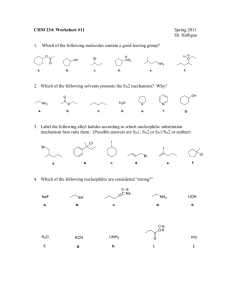
ORGANIC CHEMISTRY OCA216X ALKYL HALIDES Chapter 7 Mr. ND Thobejane Room: Building 4 G19 (NMR office) Consultation (appointment): Please make an appointment (08:00 to 17:00) Faculty of Sciences Department of Chemistry STUDY UNIT 7.1: STRUCTURE AND NOMENCLACTURE Learning outcomes Study unit: Structure and nomenclature Explain the reactivity of the C-X bond Name the alkyl halides according to the IUPAC name Faculty of Sciences Department of Chemistry NAMING OF ALKYL HALIDES • Halo-substituted alkanes are named systematically as haloalkanes. • How? By treating halogen as a substituent on a parent alkane chain. Step 1 • Find the longest chain and name it as the parent. • NB: If a multiple bond is present, then the parent must contain it. Faculty of Sciences Department of Chemistry NAMING OF ALKYL HALIDES Step 2: Number the carbons of the present chain beginning at the end nearer the first substituent, whether alkyl or halo. NB: (i) Assign each substituent a number to its position on the chain. (ii) if there are substituents at the same distance from both ends, start numbering at the end nearer the substituent with alphabetical priority. Faculty of Sciences Department of Chemistry naming of alkyl halides Step 3: Write the name • (i) List all substituents in alphabetical order. • (ii) Use prefixes di-, tri- and so forth if more than one of the same substituent's is present. • (iii) In addition, many simple alkyl halides are also named by identifying the alkyl group and followed by halogen. Faculty of Sciences Department of Chemistry STUDY UNIT 7.2: SYNTHESES AND REACTIONS OF ALKYL HALIDES Specific outcomes: • Propose reactions to prepare alkyl halides. • Predict the products of the reactions of alkyl halides. • Explain the mechanisms of nucleophilic substitution (including Walden inversion of configuration) and elimination reactions. • Use the simple reactions of methods to prepare and transform alkyl halides as part of multi-step synthetic proposals of more elaborate molecules. Faculty of Sciences Department of Chemistry SYNTHESIS AND REACTIONS OF ALKYL HALIDES • Previously studied: • Other methods of preparing alkyl halides via addition reactions of HX and X2 with alkenes in electrophilic addition reactions and alkane with Cl2. Faculty of Sciences Department of Chemistry Preparing of alkyl halides • Most general method of synthesizing alkyl halides is to prepare them from alcohols, which are formed from carbonyl compounds. • Reaction 1: treating the alcohol with HCl or HBr. For example compound 1-methylcyclohexanol is converted into 1-chloro-1methylcyclohexane by treating it with HCl. Faculty of Sciences Department of Chemistry Preparing of alkyl halides • Note: the H-X with tertiary alcohols works better. Primary and secondary alcohols reacts much more slowly. Faculty of Sciences Department of Chemistry Preparing of alkyl halides • Note: Thionyl chloride (SOCl2) or phosphorous tribromide (PBr3) is the best reagent to convert primary and secondary metabolites alcohols into alkyl halides. These reactions normally takes place in high yield. Faculty of Sciences Department of Chemistry Preparing of alkyl halides • Alkyl fluorides can be prepared from alcohols. Other reagents used are diethylaminosulfur trifluoride [(CH3CH2)2NSF3] and HF-pyridine where benzene is the nitrogen-containing analog of benzene Faculty of Sciences Department of Chemistry Reactions of alkyl halides: Grignard reagents • How to prepare Grignards reagent. • Alkyl halides reacts with magnesium metal in ether to yield alkyl magnesium halides, RMgX Grignards reagent. Faculty of Sciences Department of Chemistry Reactions of alkyl halides: Grignard reagents • Grignard reagent reacts with CO2 to afford carboxylic acids after protonation with aqueous acidCarboxylation Faculty of Sciences Department of Chemistry NUCLEOPHILIC SUBSTITUTIONS • Alkyl halides : when they react with nucleophiles/bases, such hydroxide ions, either undergo substitution of the X group by the nucleophile or elimination of H-X to yield an alkene. • A German chemist Paul Walden discovered the nucleophilic substitution reaction of alkyl halides in 1896. • He discovered that (+)- and (-) malic acid could be interconverted. Faculty of Sciences Department of Chemistry Paul walden discovery • His discovery at the time was astonishing. The (-) malic was converted into (+) malic , some reactions in the cycle must have occurred with a inversion or change in the configuration of chirality center. Faculty of Sciences Department of Chemistry Walden transformation –nucleophilic substitution reaction • Walden transformation is now referred to as nucleophilic substitution reactions. Why? • Because each step involves the substitution of one nucleophile (chloride ion Cl-or hydroxide ion OH-) by another. • Nucleophilic substitution are the most common and versatile reactions in organic chemistry. Faculty of Sciences Department of Chemistry Nucleophilic substititution • Nucleophilic substitution occurs in two major pathways (SN1 reaction and SN2 reaction • SN substitution, nucleophilic (Nu: or Nu:-) • SN reactions are the same, nucleophile reacts with a substrate RX and substitute the leaving group X:- to yield a product R-Nu. • If the nucleophile is neutral (Nu:) then the product is positively charged to maintain charge conversion. Faculty of Sciences Department of Chemistry When is reaction sn1,sn2, e1 and e2 • Consider two different reactions – Substitution and Elimination. • Four different mechanisms (SN1,SN2, E1 and E2) that begin with one class of compound (Alkyl halides). • The nature of the leaving group that is displaced by the attacking nucleophile. • The best leaving groups are those that gives the most stable anions. That is anions of strong acids. Faculty of Sciences Department of Chemistry Some nucleophilic substitutions Faculty of Sciences Department of Chemistry Sn2 reaction • SN2 Reaction occurs in a single step when the incoming nucleophile approaches from the direction 180o away from the leaving halide, thereby inverting the stereochemistry at carbon. • SN2 takes place without any intermediates. Faculty of Sciences Department of Chemistry Sn2 mechanism These are not for sale or distribution. Only for learning purposes Faculty of Sciences Department of Chemistry Rates of sn2 reaction • In chemical reactions- there is direct relationship between the rate at which the reaction occurs and the concentration of the reactants. • SN2 reaction of CH3Br with OH- to afford CH3OH takes place in a single step when substrate and nucleophile collide and reacts. • If the concentration of OH- is doubled, the frequency of collision between the two reactants doubles and therefore the reaction rate doubles. • The ‘2’ in SN2 reaction are called bimolecular- cause to the rate of the reaction depends on the concentration of two substances-alkyl halide and nucleophile Faculty of Sciences Department of Chemistry Stereochemistry of sn2 reaction Note: the incoming nucleophile attacks the substrate and begin pushing out the leaving group on the opposite site, results the configuration of the molecule inverts. (S)-2-bromobutane gives (R)-butan-2-ol. Faculty of Sciences Department of Chemistry Steric effects in SN2 REACTIONS • For the nucleophile to approach the substrate to undergo an SN2 reaction depends on the steric accessibility to the halidebearing carbon. • Bulk substrates, in which halide bearing carbon atom is difficult to approach Reacts more slowly than the carbon is more accessible. These are not for sale or distribution. Only for learning purposes Faculty of Sciences Department of Chemistry Unreactive reaction towards sn2 • Lack of reactivity due to steric hindrance. • Vinylic (R2C=CRX) and aryl (Ar-X) halides are not shown in the reactivity list cause they completely unreactive toward SN2 displacement. Faculty of Sciences Department of Chemistry Leaving groups in sn2 reactions • Variable that affects SN2 reactions. • The nature of the leaving group that is displaced by the attacking nucleophile. • The best leaving groups are those that give the most stable anions. i.e. anions of strong acids. Faculty of Sciences Department of Chemistry SN1 REACTIONS • Note most nucleophilic substitution reactions undergo SN2 pathway. Alternative SN1 reaction do occur. • SN1 reaction occur only on tertiary alcohol and only under neutral or acidic conditions (in hydroxylic solvent such as water or alcohol). • Previously studied that alkyl halides can be prepared by reaction alcohol and HCl or HBr. Faculty of Sciences Department of Chemistry SN1 REACTIONS • Previously studied in SN2 reaction that a leaving group is displaced at the same time that the incoming nucleophile approaches. • However, SN1 reaction occurs by spontaneous loss of the leaving group before incoming nucleophile approaches. • Loss of the leaving group gives a carbocation intermediate, which reacts with the nucleophile in the second step yielding a substitution product. Faculty of Sciences Department of Chemistry rates of sn1 reactions • Previously studied on SN2 that rates soley depends on the concentration of both substrate and nucleophile. • However, in SN1 reaction depends on the concentration of substrate and is independant of the nucleophile concentration. • The origin of ‘1’ in SN1 is that SN1 reactions are unimolecular, due to the fact the rate of the reactions depends only on one substance. Faculty of Sciences Department of Chemistry Mechanism of sn1 • SN1 mechanism of tert-butyl with HBr to afford an alkyl halide. H2O is the leaving group. Faculty of Sciences Department of Chemistry Stereochemistry of sn1 reaction • Since SN1 reactions occurs through carbocation intermediate, the stereochemistry is rather different from SN2 reaction. • The C+ can either react with nucleophile equally well from either side, leading to a 50/50 racemic mixture of enantiomers. • However, if only SN1 reaction is carried out on single enantiomer of chiral substrate and go through achiral carbocation intermediate the compound loses its chirality and become optically inactive. Faculty of Sciences Department of Chemistry stereochemistry Faculty of Sciences Department of Chemistry Elimination of alkyl halides • Note that two kinds of reactions can take place when a nucleophile/base reacts with an alkyl halide. Reaction 1: nucleophile/base can either substitute for the halide in SN1 or SN2 Reaction 2: it can cause elimination of HX, thus resulting in formation of an alkene. Faculty of Sciences Department of Chemistry Elimination of alkyl halides • Elimination of HX from alkyl halide is important method for preparation of alkenes. However, this is complex for several reason Regiochemistry? What products results by loss of HX from an unsymmetrical halide. Usually elimination reactions always give mixture of two products – best predict which will be the major product. Faculty of Sciences Department of Chemistry Elimination of alkyl halides – Zaitsev’s rule • 1875 a Russian Chemist Alexander Zaitsev’, predicted – baseinduced elimination reactions generally give more highly substituted alkene product. that is – alkene with the larger number of alkyl substituent on the double bond. Faculty of Sciences Department of Chemistry E1 REACTION MECHANISM • Zaitsev’s rule • Elimination of HX from an alkyl halide, the more highly substituted alkene product predominates. • Elimination reaction can take place by several different mechanism, E1, E2 1. E1 reaction, the C-X breaks first and give carbocation intermediate that undergoes subsequent base attraction of H+ Faculty of Sciences Department of Chemistry E1 mechanism The dehydrohalogenation of (CH3)3CCI with H2O to form (CH3)2C=CH2 can be used to illustrate the second general mechanism of elimination, the E1 mechanism. • An E1 reaction exhibits first-order kinetics: Mechanisms of Elimination—E1 • The E1 reaction proceeds via a two-step mechanism: the bond to the leaving group breaks first before the π bond is formed. The slow step is unimolecular, involving only the alkyl halide. Faculty of Sciences Department of Chemistry Elimination – E1 reaction • E1 reaction (elimination, unimolecular) Faculty of Sciences Department of Chemistry Acid-catalyzed dehydration – e1 process mechanism The reaction undergoes E1 reaction and involves a carbocation intermediate. 1. Firstly, strong acid protonates the alcohol oxygen Faculty of Sciences Department of Chemistry Acid-catalyzed dehydration – e1 process mechanism 1. The protonated intermediate spontaneously loses water to generate a carbocation. Faculty of Sciences Department of Chemistry Acid-catalyzed dehydration – e1 process mechanism • Loss of H+ from neighbouring carbon atom then yields the alkene product. Faculty of Sciences Department of Chemistry E1 MECHANISM Faculty of Sciences Department of Chemistry E1 MECHANISM Faculty of Sciences Department of Chemistry CHARACTERISTIC OF E1 REACTION Faculty of Sciences Department of Chemistry E1 AND E2 MECHANISM • The E1 and E2 mechanisms both involve the same number of bonds broken and formed. • The only difference is timing. In an E1, the leaving group comes off before the β proton is removed, and the reaction occurs in two steps. • In an E2 reaction, the leaving group comes off as the β proton is removed, and the reaction occurs in one step. Faculty of Sciences Department of Chemistry E2 REACTION MECHANISM • Base-induced C-H bond cleavage is simultaneous with C-X bond cleavage, resulting an alkene in a single step Faculty of Sciences Department of Chemistry E2 MECHANISM •D Faculty of Sciences Department of Chemistry CHARACTERISTICS OF E2 REACTION Faculty of Sciences Department of Chemistry Summary – Primary alkyl halide • Primary alkyl halides (RCH2X_ SN2 substitution occurs if a nucleophile such as HO, I-,Br-, RS-, NH3 or CN. • E2 elimination occurs if a strong base such as an alkoxide OH is used. SUMMARY – SECONDARY ALKYL HALIDE • A weak base or nucleophile favors S 1 and E1 mechanism and both occurs. N • E2 elimination predominates is a strong base is used. Faculty of Sciences Department of Chemistry SUMMARY – TERTIARY ALKYL HALIDE • E2 elimination occurs when a strong base is used. • SN1 and E1 occurs together under neutral or acidic conditions. Faculty of Sciences Department of Chemistry SUMMARY Faculty of Sciences Department of Chemistry SUMMARY Faculty of Sciences Department of Chemistry summary Faculty of Sciences Department of Chemistry summary Faculty of Sciences Department of Chemistry THANK YOU Faculty of Sciences Department of Chemistry