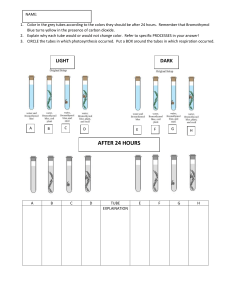
REVISION’S SHEET ERRRORS AND IMPROVEMENTS: A) Qualitative food test: ERRORS: • Difficulty in judging colours especially if the concentrations are low resulting in light colours • If heating, then temperature is not constant, as there is heat loss to the surrounding • Different molecules in the same solution,may interfere with the colours Improvements: • In case of heating,Thermostatic water bath to control the temperature and avoid the heat loss to the surrounding • Colour standard to help judging the colour • Use black card/ white card to help judging colours B) Quantitative food test: ERRORS: • In case of determining unknown concentration: it can be between 2 known concentrations • Difficulty in comparing colours • Temperature varies due to heat loss to the surrounding • In case of vitamin c :Drops fall on the walls of the test tube Improvements: • Use more concentrations with narrower range( smaller gaps between concentrations) or wider range( wider gaps between the concentrations), you can then plot a graph with the obtained results and find your unknown by reading off the graph, this provides more accurate estimate for the unknown • Use colourimeter or colour standards to help judge the colour better • Use black card or white card to enable you from observing the colour difference • Use thermostatic water bath to avoid heat loss to the surrounding • In case of introducing drops , use wider test tube • Use graduated pipette for more accurate volumes than the syringe • Repeat and take average How to make Benedicts test more reliable • Use the same volume of the substance under test. • Use the same volume of the reagent. • Leave the tubes in the same water bath for the same period of time. C) OSMOSIS How to keep fair comparison in osmosis experiments? 1-When using petri dishes , use petri dishes of the sme size. 2- When using sucrose or any other solution , put the same volume in each dish. 3- When using onion epidermis or potato, must be of the same size. 4- Leave the potato or onion epidermis in the solution for the same period of time. General precautions in osmosis experiments: 1- Cover the petri dishes during the experiment to void evaporation of water which cn affect concentration of the solution. 2- When using droppers or syringes , use separate dropper for each solution or wash and dry it after each step. 3- During preparing slide of onion epidermal cells ,lower the cover-slip gently to avoid trapping of any air bubbles. ERRORS: • Time tissues left in solutions is not enough to observe complete plasmolysis • Evaporation of solution may take place and therefore change the concentration • Difficulty in judging degree of plasmolysis • Difficulty in cutting the samples into correct dimensions • Parallex error may occur during reading the lengths IMPROVEMENTS: • Leave for longer time in the solutions • Prepare more concentrations with narrower / wider range (give examples and method of dilution) • Cover the petri dish to avoid evaporation of solution • Prepare more than one sample in each solution • Ensure that the tissues are completely immersed in the solutions D) ENZYME REACTIONS: ERRORS: • Difficulty in judging the endpoint (in amylase, the colour with iodine is not clear, in renin the coagulation is not clear, colour of litmus paper with urease is not clear) • Temperature/PH is not controlled (if they are not the factors under investigation) • Bubbles of different sizes and are all counted as one,bubbles may be too fast for counting, gas expansion interfere with the results, small bubbles may be missed as not seen (in case of counting bubbles with catalyse (potato)or with yeast) • Paper may stick to the walls and bottom of the test tube ( in case of litmus paper with urease, or paper dipped in catalyse then placed in hydrogen peroxide or paper dipped in iodine then placed in amylase using splint) IMPROVEMENTS: • Use colorimeter instead of judging colors or use black or white card against the tube for better color comparison • Measure the volume of gas using gas syringe instead of counting bubbles • Use wider test tubes( in case of beads or paper) • Control temperature using thermostatic water bath • Control PH using buffer Effect of heavy metals such as copper sulphate and lead nitrate ➢ Can act as inhibitors. ➢ May cause protein to clot or coagulate. ➢ May denature proteins. ➢ May breakdown bonds so that can alter tertiary and quaternary structures. E) AGAR: ERRORS • Difficulty in cutting the agar squares into equal dimensions • Difficulty in judging the color change • Agar is not of equal depth • Pigmentation of the agar is not even • Agar may be damaged during cutting Improvements • Use moulds for preparing agar squares with equal dimension • Use different indicator with clearer end point • Use wider or narrower range of concentrations (In case of determining unknown) • Use black card or white card below the beakers for better judgment of colors F) IMMOBILIZTION OF ENZYME (BEADS FORMATION) ERRORS • Beads are not of equal sizes • Beads stuck to the sides of the tube and to each others • Forceps causes damage the the beads • Difficulty to introduce drops using syringe • Test tube is not vertical and also test tubes are not of equal sizes (in case of catalyse beads and hydrogen peroxide) • Temperature/PH is not controlled ( if they are not the factors under investigation) IMPROVEMENTS • Use sieve with equal size holes to produce beads with equal diameter • Use wider test tubes • Stain the beads for clearer movement • Use resort stand to make the test tube vertical, use test tubes with equal sizes (in case of catalyse beads and hydrogen peroxide) • Use spatula or spoon instead of forceps • Control temperature using thermostatic water bath/ control PH using buffer G)COLOR DIFFUSION Diffusion of pigment from inside the tissue (potato/beetroot) to the solution by the influence of salt or temperature( due to the damge of cell membrane/cell wall) ERRORS • Difficulty in judging intensity of diffused color • Some pigments remain on the surface of the tissue despite washing • Temperature is not controlled • Time is not enough for the diffusion to take place • There is narrow range of concentrations IMPROVEMENTS • Use color standards with degree of color and its name for better colour describtion , or numbered colour scale with images showing the color • Use white/black card against the test tubes for comparing colors • Control the temperature using thermostatic water bath • Dry the samples with paper towel before placing them in the solutions • Leave the samples for longer time in solutions • Use wider range of concentration so the colors produced show difference H) Density of solution (a method for determining the concentration) In this method you pigment the unknown concentration by adding 2 drops of methylene blue to 5 cm3 of the unknown in a petri dish, u then take a drop of the pigmented solution and introduce it to test tubes of prepared known concentration, the drop produced will move according to its concentration: • If the drop has higher concentration than the solution, it will move down • If the drop has lower concentration than the solution, it will move up • If the drop has same concentration as the solution , it stays ERRORS • Difficulty in introducing drops by syringe • The pigmentation is too dark/ or too light • The drop ruptures (it releases a flow which may be not clear) • Too wide/narrow range of concentrations • Test tube is not vertical IMPROVEMENTS • Use dropper instead of syringe • Increase or decrease the drops of the stain ( to make the pigment lighter or darker) or use different stain • Prepare wider/narrower range of concentration • Use resort stand to make the test tube vertical I) VISKING TUBES Take care to open it using water ERRORS • Difficulty in opening and tying the visking tube • Visking tubes is too short /or too long • Difficulty in rinsing the visking tube ( water may enter inside it) • Mixing is not constant • Drops may not of equal volume • Difficulty in judging color IMPROVEMENTS • Make the visking tube longer or shorter • Dry the visking tube with paper towel before placing in the beaker • Mix using electronic mixer • Use syringe instead of dropper to introduce equal volume of solution and indicator • Use color standard/ or colorimeter TYPES OF VARIABLES: First independent variable - The variable that affects the results by the experiment for example ▪ In an experiment to investigate the effect of light intensity on the rate of photosynthesis, light intensity is the independent variable. ▪ In an experiment to investigate the effect of change in pH on the activity of amylase, pH is the independent variable. - It is represented on the X- axis of the graph. Second Dependent variable - It is the results obtained in the investigation, for example ▪ In an experiment to investigate the effect of light intensity on the rate of production of oxygen, the volume of oxygen produced per unit time is dependent variable. ▪ In an experiment to investigate the effect of change in pH on the activity of amylase by measuring the time needed for complete breakdown of a certain volume of starch, time is the dependent variable. - It is represented on the Y- axis of the graph. TYPES OF CHARTS AND GRAPHS A) Smoth curve • You will always join all your points either by ruler or hand • If there is trials and means , you will always plot the mean only, except if you were asked to plot trial 1 and trial 2 you will use a key and produce the following figure B) Histogram: in the histogram all the bars will be attached to each others, and will be of same thickness, your x axis will be inervals (e.g 12-14, 14-16) rather than categories or numbers C) Bar chart If he need a bar or histogram , he will ask you to plot a chart, if he needs a smooth curve or line graph he willask you to plot a graph • In the bar chart, your x axis will be letters or categories rather than numbers • All the bars should be of equal thickness • You will leave equal spacing between the bars , this includes the space before the first bar as the below figure (b): N.B: the bar chart may for 2 classes e.g animal a and animal b , in that case you will make the bars of the 2 classes (a and b) attached to each others but leave even spacing between the intervals as the below figure shows: INDICATORS: 1-Effects of litmus. ➢ In acidic medium its colour is red. ➢ In an alkaline medium its colour is blue. 2-Universal indicator It is a pH indicator. pH 0-3 3-6 Descripti colour on Strong Red acid Acid Orange/yel 7 Neutral 8-11 Alkaline 11-14 Strong alkali low Green Blue Violet / purple 3-Cobalt chloride paper ➢ It is used as an indicator for water. ➢ The dry cobalt chloride paper (anhydrous )has a blue colour. ➢ In presence of water (when hydrated) its colour changes to pink or mauve. 4-Hydrogen carbonate indicator. ➢ Its original colour is red . ➢ Increase in concentration of carbon dioxide makes it yellow ( increase in concentration of carbon dioxide makes the medium acidic as carbon dioxide is acidic gas) . ➢ Decrease in concentration of carbon dioxide makes it purple . ➢ ( removal carbon dioxide from the medium makes it alkaline). 5-Methylene Blue: Oxygen Indicator ➢ methylene blue indicator is blue when oxygen is present ➢ If oxygen is removed from the solution, the blue color disappears. 6-Bromothymol Blue pH Indicator ➢ Bromthymol blue changes color over a pH range from 6.0 (yellow) to 7.6 (blue) ➢ It is a good indicator of dissolved carbon dioxide (CO2) and other weakly acidic solutions 7-Potassium permanganate ➢ It is a strong oxidizing agent. It dissolves in water to give intensely pink or purple solutions ➢ Concentrated sulfuric acid reacts with potassium permengnate forming acidic solution ➢ Acidic solutions of permanganate are reduced to the faintly pink manganese(II)