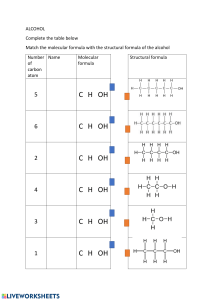
Republic of the Philippines Department of Education NATIONAL CAPITAL REGION Schools Division Office-Quezon City EMILIO JACINTO NATIONAL HIGH SCHOOL Pingkian Road, Pasong Tamo, Quezon City Demonstration Lesson Plan in General Chemistry 2 Date: May 12, 2023 12-STEM (Newton) 6:40 AM – 7:20 AM I. OBJECTIVES A. Apply and correlate the valence shell electron pair repulsion (VSEPR) theory and the modern periodic table (MPT) of chemical elements; B. Predict and reconstruct the geometry of common molecules using polymer balls and sticks and/or MoCubed app; C. Instill the importance and application of molecular design to various disciplines. II. SUBJECT MATTER A. TOPIC/SUBJECT MATTER: Molecular Geometry: Organic Compounds and their Structures B. REFERENCES: Curriculum/Teacher’s Guide page/s: page 26 Other Reference/s: Atkins, Robert C. & Carey, Francis A. (2002). Organic Chemistry A Brief Course (International Edition). Mc Graw Hill. New York. pp. 5-7 Hess, Fred C. (2015). Chemistry Made Simple, Revised edition. Doubleday and Company, Inc. New York. pp. 27, 167 Other Learning Resource/s: Chemical Bonding, Geometry of Molecules. Date retrieved from https://www.starbooks. ph/display_k12_courses/110 May 09, 2023. C. MATERIALS: paper and pen , Tablet/phone with pre-installed MPT and MoCubed app, Activity sheet powerpoint slides, marker and eraser, molecular model kit, miniature canister, Baking soda and Vinegar, Alka-Seltzer and water, Paper rocket, Inflated balloons III. Procedures A. Motivation The volunteer learner will be asked to engage in the mini demonstration titled Baking Soda Popper. They will be asked initially to predict what will happen if the common household ingredient reacts with water in the canister. SAFETY GOGGLES MUST BE WORN BY THE VOLUNTEER LEARNERS WHILE PERFORMING THE ACTIVITY. - First volunteer learner will pour in 1 teaspoon of baking soda in the mini canister. - Second volunteer learner will add water, up to about half the canister. S/he then quickly put on the lid (cover), place it upside down, and take a step back. - Observe and watch the canister. They will be jotting down some of the evidence/s of the reaction observed – GAS FORMATION/EVOLUTION to be exact. 2𝑁𝑎𝐻𝐶𝑂3 → 𝑁𝑎2 𝐶𝑂3 + 𝐶𝑂2 + 𝐻2 𝑂 They will answer “Can common household chemicals be utilized as alternative fuel?” Why or why not? Explain the reaction that happened inside the canister. B. Establishing a purpose for the lesson The second set of volunteer learner will be asked to engage in the second demonstration but this time, by using a commercially available OTC drug, Alka-Seltzer as the main reactant, then with an Emilian rocket paper will be tried to travel upwards. They will be jot down some of the evidence/s of the reaction observed – GAS FORMATION/EVOLUTION. 𝐶6 𝐻8 𝑂7 + 3𝑁𝑎𝐻𝐶𝑂3 → 3𝑁𝑎3 𝐶6 𝐻5 𝑂7 + 3𝐶𝑂2 + 3𝐻2 𝑂 They will answer “Can common household chemicals be utilized as alternative fuel?” Why or why not? Explain the reaction that happened inside the canister that enabled the paper rocket to fly. C. Presenting examples/instances of the new lesson The learners will correlate the previous lesson to the formation of the molecular products in the demonstrated reaction. – rearrangement of shared electrons thru covalent bonds among nonmetals and different nonmentals D. Discussing new concepts and practicing new skills The learners will write the Lewis structure and then predict the molecular geometry (shape) of CO2 gas that has been formed in the demonstrated mini reaction using VSEPR Theory as the guide. Valence Shell Electron Pair Repulsion (VSEPR) Theory ➢ It guides us in constructing the acceptable Lewis structure with the correct geometry of a given molecule The learners will concretize the VSEPR Theory using the provided inflated balloons. Learners will observe the natural orientation of the inflated balloons when they are grouped as: two, three, four, and five. From the natural orientation of the balloon, learners will deduce the shape as to linear, or bent, or trigonal planar, or trigonal pyramidal, and/or tetrahedral (tetrahedron). Electron Group Geometry no lone pairs in the central atom LINEAR TRIGONAL PLANAR TETRAHEDRAL 2 balloons 3 balloons 4 balloons with lone pairs in the central atom BENT TRIGONAL PYRAMIDAL E. Developing mastery (develops to Formative Assessment) The learners will master the VSEPR Theory and its correlation to writing Lewis structure and predicting molecular geometry of CO2 by employing the steps as well as completing the Chem Bonds Table. MF A CO2 Theoretical electrons B C D E No. of bonded/ shared electrons No. of bonds formed No. of unshared/ nonbonded electrons A–B 1C 1 8 = 8e Total valence electrons available for bonding 1 4 = 4e 2O 2 8 = 16e 2 6 = 12e 0 0 Charge Total 8e 24e 16e B–C 𝐶 8𝑒 = 2𝑒 2𝑒 4 (as bonds) 8e (as dots) Lewis structure (e-dot formula) Molecular geometry O=C=O linear F. Finding practical applications of concepts and skills in daily living The learners will reconstruct the Lewis structure with its appropriate molecular shape (geometry) using the ball-stick model and MoCubed app. How will you correlate then the formed structure of CO2 to the extreme heat conditions that our country is experiencing during the El Niño time or the global warning phenomenon? G. Making elaborations and abstractions about the lesson The group of learners will elaborate and concretize the concepts discussed. Each group shall show/accomplish the Chem Bond Table. Then groups I and III, will show the molecular geometry of the assigned molecule using MoCubed app; while Groups II and IV will present the molecular structures using ball-stick model. Five minutes, for completion of table and restructuring of the molecular shapes/geometry, using the assigned method. MF A Theoretical electrons B Total valence electrons available for bonding C No. of bonded/ shared electrons D No. of unshared/ nonbonded electrons E No. of bonds formed #atom1 #atom2 #atom3 Charge Total Each group of learners will be given two minutes to further explain their work. H. Evaluating learning Group No. I II III IV Assigned molecule/description Methanol, CH3OH, the major alcohol components in cooking alcohol (alcohol-based food warmer) Methane, CH4, the simplest hydrocarbon that is present in all decomposing organisms and its byproducts Methanoic acid; the compound in the ant/insect’s stings Task/Output a half-page warning material to emphasize safety on the proper use of commercial methanol An infomaterial using Canva in slide format emphasizing safety in dealing with flammable HCs A half-page campaign material in emergency treating ant/insect’s bite. Ethanol, the only alcohol that can be Create a 2-line slogan promoting consumed by human being, normally responsible drinking of alcohol used as a disinfectant. Rubric: Extent/Comprehensiveness Presentation/Discussion Analysis/Conclusion 40% (10 points) 40% (10 points) 20% (5 points) I. Additional activities for application or remediation The learners will conduct a 1-page research study in correlation of styrene, structure, to the solid waste and nanoplastic pollution in the National Capital Region, Philippines. Due will be on May 19, 2023. J. Agreement The learners will ask to conduct advanced notes taking in Introductory Nomenclature of Organic Compounds and Functional Groups. 1. What are the different classes of Organic Compounds? 2. Why are organic compounds classified as molecules? Generalize your answer based on the common elements present and the type of chemical bonds as well. 3. Write the systemic name of the first 12 simple acyclic hydrocarbons. Prepared by: EMIL L. ESCALANTE, LPT Teacher III, SHS Academic-STEM Checked/Approved by: AMMAR N. TORREVILLAS, Ed. D. School Principal II