach, and run into problems with the wave character of light. If you make the pupil so narrow that it is comparable to the light’s wavelength, the light waves spread and the image blurs. On the retina each point of the 126 Everyday Physics: Unusual insights into familiar things object is no longer represented as a sharp point but as a broader spot whose diameter increases as the pupil diameter decreases. (See the experiment below). Home experiment: Light waves fanning out Light is a wave phenomenon, and this limits the sharpness of an image. You can easily demonstrate this with a very sharp needle and a piece of aluminium foil. Pierce the aluminium foil to make a tiny hole. (The smaller the better, so use the tip of a fine darning needle to pierce the foil.) Look through the hole at text in a newspaper. Even in perfect lighting conditions the letters will appear less sharp than when you look at them with the naked eye. This is because of the wave character of light. You can demonstrate the same effect in a different way with a laser pointer. Shine the laser pointer through the tiny hole in the foil, on to a white piece of paper or a white wall – the further away the better (Fig. 4). WARNING! For safety, DON’T look directly into the laser or even into the reflected laser beam. Fig. 4: Demonstrating the wave nature of light with a laser pointer and aluminium foil. You might expect that the narrow laser beam will become narrower still because it’s passing through a tiny hole (Fig. 5), but that’s not what happens: in fact the light spot on the wall is wider than the beam of the laser pointer, and it’s surrounded by faint concentric rings. The larger diameter of the central light spot indicates that a diaphragm that is too small doesn’t give a sharper image, but in fact the reverse. Everyday Physics: Unusual insights into familiar things 127 Fig. 5: What you might expect to happen with a laser beam passing through a pinhole. The radius of the central spot on the wall turns out to be 1.22 times the ratio between the distance to the wall and the diameter of the hole, multiplied by the wavelength of the light. For example, if the distance to the wall is 2 m, the diameter of the hole is 0.1 mm, and the wavelength is 650 nm (red), the central spot is 1.22 × (2,000 ÷ 0.1) × (650 × 10-9) m = 1.22 × 13 mm ≈ 16 mm in diameter. 128 Everyday Physics: Unusual insights into familiar things 35 YOUR EYE IS MORE SENSITIVE THAN A CAMERA As experienced photographers know, natural daylight is much more intense than artificial light. If you’re taking photos indoors using a traditional camera with a film, as evening approaches you need either a flash to increase the brightness, or a tripod to keep the camera still while you expose the photograph for longer in order to make the most of the little light there is. But in the same circumstances, you can easily read a newspaper! Your eye adapts effortlessly, and you can still see quite well even when it gets darker still; if there’s a fairly bright moon, you can find your way comfortably. Everyday Physics: Unusual insights into familiar things 129 So it’s clear that your eyes have a wonderfully large capacity to adapt, but exactly how large? You can get a good approximation of the answer if you know how much fainter moonlight is than sunlight, and assume that full sunlight is the brightest light your eye can handle, and dim moonlight the faintest. You can calculate the brightness of moonlight if you know how big the moon is, how far it is from the Earth and how well it reflects the sunlight, i.e. how “white” it is. When you do the sums (see box), it turns out that full moonlight is about one million times fainter than sunlight. 130 Everyday Physics: Unusual insights into familiar things To see what that means, compare how a traditional photo camera would handle things. With a given lens, to take two photos with light intensities that differ by a factor of a million, the exposure in one case has to be a million times longer than in the other. (You can reduce that a bit by changing the aperture, but that only gives you a factor of 128 at most, with a change of 7 f-stops, because every stop allows twice as much light into the camera as the previous stop). For ease of calculation, assume changing the f-stop reduces the exposure by a factor of 100 instead of 128. Accommodating the remainder – a factor of 1,000,000 / 100 = 10,000 – would have to be done with exposure time; increasing this by a factor of 10,000 gives you an exposure of maybe 10-50 seconds. The only way a camera can manage such a long exposure is with a tripod. In contrast, your eye doesn’t need a tripod or a 10-second exposure as you walk in the moonlight: you can see just as quickly as normal. Incidentally, this calculation also illustrates how amazing our modern digital camera or smartphone is. How does your eye achieve the factor of 1,000,000? As with the camera, a small contribution comes from changing the aperture – the pupil diameter. It varies at most from 2 to 8 mm in diameter, a factor of 4, which means a factor of 42 = 16 in area. The remainder (1,000,000 / 16 = 62,500) is taken care of by the photoreceptors – the rods and cones – in your retina. These complement each other beautifully with changing light intensity. When there is a lot of light, the cones do most of the work (and they are also responsible for colour vision). When there is little light, the much more sensitive rods smoothly take over, although they aren’t very sensitive to colour, which is why things appear grey at night. Clever instrument, your eye. Comparing the brightness of the sun and moon To compare the relative brightness of the full moon and full sun you need to know the size of the moon, its distance from the Earth and its reflection coefficient (“whiteness” or “albedo”). Let’s say the solar intensity – quantity of light per square metre – at the moon is I. (We’ll assume that I is the same at the Earth, because the Earth is approximately as far from the sun as the moon is.) Then the moon, with radius R, captures πR2 × I. If the moon is perfectly white (an albedo of 1.0, i.e. 100%) this light is reflected in all directions. Let us Everyday Physics: Unusual insights into familiar things 131 assume that this is equally strong in all directions, in a “half-sphere” directed towards the Earth. If we call the Earth–moon distance a, then the moonlight at the Earth is spread over the surface of a half-sphere with radius a. The surface area of that half-sphere is 2πa2. Per square metre the amount of light is: (πR2I)/(2πa2) = ½ (R/a)2 I Compared to the direct solar intensity I it is therefore attenuated by a factor of ½ (R/a)2. With the values R = 1,700 km and a = 380,000 km this yields 1.0 × 10-5, i.e. one part in 100,000. But that’s not the whole story. The moon is not perfectly white, its albedo is only 0.1. That makes the intensity of the full moon one millionth of the full sun. By the way: an albedo of 0.1 means that the moon is a very poor light reflector: you probably knew that without realizing it. If you see the moon during daytime it is always much less bright than a white wall, even though both are illuminated by the same sunlight and both are approximately the same distance from the sun. Furthermore, the albedo depends on the phase of the moon: at full moon the albedo is largest, while at less than full moon the albedo rapidly diminishes. That is primarily caused by the shadowing effects of the rough moonscape and its grainy soil. It is only at full moon that the shadows play no role. (Strictly, you never see a really full moon, because this happens only when the sun, the Earth and the moon are exactly lined up, i.e. when there is a lunar eclipse!) 132 Everyday Physics: Unusual insights into familiar things 36 PUDDLES ON A DRY ROAD Fantastic as they are, your eyes can sometimes deceive you and make you see things that aren’t there. One example is the mirage that people often see in the summer. You are driving on the highway, the weather is beautiful, the sun is shining and you see what looks like puddles in front of you on the road. But as you get closer, the puddles abruptly disappear. Why? What happens is this: you’re looking at the road, but what you’re actually seeing is the blue sky, as if a mirror were lying on the ground. The light rays that reach your eye are not coming from the road, but from above it. They have been deflected by the air above the hot road (Fig. 1). Fig. 1: The light rays above the warm road bend, so while you think you are seeing the road surface ahead, you’re in fact seeing the sky above it. Everyday Physics: Unusual insights into familiar things 133 The reason the light bends is that the speed of light close to the warm road is just a bit larger than higher up where it’s cooler (Fig. 2). Why does this temperature gradient bend the light rays? Pretend that each dotted line in Fig. 2 is a row of people, seen from above, marching forward with their arms linked. If the people on the “warm” side take larger steps than those on the “cold” side (because the speed of the “warm” people is greater), the whole row will bend towards the cold side. In other words, the faster side is automatically on the outside of the curve, just as you saw in Waves at the beach (p13). Light rays behave the same way. Fig. 2: A light ray originating from the sky skims over the warm road surface from left to right and is deflected upwards. But why is the speed of light greater near the hot road than higher up? Air expands when heated, and gets thinner as a result. The thinner the air, the closer it is to a vacuum so the greater the speed of light, because the speed of light is greatest in a vacuum. Granted, the differences are minuscule, but over a long distance the beam is deflected by a large-enough angle to produce this effect. As you get closer to the “puddle”, you are looking too steeply downward. The deflection angle of the distant rays is too small to make it into your eye, so the “puddles” vanish, and you see the actual surface of the road (Fig. 3). Fig. 3: When you look steeply downwards at the surface, you see the surface itself rather than deflected rays from further away. 134 Everyday Physics: Unusual insights into familiar things 37 SEEING THE SUN AFTER SUNSET You’re standing on the beach. It has been a lovely day and the sun is about to set. You watch as it just touches the horizon. But that’s not what’s happening: in fact, the sun set a few minutes ago! How can that be? Everyday Physics: Unusual insights into familiar things 135 Here’s how. Light rays can be bent when the light velocity varies in different parts of the atmosphere, as you already saw in the previous chapter. There, the velocity differences were caused by temperature differences which in turn caused density differences. Density differences also occur in the atmosphere even without temperature differences, because the air pressure gradually decreases with height. Isn’t that a very small effect? Yes, it is, but even though the rays are bent only slightly (as with the mirage), if you look over a large enough distance the deflection is noticeable. In Fig. 1, the thinnest air – and therefore the highest light speed, and therefore the outer side of the bend – is on the upper side, and the rays bend downward around the horizon. This is schematically shown in the figure below. Your eye assumes that the light is travelling in straight lines, and therefore that it comes along the dotted lines, so you see the sun higher than it is. Fig. 1: The differences in air pressure cause the light rays coming from the sun below the horizon (A) to curve, so the sun appears to you to be above the horizon. Moreover the sun seems flattened. Its height is only 4⁄5 of its width. That is because the lowest rays are bent the most since they travel the longest distance through the atmosphere. You can understand this by looking at what happens when the sun is directly overhead. There is no bending of any kind, because the rays from all points on the sun travel the same distance in the atmosphere. As the sun lowers, the bending becomes stronger and stronger. The end result is that your eyes “lift” the sun a bit. You see the sun higher than it is. And since you see the lower edge raised more than the upper edge, the sun appears flattened. 136 Everyday Physics: Unusual insights into familiar things ng ) u In practice, the behaviour can be more complicated, because this “air pressure” effect and the mirage effect (p133) can occur simultaneously. When both effects combine, the setting sun can take on the most bizarre forms. A special case of this phenomenon is the “green flash” or the “green ray”, sometimes visible for a few seconds at the very last moment of sunset, if the air at the horizon is very clear. What Everyday Physics: Unusual insights into familiar things 137 happens is that the degree of the “lifting” effect above is different for different colours. Just as in a prism, the red end of the spectrum refracts the least (Fig. 2), so the red disappears first. You might expect that violet remains the longest, but violet light is scattered so much that there is hardly any left (p107). The middle – the green part – remains and delivers a flashy sunset. Fig. 2: When light passes through a prism, the red is refracted the least, and the violet refracted the most. Credit: Attribution 2.0 Generic (CC BY 2.0), Siyavula Education (www.flickr.com/ photos/121935927@ N06/13580411493) 138 Everyday Physics: Unusual insights into familiar things 38 TRANSPARENT WINDOWPANES & OPAQUE LACE CURTAINS Sometimes you can see through a window from one side and not the other, but at other times it seems to work the other way round. Lace curtains on a window are a good example. In the daytime you can see perfectly well from the inside to the outside, but not vice versa. Even a bare window shows the same effect to some extent: it is much more difficult to look in than to look out. Take, for example, a shop window: from outside it sometimes acts like a nearly perfect mirror, but you can see out from the shop without any problem.. Everyday Physics: Unusual insights into familiar things 139 Is that how light works? Does it mean the light can’t pass through the same medium depending on the time of day? Or does the transmission depend on the direction? Of course not: it is a simple question of differences in brightness. Your eyes have an enormous range for brightness (see The eye is more sensitive than a camera p129). They adapt extremely well, partly by changing the size of the pupil to regulate the amount of incoming light. But if there is a bright image close to a faint one, the bright one “wins” – it takes precedence over the faint one. You can see this if you view someone’s face with the sun directly behind them: your friend’s face seems nearly black, although a lot of light from it reaches your eye. It is simply “outshone” by the much more intense light from the background. It’s the same for lace curtain and window – a question of relative brightness. First consider a bare window. Suppose you are outside looking in. If the outside light falls perpendicularly on to the windowpane, about 4% is reflected on the front side and another 4% at the back side. So a total of 8% is reflected back to you and the remaining 92% passes through the window (Fig. 1). Now an 8% reflection may not seem like much, but it’s quite a lot if it is 8% of a bright sky or of a sunlit street scene. Compare this with the background that’s competing with it. Inside the shop is very much darker than outside (although that may not be obvious because your eye adapts when you are in the dimmer indoor light). In reality the difference can easily be a 140 Everyday Physics: Unusual insights into familiar things factor 20 to 100. You notice that when taking pictures, for example: as soon as you go indoors even in the daytime, you may have to use the flash. Fig. 1: For falling light that is more or less perpendicular to the windowpane, about 8% of the incident light is reflected and the other 92% is transmitted to the other side. So if you look through a window from the outside to the inside, you see two things simultaneously: (1) the inside scene, whose light travels – practically unattenuated – to the outside; (2) the reflection of the street scene with a brightness of about 8% of the real outside brightness. On a sunny day, the outside brightness can be 50,000 or 100,000 lux, whereas even a well-lit interior might be about 500 lux. Because of this large brightness difference, the 8% of the bright outside scene can be much stronger than the 92% of the inside scene, so you see the inside only dimly (Fig. 2). If you really want to see in, you have to bring your nose to the windowpane and shield the outside reflections with your hands: then the inside light can easily “win”. Fig. 2: When you look in a shop window, the 8% of the bright outside scene can be much stronger than the 92% of the inside scene, so you see the inside only dimly. (In this example, the outside reflection is nearly ten times brighter than the transmission from inside – 4,000 lux versus 460 lux.) Everyday Physics: Unusual insights into familiar things 141 This also explains how you can use a shop window as a mirror. If you are well lit because the street is bright, your reflected image is nice and bright. Since the light that comes from inside is faint, the display in the shop window is dark and doesn’t interfere with your reflected image. Now let’s go back to the lace curtain. Why can’t you see in during the day if there is a white lace curtain on the window? The lace curtain increases the effect of the reflection of outside light. Suppose that the curtain fabric is 50% holes; the other 50% is white thread that reflects light. Then about 50% of the outside intensity returns from the curtain to your eyes – much more than the 8% of the bare window. The light that passes from the inside to the outside is negligible compared to it, especially because half of it is blocked by the lace curtain. You therefore cannot see inside from the outside. In contrast, seeing out from inside is no problem. Even with the brightness of the outside scene halved by the curtain, the other half is more than bright enough for you to see it well. You do however see the lace curtain as a kind of white haze on the outside scene, vague because you focus your eyes on the more distant outside image. You could avoid the haze by using black lace curtains. But that would be much less effective in preventing the outside world looking in (and not to many people’s taste). And what about curtains at night, when the lights are on in the house and the outside is dark? As before you see well from dark to light, but now that is from outside to inside, and as before you see poorly from light to dark (from inside to outside). Clever idea, really, that white lace curtain. 142 Everyday Physics: Unusual insights into familiar things 39 SEEING CLEARLY UNDERWATER If you swim underwater wearing diving goggles, and the water is clean, everything is nicely in focus; without the goggles, everything is blurred – you can’t focus properly. So the goggles make the difference. But how, since the goggles don’t have lenses – just plain flat glass? Everyday Physics: Unusual insights into familiar things 143 The secret is in the convex curvature of the cornea (front side) of your eye, because both it and the eye lens proper (Fig. 1) play a part in forming the image in your eye. In normal viewing – in air – the curvature of the cornea accounts for about two-thirds of the total power of your eye’s powerful lens, i.e. more than the lens itself. Fig. 1: Both the cornea (front side) of your eye and the lens inside play a part in forming the image in your eye. The bending of the ray as it crosses the air/water (i.e. air/eye) boundary is just the same as you saw with the light ray bending at the air/water boundary of the swimming pool (see Swimming pools are deeper than they look p115) as shown in Fig. 2a. The convex curvature of the front side of your eye is therefore indispensable to seeing sharply, but when your eye is in water this doesn’t work. Optically speaking, your eyeball is like water – so optically, it’s just “water in water” and doesn’t affect the passage of the light ray. In other words, as the light ray crosses the water/eye boundary, it passes practically undeviated into the eye (Fig. 2b). Fig. 2: In air, incoming light rays are refracted at your cornea, because they are crossing an air/water boundary (a). (The angle of refraction is exaggerated for illustration purposes.) However, when your eye is in water (b), the incoming light rays pass into the eye without any deflection. You have lost the important focusing effect of the eye’s convex front side, so that the combined effect of cornea plus lens gives a focused image in air (Fig. 3a) but not in water (Fig. 3b). 144 Everyday Physics: Unusual insights into familiar things Fig. 3: In air (a) the combined power of cornea and lens focus the image sharply on your retina. However, in water (b) the focus is some distance behind the retina so the image is blurred. With goggles the eye is in air again, and the situation of Fig. 3a is restored. However this introduces another transition, at the water-glass-air boundary of the goggles. What effect does that have? It doesn’t affect focusing at all because the surfaces aren’t curved but simply flat (planar) and therefore have no lens effect. However, it does affect your vision in another way, as we see below. Why sharks can easily surprise divers “Diver attacked by shark” is a common-enough newspaper headline. Why can a diver be so easily surprised by a shark? The reason is that when you swim underwater you have a much smaller field of view than above water. As you look around, you see much less than you’re used to. If, for example, you want to see what’s happening behind you, you have to turn your head much further than you think necessary. The cause is the refraction of light. Under normal circumstances, i.e. in air, your field of view is extra large, because the light rays refract towards the centre of the eye, as they enter the eye (Fig. 4a). Underwater that changes: the light rays transmit practically straight through the water-eyeball boundary (Fig. 4b) and your field of view is dramatically restricted. Goggles do not change it that much – the water/air and air/eye transitions compensate for one another; while close up the field of view is wider, as shown in Fig. 4c, at further distances from the eye the field of vision is only slightly bigger than in water, as a comparison of Fig. 5a and Fig. 5b shows. Everyday Physics: Unusual insights into familiar things Fig. 4: In each of the three parts of the diagram the size (XY) of the image on the retina is the same. In air, you have a large field of view (a). In water without goggles (b), you have a narrow field of view, so sharks can easily sneak up on you. With goggles (c) your close-up field of view is slightly wider, but at further distances it is almost as narrow as in water without goggles, see Fig. 5. 145 Fig. 5: At greater distances, the field of view without goggles (a) is only slightly smaller than with goggles (b). The moral of this story is, if you swim underwater – especially if you have an air tank on your back that restricts your vision even further – be aware that you are not seeing everything around you – you have blind spots. To be sure nothing swims up behind you, turn your head as far as you can to either side, and avoid being surprised! 146 Everyday Physics: Unusual insights into familiar things 40 WHAT MAKES RAINBOWS? The rainbow has caught people’s imagination throughout the ages. It is beautifully pure and colourful but also mystical and elusive. And it literally is elusive – you can neither grab the rainbow nor fly to it. Can you walk under the rainbow? Walking under your “own” rainbow (i.e. the rainbow that you are looking at) is no more possible than jumping over your own shadow. But can you walk under someone else’s rainbow? To answer that question we must see what makes a rainbow: ► You need the sun (or some other strong light source) and rain (or at least water droplets – from rainfall, or a lawn sprinkler, or a waterfall). ► Whenever you see a real rainbow, it is with the sun behind you and it’s either raining now or it was recently. ► You never see a rainbow in the middle of a summer’s day when the sun is high in the sky. The lower the sun, the more of the bow you see. When the sun is just above the horizon, you can sometimes see half a circle. And if you are high enough above ground – in an aeroplane for example – you can with a bit of luck see a complete circle. (You can sometimes see a complete circle with a garden sprinkler rainbow too.) ► Often there is a second fainter bow with a reversed colour spectrum outside the “main” bow: while the main bow has blue below and red above, the secondary bow has the opposite (Fig. 1). Everyday Physics: Unusual insights into familiar things 147 ► Fig. 1: A secondary rainbow above the “main” rainbow, with the order of colours reversed. Credit: commons. wikimedia.org/wiki/ User:Alexis_Dworsky, Creative Commons Attribution 2.0 Germany 148 A careful observer will note that the sky above the rainbow is darker than below it. All these features of the rainbow are explained when we see how a rainbow arises. The rainbow displays the full spectrum of colours, so it cannot have been produced by means of reflection only. This is because for reflection, the angle of incidence is always equal to the angle of reflection, so it’s the same for all colours; therefore, reflection can’t separate white light into its individual components. It must be refraction that’s decomposing the white sunlight, just as with refraction by a prism. As we will see below, the rainbow arises from light rays that are refracted in the raindrops and leave the droplets at a specific angle. We could call that angle the “rainbow angle”: the angle between the rainbow itself and its imaginary centre, which is exactly opposite the sun (and hence is under the horizon for a real rainbow). That angle is about 42° for the main bow. Everyday Physics: Unusual insights into familiar things The sun is behind you, so light must be reaching the raindrops in front of you, and must be refracted (to decompose it into colours) and reflected (to travel back to you). Let’s see if we can work out the path of a ray through a raindrop. Fig. 2 shows a simple suitable path. (By the way, the greater part of the light exits at the back side, shown by the dotted line (CF in Fig. 2), but that is not important here.) Because the droplet is in principle spherical, this sketch is not only for the horizontal or vertical plane but for all planes through the centre of the droplet. (Different colours of the rainbow are seen at slightly different angles. This means that each colour originates from those droplets that produce just the right refraction for that specific colour. We cover this in more detail below.) Tracing or calculating the exact path of the rays is laborious. It is much easier to measure the angles with an experiment using a glass of water as a model of a water droplet. Fig. 2: A ray of sunlight (A) comes from the sun, enters the raindrop, and is refracted (B), reflected (C), refracted again (D), and travels back again (E). Home experiment: The rainbow angle With a laser pointer you can easily investigate the path of a “sunbeam” through a “rain droplet”. Use the laser pointer to imitate the sun rays, and for the raindrop you can use a cylindrical glass full of water (because optically water and glass are not very different). From the top this looks like the cross-section of the droplet in Fig. 2. Ideally choose a glass with thin walls, and at least 8-10 cm diameter. (Don’t use a wine glass, because its curvature works as a lens and spreads the laser beam.) Everyday Physics: Unusual insights into familiar things 149 Put a piece of tape over the on/off button of the laser pointer so that it stays on constantly, and raise it about 2 cm off the table (on a book or something similar). Place the pointer and the glass so that there is a white wall behind the laser pointer (Fig. 3). Start with the laser beam just missing the side of the glass (Fig. 4a). Now move the glass slowly to the left, into the beam (Fig. 4b). You will see a light spot on the wall, which originates from the reflection of the laser beam from the back of the glass. Slowly move the glass through the beam and observe the spot on the wall. It keeps moving to one side until it reaches a maximum deflection; then it comes back towards the centre again. That maximum corresponds to the rainbow angle, as we discuss below. Fig. 3: The setup for measuring the rainbow angle using a laser pointer and a glass of water. On the top right is the orange spot on the back side of the glass where the light beam reflects against the rear side of the “droplet”. On the bottom left, on the wall, you can see the same beam that left the glass at the rainbow angle. 150 Everyday Physics: Unusual insights into familiar things Fig. 4: Move the glass across the laser beam, so that the beam “scans” the whole width of the glass. The experiment shows a surprising result. As you slowly scan the laser ray across the “droplet” (the glass) and look at the light reflected backward, you see that the deflection angle (relative to the entering laser beam) never exceeds a maximum value of 42°! Fig. 5 illustrates this: both ray X and ray Z return at a smaller angle than ray Y. Fig. 5: The middle ray (Y) returns at the largest angle – the rainbow angle of ≈ 42°. Everyday Physics: Unusual insights into familiar things 151 This has two consequences when you look at a real rainbow: ► Standing with your back to the sun and looking at the light reflected by a rain shower, no light is emitted from droplets in positions beyond an angle of 42°. So, first of all, this explains why the sky outside the rainbow appears darker than inside. ► The rainbow itself arises because at the special deflection angle of 42° extra light is reflected. Because this angle is a maximum, direct neighbours on either side of ray Y return at the same angle and contribute extra intensity. This angle therefore marks not only the limit of the area where you see reflected light but also the area that is brightest. The only thing now needed for a rainbow is the colours. These arise because different colours refract differently in the droplets. That angle of 42° is for red; for other colours it is smaller, for example 40° for blue because it refracts more strongly than red. Red is therefore on the outside of the bow and blue is on the inside. In fact, you are looking at different droplets when you see different colours. Fig. 6 shows this for the case where the sun is right on the horizon. Fig. 6: Every colour of the rainbow comes from its own droplets. (These diagrams are only schematic: the observer is always at the centre of the rainbow circles.) Normally the sun is higher than in Fig. 6, and you see only a small part of a circle (Fig. 7). This also shows that you see no rainbow at all if the sun is too high; our bow is then below the horizon, so to speak. (In many mid-latitudes, the sun rises above 42° at noon only from April to September, so it’s only then that this no-rainbow effect can occur.) (See How high will the sun rise today? p25.) 152 Everyday Physics: Unusual insights into familiar things Fig. 7: The higher the sun, the less you see of the rainbow, because more of it is “underground” (a). If the sun is above 42° you can’t see the rainbow at all (b). Everyday Physics: Unusual insights into familiar things 153 SECONDARY RAINBOWS Sometimes you see a second rainbow (upper bow in Fig. 1) on the outside of the main bow. It arises from an extra reflection in the droplets (Fig. 8). For this reason the secondary bow is fainter than the main bow. Moreover its colours are reversed, because the rays now pass through the droplet in the opposite direction to Fig. 2 – they enter at the bottom and come out from the top. The red – which refracts the least – now comes to you at a smaller angle than the blue: about 52° for the red and 54.5° for the blue. With a bit of luck in your laser pointer experiment you can see at about 52° the (much fainter) maximum due to the extra reflection seen in Fig. 8. This occurs on the other side of the glass to the spot you saw earlier, and corresponds to the rainbow angle of the “secondary beam”, with the reversed spectrum. (You may have to do the experiment in a darkened room to see the fainter spot.) A variety of rainbows is possible: no two bows are the same. This is due to a number of factors that we have ignored up to now, such as interference and bending of the light, and differences in size and form of the droplets. This makes rainbows more interesting still. However, all these rainbows have one thing in common: you can’t walk under your own rainbow, which follows from the explanation above about how they are formed – your rainbow travels along with you. And while I can walk under your rainbow, I cannot see that myself. Fig. 8: The secondary rainbow arises from an extra reflection within the rain droplet. (Compare this with Fig. 2, where there is only one reflection within the droplet.) 154 154 Everyday Physics: Unusual insights into familiar things 41 WHY ARE SOAP BUBBLES SO COLOURFUL? Clean water is colourless, and adding a bit of soap doesn’t change that, but as soon as you blow bubbles, the most beautiful colours appear. You can see colours in a single soap membrane, and even puddles with oil floating on top show all the colours of the rainbow. It has to do with the fact that you’re looking at a thin layer. Everyday Physics: Unusual insights into familiar things 155 These colour effects are a good illustration of the fact that we can think of light as a wave. Light has a very small wavelength: about 1/100th the thickness of a human hair. Light shows its wave character only when it interacts with structures that are of similar size to the light’s wavelength – i.e. with very small structures. The wall of a soap bubble is very thin: a few thousandths of a millimetre, or about 1/10th the thickness of a human hair, so we won’t be surprised if bubbles display some of the wave properties of light. Waves can reinforce or weaken one another, in a process that is called interference. This is clearest for two waves that are the same height. What happens depends on whether the waves are in phase (the crest of one wave coincides with the crest of the other), or out of phase (the crest of one wave coincides with the trough of the other). If we let these interfere with each other, the result is a wave with double the height if the two waves are in phase (constructive interference: Fig. 1a), or no wave at all if they are out of phase (destructive interference: Fig. 1b). Fig. 1: Constructive interference of waves in phase (a), and destructive interference of waves out of phase (b). 156 You can easily demonstrate interference by dropping two stones about 1 m apart in a pond or a big puddle of water. The stones create circular waves, and you can see the two waves interfere. Science classes demonstrate interference more easily using a ripple tank, which is a shallow glass tank of water, and vibrating bars or paddles generating continuous waves on the surface of the water. Fig. 2 shows the interference of two circular waves; the grey bands are where destructive interference annihilates the waves. Everyday Physics: Unusual insights into familiar things Fig. 2: Interference of two circular waves in a ripple tank. Credit: Andrea Persephone at English Wikipedia Back to the soap bubble. Why do we get interference in the thin wall of a soap bubble? One part of the incident light is reflected by the front side of the bubble wall and another part by the rear side, so the reflected light that you see consists of two components (Fig. 3). Fig. 3: The light you see reflected from the soap bubble consists of two components (one reflected from the front surface of the film, and one from the back) which can interfere with one another. Everyday Physics: Unusual insights into familiar things 157 These two light waves can attenuate or amplify each other. The colours arise because each colour has its own wavelength. At a given thickness of the bubble wall one colour is reinforced, the other is weakened or even extinguished completely, so the reinforced colour predominates. The thickness of the wall varies, and so you see different colours on different parts of the bubble. Sometimes something remarkable happens just before the bubble bursts. All the colours disappear and the bubble darkens – as though the bubble feels that its end is near and gives up! In fact the bubble wall has become extremely thin – thinner than the wavelength of light. No colour can be amplified any more. On the contrary, the waves reflected from the front are extinguished by the waves from the back. The reason for this may be a surprise: light has the peculiar property that its phase reverses when it is reflected from an optically thicker medium, e.g. at an air/water interface. This happens only with the light reflected from the front side, and not with that reflected from the rear side (since that light reflects from an opposite water/air interface). This is then the only thing that affects the phase in a layer much thinner than the light’s wavelength, so the two waves now have opposite phases and extinguish each other. No light is reflected any more, and the only light you see is whatever is transmitted from behind the bubble. 158 Everyday Physics: Unusual insights into familiar things 42 WHY ARE CDS SO COLOURFUL? When ordinary white light passes through a prism it splits into all the colours of the rainbow. Even when you don’t have a prism but a thick piece of glass with oblique angles (such as the bevelled edge of a fancy mirror), you sometimes also see the whole spectrum from red to violet. Let’s recall how that arises: different colours refract to different extents – red refracts the least, violet the most. In other words, the refractive index – the measure of how much a medium refracts light – depends on the wavelength. While a CD is as flat as a coin, and light does not even go through it, you nevertheless see the whole colour spectrum if you hold the CD at an angle to the light. Why? Everyday Physics: Unusual insights into familiar things 159 The secret is in the “grooves”. These are so close to each other that their separation is of the order of the wavelength of light. The separation is 1.6 μm (1.6 micrometres, or 1.6 millionths of a metre), and the wavelength of visible light ranges from about 0.4 μm (violet) to 0.7 μm (red). That’s why unexpected things happen. The digital information is in the grooves themselves, in the form of pits, and the pits are relatively dim. Between the pits are “mirroring tracks” (or “lands”). When light reflects from them they act as sources of light, and because light is a wave, the reflected light waves can amplify or extinguish each other. With a whole series of sources at equal distance in a row (the lands), this happens in a very regular manner. Fig. 1: An example of an interference pattern (caused by two rocks thrown into a pond simultaneously). Because every one of those sources sends out waves at all angles, you can almost always find two waves extinguishing one another. But there is an exception: there is a special angle at which the waves arising from two neighbours differ by exactly one wavelength (or two or three), so they amplify one another. In fact, at this angle, waves from all the lands differ successively by exactly one wavelength, so they interfere constructively and you see a lot of light, giving the fan of bright peaks in Fig. 1. And now the colours. Because different colours have different wavelengths, the direction in which they amplify each other – and you therefore see light – differs for each colour. The longest wavelength, the red, is at the greatest angle to the vertical, and violet the least (Fig. 2). So if you let light fall on a CD from one side, you will see the whole spectrum reflected. Your CD gives you music and a low-cost optical instrument into the bargain! 160 Everyday Physics: Unusual insights into familiar things Fig. 2: Waves of different wavelengths interfere at different angles to the vertical. Everyday Physics: Unusual insights into familiar things 161 Home experiment: The CD as an optical instrument Take a bright light source that produces a fairly parallel beam, e.g. a halogen spotlight in the ceiling or the sun shining through a pinhole in a blind, and hold a CD about two metres away from it. (Tape cardboard over a small window if you don’t want to make holes in your good blinds! Cardboard is also more opaque than most blinds.) Make the room as dark as possible apart from your light source. Use the CD as a mirror (Fig. 3) to reflect the light back where it came from , but offset by about 15 cm. (Some CDs are better than others for this; a blank CD-R works best.) Observe the result on a white piece of paper or a white wall or on the ceiling itself. As with an ordinary mirror, you see a white spot of light in the middle; this is from rays reflected straight back (i.e. at 180°) and for these, the path difference between rays originating in neighbouring “grooves” is zero. This is the case for all wavelengths, so there is no separation of colours, hence the white spot. Beyond the central white spot you see a colourful circle with the whole spectrum (Fig. 4). This is caused by rays where the path difference is one wavelength: first comes the violet, because it has the smallest wavelength (as in Fig. 2); then blue, green, etc., up to red. Fig. 3: Support a CD on your fingertip under a halogen spotlight in the ceiling. In fact, you see two colourful circles. This arises because diametrically opposite parts of the CD both participate. You can easily see this is the case by covering the CD with a piece 162 Everyday Physics: Unusual insights into familiar things Fig. 4: The CD in Fig. 3 gives you a white spot and two circular colour spectra. of paper that exposes only a slot over the middle of the CD (Fig. 5a). You see two coloured bands instead of circles, one on each side of the white spot (Fig. 5b). Next, move the paper so the slot exposes a strip on only half of the CD, (Fig. 5c). Now you see only a single coloured band at each side (Fig. 5d). Fig. 5: Cover all the CD except for a slot right across the diameter (a); on either side of the central white spot you a see a double band of colour (b). Now expose only one side of the central slot (c): you see a only a single coloured band on each side (d). 5a 5b Everyday Physics: Unusual insights into familiar things 163 5c If you use a DVD instead of a CD you see the same patterns, but enlarged because the “grooves” on the DVD are closer to each other than for the CD. Provided that the light source is sufficiently bright, you can determine how much finer the DVD is than the CD, using just a ruler. Measure the distance between the central white spot and the middle of the coloured band; say this is C for the CD, and D (which is larger) for the DVD. Then the ratio D/C is approximately the ratio between the groove spacing on the CD and the groove spacing on the DVD. (We’re using the approximation that for small angles a we can write sin (a) ≈ tan (a) – although this isn’t very accurate for the DVD bands.) (See the Resources appendix for information on how to make a spectrometer using an old CD.) 5d 164 Everyday Physics: Unusual insights into familiar things 43 HOW DO HOLOGRAMS WORK? A good hologram image has an astounding degree of reality and depth. It is more than a stereo photo, which is two pictures taken about 6 cm apart (see box) and quite different from a hologram. A hologram is a single image, but a very special one: it shows an object from all directions, and you can “walk around” the imaged object, so to speak. For a hologram on film, which you look through like a large photographic slide, it’s like looking through a window at a real object that has a well-defined position behind the window; if you move your head to one side, you see the object from a different angle, and parts of it become visible that weren’t visible from the other viewpoint. The technique in holography is very different from that in normal photography. For normal photos a lens forms an image of the original directly on film or on a light-sensitive chip; the image contains information exclusively about the projected image, seen through the lens. For a hologram much more information is recorded, and instead of a lens, the image is created by the wave character of light and the associated interference (see Why are CDs so colourful? p159). To record a hologram we illuminate the object with a laser. (The laser has light of only one colour and therefore only one wavelength, which is important.) We make the narrow laser beam broader using of a set of lenses and shine the beam through a semiEveryday Physics: Unusual insights into familiar things 165 transparent (half-silvered) mirror. The part of the light transmitted through the mirror falls on the object and the light scattered by the object is captured on the film (Fig. 1). Fig. 1: Making a hologram 1 – laser light illuminates the object, which scatters the light towards the film. For a single point on the object the scattered light will behave as spherical waves. But that’s only half the story. Simultaneously with this scattered light the direct beam (via the semi-transparent mirror) is also projected on to the film (Fig. 2). Fig. 2: Making a hologram 2 – part of the laser light is intercepted by a semi-transparent mirror and also directed towards the film. The rays reflected from the mirror and the rays scattered by the object combine to create an interference pattern that is captured on the film (Fig. 3). 166 Everyday Physics: Unusual insights into familiar things Fig. 3: Making a hologram 3 – the light from the object combines with the light from the mirror to create an interference pattern that contains all the information about the object. The interference pattern due to a single point on the object results in a “grating” with gradually varying spacing. This pattern contains all information – a complete 3-D image – of the object because the scattered light (and therefore also the interference pattern) is completely determined by the object. To see how this works, consider a single point on the object. This point scatters the incident light in all directions: it emits spherical waves, just like a marble that falls into a pond makes circular waves. These spherical waves interfere on the film with the light that comes directly from the light source via the semi-transparent mirror. We can regard the direct light as a plane (flat) wave, just as the waves that wash from the sea on to the beach are also more or less plane waves. The interference pattern that arises from the simultaneous action on the film of these two waves forms a pattern of light and dark spots (an optical “grating” as it is called) indicated by the intersections with the film of the red circular rays (Fig. 3); note that the distance between the spots increases from left to right. Now let’s consider how we view the hologram, once the film has been developed. (We are dealing with a film, which transmits light through it instead of reflecting it as a CD does. But that doesn’t matter: just as it makes no difference whether we talk about a slide or about a photo when discussing the principles of photography.) We illuminate the developed film (for now the hologram of that single point) with direct laser light without any mirror. The film with its grating pattern not only transmits light straight through but also diffracts it sideways in well-defined directions just as a CD does (as Everyday Physics: Unusual insights into familiar things 167 we saw in Why are CDs so colourful? p159). Those diffracted rays come from the direction of the single point on our object (Fig. 4). Fig. 4: Viewing the hologram produced earlier. Moreover, because the grating has a progressively increasing distance between the bars, the beams of light will not be parallel but will spread out, all appearing to come from the single point on the object as you move your eye back and forth. Fig. 4 explains this: given a fixed wavelength of the light, the angle at which the outgoing rays leave the grating depends on the spacing between the neighbouring “light sources”. Your eye will have the illusion that this single point is at a very specific spot behind the film. The same argument applies to each point on the object, so your eye “sees” the whole object as though it’s really there behind the film. A surprising feature of the hologram is that each part of it contains the whole object. The pattern on even a tiny part of the hologram is formed by interference to which all points of the object have contributed. Even if you cover part of the hologram, you can still see the whole object in the remaining part. The only difference between this image and the entire hologram is that the “window” through which you observe the object is smaller. Marvellous that such a small piece of material contains so much information! 168 Everyday Physics: Unusual insights into familiar things 44 WHY DOES THE SEA LOOK SO BLUE? The sky is blue is because of light scattering. If you’re looking at the sky away from the sun, the rays that reach your eye have been deviated by the molecules in the air, and the effect of the scattering is greatest for the blue end of the spectrum (see Why is the sky blue and the setting sun red? p107). But why is the sea blue? Is it because it reflects the blue sky? No, or at least it’s not the main cause: after all, when the sky is covered with white clouds the Mediterranean doesn’t turn white all of a sudden. The same goes for mountain lakes full of clear, clean water: they often seem deep blue, and a cloudy sky doesn’t make much difference. Deep-sea diving gives us a clue about what’s happening. If you dive a few metres underwater wearing goggles, everything still seems bluish. (This is very noticeable on underwater photos: those beautiful red fish seem a bit pale now.) Underwater you have to use the flash to get accurate colours, especially the red – clearly the red part of the colour spectrum penetrates deep water less well than the blue. What’s happening is that the light is attenuated selectively by clear water at a depth of a few metres, even though it is quite transparent. (Dirty water attenuates all colours equally and does so much more than clean water.) Water acts like a colour filter that stops the red a little, and lets blue pass through nearly unhindered. In other words, it absorbs with “spectral selection”. Everyday Physics: Unusual insights into familiar things 169 Fig. 1 shows that the absorption is very small for violet and blue; it increases for green/yellow; and it becomes really dramatic for red. (Box Absorption explains what the “absorption coefficient” means.) Fig. 1: How water’s absorption of light varies with wavelength. Absorption The attenuation of light through a medium is not linear – the graph of remaining light intensity doesn’t follow a downward slanted straight line, which would mean that after a few metres it becomes completely dark. The attenuation is “exponential”, which seems reasonable when you think about what is happening: every metre of water attenuates a percentage of the remaining light intensity, not a fixed amount. An exponential decay curve starts out very steep but levels out more and more as it approaches zero. We’ll see that the curve looks like the temperature curve of a cooling cup of coffee which also goes down fastest in the beginning. The formula for attenuation is: dI = −α I dx where dI is the reduction of the intensity I, α the absorption coefficient and dx the thickness of the layer (of water in this case). The solution is I = I 0 e-αx, where I0 is the intensity at the water surface (i.e. at x = 0). 170 Everyday Physics: Unusual insights into familiar things Fig. 1 shows the absorption coefficient α, expressed as attenuation per metre. Note that this does not mean that this proportion of light is removed per metre. If it did, then for red light, where α is approximately 1 per metre, it would mean complete blackness after 1 metre. Substitute some values into the solution: α = 1 m-1, I0 = 100%, x = 1 m. That gives I = 1 e-1 = 37%, i.e. the light at 1 m deep is only 37% of what it was at the surface – it has been reduced by a factor e (just over 2.7) for each metre. After two metres that is already a factor of about 7.5, so you lose the red quickly as you dive deeper. Fig. 2 shows how the intensity of red light falls off with increasing depth. Fig. 2: Exponential curve, showing temperature against time, for a cooling cup of coffee. Fig. 3: Exponential curve, showing remaining light intensity against depth, for red light attenuated in clear water. The slight attenuation of blue light is shown for comparison. Everyday Physics: Unusual insights into familiar things 171 How does that affect the colour of the sea or a mountain lake? If you look perpendicularly into the water and the bottom is white sand or rock, what you’re seeing is actually the sunlight reflected by the bottom. (Of course you also see the light from the sky reflected by the water surface, but at perpendicular incidence that is not much – only about 2% of the incident light is reflected. So if the bottom is light and the water not too deep, what you’re seeing is mostly light reflected by the bottom.) That light must go down and up, so x in our formula is twice the depth. In the light’s journey down and up it mostly loses the red part of the spectrum, so the water looks blue! Sounds logical, doesn’t it? But that can’t be the whole story. After all, even if the water is very deep and there is no bottom in sight, the water still seems dark blue (if it’s clean). How does that light come up again out of the water into your eye? That must be through scattering: light is scattered by the water molecules themselves and by other small particles suspended in the water, just as air scatters the sun rays. Once again the scattering is strongest for the blue, as in the blue sky, so it is predominantly blue light that emerges from water. That’s why the deep ocean looks blue – as long as it’s not full of algae and green seaweed. Optically speaking, frozen water behaves much the same as liquid water, and a thick layer of ice preferentially transmits the blue side of the spectrum. That is why glacier crevices appear blue down below, and the light in a tunnel through a glacier is blue. Even a layer of snow can take on a bluish tinge from transmitted light. You can see this especially clearly when there is a deep hole in the snow. Snow typically has a grainy structure, which causes light to be scattered multiple times and therefore take a long path to reach your eye. That makes for extra absorption, so even a relatively thin layer of snow can still take on a noticeably blue colour. 172 Everyday Physics: Unusual insights into familiar things 45 WHAT’S SPECIAL ABOUT POLAROID GLASSES? The inventors of Polaroid glasses combined business sense and physics: they realized there was money to be made from the fact that light waves can oscillate in all planes perpendicular to the direction of propagation. Light waves are like the waves you get (Fig. 1) when you move the end of a thick rope or a garden hose rapidly back and forth, or up and down (or in any other direction – the plane of the oscillation can have any orientation). Everyday Physics: Unusual insights into familiar things 173 Fig. 1: Waves in a rope oscillate perpendicular to the direction of propagation. It’s the same for all electromagnetic waves for that matter. It turns out that in these waves, the oscillating electric field E can take on any plane. For simplicity let’s think of two directions: horizontal and vertical. (The oscillating magnetic field is always perpendicular to the electric field, therefore we can forget about the magnetic field.) Oscillations in an arbitrary direction can be considered as being composed of a horizontal and a vertical component. With the waving rope, you can filter out parts of the oscillation through a slot, for example a letterbox in your front door (Fig. 2, Fig. 3). What do Polaroid glasses do? The lenses consist of polarization filters, or Polaroid filters, that transmit only one component and block the other. In the analogy with the oscillating garden hose we can compare the Polaroid filters with the letterbox in your front door. (And you can rotate a filter 90° – just as you can rotate your door – if you want to get the other component instead.) The light that gets through the filter is said to be polarized in the particular plane (Fig. 4). 174 Everyday Physics: Unusual insights into familiar things Fig. 2: The vertical waves in the rope are transmitted by the vertical mail slot in the door. Fig. 3: The vertical waves in the rope are transmitted by the vertical mail slot but not by the horizontal slot. Everyday Physics: Unusual insights into familiar things 175 Fig. 4: In polarized light, the oscillation of the electric field is all in one plane – in the vertical plane for vertically polarized light (a), and in the horizontal plane for horizontally polarized light (b). A Polaroid filter is really only useful when the light is significantly polarized and when there is an advantage to stopping one of the two directions. Ordinary light is barely polarized at all: all oscillation directions occur. In that case the Polaroid glasses cannot do more than stop half the light, e.g. the components of the oscillations in the vertical plane. These glasses then behave like a normal attenuator, i.e. like ordinary sun glasses. But in certain situations, Polaroids work especially well: they can remove troublesome dazzles and help you to see through a window or into water. Now they are making use of the fact that reflected light is often partly polarized, especially when it is reflected by water or glass, and then the Polaroid glasses particularly attenuate the reflected light, which for example cuts down the dazzle of oncoming headlights reflected from a wet road surface at night. If the reflecting surface is nice and smooth, the extent of polarization depends on the incident angle. There is even one incident angle – if you look a little under 45°, see box – for which reflected light is completely polarized. In that case you can completely 176 Everyday Physics: Unusual insights into familiar things suppress that reflection with Polaroids, for example allowing you to see clearly something underwater without the image being overwhelmed by reflection from the bright sky. What happens at the two extreme angles of incidence: perpendicular to and grazing along the surface? For perpendicular incidence it is simple. Because all oscillation directions are equivalent (since there is symmetry around a line perpendicular to the surface) there can be no difference between the several polarizations: they all are reflected equally. From a water surface about 2% is reflected, and from glass 4%. Because a windowpane has a front side and a back side, each of which reflect, a total of 8% of the incident light is reflected. For grazing incidence there is also no difference between the two polarizations: both are completely reflected. So if you look over water at grazing incidence, the surface acts like a perfect mirror and you see the sky as clearly in the water as above it. The same goes for glass: if someone stands with their nose against a shop window, you see the mirror image of that nose as clearly as the nose itself (as long as your eye is very close to the window). So Polaroid glasses really work for reflections from water and glass but only if you view the surface at an angle. Interestingly, pilots do not wear Polaroid glasses because they do not want to miss the reflections – the reflected flash from another aircraft may be a useful warning that it’s too close. Light reflection Fig. 5 shows how the percentage of light (the reflection coefficient R) reflected at the surface of a transparent medium depends on the angle of incidence. (Grazing incidence is 90° and incidence perpendicular to the surface is 0°.) There are two different curves for the two polarization directions: one with the electric field parallel to the plane of incidence (//, blue in Fig. 5, “parallel polarization”) and one perpendicular to that (⊥, red in Fig. 5, “perpendicular polarization”). The two curves differ substantially. In particular, the blue curve has no reflection at all at a certain angle (the “Brewster angle”), so at that point the only light that is reflected is from the red curve – it is therefore completely polarized. For an air/glass transition (drawn in Fig. 5) that angle is 56°; for an air/water transition it’s 53°. Why does the reflection at this angle disappear completely? Everyday Physics: Unusual insights into familiar things 177 Fig. 5: The percentage of light reflected from an air/glass surface depends on the angle of incidence, where grazing incidence is 90° and incidence perpendicular to the surface is 0°. The two directions of polarization are shown by the red (perpendicular) and blue (parallel) curves. The reflection disappears because of how reflected light comes about. When a light ray hits the molecules just under the surface, the molecules act as small dipole antennas, with the dipole directed along the electric field. The light ray bends (Fig. 6), and the molecules responsible for the reflection will be in that bent ray. Now look at the parallel polarized light. Its electric field by definition lies in the plane of incidence (the plane defined by the incident ray and the perpendicular to the surface). The field Fig. 6: Snell’s law: v1 and v2 are the respective velocities of light in air and water. Credit: After nl.wikipedia.org/wiki/User:Sawims 178 Everyday Physics: Unusual insights into familiar things oscillates in that plane and perpendicular to that (bent) light ray, and so do the dipoles induced by the field. But then no radiation can be transmitted in that direction because dipoles cannot transmit radiation along their own axis. Conclusion: at that angle the reflection must be zero. As the direction of the bent ray depends on the refractive index, the Brewster angle must also depend on that. Simple trigonometry shows that the tangent of the Brewster angle is equal to the refractive index, n, as is quite simply derived using Snell’s law (sin i / sin r = n, with i and r being the incident and refracted angles). This also explains the different behaviour between water and glass: for perpendicular incidence (red curve) water and glass have different values of R (the reflection coefficient). That is due to the difference in refractive index: n = 4/3 or 1.33 for air/water and about 1.5 for air/glass. The reflection coefficient for perpendicular incidence is given by R = (n – 1)2 / (n + 1)2. For water this becomes (1/3)2 / (7/3)2 = (1/7)2 = 1/49 = 2%. For glass (1/2)2 / (5/2)2 = (1/5)2 = 1/25 = 4%. Because of the squaring the reflection for the transition air/glass is the same as for glass/air. A normal windowpane therefore attenuates a perpendicular beam in total by 8%. Everyday Physics: Unusual insights into familiar things 179 180 Everyday Physics: Unusual insights into familiar things 46 WHAT DO YOUR EARS HEAR? Sound is basically periodic pressure fluctuations of the air (or more precisely, periodic compression and expansion of the medium through which the sound travels). In the days of 78-RPM records you could demonstrate this easily by sticking the corner of a postcard in the groove of a rotating record and then you could hear the music! The postcard just followed the oscillations of the groove that represent the pitch of the recorded music. The postcard pushed and pulled the air at the corresponding frequency and that was it. It was by no means hi-fi quality, and not very loud, but it worked. Everyday Physics: Unusual insights into familiar things 181 Fig. 1: Sound intensity, in dB (left axis) and W/m2 (right axis) at different frequencies. Credit: After https:// en.wikipedia.org/ wiki/File:Octave_ Designations_4.svg, Wikipedia Tagrich1961 182 How loud must sound be in order to be heard by your ears? It depends on the pitch, or frequency. At a few thousand hertz (hertz or Hz means “vibrations per second”) your ears have the highest sensitivity, and can detect quite low intensities. We can express the intensity of sound in terms of energy flow per unit area, or watts per m2: the further you are from a loudspeaker, the more “diluted” the watts that the loudspeaker produces, i.e. the fewer W/m2. In that respect sound behaves just like light: the further away you are from the lamp, the dimmer the illumination on your book. At the frequency where your ears are most sensitive, you can just hear some 10-12 W/m2 (a millionth of a millionth). Everyday Physics: Unusual insights into familiar things Fig. 1 illustrates various intensities of sound at different frequencies, for the average person. The horizontal axis shows the frequency, or pitch, on a logarithmic scale. (A piano keyboard is a logarithmic scale. As you ascend the musical scale in octaves, you don’t go up in fixed steps of frequency, i.e. not 100 Hz, 200 Hz, 300 Hz… In fact each octave is a doubling of frequency, so the octaves are more like 110 Hz, 220 Hz, 440 Hz, 880 Hz… This is why Fig. 1 can accurately show a piano keyboard and its associated frequencies along the horizontal axis.) On the vertical axis is the sound intensity. It is proportional to the volume control of a stereo system. The vertical axis has a logarithmic scale also: the axis on the right gives the intensity – in W/m2 as explained above. The axis on the left gives the intensity in decibels (dB). Each 10 decibels – or 1 Bel – represents an increase of a factor of 10 in intensity. (By the way, the unit is named after Alexander Graham Bell, the inventor of the telephone). The 0 dB reference level is, somewhat arbitrarily, put at the lowest level normally audible: 10-12 W/m2. Everyday Physics: Unusual insights into familiar things 183 Each of the solid curves represents a certain subjective impression of loudness. The lowest curve represents the threshold of hearing, i.e. the minimum intensity at which you can just hear the sound. In the most sensitive region, around 3,000 or 4,000 Hz, it is about 10-12 W/m2, as we said. Notice that the threshold curve rises steeply at very low and very high frequencies: this means that you need a higher intensity (more watts per m2) before you can hear these frequencies. Evidently, your ears are less sensitive to such high and low frequencies. The next five solid curves are similar but each one indicates the intensity needed to create a certain subjective loudness level for your ears. In musical terms, from the bottom up they correspond to pianissimo (pp), piano (p), mezzo forte (mf), forte (f) and fortissimo (ff) respectively. Finally, the uppermost curve corresponds to the pain threshold, where the intensity is so high that the sound starts to be painful. The numbers in Fig. 1 show that your ears are extremely sensitive – we are dealing with unimaginably small intensities. One millionth of a millionth of a watt per square metre is terribly little. Compare it with the lighting in your living room: if the lamps total 200 watts, for example, and they illuminate an area of 20 m2 the average intensity is 10 W/m2. Even the energy of disco noise is feeble by comparison! 184 Everyday Physics: Unusual insights into familiar things 47 WHY ISN’T THERE MORE NOISE POLLUTION? Excuse me? The last thing you want is more noise. But if you look carefully at Fig. 1 (repeated from the previous chapter), the above question makes sense, because Fig. 1 shows the immense range of intensity your ears can manage. As the vertical axis shows, it’s approximately a factor of 1012 (a million million) at the mid-range of around 1,000 hertz. In a way this is not altogether surprising: while a choir of 100 singers is a lot louder than a single singer, you don’t perceive an enormous difference with your ears. In fact you find these 100 singers about 4 times louder than a soloist, not 100 times louder. The large intensity range your ear can handle is worrisome in the context of noise pollution. In principle, the intensity of sound is inversely proportional to the square of the distance to the source. Your ears can handle an intensity difference of 1 million squared. So if you were very close to a source of noise that begins to hurt your ears – say an aircraft at take-off, or a disco loudspeaker – you would have to move a million times further away from the source in order to hear it no longer. That ought to make life awfully noisy: you would, so to speak, be troubled by the din in Durban and the noise in Nagoya. In real life, however, there’s much less of a problem, thanks mostly to the absorption of sound in the air. For every compression and expansion, a tiny bit of sound energy is lost, with the high tones Everyday Physics: Unusual insights into familiar things 185 suffering the greatest loss. You especially notice that with thunder: close-up, you hear a lot of sharp high tones in a thunder, but from a long distance away the same thunder clap just sounds like a dull rumble. Fig. 1: Sound intensity, in dB (left axis) and W/m2 (right axis) at different frequencies. 186 But fortunately even the low tones are attenuated in the atmosphere. In addition, the sound is weakened by intervening obstacles. Finally, the Earth is not flat, and over long distances the curvature of the Earth helps reduce the intensity. Everyday Physics: Unusual insights into familiar things 48 THE ENERGY COST OF TALKING Fig. 1 (repeated here from earlier) also illustrates something interesting about sources that produce sound. Take, for example, sound with an intensity of 60 dB; this is roughly the intensity of the average conversation, or music in your living room. The righthand scale shows that this corresponds to 10-6 W/m2 (1 microwatt/ m2 ). Suppose the source of this sound – a loudspeaker or somebody talking – is fairly close, so that the energy is spread out over a surface of, say, 10 m2. This means that the source is producing about 10-5 watts, or 10 microwatts of sound, which is very little indeed. It means, for example, that old-fashioned radios, which drew 50 to 100 W of electricity from the outlet, converted only a tiny bit of that energy into sound – the rest, well over 99% – was emitted as heat. So we could consider those old radios as electric heaters with a tiny bit of noise as a by-product. Everyday Physics: Unusual insights into familiar things 187 Fig. 1: Sound intensity, in dB (left axis) and W/m2 (right axis) at different frequencies. 188 If the sound is produced in the form of speech, then the average talker in a living room situation also produces some 10 microwatts. Just for fun, let’s calculate how much energy a teacher delivers during their entire career, or how much energy you produce talking over your whole life. Take the extreme case and assume that you talk 24 hours a day, 365 days a year. That’s 24 × 365 hours or, if we exaggerate a bit for simplicity, 25 × 400 = 10,000 hours a year. If you live for 100 years, the total is at most 106 or one million hours. So the total energy you spend talking is 10-5 W × 106 hours = 10 watt hours or 0.01 kWh. Even with a sky-high electricity cost of 20 pence per kWh unit, the energy value of talking non-stop for your whole life is less than a penny! Everyday Physics: Unusual insights into familiar things Music reproduction and the “loudness” control The shape of the curves in Fig. 1 is important: the lower curves rise steeply on the left, i.e. at low frequencies. That affects how your stereo equipment at home reproduces sound. Suppose you are listening to music and the volume control is up high, giving a sound intensity of 10-2 W/m2 in your living room; this corresponds to the ff curve (see What do your ears hear? p182) in Fig. 1. The curve – which indicates the subjective loudness level – is relatively flat here, so you perceive all frequencies as about equally loud, from very low to very high, so the sound has almost the same natural “colour” that it would have in the concert hall. What happens if you turn down the volume, so that it corresponds to the 10-10 W/m2 (pianissimo) curve in Fig. 1? Notice what happens at the lower frequencies: below about 200 Hz the intensity is below the threshold, so you don’t hear all the low tones – unless you selectively adjust the sound intensity for that low frequency region. On some hi-fi systems there is a special control to do just that. (It’s sometimes called the “Dolby volume” or “loudness control” – not to be confused with the volume control.) It selectively increases the amplification for the low frequency region (and also somewhat for the very high frequencies) when you turn the volume down. In this way the richness of the music is preserved over the entire frequency range. Everyday Physics: Unusual insights into familiar things 189 Home experiment: Increasing the volume of a tuning fork To get a loud sound from a tuning fork after striking it you have to press the handle of the tuning fork against a table or other big hard surface. Otherwise it’s not very loud, which isn’t surprising for the following reason. For example, consider the tone produced by a 440 Hz (“A”) tuning fork. Its wavelength is ¾ metres, which is much bigger than the tuning fork itself, and the pressure fluctuations have time to “shortcircuit” around the prongs of the fork: while one side of a vibrating prong wants to increase the air pressure, the other side wants to decrease it. The two sides work against each other, and the result is you don’t get much sound. The same short-circuit effect causes a loudspeaker without a housing to produce only high tones, because for the low ones it is just too small. For most people that is not at all obvious: we often think more in terms of resonances and the sound boxes of musical instruments and loudspeakers. Here’s a nice experiment to demonstrate this shortcircuit effect. You need a tuning fork and a large stiff envelope or a piece of cardboard. Cut out a rectangular slot in the cardboard, big enough to take one prong of the tuning comfortably, so that you can easily hold the resonating fork in the slot without touching the cardboard (Fig. 2). Strike the tuning fork and gently move one prong into the slot, as shown. Sure enough, now the fork sounds much louder. The short-circuit paths have been adequately damped, so that the pressure variations on the outside of the cardboard now effectively originate from one side of one prong of the fork. This arrangement is comparable to a loudspeaker mounted in the middle of an old door. Fig. 2: Placing a tuning fork in a slot cut in thick cardboard prevents the sound waves “shortcircuiting” to the other side of the fork and reducing the volume. 190 Everyday Physics: Unusual insights into familiar things 49 HOW CAN YOU TELL WHERE A SOUND IS COMING FROM? How do your ears determine the direction a sound is coming from? For that you need both ears. Because of their different positions, each of your ears receives the sound slightly differently. Consider a sound coming exactly from your left-hand side: not only does your left ear hear the sound somewhat louder than the right (because it’s closer), it hears it earlier too. So two mechanisms are at play: intensity difference and time difference, as shown schematically in Fig. 1. Everyday Physics: Unusual insights into familiar things 191 Fig. 1: When a sound comes from the left, your left ear hears it louder (a) and earlier (b) than the right ear. (TL is the travel time of the sound to the left ear, TR the time to the right.) Consider the time difference first. If a sound comes from one side, the nearer ear hears the sound a bit earlier than the other ear. This mechanism works only for low tones (long wavelengths); to see why that is, we have to consider the length of the wave. The length of a single wave is the speed divided by frequency. The speed of sound is just over 300 m/s; a fairly low tone has a frequency of about 100 Hz (i.e. 100/s), so the wavelength is 300 m/s divided by 100/s = 3 metres. Such a long wave is much bigger than the distance 192 Everyday Physics: Unusual insights into familiar things between yours ears, so each of your ears will perceive the same wave, but in a slightly different phase: one ear hears the beginning of the wave earlier than the other (Fig. 2).Your brain observes that time difference with great sensitivity and can work out which direction the sound is coming from. For a mid-range tone where the wavelength is equal to the distance between your ears – about 15 cm – the sound waves from the side hit the ears in the same phase (although with a whole wavelength difference). Because there’s no phase difference, the ears can’t distinguish between this sound and sound that comes exactly from the front or the back, so your brain can’t tell where it came from. For this wavelength and even shorter ones, the time difference is no good for determining direction. Fig. 2: For sound with long wavelengths, your two ears hear different phases of the wave. However, there is a second mechanism, which does work for these short waves – the intensity difference. Short waves, in particular those smaller than your head, show an intensity difference between right and left. Take a tone a hundred times as high as before (i.e. 10,000 Hz instead of 100 Hz). Instead of 3 m the wavelength is only 3 cm, much smaller than your head. When the sound comes exactly from the left, your head is in the way of your right ear: your head forms a “sound shadow” and your right ear hears a less intense sound. Measurements show that the intensity difference can easily be as much as 25 dB; your ear can detect such big differences without any problem, and your brain can then work out the direction of the sound. It is amazing how precisely you can determine the direction from which the sound comes. Careful measurements have Everyday Physics: Unusual insights into familiar things 193 demonstrated that you can do it with a precision of about 2° – the angle between the ends of a matchbox at a distance of three metres! By the way, neither of these two mechanisms explains how you manage to determine whether the sound is coming from the front or the back, which in fact you are able to do. Probably the shape of your ear shell plays a part, because it’s not symmetric front-to-back. (In fact, if you temporarily make the ear shell symmetric by means of an artificial attachment, it’s much harder to differentiate between front and back.) Moreover, there is yet another aid: you turn your head to one side when you’re not sure whether the sound is from front or back. Turning your head changes the front/back distinction into a left/right distinction, and your ears can handle that with a good deal of precision. Similarly, to distinguish between up and down you rely on the shape of your ear shell and of your head. It helps to nod your head when you want to better localize sound in the vertical direction. 194 Everyday Physics: Unusual insights into familiar things 50 DISCRIMINATING BETWEEN DIFFERENT VOICES: THE COCKTAIL PARTY EFFECT Background noise can make it difficult to hear a conversation, especially for older people, even if their hearing is good otherwise. It becomes hard to isolate one conversation from a sea of voices. This is sometimes called the cocktail party effect. How does it work and why do we have more trouble with it as we age? Everyday Physics: Unusual insights into familiar things 195 First of all, it is a complex phenomenon and it’s not just sound that’s involved, because lip-reading is also important in following a conversation – it has been shown experimentally that blind people have even greater problems following a conversation in a noisy environment. However, for older people the cause of the problem is primarily acoustic: the loss of sensitivity to high frequency is key. Fig. 1 shows how large that loss is for ages 30-85, over the frequency range from 250 to 8,000 Hz. Age 15 is taken as the baseline, representing no hearing loss. The curves are averages taken over large numbers of people. You can see that the loss increases with age, as expected, but what is striking is that the loss becomes dramatic for the higher tones. At 8,000 Hz there is typically a loss of 35 dB at the age of 60, and it increases by 10 dB each 5 years. Fig. 1: Hearing loss with increasing age. Credit: Dr JAPM. de Laat, Audiological Centre, LUMC, Leiden, The Netherlands, 2002) Comparing Fig. 1 with the graph on page 182, it’s clear that by the time we reach the age of 80 we are practically deaf to high tones above about 8,000 Hz. Why does that make it so difficult to follow a specific conversation in a hubbub? 196 Everyday Physics: Unusual insights into familiar things ► Consonants such as p, t, k, f and s contain highfrequency components, which you can’t hear properly, so can easily confuse one for another, or even miss them entirely. ► Not hearing some of the words reduces your overall comprehension of the conversation you’re interested in. ► It becomes more difficult to isolate that one conversation from the surrounding babble, because your directional hearing (see How can you tell where a sound is coming from? p191) deteriorates. ■ While the time difference mechanism works well for low tones, as we saw earlier, it’s not so good in enclosed spaces, because in many function venues the walls, floor and ceiling reflect low frequencies well (because all kinds of surfaces absorb low frequencies less than high ones). The result is that low tones reflect and continue to reverberate, so the reflected sound dominates, making it hard to determine the direction of the original sound. (We know that from our speaker set-up. For stereo we need at least two speakers, and the effect is provided primarily by the high tones. The low tones don’t contribute much to the stereo effect, so a single subwoofer is sufficient.) Overall, in enclosed spaces low tones give little directional information. Everyday Physics: Unusual insights into familiar things 197 ■ As we saw, the intensity difference mechanism works particularly well for the high frequencies. However, Fig. 1 shows that that is precisely where ears fail with age. ► Therefore, neither of the two mechanisms works well as you age, so you have little directional information to help isolate one specific conversation from the hubbub of voices. (The Technical University of Delft has developed a device that does help. It’s a pair of glasses that selectively amplifies sound from the direction you’re looking it. It works surprisingly well and makes it much easier to hear the person you’re talking to.) If you don’t feel like wearing glasses, you can still switch to lipreading – hearing with your eyes and seeing what is being said. 198 Everyday Physics: Unusual insights into familiar things 51 DO YOU HEAR BETTER AT NIGHT? The night is quieter than the daytime: there’s less traffic, no children playing, no noise from the neighbours. So it’s not surprising that overall you notice faint noises more at night than in the daytime. But not all nights are the same – on some nights you hear the distant highway much more clearly than on others; one night you clearly Everyday Physics: Unusual insights into familiar things 199 hear the church bell, but not another night. This happens even when there is no wind, so wind can’t be the explanation. Maybe temperature has something to do with it – perhaps it bends sound as happens with light (which we saw in Puddles on a dry road, p133). Sound does bend, as with all waves propagating in a medium. Bending depends on the propagation speed, and the speed of sound is more strongly temperature-dependent than the speed of light. In air, the speed of sound is related to the thermal velocity of the molecules: they have to pass on the pressure disturbance, and they can’t do that any faster than the speed at which they themselves move. Because the thermal speed of the molecules increases with temperature, the speed of sound increases with temperature. (That’s true for light too, although the underlying physics is very different.) 200 Everyday Physics: Unusual insights into familiar things On a sunny day, the air just above the ground is warmer than higher up, so the speed of sound is also greater there. Therefore, you’d expect sound to bend, in the same way as we saw light does when it creates mirage puddles. For sound waves moving horizontally, the warmest side of the wavefront is the fastest, and the fastest side automatically forms the outer curve, just as we saw with light waves and waves on the beach (Fig. 1). Therefore, sound from a source just above ground curves away from the Earth (Fig. 2). As a result, if you’re standing some distance from the source the sound tends to go over your head, and you hear the sound less well. Fig. 1: Light rays skimming over the warm road surface are deflected upwards. Fig. 2: Sound travels faster through the warm low air than through the colder air higher up, causing the sound waves to curve upwards from warm ground. A kind of sound shadow can even form where little sound penetrates (Fig. 3). The lowest sound wave is the one that just skims over the ground. This wave then curves up from the ground, and if you’re far enough away it goes completely over your head, and you hear it less. Everyday Physics: Unusual insights into familiar things 201 Fig. 3: If you stand in the “sound shadow” caused by the bending of sound waves on a warm sunny day, you will hear less sound. On a cloudless night things are precisely reversed. The air just above the ground is colder than higher up, because the ground easily radiates its heat to the clear sky. The sound rays now curve down towards the Earth. You hear the sound source better or – if the noise is troublesome – the sound pollution is worse. So if you hear a faraway barking dog better at night than in the daytime, you’re not imagining it – it’s probably just a matter of temperature. 202 Everyday Physics: Unusual insights into familiar things 52 CAN THE WIND BLOW SOUND TO YOU? Sometimes in Swiss and Austrian mountain villages, when the wind blows from a neighbouring valley, you can hear the bells from the far side of the mountain. Similarly you hear the noise from a distant motorway better when the wind blows from there. It might appear the that wind is catching the sound and blowing it to you, but that isn’t so. The real reason (which isn’t obvious) is twofold. First, wind direction influences the effective speed of sound – the speed decreases against the wind (Fig. 1a), and increases in the same direction as the wind (Fig. 1b). Fig. 1: When sound travels into the wind, its effective speed is lower (a). When sound travels with the wind, its effective speed is higher (b). The second part of the reason is that wind speed increases with height. Close to the ground the wind speed is relatively low. Higher up the wind blows harder. (Think of wind turbines: the higher they are, the better.) So, with the wind, the speed of sound Everyday Physics: Unusual insights into familiar things 203 increases with height, so the sound waves curve downward (Fig. 2a). However, for the same reason, against the wind the effective speed of sound decreases with height. That gives an upward-curving bending pattern (Fig. 2b). This looks the same as the left-hand side of Fig. 2 in the previous chapter, because in both the speed of sound decreases with height. The sound waves therefore curve upward. Fig. 2: Wind speed increases with height. Therefore, with the wind, the sound gets faster with height and therefore bends downwards (a). Against the wind, sound gets slower with height and therefore bends upwards (b). So, a downward-curved ray can pass above obstacles in its way, which explains the tolling bells in the mountain village (Fig. 3). It also explains why the motorway is more of a nuisance when the wind blows from there: the “sound rays” are concentrated as they bend towards you. In the next chapter we’ll see whether noise barriers can help. Fig. 3: A downwardcurving sound wave can avoid obstacles and be heard further away than you’d expect. 204 Everyday Physics: Unusual insights into familiar things 53 DO NOISE BARRIERS WORK? Even people living very close to a busy highway like to have quiet conversations and undisturbed sleep. Noise barriers can help. An obstacle – such as a noise barrier – gives rise to a sound shadow, analogous to a light shadow. Inside the sound shadow the sound is a lot fainter than outside it. (That’s quite noticeable when, for example, you cycle on a sunken cycle path. When you come back above ground and can see the cars, the traffic noise is noticeably louder.) The same applies to a noise barrier: you hear less noise as long as you are in the “shadow” of the barrier (Fig. 1). Everyday Physics: Unusual insights into familiar things 205 Fig. 1: When you are in the sound shadow cast by a noise barrier, the sound you hear is a lot fainter. But when it’s windy, noise barriers can be particularly ineffective. The wind speed is always much less than the speed of sound, so why does it make such a difference? The reason is not the wind speed itself but the fact that the speed can vary strongly with height – certainly close to noise barriers. The wind speed increases with height – there is a “velocity gradient” in the vertical direction. As we saw in the previous chapter, this means that the sound waves curve down when travelling with the wind, and the sound reaches areas behind the barrier that would normally be in the “sound shadow” (Fig. 2). In fact, just above the barrier the velocity gradient is extra large and the curvature therefore more pronounced, making the sound shadow smaller still. Measurements at the labs of the TNO-TPD research centre in Delft showed that at moderate wind speeds (4-5 m/s) the noise behind the shield can be 5-15 dB stronger than when there’s no wind. Fig. 2: When the wind is blowing from the source of the noise, the sound waves are bent downwards, and the sound shadow cast by the noise barrier is much smaller, so you hear more noise. The only consolation is that when the wind turns and blows away from the house towards the highway, the effect is reversed. Then you have less noise than when there is no wind. 206 Everyday Physics: Unusual insights into familiar things 54 CAN YOU HEAR WHETHER THE CURTAINS ARE CLOSED? Unfurnished empty rooms sound hollow; the acoustics are poor because the walls reflect sound well and don’t attenuate it very much. You notice the same thing in an enclosed swimming pool, or in a restaurant with bare walls and a concrete or tile floor. The sound bounces back and forth many times before it dies out, so in the restaurant there is so much background noise ricocheting around that it almost drowns your own conversation. Everyday Physics: Unusual insights into familiar things 207 Fortunately living rooms usually have better acoustics. The sound is muffled fairly efficiently by – among other things – the carpets and curtains. This is because soft surfaces absorb sound better than hard ones: you’ve probably noticed that thick curtains absorb sound much better than a bare window. The reason is that thick curtains have a large microscopic surface, and it is at surfaces that absorption takes place. Here’s an interesting question: imagine you’re wearing a blindfold and enter a room when it is pitch black. You don’t know whether the curtains are closed or not. But if you make a noise, by speaking or stamping your foot say, and you listen carefully, then you should be able to hear whether the curtains are open or closed. Right? Surprisingly, the answer is no. In fact open curtains muffle sound as well as closed ones, because indoor sounds are muffled differently from how the wall of a house attenuates sound coming from outdoors. For a wall it’s the mass that matters most: thick heavy walls insulate sound better than thin light ones. That’s logical: heavy masses are more difficult to get vibrating than light ones, so a heavy wall transmits less sound to annoy the neighbours on the other side. But for the muffling of sound inside a room, materials that are very porous (and thus have a large surface area on a microscopic scale) are most effective. As we saw earlier, the reason is that sound waves are absorbed at surfaces – primarily by friction of the vibrating air – and eventually transformed into heat. Materials with open structures absorb well because they have large surface areas. Examples are glass wool, thick textiles and even plastic foam with an open cell structure (unless it’s painted – because paint can close the open structure and spoil the soundabsorbing properties). So thick curtains are good absorbers. It doesn’t matter whether they are spread over a large area or whether they hang in a bundle in a corner, because the amount of porous surface is unchanged. Of course, they must not be in a cupboard (which probably has a door with a hard surface) but as long as they are easily accessible to the sound they are good mufflers, and the thicker the curtain the better. Therefore, the overall conclusion is: you can’t hear whether the curtains are open or closed, but you can tell if they have been taken away for cleaning! 208 Everyday Physics: Unusual insights into familiar things 55 DO – RE – MI … THE PHYSICS OF MUSICAL SCALES Musical scales are interesting: you may find it pleasant when certain combinations of pitches are produced, but not others. To explain why, we need to look at how a musical instrument generates its notes. Notes, tones, overtones, and timbre A guitar or violin string is stretched tight between two fixed points. At rest, it’s not vibrating (Fig. 1a) and it produces no sound. When you pluck the string or play it with a bow, the string vibrates. The Everyday Physics: Unusual insights into familiar things 209 most common vibration is the one where the middle of the string vibrates the most (Fig. 1b). There the size of the wave – its amplitude – is greatest. The tone or note produced by this vibration is called is the fundamental and is the lowest tone the string produces; let’s say this frequency is f. Fig. 1: Fundamental and overtones of a vibrating string. 210 Everyday Physics: Unusual insights into familiar things A string can also produce what are called overtones (Fig. 1b-1e) with frequencies higher than the fundamental tone. Take the first overtone – the 2f vibration – for example. You can hold the string in the middle without affecting anything because that point does not vibrate. Now the length of vibrating string is only half as long as before (though there are two almost-separate lengths), so its frequency, or pitch, is doubled. Similarly the second, third, etc. overtones have frequencies 3f, 4f, etc. (The fundamental tone is also called the first harmonic (1f ), the first overtone is the second harmonic (2f ), and so on.) Which vibration will the string produce? Usually it produces a mixture of the fundamental and a number of overtones. That mix determines the colour of the tone, its timbre. For a guitar, for example, you can influence this by where you pluck the string. If you pluck it in the middle, the fundamental tone dominates; pluck it close to an end, and many overtones are added, and the sound is then coarser. (Flamenco guitarists use this feature a lot to produce the distinctive flamenco sound.) Everyday Physics: Unusual insights into familiar things 211 The frequency at which the string vibrates increases if you tighten the string. The reason is that when you pluck the string now, the force on it to return to its resting (middle) position is greater – the string therefore moves faster and will be back in the middle quicker. In this way, the frequency of the vibration increases, and you get a higher pitch. For the same reason a short string has a higher frequency than a long one at the same tension: for a short string, a deviation of 1 mm from the centre causes a larger force pulling the string back to the middle than for a long string. The pitch of the note generated by the string is also affected by the thickness of the string, or rather its mass. A heavy string vibrates more slowly – and gives a lower pitch – than a light string (see box). If you look inside a piano, you can easily see the difference in thickness between the strings for the low notes and those for the high notes. You can see the same thing on a violin or guitar, though it’s not as obvious. String weight and note frequency A heavy string gives a lower pitch than a light string. The reason is that the heavier the string, the more slowly it accelerates for a given force, and the slower the vibration, the lower the pitch. The frequency f of a vibrating string is f = (1/2L) × √ (F/μ), where L is the length of the string, F the tension force, and μ the mass per metre. Relationships between notes We said that certain combinations of notes can be pleasant, but not others – at least to western ears. It turns out that the notes that sound well together are the overtones for a specific fundamental. Take dohmi-sol (the major triad C–E–G in musical terms). These sounds are all overtones of the C two octaves lower. Their respective frequencies are 4, 5, and 6 times that of lower C. They relate as 4:5:6. The whole musical scale is that way – all simple ratios. But not quite. Consider the guitar. Metal strips called frets are embedded in the fingerboard along the neck of the instrument. When you press a string down between two frets, it shortens the string to a specific length to give you a specific pitch. If you press the 212 Everyday Physics: Unusual insights into familiar things string at the twelfth fret (just where the sound box begins), the string length is halved and the pitch is doubled – you get a note exactly one octave higher. Look closely at the guitar neck (Fig. 2). Notice that the distance between frets gets smaller the closer you come to the sound box, as shown diagrammatically in Fig. 3. Because the frets are at right angles to the strings, they affect all strings equally, although different strings have been tuned to very different pitches. It looks as though every segment between frets reduces the string length by a fixed percentage and therefore adds a fixed percentage to the pitch. That is indeed the case; the proof is that by fitting a capo which clamps all the strings at any desired fret (Fig. 2) and shortens the length of an “open” string, you can play exactly the same chords as before but now everything sounds a half tone higher per fret shortened. Fig. 2: A capo on a guitar to shorten all the strings and raise their open-string pitch. Everyday Physics: Unusual insights into familiar things 213 Fig. 3: The separation between the guitar frets gets successively smaller as you move from the head to the sound box. Now let’s work out how much the pitch is raised per segment. Say the “increase factor” is h, i.e. if the note at one segment is f Hz, then the note at the next segment is f × h, then at the one after that it’s f × h × h, and so on. We know the pitch is doubled (i.e. is 2f) after 12 segments, so h12 = 2, and using your calculator shows that h = 1.05946. That looks weird! With such a crazy number how will you get simple ratios between two notes? For example, how do you get the interval doh-mi, which is 5:4 or a ratio of 5/4 = 1.25? Four segments on the guitar gives (1.05946)4 = 1.260 rather than the exact 1.25 you wanted. For the interval doh-sol you want the ratio 6:4 that is 1.5. On the guitar the seventh segment gives (1.05946)7 = 1.498, which is very close but not exactly right. 214 Everyday Physics: Unusual insights into familiar things So the tones are always slightly out of pitch. However, the big advantage is that you can start at any pitch (i.e. choose any suitable note to be “doh”) so an accompanist can choose the starting pitch to suit a singer’s voice. What about the notes on a piano? If again you want to be able to start a tune at any pitch, you must make a small concession just as with the guitar, and fudge the pitch a little. You have to tune the piano so that the ratio between successive keys (“half tones”) is always 1.05946; this is called “equal temperament” tuning as opposed to the whole-number ratio scheme (so-called “natural tuning”). A variety of equal temperament was for the first time applied by JS Bach in his Das Wohltemperierte Klavier. Equal temperament is now generally used on keyboard instruments. How are the other notes affected when you change from one method of tuning to the other? Fig. 4 shows that the differences are remarkably small: nowhere are the “errors” that you make by going from the natural to the even tuning larger than 0.9%. Really musical people can hear the difference, but it is not huge. Bach didn’t think so either. Fig. 4: Comparison of the relative frequencies using the two methods of tuning. C D E F G A B C Natural: 1 9/8 5/4 4/3 3/2 5/3 15/8 2 or 1 1.125 1.250 1.333 1.500 1.667 1.875 2 Even: 1 1.122 1.260 1.335 1.498 1.682 1.888 2 Difference 0 0.003 0.01 0.002 -0.002 0.015 0.013 0 0.0% 0.3% -0.8% -0.1% 0.1% -0.9% -0.7% 0.0% Difference % Everyday Physics: Unusual insights into familiar things 215 Home experiment: The two tones of a mug Put an empty mug with a handle on the table and gently strike the outside rim with a spoon. The mug produces a certain tone. Whether you strike exactly opposite (180° from) the handle or exactly on the side (at 90°), the pitch is the same. But now strike a spot just between those two (at 45° or 135°): the pitch is clearly higher. Why? Same mug, same spoon and yet a different pitch. The explanation must have something to do with where you strike the mug relative to the handle. What would happen if the mug had no handle? Fig. 5 shows its simplest vibration, seen from above: the original circular shape becomes slightly oval periodically, first in one direction (red oval) then in the other one (blue oval). The largest amplitudes are found at four opposing locations: the maxima of the vibrations, and one of these is where you struck the spoon. There are four points that do not vibrate at all; these are called nodes, which are at +/− at 45° and 135° from the point you struck (marked “N” in Fig. 5). If you add a handle at one node, the vibration won’t be affected much, because the node isn’t vibrating. However, if you stick on a handle on to a maximum, during a vibration the handle has to be dragged back and forth. That slows the vibration and therefore lowers the pitch. Fig. 5: How a circular mug vibrates. It’s worth trying this with a few different mugs. Some work better than others, probably due to the quality of the porcelain. 216 Everyday Physics: Unusual insights into familiar things 56 WHY ORCHESTRAS GO OUT OF TUNE When several musicians play together, they must ensure they are all in tune. How they do that depends on the type of instrument. Everyday Physics: Unusual insights into familiar things 217 As we saw in Doh-re-mi: the physics of musical scales (p209), the pitch of a string is determined directly by three parameters: (1) the length of the string, (2) its mass per unit length, and (3) its tension. The speed of sound in air increases as the temperature increases, but this makes no difference to how the string vibrates – and therefore makes no difference to the frequency. Even if the concert hall warms up, it doesn’t matter. Wind instruments are different. Consider an organ pipe. Here the wavelength of the sound is fixed; because of the pipe’s construction, it generates sound waves of a specific length. Now pitch = speed of sound ÷ wavelength, so the pitch does depend on the speed of sound. (A different way of looking at this is that the pitch is determined by how long a sound wave takes to go back and forth in the pipe. The more often that happens per second the higher the pitch, so increasing the speed of sound increases the pitch.) 218 Everyday Physics: Unusual insights into familiar things ► The only one of their three parameters that string players can adjust quickly is the tension, so they tune their instruments by loosening or tightening the strings. ► Wind players tune by adjusting the length of their instrument, typically by sliding a flexible joint in the instrument in or out to shorten or lengthen the tube. ► Organists are out of luck – they can’t adjust all those pipes! All they can do is ask the rest of the orchestra to keep in tune with them. Now suppose the temperature increases. As we saw in Do you hear better at night? (p199), the speed of sound increases with temperature. The air molecules move faster; they can pass on pressure waves more rapidly and so the sound speed increases. (In fact it is directly proportional to the average speed of the molecules.) And for a fixed wavelength, as in a wind instrument, a greater sound speed means a higher pitch. So the higher the temperature the higher the pitch. To minimize temperature changes when playing, wind players warm up their instrument beforehand with their breath. Therefore, some practice before the concert is not only useful to loosen up the player’s fingers but to warm up the instrument too. All in all it’s surprising that so many concerts sound wonderful. It’s just as well that while they are playing, all those musicians don’t worry about the laws of physics! Everyday Physics: Unusual insights into familiar things 219 220 Everyday Physics: Unusual insights into familiar things 57 WHY DO EGGS EXPLODE IN THE MICROWAVE? Cooking in a microwave differs from cooking in a traditional oven in lots of ways: ► The cooking time in a microwave depends on the amount of food you put into it: two portions take twice as long as one. That is actually quite reasonable. For example, if your oven supplies 700 watts of microwave power and if that is distributed over twice the quantity of food, it’s not surprising it takes twice as long. ► The food is heated from inside rather than from the outside. ► There can be large temperature differences between parts of the food, if the food is stationary in the microwave. ► Strange things can happen with metal foil and with decorated porcelain. ► Thawing frozen food is slow – there’s even a separate setting for it. ► Whole eggs explode! The fundamental difference from an ordinary oven is that in the microwave the heating occurs not by hot air but by electromagnetic waves – hence the name microwave. The waves are in fact radio waves, with a frequency higher than FM radio – Everyday Physics: Unusual insights into familiar things 221 about 2,450 MHz (2450 million hertz, or 2.45 GHz) compared to FM’s 100 MHz. The microwave wavelength is around 12 cm. (Divide the speed of light by the frequency to get the wavelength: 300,000,000 m/s ÷ 2,450,000,000 Hz = 0.12 m.) And just like ordinary radio waves, microwaves penetrate most materials including (dry) crockery. (You know radio waves penetrate most materials because you can listen to a radio almost anywhere – in the house, in the car, etc.) Because of their very high frequency, microwaves are strongly absorbed by water. The reason is that water molecules have one electrically positive end and one electrically negative end: they are small electric dipoles. The water molecules therefore want to orient themselves along the electric field of the waves, like a compass needle does in Earth’s magnetic field. However, the electric field of the electromagnetic waves alternates as the wave travels along from the source, and it alternates at the frequency of the microwaves. So the molecules flip back and forth at a frequency of 2.5 billion times per second; they collide with neighbouring molecules, and make them move even more. So the temperature rises. The net effect is that all the energy of the electromagnetic waves is absorbed by the water, and the water heats up. (Other liquids whose molecules are electric dipoles heat up as well as water.) Now it becomes clear why thawing is so slow. In ice (and therefore in frozen food) water molecules do not move freely: they are locked inside ice crystals. That limits their movement and therefore the process of heating. Only when a layer of water is formed that absorbs the waves efficiently does the heating speed up. (An amusing stunt is to try to boil water in a cup made of ice before the cup itself melts; put the whole lot in a big enough bowl so you don’t risk flooding your microwave. Again, the reason this stunt is possible is the relatively poor heat conduction of ice – something inhabitants of igloos figured out a long time ago.) Meals in the microwave can heat unevenly because the wavelength of the microwaves (12 cm) is smaller than the oven (typically 30 cm × 30 cm × 20 cm); the waves are reflected by metal walls, and we get wave interference. Wherever the waves reinforce each other it becomes extra hot and where they attenuate each other it remains relatively cool. A rotating turntable reduces 222 Everyday Physics: Unusual insights into familiar things the unevenness: the food goes through “warm” and “cold” spots alternately. In addition a small rotating metal fan promotes even heating – not because it stirs the air but because it reflects microwaves in all directions, so even if the food does not move, the “warm” and “cold” spots wander about the oven. Gold edges or similar decorations on the crockery can get damaged or even cause sparking, whereas a metal spoon hardly heats up. That seems strange because they are all metals and all good conductors. When they are exposed to electromagnetic waves, currents run through them, and the current in turn heats the metal just as in an incandescent lamp. Why is there such a huge difference between the gold edge and the spoon? The answer is that at such high frequencies currents only run in the “skin” of metals, i.e. in a very thin layer of about one micron (one thousandth of a millimetre) whereas nothing happens deeper inside the metal. For the gold layer that is catastrophic: all the heat produced in that thin layer has nowhere to go; the temperature rises very swiftly; melting, evaporating, and sparking is then possible. It is different for thick objects such as spoons: because the spoon is a good heat conductor, the heat can spread over the whole thickness of the spoon and the temperature rises only modestly. However, you still have to be careful with forks and knives (as opposed to spoons). The sharp extremities can work as lightning conductors or lightning rods: the electric fields at the sharp extremities become so strong that an electrical discharge can easily occur, resulting in a rain of sparks and a damaged fork. The current only in the skin also explains why the waves are reflected from the walls of the microwave. Whereas the metal wall is maybe 2 mm thick, the microwaves penetrate only about 2 microns. A tiny bit of the microwave energy is absorbed in that thin layer; the rest is reflected (by the moving electrons that form a kind of screen for the electric field). The door also reflects, because the holes in the metal mesh on the door are much smaller than the wavelength of 12 cm. No radiation escapes, provided the door is properly closed. Why do microwave ovens use the 2.45 GHz frequency (12 cm wavelength)? Is it an exact resonance frequency of the H2O molecule? No, that’s not the case. It is just a frequency at which water Everyday Physics: Unusual insights into familiar things 223 absorbs the radiation well (otherwise the food wouldn’t heat up), but not so well that the outside of the food heats up the before the inside has had time to cook. (Furthermore, there are international agreements that allocate frequencies of radiation for specific uses, including this one.) What happens if you turn on the oven while it is empty? Then there is nothing to absorb the waves. The microwave tube that emits the waves cannot dissipate its energy and can be easily damaged. Finally, why do eggs explode in the microwave? If there is nothing else in the oven, all the energy goes into the egg as heat. The egg reaches 100°C very quickly but the heating continues, so the water in the egg boils, and swiftly builds up pressure as in a pressure cooker. As there is no safety valve on the egg, the pressure keeps increasing until the shell explodes. That makes a big mess. And it is somewhat disrespectful to the chicken. 224 Everyday Physics: Unusual insights into familiar things 58 CAN YOU COOL YOUR HOME WITH YOUR FRIDGE? It sounds like a good idea: beat the heat using the fridge. This could be useful in a heat wave, and maybe even give a second useful life to an unused refrigerator: switch it on, leave its door open and there you are! Everyday Physics: Unusual insights into familiar things 225 Unfortunately, it doesn’t work. Even worse, it will act as a heater instead of a cooler. That sounds strange – a refrigerator as a heater. But if you think about it carefully, it’s reasonable. All a refrigerator does is transport heat from its inside to its outside. It is literally a heat pump. All the heat that it removes from its inside is dumped neatly outside the case – into that dusty black grid of pipes on the back of the fridge; this heats up the grid; the grid in turn releases its heat to the air. So far the net effect seems zero, as far as your home is concerned. You are simply moving heat, not removing it. But how does the fridge transport the heat? It uses a (normal) pump to circulate the cooling fluid (Fig. 1). The fluid is made to evaporate in pipes (blue in Fig. 1) inside the cold compartment of the fridge, thereby cooling it. The vaporized fluid condenses outside the fridge in the black grid (red in Fig. 1) – thus heating it up; the heat is carried away by the air into the rest of the room. Suppose the pump is driven by a 300 W electric motor, i.e. it draws 300 watts of power from the outlet. All this energy is turned into heat: the electric motor and the pump both get hot. Overall, while the fridge makes some places cold (the food compartment) and others hot (the motor, and the dusty grid of pipes, and the room), the net overall effect is that the fridge is a 300 watt electric heater if it runs continuously – which it certainly will do if you leave the door open! Fig. 1: In a fridge, the pump compresses (and thereby heats) and circulates the cooling fluid. The hot fluid discards heat through a grid of pipes and cooling fans (red), and the fluid evaporates in tubes inside the fridge (blue) and cools the interior. 226 Everyday Physics: Unusual insights into familiar things Is there any way to make that fridge cool your home? Sure, but you ensure that the heat released is dumped outside your home. You can do that by mounting the fridge facing inwards in an open window and removing the door (Fig. 2), so that the heat in the grid of pipes is carried away by the wind outside. In effect what you’ve done is install your own air conditioning unit (and made the neighbours wonder about you). Fig. 2: A fridge in a window, acting as an air conditioner. Everyday Physics: Unusual insights into familiar things 227 So if a refrigerator is a heat pump, could you use it to heat your home? Sure: reverse the fridge in the window and leave the door off. The black grid of pipes then heats the room. The cool interior of the fridge, at a much lower temperature, is warmed by the outside air (Fig. 3). The net effect is to pump heat from the outside world into your room. It is heat stolen from the wind. If the outside air is not too cold and you seal and insulate the gaps around the fridge in the window, you have, believe it or not, a much more efficient heating system than a normal electric heater. This is because a heater produces heat, whereas a heat pump merely moves heat. The ratio between the heat moved and the energy needed for doing this depends on the temperature difference and is generally much greater than 1. It is called the Coefficient Of Performance (COP). Fig. 3: A fridge in a window, acting as an air-source heat pump. 228 Everyday Physics: Unusual insights into familiar things 59 CURVE BALLS, BACKSPIN AND TOPSPIN Why are those volleys or drop shots so successful in tennis or table tennis? And why does a ping-pong ball stay in place above a blowing hairdryer instead of blowing away? The answer to these questions is non-trivial, all the more because these particular phenomena sometimes contradict our intuition. The explanation lies largely in Everyday Physics: Unusual insights into familiar things 229 Bernoulli’s law which says that in a gas flow, the pressure is lower where the flow speed is higher and vice versa. This law holds for every flowing medium, including liquids. It’s a consequence of the law of conservation of energy. Bernoulli’s law has many applications, from flow rates in an oil pipeline to the pitot tube on the nose or wing of an aircraft to measure the air speed. But the law also explains many everyday phenomena like those above, and some can be illustrated by spectacular experiments. First we’ll consider a tennis ball travelling to the right (Fig. 1). With topspin the ball rotates as well as moving forward: the top of the ball shown in Fig. 1 rotates to the right. A rotating ball flying through the air with speed V behaves the same as a stationary ball with the air flowing past it in the other direction with speed V (Fig. 2), and looking at it from this vantage point makes it easier to analyze what’s happening. Fig. 1: A tennis ball moving to the right with velocity V. Fig. 2: A stationary tennis ball, with air flowing past it to the left, at velocity V. The physics is the same as in Fig. 1. As Fig. 2 shows, the bottom of the topspun ball rotates to the left, i.e. in the direction of the air flow, so the net flow speed is thereby increased. Bernoulli’s law says higher speed means lower pressure. On top the reverse is the case: the ball turns against the air flow, the 230 Everyday Physics: Unusual insights into familiar things air speed is reduced, and so pressure is higher here. High pressure above and low pressure underneath pushes the ball downwards, increasing the total downward force (Fig. 3). The end result is that the topspun ball makes a tighter curve than a ball without the spin (Fig. 4). Exactly the same applies to a baseball with side spin, except the side spin makes the ball curve sideways rather than down, which explains the curve ball. Fig. 3: The downward forces on (a) a ball without spin, and (b) a ball with topspin. G = pull of gravity. P = downward push because of aerodynamics. Fig. 4: Side view of a tennis ball passing over a net, struck with topspin (red line) and without spin (blue line). Everyday Physics: Unusual insights into familiar things 231 The description above is not complete because many rotating objects in a flow of air or water are affected by turbulence, which is explained by what is called the Magnus effect. An illustration of this effect is given in the Home experiment below. Home experiment: The curved path of a rolling cylinder Objects rotating in air or water are affected by whether the object is spinning, as we saw above with the baseball and tennis ball. In cases in which turbulence dominates, this is called the Magnus effect. This describes the sideways force, experienced by a rotating object as it moves through a gas or fluid. To demonstrate the effect in water, take a solid cylinder of PVC or acrylic, 2 cm in diameter and about 5 cm long. (The choice of material is important: high-density materials such as Teflon or solid metal don’t work well because they sink too fast.) Let the cylinder roll down a slanted board into a tank filled with water (Fig. 5). An aquarium is best, so you can view from the side what’s happening. The board should nearly touch the surface of the water, near the middle of the tank. The cylinder is rotating when it enters the water, having started to rotate as it rolled down the board. In the water you might expect the cylinder to continue its forward motion, and hit the bottom some distance towards the right of the tank. In fact the cylinder curves backward and hits the bottom to the left of the place it entered the water. Fig. 5: Demonstrating the Magnus effect. You might expect the cylinder to hit the bottom to the right of (a) which is directly below where it entered the water, but in fact it hits the bottom to the left, at (b). 232 Everyday Physics: Unusual insights into familiar things Fig. 6: Demonstrating the Magnus effect in air. Wind up a paper cylinder on two threads tied to a stick. If you don’t have a suitable water tank, here’s a simpler version of the experiment (or view the video – see Resources appendix). Make a paper cylinder by rolling up a sheet of paper. Take a wood or plastic rod/stick about 1 m long, and tie two pieces of light sewing thread near one end, about 15 cm apart (Fig. 6). Roll the threads around the cylinder about 3 cm from each end, so that the cylinder is gradually wound up the threads. (Fig. 7a). Hold the stick parallel to and about 1 m above the floor. Now let go of the cylinder. The unwinding threads spin the cylinder as it falls, and the cylinder describes an elegant curve in the air (Fig. 7b) – thanks to the Magnus effect. Everyday Physics: Unusual insights into familiar things 233 Fig. 7: End view of the paper cylinder wound up on the two threads, ready to be released (a). The cylinder unwinding as it falls (b), describing an elegant curve due to the Magnus effect. Winding in the opposite direction (c) results in an opposite curve. 234 Everyday Physics: Unusual insights into familiar things If you wind the cylinder up on the other side of the threads (Fig. 7c), the curve will be to the left of the vertical instead of to the right as it is in Fig. 7b. A famous real-world demonstration of this effect was Anton Flettner’s Baden Baden – the “sailing ship without sails” that crossed the Atlantic in 1926. Instead of sails it had two “rotors” – vertical rotating cylinders, driven by electric motors. (The energy needed to drive rotors reduced the overall efficiency, so the idea never really caught on, although there have been a few recent ships powered by “Flettner rotors”.) Another illustration of the Magnus effect was the bouncing bomb used in World War II to destroy dams on German water reservoirs, in the so-called “Dambuster Raids”. The bomb was set spinning before being dropped, and was dropped just short of the dam. Because of the spin, the bomb as it sank curved in towards the dam, and was touching the dam when it exploded. For videos, see Resources appendix, Magnus effect. Home experiment: Sucking by blowing! In a nutshell, Bernoulli’s law says that where the air flows fastest, the pressure is lowest. You can easily create an air flow with a hairdryer, and perform a couple of surprising demonstrations of objects being drawn to the low-pressure (highest velocity) side of the flow. First is a fairly well-known trick with a ping-pong ball. Switch on the dryer (on a “cold” setting if it has one) and point it vertically upwards. Place a ping-pong ball in the air stream about 30 cm above the dryer. Most people expect the ball to blow away, but in fact it remains suspended above the dryer (Fig. 8a). Even more surprising is if you hold the dryer at an angle to the vertical (Fig. 8b) – the streaming air appears to suck the ball towards it and the ball still doesn’t fall down. The reason is that the air speed in the centre of the flow is highest, so the pressure is lowest. If the ball leaves the centre, it will be pushed back by the higher pressure outside the centre. Everyday Physics: Unusual insights into familiar things 235 a) b) Fig. 8: The ping- pong ball floats in the air flow above the dryer (a), and remains there even if you angle the dryer (b). Here’s an even more surprising demonstration. Make two disks of foam plastic / expanded polystyrene about 30-40 cm in diameter, one about 1 cm thick, the other 3 cm thick. In the middle of the thicker disk cut a hole that tightly fits the dryer nozzle and push the dryer’s nozzle into it (optionally making it more secure with masking tape or sticky tape). Point the dryer – with attached disk – vertically upwards, but don’t switch it on yet. Place the second disk on top of the first, and push a match or cocktail stick through the middle of the second disk to keep it roughly concentric with the first. Switch on: nothing special happens. Now ask your audience what will happen when you turn the dryer upside down. Everyone guesses wrong! The disk doesn’t fall off, but continues to float under the “fixed” disk instead. Thanks to Bernoulli’s law the air between the disks has a high speed and therefore a relatively low pressure. In contrast, the outside air has no speed at all, so it has a higher pressure which presses the disks together. 236 Everyday Physics: Unusual insights into familiar things Fig. 9: Even though the hairdryer is blowing air, the loose bottom disk is sucked against the disk fixed to the dryer, and doesn’t fall off. Home experiment: Oddly bouncing ball It looks impossible: you bounce a little ball off the floor against the underside of a table and it returns (Fig. 10) as though you had thrown it against a wall, almost exactly back into your hand. How can that be, since the underside of the table is completely flat? Why didn’t the ball continue and come out at the other side of the table? Clearly something non-obvious is at work here. In fact this is not an ordinary ball but a superball (often sold in science museum shops). It’s about the size of a squash ball, but bounces much better. If you drop it from a height, it bounces back to nearly the same height. Everyday Physics: Unusual insights into familiar things 237 But there is more: when rotating around its own axis, that rotation (the angular momentum) is reversed during a collision with a solid surface. This is a special feature of a superball and it explains the strange bouncing behaviour under the table. What happens is this. At the first bounce (on the floor, Fig. 10), the ball loses part of its forward motion and converts it into rotation so that it looks like a tennis ball with lots of top spin (Fig. 1, p230). At the second bounce (against the underside of the table), that rotation brakes the forward motion, as is the case with a tennis ball with spin. But here the effect is even stronger: the ball actually starts to rotate the other way. Therefore, the effect is twice as large, causing the ball to return the way it came. Then, at the third bounce (against the floor) this new rotation of the ball repeats that effect once more so that the ball comes back more or less on its original path. Fig. 10: The superball bounces back the way it came (solid line), instead of coming out the other side as expected (dotted line). What happens if you use an ordinary ball instead? When it collides with the ground or the table it converts a much smaller part of its forward speed into rotation (spin). The same laws of physics apply to both types of ball, but the exchange of linear momentum and angular momentum is different for the two, so that at every bounce the ordinary ball slows down less. The result is that halfway under the table the ball gets stuck bouncing up and down under the table and doesn’t come out the other side. What if you use a perfectly smooth ball? That doesn’t start rotating at all and therefore doesn’t slow down on collision. It will zigzag between table and floor and reappear on the other side. (If you don’t have a smooth ball, you can take an ordinary ball and smear it with soap to make it smooth.) 238 Everyday Physics: Unusual insights into familiar things 60 HOW MUCH POWER CAN YOU GET FROM SOLAR ENERGY? The sun radiates so much energy that if we express it in common energy units, we get a number so large it’s hard comprehend. Einstein’s famous equation E = mc2 says energy and mass are equivalent, so instead of expressing the sun’s energy output in watts, we can express it in radiated kilograms per unit time instead. If we do that, it turns out that the sun loses more than 4 billion kilograms per second – about the mass of 3 million compact cars – every second. And it has been doing that for billions of years! Everyday Physics: Unusual insights into familiar things 239 The sun is an unimaginably gigantic stove. It’s impossible to imagine a fire 150 kilometres away from where you live that’s so big it keeps you as warm as the sun does. Let alone at 150 million kilometres – nearly 4,000 times the circumference of the Earth! What can we do on Earth with all that solar energy? First, let’s calculate how large the energy flow is (see box): we find it’s 1,350 watts per square metre just above the atmosphere or – because the atmosphere absorbs some of the energy as it passes through – just over 1,000 watts per square metre at the Earth’s surface on a cloudless day. The solar radiation per square metre is therefore the equivalent of a 1,000 watt electric heater. The average radiation per horizontal square metre, averaged over day and night and over summer and winter, turns out to be about 100 W in the UK; between 150 and 250 in the US; between 200 and 250 in Australia; and about 150 in New Zealand. The description above is not complete because many rotating objects in much a flow ofsolar air or energy water arereaches affected bythe turbulence, How Earth? which explained by what is called the Magnus effect. An illustration of this The surface of the sun behaves like a radiator with a temperature, T, of 5,750K (kelvin). effect is given in the Home Experiment below. Once you know that, you can calculate the total emitted energy flow. According to the Stefan–Boltzmann law, that energy flow is proportional to the surface area <box start> of the sun and to the fourth power of the temperature. The proportionality constant Home experiment: The curved path of a is σ, the Stefan–Boltzmann constant. So the total energy flow in watts is: rolling cylinder 2 (4 π R ) σ T4, where R is the sun’s radius. Objects rotating in air or water are affected by whether At Earth, at a distance a from the sun, thebaseball energy isand distributed over an imaginary thethe object is spinning, as we saw above with 2 π a (Fig. 1) so the energy per square sphere of surface area 4 tennis ball. In cases in which turbulence dominates, this is metre is called effect. This describes the sideways force, (4 πthe R2) Magnus σ T4 experienced by a rotating object as it moves through a gas or fluid. 4πa2 or σT4 (R/a)2. Now substituting the radius of the sun (R = 7.0 × 108 m), the sun– To demonstrate effect in9 water, take a solid cylinder and of the value of the constant m), the solar temperature Earth distance (athe = 150 × 10 PVC or acrylic, 2 cm -8 -2 in -4 diameter and about 5 cm long. (The (σ = 5.7 × 10 W·m ·K ), we then find an energy flux density of 1,350 W/m2. choice of material is important: high-density materials such as Teflon metal don’tthat workthe well because they sinkover is perpendicular to the Note that or thesolid “square metre” energy is measured too fast.) solar Let the cylinder roll down a slanted boardBecause into a of different angles of the sun’s incident rays, not a square metre of ground. incidence at different latitudes and at different times of year, the 1,000 W/m2 is distributed over a wider area (Fig. 2). 240 Everyday Physics: Unusual insights into familiar things Fig. 1: At the Earth, at a distance a from the sun, the energy is distributed over an imaginary sphere with a surface area of 4πa2. (Not to scale.) Fig. 2: The calculated energy flow is per square metre perpendicular to the incident solar rays. On the ground, the same 1,000 W of energy is spread over a wider area – e.g. over 1.1 m2 in summer (left), or over 4 m2 winter (right). Everyday Physics: Unusual insights into familiar things 241 Fig. 3: Nuna7, winner of the 2013 World Solar Challenge car race. Credit: Creative Commons AttributionShare Alike 3.0 Unported, Wikimedia user GTHO 242 You can capture this energy in order to heat your home. The simplest way to do this is to let the sun shine into your windows, or into a conservatory which warms the air entering your home. The most efficient way is to use solar-thermal collectors, which are up to 75% efficient, depending on the installation and the type of application. You can also use sunshine to generate electricity, using solar panels of photovoltaic cells. However, they are less efficient: common silicon solar cells reach a maximum of about 15%, so of the 1,000 watts incident solar power, these cells give you 150 watts of electric power at most. More advanced cells (such as used in space technology, or for the “Nuna” cars (Fig. 3), many-times winners of the World Solar Challenge car race in Australia) reach 25%, but they are very expensive; leading-edge research cells in the laboratory have reached an efficiency of >45%. Everyday Physics: Unusual insights into familiar things Another way to “harvest” solar power is biomass – burning plants or trees to provide heat or, indirectly, electricity. To determine how useful this is, you need to know how fast things grow, which in turn is determined by the efficiency of photosynthesis. That of course depends on the kind of crop, but in temperate areas with adequate water a reasonable estimate for the annual average for photosynthesis is 1%, i.e. for each 100 kWh of incident energy, you grow enough to burn to give 1 kWh of heat. That’s not a lot, but an advantage of biomass over photovoltaics is that you get “energy storage” for free: you can easily store wood and burn it when it suits you, whereas storing large amounts of electricity is very expensive. In terms of oil “energy equivalents”, the yield of biomass is about one litre of oil per square metre per year. Everyday Physics: Unusual insights into familiar things 243 Land area needed for power generation For large-scale photovoltaic and for biomass, the area of land required is important. How much space is needed for a large solar electric power station? Photovoltaic cells produce electricity directly. For biomass we have to take into account the transformation from heat to electricity. Consider a 1,000 MW (1 Gigawatt, 1 GW) power station generating electricity, averaged over the year. The space needed with current technologies turns out to be large: ► For photovoltaic cells (with 15% efficiency), area needed = 60 km2 (the area of the island of Manhattan, NY). ► For power generated from biomass (with 1% efficiency), area needed = 3,000 km2 (a little less than the area of Long Island, NY, or a little more than the country of Luxembourg). If, for example, the Netherlands wanted all its energy to come from biomass – by continually sowing and harvesting – it would need an area bigger than the Netherlands and Belgium combined. It’s clear, therefore, that much research is needed, both in the production of cheap but high-efficiency solar cells, and artificial photosynthesis with greater efficiency than nature’s. This is a suitable task for our politicians to invest in, provided of course they ever look beyond the next election. 244 Everyday Physics: Unusual insights into familiar things 61 THE MYSTERY OF THE WANDERING CARPETS Wall-to-wall carpets do not wander about, but their smaller relatives do: loose rugs and runners that are not tacked down can slowly get out of place, and have to be put back every so often. Curiously, they always move in the same direction, even if the floor is perfectly horizontal. Why is there a preferred direction? Everyday Physics: Unusual insights into familiar things 245 It is because of the laws of mechanics. If you walk over a carpet at constant speed, nothing happens. Granted, there is a backward force when you push off with your back foot, and a forward force when your forward foot lands, but they even out so the net force is zero (Fig. 1). Even on the most slippery surface the carpet stays neatly in place. But as soon as you stop, the carpet and the floor have to deliver enough friction for you to brake. The carpet therefore tends to shift in the same direction you’re walking in. Even if it can move only a millimetre, it will. Fig. 1: (a) When you step on to the rug, the force tends to move the rug forward. (b) When you step off the rug, the force tends to move the rug backward. But isn’t that shift compensated for as you leave the carpet, when you walk out of the room for instance? It depends: yes, if you continue walking in the same direction as before; that’s what happens to a doormat when you walk in after wiping your feet. But it’s different if after stopping you go back the way you came: the “pushing off” as you resume walking repeats the effect caused by your stopping. Think of a rug in the middle of a room that has only one door: the rug will wander away from that door, even if it lies in the middle of the room. In a room with two doors, the rug will drift away from the door that has the most use. So show me how a carpet drifts over time, and I will tell you the typical walking pattern of the people who use that room. 246 Everyday Physics: Unusual insights into familiar things 62 COMMON MISCONCEPTIONS ABOUT THE RADIOMETER Science museums and even shop window displays often display a radiometer. It consists of a glass bulb with a small horizontal rotor on a vertical axis (Fig. 1). Each vane is black on one side and white or silver on the other. When illuminated brightly, the rotor turns like crazy – see the Resources appendix for a video. Everyday Physics: Unusual insights into familiar things 247 Fig. 1: The radiometer. Credit: https://commons. wikimedia.org/wiki/ User:Haade, CC-BY-SA3.0 <image>crookesradiometer=haade.jpg A common misconception (repeated even in a popularscience magazine a few years ago) is that the rotation is caused by the pressure of the light. Granted, light does exert a pressure: to put it in scientific terms, photons – the quanta or “particles” of light – have momentum. So the argument goes that the light rays bounce off the shiny side of the vanes, but are absorbed by the black side (Fig. 2). The result is a net force that turns the rotor. But in fact that isn’t correct. The light pressure is tiny. (The value of the light pressure, given by a detailed calculation, is the number of watts per square metre divided by the speed of light.) Even if the vanes are in full sunlight, the force on a vane is a few tenths of a billionth of a newton (10-7 N). That’s equivalent to the weight of a tiny piece of hair about 0.01 mm long – so small you can hardly see it. Such a small force couldn’t conceivably turn the rotor 248 Everyday Physics: Unusual insights into familiar things so quickly, not even in a vacuum. Moreover, the mill turns the wrong way to be explained by light pressure: it isn’t the shiny side that is pushed back, but the black side. Fig. 2: The wrong explanation of why the radiometer vanes rotate. The real mechanism is completely different. The only effect of the incident light is to heat the sides of the vanes. The heating is uneven: the black side gets a bit hotter than the shiny side. The force that makes the rotor turn is actually delivered by gas molecules. The glass bulb is only a partial vacuum and contains some gas at low pressure. In gas at sufficiently low pressure, funny phenomena (like this) occur that don’t occur at normal pressures. Let’s look at exactly what’s happening. The gas pressure is so low that the molecules hardly ever collide with each other; mostly they bounce from wall to wall inside the glass bulb, and occasionally hit the vanes. Because the bulb is symmetrical, there’s no preferred direction of travel for the molecules, so the number of molecules striking the shiny side of the vanes is the same as the number striking the black side. And the molecules have equal speeds, since they have just bounced off the same glass wall. In other words, when Everyday Physics: Unusual insights into familiar things 249 molecules strike the vane surface, they deliver equal momentum whether the surface is black or shiny; so far, there is no net force to move the vane. But the above is only for the striking of the vanes; things are different when a molecule bounces back off a vane. During the nanosecond or so when a molecule is touching a vane surface, the molecule takes on the temperature of the surface, and that temperature determines the take-off speed. Molecules leave the black (hot) surface at higher speeds than the shiny (cooler) surface – they push off from the black surface more strongly than the shiny one (Fig. 3). Therefore, it’s the black surface that is pushed back, not the shiny one, which is the opposite of what would happen if it was light pressure that caused the rotation. Fig. 3: It is the air molecules that rotate the vanes. Molecules striking the (hotter) black surface rebound at higher speed than molecules rebounding from the (cooler) white surface. This phenomenon has been studied in detail, including the case of higher pressures where the molecules start colliding with each other, to determine the optimum pressure to make the rotor turn fastest. (In 1924 Albert Einstein published a paper about this – see Resources appendix.) 250 Everyday Physics: Unusual insights into familiar things 63 IS THICK GLASS A BETTER INSULATOR THAN THIN GLASS? Glass is a poor heat conductor. The handle of a glass teacup doesn’t get nearly as hot as the handle of a tin mug. If glass insulates so well, could you double the insulation value of a window by making the glass twice as thick? Everyday Physics: Unusual insights into familiar things 251 The answer is no. Even though doubling the thickness of the glass doubles the heat resistance, it makes no difference to the window. Surprisingly, even a cellophane film of 0.1 mm thick works just as well as thick glass, which is why this sort of film is used as a temporary form of double glazing. The reason is that air insulates far better than glass – it conducts heat about 40 times less well than glass. In a window, 99% of the insulating capacity is due to the air layers adjacent to the pane. Doesn’t the air need to be still to insulate well, whereas air flows all the time, especially on the outside of a window where the wind blows? Indeed there is flow – due to convection – even inside. This is a good thing, because with this air flow the temperature in the room becomes reasonably even: warm air above the radiator expands and rises, flows along the ceiling and comes down on the other side of the room. Without this “natural” convection there would be even bigger temperature differences in your rooms than there are now – warm at one chair, cold at the other. However, this doesn’t affect the insulation properties of the window, because heat is transported by convection only in the direction of flow, since it is the travelling air that takes the heat along. In a layer adjacent to the windowpane, air can only flow parallel to the pane: it cannot go into it. This layer usually has a thickness of a few millimetres, and in this layer the heat transport must take place by conduction, as if the air really were stationary. And a stationary air layer insulates 40 times better than a glass layer of the same thickness as we said above. In fact the air has a double benefit, because there are two layers, one on the inside of the glass, and one outside. The exterior layer is slightly less effective – slightly less thick – than the interior one, due to wind. But together these layers are some 4 to 5 mm thick, thicker than most panes of glass, so they easily outperform glass in the insulation contest. Because air is a much better insulator than glass, in a window we can regard the glass as a good conductor within which the temperature is practically homogenous; there is no temperature difference between the interior and exterior of the pane (Fig. 1, 252 Everyday Physics: Unusual insights into familiar things inset). The temperature gradient occurs exclusively in the insulating air layers, so the temperature profile is as shown in Fig. 1. It’s clear that the temperature of a single pane is quite low. With the external temperature at −10°C and the interior temperature at +20°C, the temperature of the glass can go below freezing, and when the winter is very cold you can see frost on the inside of single-glazed windows! Double glazing doubles the number of air layers, as long as the in-between space is at least about 6 mm. (A much wider cavity doesn’t improve the heat insulation, because convection limits the insulating action to the layers adjacent to the panes. However, a wider cavity does improve the noise insulation.) Fig. 2 shows the temperature profile, with the same inside and outside temperatures as in Fig. 1. The insulating value is practically doubled: you now have four air layers instead of two, and an additional advantage is that the temperature of the inner pane is much higher than in the case of single glass. That adds comfort, because it reduces the draughts from the window. Everyday Physics: Unusual insights into familiar things Fig. 1: The temperature profile across a single-glazed window. Inset: note that the temperature on the inside surface of the window is almost exactly the same as on the outside surface. 253 Fig. 2: Double glazing insulates a room more effectively than single glazing, and the much higher temperature on the interior side of the glazing reduces draughts from the window. The curve in the temperature profile symbolizes the onset of convection between the panes. Radiation also affects your comfort (see How do you keep your temperature constant? p49). The side of your body facing the cold window will be colder than the other side, and this unequal cooling can feel unpleasant. What about “secondary double glazing” – a removable pane of glass, sometimes in a light frame, that’s temporarily fixed over the window? As is clear from the above, these are just as effective as real double glazing. In the US, these are called storm windows and can be installed inside or outside. In the UK they are almost always installed inside. Instead of an extra pane of glass, you can get purpose-made very thin plastic film (like cling film / food wrap) to stretch over the inside window frame and tape on at the sides. This also works well. The only real requirement is, after all, that the extra surface is impermeable to air. Thickly woven curtains that close well are nearly as good in this respect. 254 Everyday Physics: Unusual insights into familiar things 64 IS THERE A VACUUM INSIDE DOUBLE-GLAZED WINDOWS? Double glazing is a great invention, substantially improving the insulation of our homes, as we’ve just seen. It works because you’re adding an extra layer of air between the two panes of glass, and air is a good insulator. Even better would be to completely evacuate the space between the two panes – after all, a vacuum is the best possible insulator. That sounds good, but how big is the force on such a pane, and can the panes stand it? Let’s compute. Atmospheric pressure corresponds to a mercury column of 76 cm. Mercury is very dense (13.6 times denser than water), so the weight of such a mercury column on 1 square centimetre is 76 cm × 13.6 g/cm3 = 1,033 g/cm2 or more than a kilogram! That’s an awful lot and really adds up for a large surface. Take a window that’s one metre square (which isn’t particularly big as windows go): its area is 10,000 cm2. So assuming there’s a vacuum on the other side of the glass, the force on the pane is a weight of 10,000 kg, the same as about 8 small cars. So a vacuum between the panes is impossible – there’s no way the glass could withstand the pressure without breaking. Unless you play a trick. You can get evacuated double glazing panels that have tiny transparent pillars inside to keep the panes from collapsing. The manufacturer says “they are 0.5 mm diameter and 20 mm apart, [and] when viewed in transmission at a distance of 1-2 metres they become imperceptible.” (See Resources appendix.) Everyday Physics: Unusual insights into familiar things 255 The above example illustrates the enormous force of atmospheric pressure on a relatively large area, which was first demonstrated by Guericke’s famous 1654 experiment in Regensburg: he placed two hemispheres together (Fig. 1), evacuated them, and teams of horses couldn’t pull them apart. Another example is an oldstyle TV with its deep picture tube or “cathode ray tube” (CRT) which was also a feature of old computer screens. The tube has to maintain a vacuum to allow the electron beam to move unhindered and form the picture on the screen; it must therefore be able to withstand the external pressure without imploding in the middle of your living room. The atmospheric force on the front of a TV with a screen of 40 by 50 cm (that is 2,000 cm2) is 2,000 kg, so the glass must be very thick to be strong enough. This explains why for half a century TVs hardly became lighter in spite of the miniaturization of all other electronic components: it was simply the weight of the glass. Back to double glazing. Could you get at least some of the benefit of a vacuum by lowering the internal pressure a bit, e.g. to 0.9 or 0.8 bar, which might reduce the heat loss by 10 to 20%? Alas, no: it turns out that the heat conduction of a gas is independent of the pressure. Here’s why. Say you halve the pressure. Then there are only half as many molecules left to transport the heat, so you might 256 Everyday Physics: Unusual insights into familiar things Fig. 1: Guericke’s original “Magdeburg” hemispheres (named after Guericke’s hometown, where he later repeated the demonstration) and vacuum pump, in the Deutsches Museum, Munich, Germany. Credit: Creative Commons AttributionShare Alike 3.0 Unported, LepoRello (Wikipedia) think the heat conduction would be halved. However, that’s not true: each molecule now transports heat twice as efficiently because the molecules hinder one another less – the length of the average path between two collisions is doubled. The end result is that decreasing pressure does not affect the heat losses. Insulation is improved only when the pressure becomes so small – far below a thousandth of an atmosphere – that the molecules hardly collide at all, but fly from pane to pane unhindered. Then, the heat conduction becomes proportional to the number of molecules, and does decrease with reducing pressure. Everyday Physics: Unusual insights into familiar things 257 Home experiment: Greedy balloon? When you’re inflating a balloon, the first few puffs are the hardest, even if you’ve stretched the balloon a few times, but as soon the balloon reaches a certain size, it’s easier to inflate. This is surprising, because it’s the opposite of what happens when you stretch a rubber band: it’s easiest at the beginning. Similarly it’s easiest at the beginning when you stretch an uninflated tyre, so it can’t be the type of rubber that makes it hard to inflate the balloon; something else is happening here. More surprising still is what happens if you connect two identical balloons with a piece of flexible tubing or garden hose that you can squeeze shut with your fingers (Fig. 2). Attach the first balloon to the tube, and inflate it to a reasonable size (roughly twothirds of its maximum size); now squeeze the tube shut between two fingers. Inflate the second balloon somewhat less (about half the size of the first one), and attach it to the other end of the tube (Fig. 2a). And now for the exciting moment: what happens when you release your fingers on the tube, so the air can flow freely between the two balloons? You’d probably expect that the air will distribute itself equally between the two balloons, and they will end up the same size. After all, that’s what happens when you pull on two elastic bands that are tied together: they stretch equally far. However, that’s not what happens here (Fig. 2b): the larger one grows and the smaller one shrinks! 258 Everyday Physics: Unusual insights into familiar things The explanation is not that one balloon is greedier than the other. In fact it relates to something you notice when inflating an empty balloon: when the balloon is small it needs a lot of pressure to inflate. To see why that is, we can view the inflating as pushing two imaginary halves of the balloon away from each other. To do that you must apply a force, F say, that’s just big enough to stretch the rubber. You may remember that force is equal to pressure times area (because by definition, pressure is force per unit area). Now the area – the cross-section of the balloon – increases with the square of the diameter: twice as large a balloon has an area four times as large an area of cross-section. This bigger area requires less pressure for the two imaginary halves to be pushed apart, so the balloon grows more easily. (We tacitly assumed that the counteracting forces of the rubber remain constant in the process. This isn’t quite true, but these forces increase only slightly, as long as we are within the elastic range of the rubber.) Everyday Physics: Unusual insights into familiar things Fig. 2: Two balloons connected by a tube: (a) with the connecting tube closed, and (b) with the tube open. 259 Thinking about inflating a balloon that has a long sausage-like shape might remind you of what we’ve just explained. When you blow into a sausage balloon (Fig. 3a), as it inflates it doesn’t thicken evenly along its length but in fact inflates at one place (Fig. 3b) until the “elastic limits” are reached, i.e. until the rubber there is stretched so much that it’s much harder to stretch any more. Only then does it become thick over the whole length (Fig. 3c). Fig. 3: Inflating a sausage-shaped balloon (a) causes the balloon to inflate initially at one place (b) and not evenly along its length; only when fully inflated (c) is the balloon even along its whole length. 260 Everyday Physics: Unusual insights into familiar things 65 CAN YOU FEEL ENERGY CONSUMPTION? Everyone knows you can measure energy consumption, or look in the instruction manual to find the rating of your electrical appliances. But in order to make a rough estimate, that isn’t necessary – most of the time you can feel the energy use via the heat an appliance produces. After all, according to the law of conservation of energy, the energy has to go somewhere and that’s nearly always in the form of heat, so in effect a 1,000 W vacuum cleaner is a 1,000 W heater, a 60 W lamp is a 60 W heater, and so on. If you want to know if your DVD player, the hand-mixer or internet router use much Everyday Physics: Unusual insights into familiar things 261 energy you need only put your hand on it to find out. Appliances that remain cool cannot be big energy consumers (although even modest energy consumption used 24/7 does add up over time). If an appliance has a ventilator for cooling, that’s a bad sign, because it suggests that the manufacturer expects the appliance to produce a lot of heat. And things designed to produce heat – central heating units, boilers, washing machines, dishwashers – are the really big consumers. Here’s an example of estimating energy use – for an electric shaver. It can’t use much energy, because the only place that the energy can go is conversion into heat – from the electric losses and mechanical heat of friction in the shaving head. That cannot be much: during the whole shaving operation of a few minutes the head barely heats up, and the amount of heat generated is much less than you’d need to heat a basin of water for a wet shave. So we can estimate with a rough calculation that an electric shave uses about as much energy as running the hot water tap for one second. A wet shave doesn’t save energy – quite the opposite, unless you shave with cold water. Of course, when considering electricity, you must also take into account the efficiency of the power-generating station, and any further losses that occur before the current gets into your house. For every joule (one watt for one second) of electricity that comes into your house, nearly three joules of energy were consumed at the power station, assuming it runs on natural gas, coal, oil or nuclear, rather than solar or wind. So we have to multiply the electricity consumption by three for a true whole-energy comparison with, e.g., the gas used in the central heating unit that has an efficiency of nearly 100%, or with the car that runs on petrol or diesel fuel. Fig. 1 shows a rough comparison of various “appliances”. It gives round numbers for the power in watts, and the energy use in terms of litres of oil or petrol per hour, or cubic metres of natural gas per hour. The figures assume that the appliance is used 24 hours/ day, at the specified power. The conversion losses are included. So the energy use of a car in terms of power (watts) is taken as five times the mechanical power of the engine, because the engine efficiency is taken to be 20%, i.e. the energy delivered by the engine is only 20% of the energy in the fuel. 262 Everyday Physics: Unusual insights into familiar things Power in watts Equivalent energy consumption in m3/hour of natural gas, or litres of oil / hour Clock (3 W electric) 10 0.001 Shaver (10 W electric) 30 0.003 Candle; small pilot flame; a human adult 100 0.01 Lamp; laptop (100 W) 300 0.03 PC, TV (200 W) 600 0.06 Large gas ring 2,000 0.2 Vacuum cleaner (1,000W) 3,000 0.3 Kitchen instant water heater (on natural gas) 10,000 1 Midsize central heating unit 20,000 2 Generous shower 20,000 2 Car (100 km/h, 16 kW) 80,000 8 Full car, per person 20,000 2 Full airliner, per passenger 400,000 40 The table shows some interesting points. The energy used in a day by an old-fashioned pilot flame or a candle is enough to watch TV all evening, or to shower for ten minutes. With the energy used in a day by a 100 W electric appliance, you can drive ten kilometres. A full airliner uses 20 times as much fuel per passenger per hour as a full car on the motorway. Of course, the aircraft goes 10 times as far as the car in an hour, so it uses only twice as much fuel per kilometre; therefore driving to your holiday destination in a half-full car uses about as much fuel as flying there on a full plane. Everyday Physics: Unusual insights into familiar things Fig. 1: Comparison of whole-energy use of various appliances. 263 Rules of thumb for the running cost of appliances One watt (1 W) used constantly for a year = 1 W × 3,600 s/hour × 24 hours/day × 365 days/ year = 31,536,000 watt seconds (Ws) = 31,536 kilowatt seconds (kWs) = 31,536/3,600 kWh = 8.7 kWh ≈ 10 kWh. In other words, if you run something for a year that uses 1 W, it uses about 10 kWh/year. Now for the cost: ► If a unit (i.e. 1 kWh) costs 10 pence or cents or whatever your currency is, the yearly cost of your 10 kWh is about £1 – or $1 or, etc. Using this, you can easily estimate the annual cost of an appliance. If you leave a 100 W bulb burning in the basement all year, that costs you about 100 × £1 = £100. If you leave your 8 W internet router switched on 24/7, it will cost you £8 per year. (If a unit costs 15 pence or cents instead of 10, the yearly cost is 1.5 times the above, so just multiply up accordingly: e.g. your 8 W internet router will cost 8 × 1.5 = £12/year.) ► 264 Everyday Physics: Unusual insights into familiar things 66 IS A BLACK CENTRALHEATING RADIATOR BETTER THAN A WHITE ONE? Most central heating radiators are white or another light colour that suits the interior decor. You might think that’s silly – after all, don’t radiators yield much of their heat by radiating, and doesn’t a black surface absorb and radiate heat best? (And that’s why you use a chromium coffeepot, and pack your baked potatoes in aluminium foil, because they radiate heat poorly.) It’s true that a surface that absorbs radiation poorly, such as a white surface, also emits radiation poorly. (In technical terms, the absorption coefficient is equal to the emission coefficient.) Everyday Physics: Unusual insights into familiar things 265 That is actually correct but there is a snag. A surface with a low absorption will indeed also have a low emission, but at that wavelength. A surface that you perceive as “white” has little absorption for visible light, so that also goes for that white radiator. But a heating radiator doesn’t emit any visible light: it radiates in the far infrared, with a wavelength about 20 times longer than visible light (about 10 μm compared to 0.5 μm). And the absorption properties at these very different wavelengths can also be very different. The surfaces of most radiators are painted and, in the infrared region we are talking about, most types of paint surfaces are efficient radiators: they are “black” at those wavelengths even though are “white” at visible wavelengths. White paint on a radiator is practically as efficient as black paint. There are exceptions: metallic paints such as aluminium. These are fairly “white” in the infrared and therefore poor heat radiators, just as is the case for most metals themselves. So that shiny metal coffeepot is certainly clever, and so is the aluminium foil: they radiate heat poorly and keep the contents warmer for longer. What if you have an aluminium-painted radiator at home? Well, it’s not as bad as it seems. The heat that would have been radiated from a different radiator isn’t lost – it just goes back to the central heating boiler. However, the temperature of the radiator has to be raised in order to have the same heat emission. That means a higher temperature of the water in the system, which reduces the efficiency of the central heating unit somewhat. Not ideal, but no reason to rush for your paintbrush right away. Do radiators radiate? Many internet articles say that radiators don’t radiate much, and that instead they yield most of their heat by convection. Is that true? For radiators consisting of three panels, each behind the other, it is true – about 80% of the heating is by convection. However, for single-panel radiators, about 50% is by convection and 50% by radiation. (The radiation percentage in multi-panel radiators is lower because the front panel blocks radiation from the middle and back panels.) 266 Everyday Physics: Unusual insights into familiar things 67 DOES BLACK PAINT GET HOTTER THAN WHITE? South-facing doors and window-frames have a rough time when the sun shines. Wood is a poor thermal conductor and can’t conduct away the solar heat easily; the paint gets hot, expands and shrinks, and the surface has to be repainted frequently. Everyday Physics: Unusual insights into familiar things 267 A reasonable question to ask is does the colour of the paint matter? Does white paint stay cooler than other colours? At first you might say yes, because white surfaces absorb less solar heat than dark ones, so they stay cooler. But in fact it’s not that simple. Aren’t surfaces that are poor absorbers also poor emitters? In other words: the net effect of colour on temperature may well be zero. In fact the first, intuitive answer is correct: colour does matter. You can prove it with a simple measurement. Around noon on a sunny day in June, my digital thermometer told me that a white window frame was 43°C and a dark green one was 66°C. That’s a significant difference. (The real difference may be larger, since touching the surface with the temperature sensor drains away a bit of heat from the painted wood, yielding readings which are too low.) So why is the net effect of colour not zero? The reason is that our analysis above was incomplete: the crucial point is that whereas a lot of the absorption by the paint is in the visible range, the emission (as discussed in the previous chapter) is in the far infrared, where the paint’s emission properties may well be different. And sure enough, they are. In the infrared, almost all painted surfaces behave as if they were black, so while different-coloured paints may have different absorption for the incoming visible light, they are almost identical for the infrared emission. The overall effect is that white paint therefore remains cooler because it absorbs less radiation than black, while emitting just as efficiently. This effect is like two buckets, both having the same large hole in the bottom. One is being continuously filled by a small jet of water and the other by a larger jet. When the levels reach equilibrium, the water level (corresponding to “temperature”) in the bucket filled by the larger jet will be highest. And sure enough, that’s the “black bucket”. Correspondingly, when warmed up by the sun, white paint takes less time than black to reach the point where absorption and emission are balanced. (The black paint has to get hotter before the two balance.) 268 Everyday Physics: Unusual insights into familiar things 68 DOES BABY REALLY NEED A HOT WATER BOTTLE? People who use a lot of matches notice a problem with big economypack matchboxes: the striking surface often wears out too quickly so you can’t use the rest of the matches. Why does this happen: on a bigger box, isn’t the striking surface bigger too? (Don’t worry, this really does have something to do with Baby’s hot water bottle!) The secret is in the difference between volume and surface. The number of matches in the box is proportional to the volume Everyday Physics: Unusual insights into familiar things 269 (and in all your boxes the matches are the same size). Twice as big a box means twice the length, twice the width, twice the height, i.e. 2 × 2 × 2 = 8 times the volume. So your big box contains eight times as many matches as the small one. Now consider the striking surface, or two striking surfaces if there’s one on each side. Their area has become twice as long and twice as wide, therefore 4 times as large. However, you have 8 times as many matches, so you need the striking surface to last twice as long, which sometimes it doesn’t. This is just one example of the importance of the area/volume ratio when comparing objects that are similar but of different sizes. For example, this ratio explains why animals don’t become much bigger than they are. Elephants’ legs already have to be very thick to support so much weight. Making an elephant 2 times as big would mean that the cross-sectional area of its legs would be 4 times bigger, whereas its weight would be 8 times bigger; so mega-elephants’ legs wouldn’t be able to support this double load per unit area. In contrast, a ladybird has extremely thin legs for a relatively stout body; as beasties become smaller their weight decreases disproportionally rapidly, so skinny legs easily support them. Similarly, an ant can carry eight times its own weight (but don’t expect this from an elephant!). The same area/volume argument applies to aircraft too. Simply doubling all dimensions of a Boeing 747 would result in an aircraft that couldn’t fly: the 4-times-greater-area wings would be too small to carry the 8-times-greater weight. Now back to Baby. For people, the area/volume ratio is really important. We’ll continue to assume that when a child becomes twice as big, every part of their body becomes twice as big. So then their surface area is 4 times as big but their weight is 8 times as big. Adults therefore have a relatively large body volume with respect to their surface area, whereas Baby has a large surface area with respect to volume. Baby therefore cools down faster than an adult, because the amount of cooling is determined by the surface area, whereas the heat content (or heat production) is determined by the volume. Conclusion: when it’s cold, wrap up Baby warmly. 270 Everyday Physics: Unusual insights into familiar things 69 THE LID ON THE SAUCEPAN The science section of a newspaper posed the following problem: when you boil water in a saucepan (or cook something in boiling water), you think that by putting the lid on the pan you’re reducing the amount of moisture that’s evaporating into your house. However, when the editor tested this by experiment, they found it made no difference. A fixed quantity of water evaporated, whether the lid was on or not. This seems strange: with the lid on the pan a large part of the water vapour condenses inside the lid and runs back into the pan. Nevertheless the net evaporation is the same as without the lid! Everyday Physics: Unusual insights into familiar things 271 The law of conservation of energy explains this. What exactly happens in a pan full of boiling water? ► Your gas burner or electric ring provides a fixed quantity of heat per second to the pan. ► Once the water has been heated to boiling point, exactly the same amount of heat must be removed (i.e. go somewhere else) because as long as the water is boiling, the temperature remains constant at 100°C. (This wouldn’t be true if the pan contained ball bearings or iron filings. In that case, the pan and contents would continue to absorb heat, and the temperature would continue to rise.) ► A small part of that heat is released by the pan to the environment by means of conduction and radiation. We call that the heat losses. The rest of the heat – most of it, in fact – is used to evaporate water. When you put the lid on the pan, you don’t change the conduction and the radiation, because they are determined by the temperature of the pan, which remains constant (at 100°C). So the quantity of water that evaporates must remain constant, too. (It is even probable that the shiny lid reduces radiation heat losses, so that more heat remains and evaporation increases!) The only thing changed by the lid is that the water vapour escapes with some force between lid and pan. But that doesn’t change the quantity of vapour. Now, all this seems to contradict common experience: everyone “knows” that it’s more economical and reduces the dampness in the kitchen if you keep the lid on the pan when you boil water. So what’s the explanation? As you’ve probably guessed, it’s because the above description isn’t complete: almost intuitively, you lower the gas burner or cooking ring to the point where the water just barely boils. Then the cooker is supplying only the energy to compensate for the conduction and radiation heat losses, and hardly any for evaporation. Without a lid, there would be a lot of extra evaporation because the air circulation around the pan blows the vapour away and more heat is required to replace it. (This is why people put a saucer on the top of cup of hot coffee that they want to keep warm.) 272 Everyday Physics: Unusual insights into familiar things 70 WHY DOES THE AIR GET SO DRY IN WINTER? The air is dry when it freezes outside, which you might notice because newspaper rustles, and wood is more inclined to crack. How come? In winter does dry air flow into the country from somewhere else? No. Suppose the outside temperature is below freezing, and it rains. The air is full of water droplets; it cannot become more humid; it contains as much water vapour as it can hold. The relative humidity (the amount of water vapour the air contains divided by the amount that it can contain) is practically 100% (see Why are some alpine winds so warm? p5). Now go indoors, where it is 20°C, say. Ultimately, all the inside air has come from outside – whether through cracks, or deliberate ventilation. As the air enters and warms up, the quantity of water vapour in the air remains the same. However, warm air can contain more water vapour than cold air, so the relative humidity goes down: the air becomes drier. What’s surprising is how dry the air has become: it’s much drier than you would expect. The explanation is the steep shape of the vapour pressure curve. (The vapour pressure is the maximum pressure that water vapour can have at a certain temperature; if the pressure were higher the vapour would condense.) The curve (Fig. 1) gives the value of the maximum water vapour pressure at different temperatures. Since we are discussing water vapour in air, we can put it more precisely: the graph gives the maximum contribution from water molecules Everyday Physics: Unusual insights into familiar things 273 Fig. 1: Vapour pressure curve for water, from 0°C-20°C. to the total ambient atmospheric pressure (most of which is due to nitrogen and oxygen). The vertical axis shows the pressure in millibars (or in hectopascals, which are the same). The growth in the curve is nearly exponential: it doubles approximately every 10°C (rising from 6 mbar to 12 mbar in 0°C-10°C, and from 12 to 23 mbar for 10°C-20°C). That means that warming air from 0°C to 10°C allows it to hold twice as much water vapour, and at 20°C it can hold about four times as much as at 0°C. So even if air at 0°C is saturated with water vapour, when you heat it to 20°C the relative humidity can’t exceed 25%, which means that the air at 20°C is quite dry. That’s why the interior air is very dry when the outside temperature is very low. Dry air in winter is not due to the relative humidity of the air outside; it’s because it is cold outside. (Of course, you can accidentally or deliberately make your interior air moister. In some “passive houses” the ventilation system always leaves the air very dry, so some owners leave their washing to dry inside the house to moisten the air.) Fig. 1 also shows how easily water condenses on cold surfaces if the interior air is not very dry. Suppose the inside temperature is 20°C and the relative humidity is 50%; that means the water vapour pressure is 50% of 23 mbar = 11.5 mbar. (Point A on Fig. 1 shows where the 23 mbar – the maximum possible vapour pressure at 20°C – comes from.) Assume also that you have a window, or the inside surface of a poorly insulated outside wall, at 10°C. When the 20°C air cools down to 10°C, its water vapour pressure is still 11.5 mbar, but now the maximum possible vapour pressure is 12 mbar (point B on Fig. 1), so the relative humidity is 11.5⁄12 = 96%. If you have a small source of extra moisture, the level reaches 100% and you’ll get misty windows and mouldy walls; to avoid this you need some ventilation to remove the extra moisture. Fig. 2 shows the vapour pressure curve for a larger temperature range. At 100°C the water vapour pressure reaches 1,000 mbar (actually 1,013 mbar) which is normal atmospheric pressure, which is why the vapour from water boiling in a pan can lift the lid off. Just below freezing, the steep trajectory of the vapour pressure curve continues (Fig. 3). The bottom left of Fig. 3 explains why it hardly ever snows in Siberia in the middle of winter. It’s not because it’s too warm but because it is too cold: below −30°C there just isn’t enough water vapour in the air to make snow. 274 Everyday Physics: Unusual insights into familiar things Fig. 2: Vapour pressure curve for water, from 0°C-100°C. Fig. 3: Vapour pressure curve for water, from −30°C to +20°C. Everyday Physics: Unusual insights into familiar things 275 If the air is too dry… If the air is too dry, you can bring extra moisture into your room. However, it’s not a oneoff operation: because ventilation is continuous, you must add water vapour continuously too – it’s like fighting a losing battle. For a typical level of ventilation that’s not so easy: a water container hung on a radiator isn’t very effective unless you find it evaporates many litres of water per day. It’s easy to estimate how much water you must evaporate to raise the relative humidity in a room of volume 100 m3 (3,500 ft3), say. First you need to know how many m3 (or ft3) of water vapour you get by evaporating one litre (a quarter gallon) of water. When you evaporate a litre of any liquid, you always get about 1 m3 (35 ft3) of vapour. (The precise number depends on the liquid; it varies from about 0.7 m3 to 1.3 m3, but for simplicity we’ll stick with 1 m3.) Suppose you want to raise very dry air of, say, 10% relative humidity to 60% – an increase of 50 percentage points. That means you have to add half the maximum amount of vapour that the air can contain. Point A on Fig. 1 shows that at 20°C that maximum amount is 23 mbar, so you have to add half of that, i.e. 11.5 mbar, or 10 mbar in round numbers. The atmospheric pressure is about 1,000 mbar. You’re adding 10 mbar of vapour, so you’re replacing 1% of the air molecules in the room with water molecules. The volume of your room is 100 m3 so you need 1 m3 of water vapour, and we saw above we get that from one litre of water. But that only humidifies the air that’s in the room now. A typical level of ventilation is one “air change per hour”, which means you’ll need one litre of water per hour – about 2 bucketfuls per 24 hours. In practice, considerably less humidification will make the room more comfortable, but don’t expect a miracle from a bowl of water on the radiator. 276 Everyday Physics: Unusual insights into familiar things 71 WHY DON’T YOU DIE OF HEAT IN THE SAUNA? The air in a sauna is extremely dry, due to the heat. If the external air is at 0°C, there is so little water vapour in the air that when this air enters the sauna and is raised to 90°C, the relative humidity (RH) is only about 1% (see box). Even if the air that feeds the sauna comes in at 20°C, the RH in the sauna will hardly reach 3% – unless somebody does something deliberately to raise the humidity. Everyday Physics: Unusual insights into familiar things 277 <box> Relative Relative humidity in the saunahumidity in the sauna Assume that the coming the sauna at 0°C and is Assume that the air coming into the sauna is atair0°C and is into saturated with is moisture. Point saturated with moisture. Point C in Fig. 1 shows that the C in Fig. 1 shows that the water vapour pressure is at most 6 mbar. When that air is then water vapour pressure is atthe most 6 mbar. that the air heated to 90°C in the sauna, the vapour pressure remains same, 6 mbar.When However, is then heated to 90°C in the sauna, the vapour pressure maximum water vapour pressure at 90°C is about 700 mbar (D in Fig. 1), so the relative humidity = 6 ÷ 700 ≈ 1%. remains the same, 6 mbar. However, the maximum water vapour pressure at 90°C is about 700 mbar (D in Fig. 1), so Fig. 1: Point C shows the maximum water vapour pressure (6 mbar) at 0°C, and point D (700 mbar) for 90°C. How warm the sauna really is becomes clear if you swing your arms back and forth. Normally that would cool them down but it doesn’t: they get warmer, especially if they are still dry. Even though the wooden benches are at 90°C, you can sit on them comfortably because of the poor heat conduction of wood. (This poor heat conduction makes the heat-flow from the benches to your body very small; the slow heat-flow means that your body can easily dissipate the small amount of heat it receives. In contrast, if the benches were metal, you’d get burnt, nearly as badly as with a pot of boiling water.) 278 Everyday Physics: Unusual insights into familiar things If you touch the glass of the door, it feels so hot it almost hurts, although its temperature is much less than 90°C (see Fig. 1a, Is thick glass a better insulator than thin glass? p251). In spite of the 90°C heat, you can stand the sauna for quite a while, for two reasons. First and most importantly, the layer of air very close to your skin is insulating (see How do you keep your temperature constant, p49). Second, because it is so dry in the sauna, your sweat readily evaporates and cools you. Even so, the evaporation can’t stop you gradually getting hotter: the heat flux into your body is too big for that. It amounts to about 1.5 kW, as much as an electric heater produces! To cool that off by evaporation, you’d have to evaporate two litres per hour (see box), and your body can’t keep pace with that. When someone throws water on the coals in the sauna, you feel a brief surge of heat. This has a number of causes. First, hot steam at 100°C is produced, and that’s higher than the ordinary sauna’s normal temperature. Next, that steam moves the air around, sweeping away the insulating layer and bringing the heat closer to your skin; this is especially pronounced when the skin is still dry so that evaporation isn’t cooling you. Moreover, this newly formed steam moistens the air and reduces the cooling effect of evaporation. Everyday Physics: Unusual insights into familiar things 279 And water vapour conducts heat better than air, so the now-moist air layer near the skin transports more heat to your body. Finally, there is another surprising factor: the latent heat released in the condensation of water vapour on to the skin is probably the most important contribution of this heating effect when you throw water on the coals. The reason is that your skin is the coldest place in the whole sauna, and the humidity can easily reach 100% near the skin, so condensation is inevitable, and the latent heat of condensation is large. In the sauna In the sauna your body has to cope with a lot of conducted heat and radiated heat. The conduction contribution can be written in terms of your surface area A, the heat conductivity of air λ, the temperature difference between the sauna air and your skin ΔT, and the insulating air layer d around your skin. The conducted contribution then is: A λ × ΔT / d. Substituting A = 1.7 m2, λ = 0.025 W⋅m-1⋅K-1, ΔT = 50 K and d ≈ 3 mm yields 700 watts. The radiated contribution is A σ (T4sauna − T4skin). Substituting A = 1.7 m2, σ = 5.7 × 10 W⋅m-2⋅K-4, Tsauna = 363 K and Tskin = 310 K, we get 790 watts. So the calculation shows that in the sauna conduction and radiation are about equally important, as they are under normal circumstances. However, in the sauna the heat-flow goes the other way, i.e. you’re taking in heat, not losing it. Moreover, the heat-flows are much larger in the sauna, because of the large temperature differences and the fact that in the sauna you are appropriately dressed (well, appropriately undressed) so there are no insulating clothes to reduce the heat-flow. -8 Thus in total your body takes in about 1,500 watts, which you have to get rid of by evaporation. The “latent heat of evaporation”, i.e. the heat needed to change liquid water to vapour at the same temperature, is 2.4 kJ per gram at body temperature. To get rid of 1,500 W, the quantity of water to be evaporated per second is 1,500 W / 2.4 kJ per gram = 0.62 gram per second = 0.62 ml/s = 0.62 ml × 3,600 seconds/hour = 2.2 litres (half a gallon) per hour. In practice it will be somewhat less because some heat goes to raising your body temperature a little bit, especially at the start when you have just entered the sauna. 280 Everyday Physics: Unusual insights into familiar things 72 THE WINE-MIXING PROBLEM This is a typical science quiz problem. You have two identical glasses, one containing red wine and the other white. Take a spoonful of wine from the red glass and put it into the white and stir, making it a little rosé. Now take an exactly equal spoonful of this rosé and put it back into the red. Which of the two wines is now most “contaminated” by the other? To most people the answer isn’t obvious. The spoon of red that went into the white was, after all, pure red, while the spoon of white that went into the red was already a bit rosé, so isn’t the spoon of pure red “more contaminating”? On the other hand, that second spoon of rosé went into a glass that was less full, so it might have had more of an effect! Which effect wins? Everyday Physics: Unusual insights into familiar things 281 The answer is that each glass is just as contaminated as the other. You can see why this is so in several different ways. The first way is to forget the details and just look at the end result instead. You haven’t destroyed or created any wine, so you have exactly the same total amount as you started with. Any red wine not in the white glass must therefore be in the red glass; and as the white glass contains the same volume now as at the start, any red wine in it has replaced the same amount of white wine, which must now be in the red glass. So both glasses have exactly the same level of “contamination”. A second, more abstract, way of addressing the problem is to consider the case where you have 100 red balls in one glass and 100 white balls in the other. Move a scoop of red balls to the white glass, and move a scoop of red/white mix back to the red glass. If there are now 2 white balls in the red glass, there must be 98 red balls there (because it’s full) and the only place the other 2 red balls can be is in the other glass. This also makes it very clear that it doesn’t matter whether you thoroughly mix the red and white balls (wine) before transferring between the glasses: it’s always true that any red balls not in the red glass are in the white, and vice versa, whatever the concentrations are. The third and final way is to calculate exactly what is transferred from one glass to the other. While the calculation isn’t complicated, it’s long-winded because of all the fractions, so we’ve shown it only in an appendix (p296). 282 Everyday Physics: Unusual insights into familiar things 73 THE WANDERING TEA LEAVES Stir your cup of tea, stop stirring, and then what do you see? When the liquid has stopped moving, the tea leaves have gathered in the middle at the bottom, but wouldn’t you have expected the centrifugal force to push the heavier-than-water leaves or sugar to the outside? Everyday Physics: Unusual insights into familiar things 283 Centrifugal force does of course play a role; the circulating liquid does want to go to the outside. However, the centrifugal force is not equally strong at every height (because the circulation speed is not the same everywhere), and friction also plays a part: ► At the top, when the tea is stirred it rotates unhindered and the centrifugal force can do its work, so the liquid moves from the centre towards the edge. ► Near the bottom things are different. The rotation speed is smaller and therefore the centrifugal force is less. This is because the rotating tea is hindered by friction against the cup’s bottom. The result is that something unexpected occurs. At the top the tea is pressed towards the outside with a lot of force; at the bottom the tea is pressed outwards with a smaller force. So the larger force at the top “wins” and the tea at the top flows towards the outside. From there it has to go down along the outside, and it comes in again towards the middle at the bottom where the centrifugal force is lowest. So overall, in addition to a rotating motion in the horizontal plane, there is also a circulation pattern in the vertical cross-section of the cup (Fig. 1). Fig. 1: In the teacup, the centrifugal force is greater at the top than at the bottom – due to friction at the bottom – so the tea circulates as shown, and the tea leaves collect in the middle at the bottom. 284 Everyday Physics: Unusual insights into familiar things The circulating tea takes the tea leaves with it – towards the outside at the top, then down the walls, and back into the middle at the bottom. When the flow speed is high, the leaves keep on circulating, but when the flow reduces and is no longer strong enough to lift the leaves back to the top, they are swept into a nice pile in the middle. Thus stirring does eventually bring some order into chaos. Everyday Physics: Unusual insights into familiar things 285 ACKNOWLEDGEMENTS It is a pleasure to thank all those who reviewed the manuscript for accuracy or comprehensibility and those who contributed in other ways to the realization of this book: Ellen Backus, Rinus Boone, Barry Cats, Daniëlle Duijn, Loek Eenens, Huub Eggen, Eric Eliel, Martin van Exter, Jan, Ineke and Annette Heijn, Hanneke, Reinoud and Francine Hermans, Marjon de Hond, Gert ’t Hooft, Marieke Huijvenaar, Vincent Icke, Frank Israel, Stuart Johnson, Jan de Laat, Henriette van Leeuwen, Jan Lub, Tjerk Oosterkamp, Gijs Pellinkhof, Henk and Linda Petersen, Ellie van Rijsewijk, Edith van Ruitenbeek, Jean Schleipen, Caesar Sterk, Fokke and Free Tuinstra, and the enthusiastic audience at my “Everyday physics” lectures for the University of the Third Age (HOVO). Thanks to Professor Fokke Tuinstra, Delft University of Technology, for material on bike side winds and on bike-wheel splashing. Jo Hermans 286 Everyday Physics: Unusual insights into familiar things HANDY REFERENCE DATA The Earth Circumference: 40,000 km Atmospheric pressure at sea level: on average 1.013 bar (1 bar = 105 pascal) or about “1 kilogram per cm2” Density of the air at 20°C: 1.2 kg/m3 Earth’s atmosphere: the effective thickness is approx. 8 km. The atmospheric pressure falls with height approximately according to the formula e-h/8 km; it halves roughly each 5.5 km. Around the Earth Orbit time of “low” satellites (just outside the Earth’s atmosphere): approx. 90 minutes Orbit time of GPS-satellites: 12 hours (at a height of 20,000 km) Orbit time of a satellite in geostationary orbit, 35,786 km above sea level and directly above the equator: 24 hours Orbit time of the moon: 27.3 days (at a height of approx. 380,000 km) The moon Average distance to the Earth: approx. 380,000 km Diameter: approx. 3,500 km Apparent diameter in degrees: ½ degree The sun Average distance to the Earth approx. 150 million km At the beginning of July the sun is a good 3% further away than at the beginning of January Everyday Physics: Unusual insights into familiar things 287 Diameter: 1.4 million km Apparent diameter in degrees: ½ degree Mass: 2 × 1030 kg Temperature at the surface: approx. 5,750 K or approx. 5,500°C Emitted energy flow: 3.8 × 1026 watts Emitted energy flow, translated into equivalent loss of mass: 4 billion (4 × 109) kg per second Solar energy reaching the Earth Just outside the Earth’s atmosphere: approx. 1,350 W/m2 (measured perpendicularly to the sun rays). At the Earth’s surface: approx. 1,000 W/m2 (measured perpendicularly to the sun rays). In the northern United States and in central Europe, the average on a horizontal surface, over the entire year and 24 hours per day, is typically 150 W/m2. Energy (For the definitions of energy, work, heat and power see The human engine p43) Energy content of: ► Oil, fat: approx. 40 MJ/kg ► Petrol/gasoline: 48 MJ/kg or 35 MJ/litre ► Natural gas: 35 MJ/m3 (higher value) or 32 MJ/m3 (lower value) (The “higher value” includes the condensation heat of the water produced in the chemical reaction of combustion. This water is in the vapour phase, so by condensing it out of the exhaust gases, which is what a “condensing boiler” does, we get extra useful heat.) 288 Everyday Physics: Unusual insights into familiar things Light Speed of light in a vacuum: 299,792 km/s, i.e. about 300,000 km/s or 300,000,000 (3 × 108) m/s Speed of light in air is only 0.03% less Speed of light in water: 225,000 km/s Sound Speed of sound in air (15°C) : 340 m/s or 1,200 km/h Speed of sound in water (15°C): 1,470 m/s or 5,300 km/h Speed of sound in a vacuum: in a vacuum there is no sound! Prefixes multipliers kilo k 103 thousand mega M 106 million giga G 109 billion tera T 1012 trillion peta P 1015 quadrillion dividers milli m 10-3 thousandth micro μ 10-6 millionth nano n 10-9 billionth pico p 10-12 trillionth femto d 10-15 quadrillionth Everyday Physics: Unusual insights into familiar things 289 Rules of thumb Speeds To convert miles or kilometres per hour, to metres/second: 100 mph = 44.7 m/s ≈ 50 m/s, i.e. 2 mph ≈ 1 m/s 2 miles ≈ 3 km, so 3 km/h ≈ 1 m/s ► I cycle at about 5 m/s, which is about 10 mph. ► The New Horizon satellite is travelling at 36,000 mph, or about 18,000 m/s = 18 km/s. For comparison, a rifle bullet travels at about 1 km/s and weighs about 10 g, so if New Horizon bumps into a 10 g piece of space debris, it’s like being shot with a very very fast bullet. Rain A rainfall of 1 mm is the same as 1 litre of rain per square metre. The advantage of the litres/m2 expression is that it’s easy to see what volume of water a property will have to handle. For example, if the area of your house and garden is 500 m2, and you have a rainfall of 8 mm, then the volume of water that has to flow away is 4,000 litres, which is 4 cubic metres. 290 Everyday Physics: Unusual insights into familiar things Circles and spheres How many litres of water do you need to fill a spherical goldfishbowl? (Not that it’s advisable to keep goldfish in that sort of bowl.) How many square metres is a round plot, so you can work out how much grass seed to buy? While it’s handy to know the proper formulae for circles and spheres by heart, it’s not essential. In everyday life when you don’t have to be very precise, remembering a few simple images is enough. Take the volume of a sphere. Imagine a cubic box that fits the sphere exactly. What fraction of the cube does the sphere occupy (Fig. 1)? Fig. 1: Estimating the volume of a sphere by fitting it within a cube. At the centre points of the six sides of the cube, the cube and sphere coincide, there is no space in between. However, in each of the eight corners, there are fairly large parts of the cube that the sphere doesn’t touch. At a guess, if we add up the volume of these eight pieces, they might be half the volume of the cube. Everyday Physics: Unusual insights into familiar things 291 Would that be a good guess? Look at the formula for a moment. The volume of a sphere of radius r is 4⁄3 π r3. The value of π is 3.14, which we’ll round down to 3 as we’re not being too precise, so the volume is about 4⁄3 × 3 × r3 = 4 r3. Now for the cube: each side is 2 r long, so the volume is (2 r)3, i.e. 8 r3, which is double our approximation for the sphere. Conclusion: the volume of a sphere is about half the volume of its surrounding cube, to within a few per cent, because the inaccuracy of our calculation was small – rounding 3.14 to 3.00, an error of 14 in 300 or about 5%. Fig. 2: Estimating the area of a circle by fitting it within a square. The top-left quarter (marked with blue dashed lines) is highlighted in Fig. 3. Fig. 3: The top-left quarter of the circle of Fig. 2. You can use the same approach for the area of a circle. Draw a square around it that fits it exactly (Fig. 2). Clearly the circle doesn’t cover the whole square, but it does cover much more than half, because in Fig. 3 the triangle marked (A) is half the area, and area (B) looks about the same size as area (C), so ¾ would be a good guess. Check this using the formula: the area of a circle is π r2, or approximately 3 r2. The area of the surrounding square is (2 r)2 or 4 r2. So our guess above of ¾ is good to within a few per cent. Easiest of all is the circumference of a circle. Fig. 4 shows that the circumference of the circle is not much longer than the circumference of the hexagon that fits inside exactly. The six triangles are all equilateral (because the vertices are 360 ÷ 6 = 60°), so the sides are equal to the radius. Therefore, the circumference of the hexagon is 6 r and the circumference of the circle is about the same – six times the radius (or three times the diameter), again to within a few per cent. If you have to be really precise and need the value of π to many decimal places, use the mnemonic aid: “How I want a drink, alcoholic of course, after the heavy lectures involving quantum mechanics.” Count the numbers of letters in each word, which gives you 3.14159265358979. Or if you only want the decimal digits after the initial “3.”: Fig. 4: Estimating the circumference of a circle using a hexagon. 292 I wish I could determine pi. Eureka, cried the great inventor. Christmas pudding, Christmas pie, Is the problem’s very centre. Everyday Physics: Unusual insights into familiar things Pi Here’s π to a thousand decimal digits: 3.14159 26535 89793 23846 26433 83279 50288 41971 69399 37510 58209 74944 59230 78164 06286 20899 86280 34825 34211 70679 82148 08651 32823 06647 09384 46095 50582 23172 53594 08128 48111 74502 84102 70193 85211 05559 64462 29489 54930 38196 44288 10975 66593 34461 28475 64823 37867 83165 27120 19091 45648 56692 34603 48610 45432 66482 13393 60726 02491 41273 72458 70066 06315 58817 48815 20920 96282 92540 91715 36436 78925 90360 01133 05305 48820 46652 13841 46951 94151 16094 33057 27036 57595 91953 09218 61173 81932 61179 31051 18548 07446 23799 62749 56735 18857 52724 89122 79381 83011 94912 98336 73362 44065 66430 86021 39494 63952 24737 19070 21798 60943 70277 05392 17176 29317 67523 84674 81846 76694 05132 00056 81271 45263 56082 77857 71342 75778 96091 73637 17872 14684 49010 22495 34301 46549 58537 10507 92279 68925 89235 42019 95611 21290 21960 86403 44181 59813 62977 47713 09960 51870 72113 49999 99837 29780 49951 05973 17328 16096 31859 50244 59455 34690 83026 42522 30825 33446 85035 26193 11881 71010 00313 78387 52886 58753 32083 81420 61717 76691 47303 59825 34904 28755 46873 11595 62863 88235 37875 93751 95778 18577 80532 17122 68066 13001 92787 66111 95909 21642 01989 The truth about exponential growth Just about everyone can misjudge the long-term effect of exponential growth. Exponential growth is growth where a fixed percentage is added every time period. So compound interest of 5% per year gives exponential growth. Inflation of 322% per month, as there was in the 1923 hyperinflation in Germany, is also exponential growth. Test yourself: you have two seconds to answer the following question. Which would you prefer, $50,000 a day for the next month, or ¢1 today, ¢2 tomorrow, ¢4 the day after, ¢8… for the next month? Everyday Physics: Unusual insights into familiar things 293 Now give yourself 20 seconds, to estimate (without a calculator) how much your ¢1 will grow to after a month. (This is just a variant of the traditional story of the grains on the chessboard. Before a game of chess, a king asked a sage to name his own reward; the sage asked for 1 grain of rice on the 1st square, 2 grains on the 2nd, 4 grains on the 3rd, 8 grains on the 4th, etc.) The answer to what your ¢1 becomes is – assuming 29 days of doubling – $5.4 million. (The royalty-bankrupting rice amount is 1.8 × 1019 grains, or 18 billion billion!) The above is about rapid doubling, but even growth with a fixed small percentage can be treacherous. In 1626 Peter Minuit bought Manhattan for $24. The current estimated value of Manhattan is about $250 billion, that is about ten billion times as much. Surely such a fantastic investment was much better than the optimistic 7% per year that the stock market yields over the long term? To see if that’s correct, calculate how much the stock market would have yielded. You can use two simple tricks to calculate the answer quickly. The first trick is “the rule of 70”. This is based on the fact that money invested at 1% compound interest does not need 100 years to double; because of the effect of compounding, it needs only 70 years. The rule is: The doubling time is equal to the number 70 divided by the growth percentage. So, at a rate of 2% your investment doubles in 70 ÷ 2 = 35 years, and at 7% it doubles in 10 years. The rule gives a very good approximation (although it goes wrong for large percentages). Accordingly, you can regard every exponential growth as a process of doubling. The mathematical background here is that the natural logarithm of the number 2 is approximately 0.70, i.e. ln 2 (or loge 2) = 0.693. The second trick relies on the fact that when you repeatedly double your investment, the amount increases according to the series 2, 4, 8, 16, 32. 64, 128, 256, 512, 1024… so after 10 doublings, your investment has increased by a factor of 1,024, giving us our second rule of thumb: Ten doublings increase your investment by a factor of 1,000. These two simple rules make it easy to estimate the consequences of a process of exponential growth. Look at Peter Minuit’s investment in Manhattan as an example. If he had invested his money at 7% compound per year: 294 Everyday Physics: Unusual insights into familiar things 1. According to the “rule of 70” his money would have doubled every ten years. In 380 years that amounts to 38 doublings. 2. According to the second rule, the first 30 doublings yield three factors of 1,000, i.e. 1,000 × 1,000 × 1,000, that is a factor one billion (1 bn). That still leaves 8 doublings, which give a further factor of 256, giving a total factor of 256 bn, about 25 times better than the 10 bn factor actually achieved, i.e. $24 × 256 bn = $6,144 bn instead of the actual $250 bn. So poor old Peter Minuit didn’t make the best investment decision: he ought to have shopped around for a fund with a yield of 7% compounded per annum, and invested his $24 there instead. Here’s an even more surprising example – world population. Around 1970 the population was growing at a little less than 2% per year. While that doesn’t sound very much, you can see what its long-term effect will be, using a thought experiment. Suppose that the world started very modestly 2,000 years ago, with only two people – maybe Adam and Eve at the time of Christ, to jumble our biblical history a bit. And suppose the growth since then has been 2% per annum. Would the population be much lower than the current 8 bn, or about the same, or even higher? Let’s use our rules to make a quick estimate. The “rule of 70” says 2% per annum yields a doubling every 35 years, or about three doublings per century, which is 60 doublings in 20 centuries. Our second rule says 60 doublings give us 60/10 = 6 factors of 1,000, one after the other, or 1018, i.e. one billion billion! Even though a precise calculation gives something slightly less – 3 × 1017 people, starting with two people in the year zero – that’s still 50,000,000 times as much as the actual population. Clearly the world population actually grew very much less than 2% per annum over the centuries. Until the year 1600 the growth was probably lower than 0.1% per annum. However, the most important conclusion from this example is that exponential growth, even with a modest percentage, in the long term leads to impossible situations. For example, if the world population really did reach 3 × 1017 we’d have 2,000 people per square metre (or only 600/m2 if we spread them over the oceans too). The “rule of 70” gives a nice insight into what happens within a human lifetime which, for simplicity, we’ll say is 70 years. Each 1% growth of anything then means a doubling in “a lifetime”, and the growth percentage gives the number of doublings. E.g. if the volume of rubbish incinerated goes up by 5% per year, then our grandchildren will need 25 = 32 times as many incinerators as we have today. In brief: while we regard growth as indispensable, long-term growth at a constant compound rate is impossible. Everyday Physics: Unusual insights into familiar things 295 APPENDIX: THE WINE-MIXING PROBLEM – CALCULATION This appendix shows the third – detailed calculation – way to see that the red and white wine glasses in The wine-mixing problem (p281) are equally contaminated. For simplicity assume that each glass contains 100 ml of wine and that your spoon contains 7 ml. (In fact it doesn’t matter what size the spoon is, as you’ll see.) The table below shows the number of millilitres in the white (W) and red (R) glasses respectively, at each stage of the process. WHITE GLASS RED GLASS W R R W Initial situation 100 0 100 0 After one spoon of red added to white: 100 7 93 0 At this point the white glass contains 107 ml, of which 100 are white and 7 are red. So in the white glass: ► The W concentration is 100:107, i.e. 1 ml of this rosé contains 100/107 ml of white, and a 7 ml spoon contains 7 × (100/107) = 700/107 ml of white. ► The R concentration is 7:107 i.e. 1 ml of this rosé contains 7/107 ml of white, and a 7 ml spoon contains 7 × (7/107) = 49/107 ml of red. These white-glass concentrations remain unchanged because you don’t dilute the white glass any further. Now use these concentrations to see how much red and white are transferred to the red glass. To keep it simple, just look at the contamination in both glasses: in either glass there is 700/107 ml of the “wrong” kind. QED. 296 Everyday Physics: Unusual insights into familiar things WHITE GLASS W Remove one spoon of “white” (actually rosé) from the white glass 100 − 7 × (100/107) = 100 – (700/107) And add it into the red glass: (unchanged) 100 – (700/107) R RED GLASS R W 7 – (49/107) = 700/107 93 (unchanged) 0 (unchanged) (unchanged) = 700/107 93 + (49/107) = 100 – (700/107) 0 + (700/107) = 700/107 Everyday Physics: Unusual insights into familiar things 297 TWO MORE EXPERIMENTS Home experiment: Twin divers Here’s a trick that was popular at children’s parties in the past. The “magician” holds in her hand a sealed, flat-sided glass bottle filled with water, containing a primitive model diver that can be sent to the bottom of the bottle on command, and back again on command. What the children see is no magic trick but the stealthy use of the laws of physics. The trick is easier to demonstrate with a plastic water bottle. The “diver” consists of a vertical glass tube open at the bottom only; an eye- or nose-dropper from the drugstore complete with squeezer works very well. Fill the bottle nearly to the brim with water, and suck enough water into the diver so that it just barely floats in the bottle. Cap the bottle, and you’re in business. Squeeze the bottle: the pressure increases, more water enters the diver and it sinks to the bottom! Squeeze less hard and it rises to the surface again. (To do this with a cylindrical glass bottle is impossible, but you can do it with a flat-sided glass bottle, and it’s much more impressive than with a plastic bottle, although it requires a fairly strong hand! The alternative is to seal the top of the bottle with a piece of balloon and discreetly put your thumb on it to increase the pressure.) You can make a game out of this by using two divers in the same bottle, each with a hook of bent copper wire, one facing up and one facing down (Fig. 1). Fill the down-hook diver as before, but fill the up-hook diver with enough water so that it just hovers on the bottom of the bottle. The challenge is to rescue the lower diver with the upper one. It’s not so easy, so the child who manages it is rewarded with the biggest piece of birthday cake, and later goes on to study physics. 298 Fig. 1: Rescuing a “diver” in a bottle by manipulating the pressure. Everyday Physics: Unusual insights into familiar things S Home experiment: Super simple electric motor You can build an electric motor in five minutes! The only part you need that you wouldn’t find around the house is a small super-magnet in the form of a flat disk or – easier still – of a sphere of about 2 cm diameter. You can buy these cheaply on the internet – search for “super magnet”. Any handyman probably has the others parts: a short (< 25 mm) flat-headed steel screw, an AA or AAA battery, and a piece of copper wire with bare ends. Fig. 1 shows how to assemble your motor. (It also shows the field lines of the magnet.) The magnet sticks on to the screw, and if the screw isn’t too long, the point of the screw sticks to the bottom (the negative terminal) of the battery. Fig. 1: How to connect the components of your motor, showing the magnetic field from the spherical magnet. With the battery in one hand and the wire in the other, close the circuit by holding one bare end of the wire against the top of the battery (the positive terminal) and the other bare end loosely against the side of the magnet as a kind of sliding contact. Now the screw and magnet start to rotate like mad. Friction is minimal, thanks to the pointed screw and the smooth bottom of the battery. But why does the magnet rotate? It is because of the Lorentz force, a force that works perpendicular to a current in a magnetic field. The magnetic field is vertical in this case and the sliding contact ensures the horizontal current (shown by the white arrow in Fig. 2). Fig. 2: The current flows horizontally from the lower end of the wire into the spherical magnet. With the magnetic field directed vertically, the Lorentz force acts perpendicular to both and makes the sphere turn. Everyday Physics: Unusual insights into familiar things 299 RESOURCES ► Animated image of GPS satellites orbiting the Earth: https://commons.wikimedia.org/wiki/File:GPS24goldenSML.gif ► Camera obscuras that you can visit: Public access: Camera obscuras open to the public https://en.wikipedia.org/wiki/Camera_obscura#Public_access ► How to make a spectrometer using an old CD: Papercraft Spectrometer Intro Kit https://publiclab.org/wiki/papercraft-spectrometer ► The Magnus effect: ■ Video of rolled paper cylinder: https://upload.wikimedia.org/wikipedia/commons/e/e1/08._Цилиндар_што_паѓа_и_ ротира.ogv (or search Wikipedia for “Magnus effect”, and click on the relevant video on the right-hand side). ■ Video of a home-made Flettner rotor-powered toy car: Flettner Rotor Sail Cart // Homemade Science with Bruce Yeany https://www.youtube.com/watch?v=XsGin7CFaF8 ■ Video: What Happens When a Spinning Basketball is Thrown Off a Dam! https://www.youtube.com/watch?v=QtP_bh2lMXc ■ The bouncing bomb: Article: https://www.smithsonianmag.com/history/how-british-engineer-made-bombthat-could-bounce-on-water-180969095/ Video: Dambusters | Building the Bouncing Bomb | 2 of 2 https://www.youtube.com/watch?v=8IeGYkwVIWw ► Crookes radiometer video: https://en.wikipedia.org/wiki/Crookes_radiometer and click on the video in the Thermodynamic explanation section. ► Albert Einstein’s autograph manuscript of his paper on how a radiometer works: Zur Theorie der Radiometerkräfte https://alberteinstein.info/vufind1/Record/EAR000034044 (Click on the image of the document, to read it.) ► Evacuated double glazing description: https://www.pilkington.com/en-gb/uk/products/product-categories/thermal-insulation/ pilkington-spacia 300 Everyday Physics: Unusual insights into familiar things CREDITS ► Jo Hermans: p299. ► Mad-i-Creative: p67*, 69*, 96, 126, 155, 201*, 251, 255, 265* (x2), 281* (x2). ► Pexels: https:/www.pexels.com/ p17, 19, 37, 47, 49 (x2), 59, 83, 89, 112, 113, 119, 161, 169, 180, 181, 189, 191, 195 (x2), 197, 199, 207 (x2), 211, 225, 244, 245, 267. ► Pixabay: https:/www.pixabay.com/ p7, 8, 10, 11, 15, 21, 31, 33, 44, 51, 57, 67, 67*, 69*, 90, 94, 96*, 98, 99, 101 (x2), 103, 115, 117, 119, 126*, 129, 133, 147, 155, 158, 159 (x2), 173 (x2), 185, 191, 205, 209 (x2), 214, 215, 231, 239, 245, 261, 265*, 267, 269 (x2), 287, 288 (x3). ► Pxfuel: https:/www.pxfuel.com/ p21, 33, 99, 105, 107, 199, 203, 220, 221, 265*, 271, 273, 277 (x2), 281* (x2). ► Unsplash: https:/www.unsplash.com/ p0, 1, 5, 13, 19, 25, 27, 29, 32, 50, 53, 55, 59, 60, 63 (x2), 65, 71, 75 (x2), 79, 84, 86, 87 (x2), 93 (x2), 107, 108, 113, 115, 123 (x2), 129, 130, 135 (x2), 137, 139 (x2), 140, 143 (x2), 146, 183, 187 (x2), 201*, 217 (x2), 218, 225, 227, 229 (x2), 239, 251, 261, 271, 283 (x2), 287. ► Wikimedia Commons: https://commons.wikimedia.org/ p37, A man walking. Photogravure after Eadweard Muybridge, 1887. Credit: Wellcome Library, London, CC BY 4.0. p165, Hologram on a 100-Euros bill, Heike Löchel / CC BY-SA 3.0. p205, Circular road, in Třebíč, Czech Republic, Credit: Frettie, CC BY 3.0. p235, Bouncing bomb tranining - IWM FLM. Credit: Official film maker / IWM staff photographers / Public domain. p247, A collection of Crookes radiometers at the Royal Society, London. Credit: The wub, CC BY-SA 4.0. p247, Radiometer Crookes - Oberweißbach-Thüringen. Credit Nieuw~commonswiki, CC BYSA 3.0. p256, 2016-03-05 Ablenkeinheit by DCB. Credit: DCB, Wikimedia Commons, CC BY-SA 3.0. p290, National Weather Service Standard Rain Gauge measuring tube and measuring dip stick in use. Credit: Famartin, CC BY-SA 4.0. * Edited by Mad-i-Creative Everyday Physics: Unusual insights into familiar things 301 INDEX Page numbers in bold refer to boxed text and home experiments; page numbers in italic refer to Figures and captions. A B absorption coefficient 170–1, 265, 268 acceleration 15, 16, 23, 48, 62, 82, 91 aerodynamics 22, 231 air dry see dry air insulating property 252, 255, 279, 280 air density 22, 23, 62, 89, 136, 287 air molecules 108–9, 109, 114, 200, 219, 249–50, 250 air pressure 5, 22, 30, 30, 136, 255, 256, 276, 287 air resistance 15, 17, 20, 22, 23, 59, 60–1, 61, 62, 63, 66, 70, 87, 89–92, 91, 92 air resistance coefficient 60–1 aircraft area/volume ratio 270 energy consumption 263, 263 aluminium foil 265, 266 angular momentum, conservation of 37–8, 38–41, 39, 40, 41, 238 ants 270 area/volume ratio 269–70 atomic clocks 3–4 baby, keeping warm 270 Baden Baden sailing ship 235 balloons 17 inflation 258–60 balls bouncing balls 237–8 bowling balls 38 curve balls 231 superballs 237–8 suspension in hairdryer airflow 235–7 topspin 230–1, 230, 231 Baumgartner, Felix 23 Bell, Alexander Graham 183 Bernoulli’s law 62, 230, 235, 236 bicycle pumps 5 bikes/biking see cycling biomass 243, 244 black paint 265–6, 268 bloodstream 74 blue sea 169–72 blue sky 29, 107–10, 111–12, 133 body temperature 8, 10, 50–4, 280 bouncing bomb 235, 300 Brewster angle 177, 179 302 C caesium atoms 3 calorie 43 Everyday Physics: Unusual insights into familiar things camera obscuras 120, 122, 300 candles energy efficiency 99–100, 106, 263 wax burn per hour 99, 100 carpets, wandering 245–6, 246 cars and driving air resistance (drag) 87, 89–92, 91, 92 braking distance 95, 97 energy consumption 46, 87, 91, 92, 262, 263, 263 engine power 47 “Nuna” cars 242, 242 “puddles” on a dry road 133–4, 133, 134 radiator, frozen 7–8 rolling resistance 87–9, 89, 90–2, 91, 92 traffic density 93–7, 95, 97 tyres 87, 88 cathode ray tubes (CRTs) 256 CDs 159–64, 167–8 centrifugal force 79, 80, 81, 82, 283–5, 284 cigarette smoke, colour of 113–14 circles and spheres 3 area estimation 292, 292 circumference estimation 292, 292 volume estimation 291–2, 291 clocks, atomic 3–4 cloud colours 114 Coefficient of Performance (COP) 228 coffeepot, chromium 265, 266 colours blue sea 169–72 blue sky 29, 107–10, 111–12, 133 CDs 159–64 cloud colours 114 LED colour spectrum emission 106, 111 rainbows 147, 148, 149, 152, 154 setting sun 110, 112, 137–8 soap bubbles 155–8 condensation 6, 271, 274, 280 conduction 24, 50–1, 52, 222, 223, 252, 257, 267, 272, 280 convection 53, 252, 253, 266 curtains insulating property 254 lace curtains 139, 140, 142 sound absorption 208 curve balls 231 cycling air resistance 59, 60–1, 61, 62, 63, 66, 70, 90 back-splashing 79–82, 80, 81, 82 echelon 72, 72 efficiency 55–8, 56, 62, 75 fast 63–6 home trainers 48, 51–2, 53, 200 HPVs 61, 64, 65, 66 minimising journey time 76–8, 76, 77 on the moon 67–8 power 53, 64–5, 64 in the rain 18, 83–6, 85 rolling resistance 59, 60, 61, 62, 67, 68, 90 side winds 69–72, 70, 72 speed records 64, 90 super bikes 61, 62, 63, 68 tail wind 69, 83 Everyday Physics: Unusual insights into familiar things 303 tyres 60, 62 wind chill 7, 9 D daylength, seasonal 34–5, 34 decibel (dB) 183 dipoles 178, 179, 222 Dolby volume 189 double glazing 252, 253, 254, 255–7 evacuated 255–7, 300 secondary glazing 254 drag coefficient 22, 23, 60–1, 62, 90 dry air humidification 276, 279–80 mountain air 5–6, 6 saunas 277–80 winter air 273–4 E ears and hearing age-related hearing loss 196, 196, 198 cocktail party effect 195–8 directional hearing 191–4, 192, 193, 197 hearing glasses 198 hearing threshold 182, 184 intensity difference mechanism 191, 192, 193, 198 lip-reading 196, 198 night-time 199–200, 202 sound sensitivity 182, 182, 184, 185 time difference mechanism 191, 192–3, 197 Earth atmosphere 20, 29, 30, 110, 110, 287 304 axis 25, 25, 26 data 287 orbit 26–7, 28, 36 eggs in the microwave 224 Einstein, Albert 4, 239, 250 electric motors 226, 299 electric shavers 262, 263 electricity 24, 44, 48, 100, 223, 243, 244, 262 electromagnetic fields 108, 109, 222 electromagnetic waves 174, 221–2, 223, 224 elephants 270 emission coefficient 265, 268 energy 288 consumption, feeling 261–2 definition 43 energy consumption comparisons 262–3, 263 human engine 45–8 law of conservation of energy 24, 230, 261, 272 resting energy expenditure 45, 49, 52, 53 work and 43 equator 25, 26, 29 equinoxes 27, 27 evaporation 6, 50, 51–2, 52, 53, 271, 272, 279, 280 exponential growth 293–5 eye cornea 144, 144 lens 123, 124, 125, 126, 144, 144 photoreceptors 100, 123, 131 sensitivity 102, 102, 103, 106, 129–31, 140–1 see also vision Everyday Physics: Unusual insights into familiar things F falling process 15–22 slow fall 24 fats, dietary 46 Fermat’s principle 77–8 figure skating 38 fog drops 19–20 Föhn 5, 6 food, conversion into heat 45 force unit of 43 work and 43, 47 friction 12, 15, 16, 20, 62, 88, 246, 262, 284 fridges 225–8 cooling your home with 227, 227 heat pump action 226, 226, 228, 228 heating your home with 228, 228 G glaciers 172 glass heat conduction 251 insulation properties 251–2 see also windows glasses hearing glasses 198 Polaroid glasses 173–7 reading glasses 124, 125 GPS (Global Positioning System) 1–4 gravity 14, 15, 19, 20, 22, 23, 48, 57, 62, 80 lunar 67 grazing incidence 177, 178 Guericke, Otto von 256 Gulf Stream 35 H hairdryer airflow 229, 235–7 heat definition 43 latent heat 280 heat pump 226, 226, 228, 228 hertz (Hz) 182 hiking 5, 48 holograms 165–8 home experiments angular momentum, conservation of 38–41 balloon inflation 258–60 blue sky and red setting sun 111–12 bouncing ball 237–8 CD as an optical instrument 162–4 electric motor 299 hairdryer airflow 235–7 Magnus effect 232–5 mug, vibration and pitch of a 216 rainbow angle 149–50 reckless wine glass party trick 41 “rotating string” blowpipe 73–4 the slow fall 24 tuning fork, increasing the volume of a 190 twin divers 298 wave nature of light 127–8 household appliances energy consumption 263 running costs 264 see also specific appliances human engine 45–8 heat loss/removal 8, 9, 10, 50–1, 52, 53, 54 Everyday Physics: Unusual insights into familiar things 305 humidity 6, 273, 274, 276, 277, 280 humidification 276, 279–80 relative humidity (RH) 6, 273, 274, 276, 277, 278 hypothermia 10 I ice 172 slipperiness 11 thawing 12, 222 ice skating 11–12 infrared 100, 114, 266, 268 insulation 8, 251–2, 253, 254, 255–7, 279, 280 interference 156–8, 156, 157, 160, 160, 161, 165, 166, 167, 167, 168, 222 J joule ( J) 43, 44, 45 K kilocalorie (kcal) 43, 45 kilowatt-hour (kWh) 44 kinetic energy 24 Kittinger, Joseph 22 L ladybirds 270 laminar flow 20 Lenz’s law 24 Leonardo da Vinci 122 light absorption coefficient 170–1, 265 attenuation 169–70, 171, 171 306 brightness, relative 140, 141, 141 Fermat’s principle 77–8 polarization 174, 176–7, 176, 178–9, 178 Snell’s law 78, 78 speed of light 3, 78, 134, 289 white light 106, 114, 148, 159 light bulbs/lamps fluorescent lamps 99, 105–6, 106 incandescent bulbs 99, 100, 101–4, 102, 106, 126 LED lamps 100, 105–6, 106 light waves 108–9, 126, 127–8, 156, 248 interference 156–8, 156, 157, 160, 160, 161, 165, 166, 167, 167, 168 oscillation 173–4, 174, 175, 176, 176, 177 prism, passage through a 138, 138, 159 reflection 140, 141, 142, 148, 149, 154, 158, 162, 172, 176, 177–9 refraction 77–8, 108, 115–18, 116, 118, 133–4, 133, 134, 136, 138, 138, 144, 145, 148, 149, 149, 159 scattering 108, 109–10, 109, 111–12, 113–14, 166, 166, 167, 169, 172 lip-reading 196, 198 lumens 100 M Magdeburg hemispheres 256, 257 magnets 24, 299 Magnus effect 232–5, 300 matchboxes 269–70 mechanical energy 48, 49, 62 Everyday Physics: Unusual insights into familiar things megajoule (MJ) 44 microwave cooking 221–4 mirage effect 133–4, 136, 137 moon albedo 131, 132 brightness 130, 131–2 cycling on the 67–8 gravity 67 moon data 287 reflection coefficient 131 multipliers and dividers 289 muscles 47, 48, 49, 62, 124, 125 muscular work 49, 52, 53, 57 music and musical instruments musical scales 183, 209 stringed instruments 182, 182, 209–15, 210, 213, 214 wind instruments 218, 219 N Newton, Isaac 15 newton (N) 43 noise barriers 205–6, 206 noise pollution 185 O oscillation 173–4, 174, 175, 176, 176, 177 ozone 30 P painted surfaces 265–6, 268 parachutes 23 “passive house” ventilation 274 photography 104, 114, 122, 124, 125, 126, 129, 131, 141, 165, 167 photons 248 photoreceptors 100, 123, 131 photosynthesis 243, 244 photovoltaic cells 242, 243, 244 pi (ϖ) 293 ping-pong ball, and hairdryer airflow 235–7 pirouette principle 38 pitch (frequency) 181, 182, 182, 183, 189, 197, 198, 210, 211, 212, 212–15, 215, 216, 218, 219 Poiseuille’s law 74 Polaroid glasses 173–7 power, definition 43–4 power stations 100, 244, 262 prism 138, 138, 148, 159 R radiation 50–1, 53, 54, 100, 102, 105, 179, 254, 265, 266, 272, 280 see also specific forms of radiators black vs white 265–6 electric 100 radiation/convection 266 radio waves 3, 221–2 radiometer 247–50, 300 radios 187 rainbows 147–54 colours 147, 148, 149, 152, 154 darker sky above 148, 152 features of 147–8 no-rainbow effect 152, 153 rainbow angle 148, 149–52, 150, 151, 154 secondary rainbows 147, 148, 154, 154 raindrops 14, 15–18, 18, 83–6, 149, 149 Everyday Physics: Unusual insights into familiar things 307 rainfall 19, 290 Rayleigh scattering 109–10, 111–12, 113–14 reading glasses 124, 125 reckless wine glass party trick 41, 41 records, 78-RPM 181 reflection 29, 140, 141, 142, 148, 149, 154, 158, 162, 172, 176, 177–9, 197, 207 reflection coefficient 131, 177, 179 refraction light 77–8, 108, 115–18, 116, 118, 133–4, 133, 134, 136, 138, 138, 144, 145, 148, 149, 149, 159 sound 200, 201, 201, 202, 202, 204, 204, 206, 206 refractive index 159, 179 Reichert, Todd 66 relativity theory 4 resources 300 resting energy expenditure 45, 49, 52, 53 Reynolds-slip 62 ripple tank 156, 157 rolling resistance cars 87–9, 89, 90–2, 91, 92 coefficient 60, 68, 88, 92 cycling 59, 60, 61, 62, 67, 68, 90 Rompelberg, Fred 66 “rotating string” blowpipe 73–4 S satellites GPS satellites 1–4, 2, 287, 300 New Horizon 290 orbit times 287 saunas 277–80 308 shock waves 23 skiing 7, 9, 9, 29 skydiving 21–4 Snell’s law 78, 78, 178, 179 snow 172, 274 soap bubbles 3, 155–8 sodium street lights 106 solar eclipse, observing 120–1, 121, 122 solar panels 242 solar power, harvesting 242–4 solar radiation 29–30, 30, 35, 35, 103, 239–40 energy flow 240–1, 288 solar-thermal collectors 242 solstices 26, 27, 27, 35–6 sonic boom 23 sound absorption 185–6, 197, 208 intensity 182–4, 182, 185, 186, 187, 188, 189, 191, 193, 198 noise barriers 205–6, 206 noise pollution 185 pitch (frequency) 181, 182, 182, 183, 189, 197, 198, 210, 211, 212, 212–15, 215, 216, 218, 219 reflected sound 197, 207 sound barrier 22, 23, 68 “sound shadow” 193, 201, 202, 205, 206, 206 wind-blown 203–4, 203 see also ears and hearing; music and musical instruments sound, speed of 23, 200–1, 203–4 in air 289 temperature-dependent 200–1, 218, 219 in water 289 sound waves 190, 192–3, 218 Everyday Physics: Unusual insights into familiar things amplitude 210, 210, 216 refraction 200, 201, 201, 202, 202, 204, 204, 206, 206 spectacles see glasses spectrometer 164, 300 speed acceleration 15, 16, 23, 48, 62, 82, 91 conversions 290 terminal velocity 22, 23 speed skating 90 squash (sport) 43 squinting 126 stair climbing 38, 47–8, 48, 49, 57 Stefan-Boltzmann law 54, 240 stereo equipment 189, 197 stringed instruments 182, 182, 209–15, 210, 213, 214 fundamental and overtones 210, 210, 211, 212 harmonics 211 pitch 210, 211, 212, 212–15, 215, 218, 219 timbre 211 tuning 215, 215, 217–19 summer and winter temperatures 33–6 sun brightness 130, 132 elevation 25–8, 25, 26, 27, 29, 30, 35, 110, 136, 152 “green flash” phenomenon 137–8 parallel light beams 31–2, 31 radiation see solar radiation setting sun 110, 112, 135–8, 136 solar eclipse 120–1, 121, 122 sun data 287–8 sunlight, filtered 119–20, 120 surface temperature 103, 105, 288 winter sunshine 34–5, 34, 35 sunburn 29, 30 surface tension 14, 17 swimming 29, 53 goggles 143, 145, 145, 146 shark attacks 145–6 underwater vision 143–6, 146 swimming pools 115–18, 116, 118, 144 T talking, energy cost of 187–8 tea leaves in a cup 283–5 temperature body temperature 8, 10, 50–4, 280 seasonal temperatures 33–6 wind chill 7–10 ten-pin bowling 38 tennis ball, topspun 230–1, 230, 231 terminal velocity 22, 23 thunder 186 tree height, estimation 120 tuning fork 190 TVs 256, 263 twin divers 298 U ultraviolet (UV) radiation 29, 30 V vacuum 15, 134, 249, 255–7 Everyday Physics: Unusual insights into familiar things 309 vacuum cleaners 47, 261, 263 vision accommodation 124, 125 mirage effect 133–4, 136, 137 sharp 123–6, 124, 125 squinting 126 underwater 143–6, 144, 145, 146 see also eyes W walking angular momentum 37–8, 75 efficiency 55, 56, 57 up stairs 38, 47–8, 48, 49, 57 water boiling 51, 271–2, 274 fog drops 19–20 molecules 222, 273–4 raindrops 15–18 waves 13–14 see also condensation; evaporation; ice; water vapour water vapour 6, 273, 276 heat conduction 280 vapour pressure 278, 278 vapour pressure curve 273, 274, 274, 275 watt (W) 43–4 waves, water interference 156, 156, 157 parallel to the beach 13, 14 speed 14 weight-loss diets 46 white light 106, 114, 148, 159 white paint 265–6, 268 Whittingham, Sam 66 wind 50, 200 310 mountain winds 5–6 side winds, cycling and 69–72, 70, 72 wind chill 7–10, 9 wind speed 9, 9, 203, 203, 204, 206 wind instruments 218, 219 wind turbines 203 windows insulation 251–2, 253, 254, 255–7 as mirrors 142, 177 reflection 140, 141, 177 temperature profile 253, 253, 254 transparency 139, 140, 141, 141, 142 see also curtains wine-mixing 281–2, 296–7 wood, and heat conduction 267, 278 work, definition 43 world population growth 295 World Solar Challenge car race 242 Everyday Physics: Unusual insights into familiar things Also published by UIT Drugs without the hot air This second edition of David Nutt’s brilliant and straightforward book explores the science of legal and illegal drugs, how they work, why people take them and the effects drugs have. Food and Climate Change without the hot air 25% of greenhouse gas emissions come from our food. Here’s how to reduce this -- from breakfast to lunch, and snacks to supper.† Sustainable Materials “Instead of the usual ya-boo about sustainability, this is a pragmatic guide to getting more value from less stuff. Researched with long-term co-operation from industry, it emphasizes facts and evidence” - BBC News Magazine Solar Energy The design of photovoltaic systems for real-life applications, with the physics behind solar energy generation.† Sustainable Energy - without the hot air* “May be the best technical book about the environment that I’ve ever read. This is to energy and climate what Freakonomics is to economics.” Cory Doctorow, Boing Boing Foreign-language titles based on ‘Sustainable Energy without the hot air’ Erneuerbare Energien Energía Sostenible - ohne heisse luft† Christian Holler, Joachim Gaukel 9781906860882 - sin malos humos† Javier Pascual et al. 9781912934188 (EPUB) *Free PDF from withouthotair.com Kindle and EPUB ebooks available free from the usual ebook retailers. †