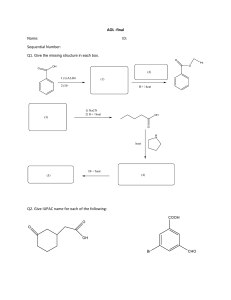
EXPERIMENT A
PRECIPITATION REACTIONS: IDENTIFICATION OF INORGANIC
COMPOUNDS IN AQUEOUS SOLUTION
A precipitation reaction is a reaction that yields an insoluble product—
a precipitate—when two solutions are mixed. We described a precipitation
reaction in which a colorless solution of silver nitrate was mixed with a
yellow-orange
solution
of
potassium
dichromate
to
give
a
reddish
precipitate of silver dichromate:
AgNO3(aq) + K2Cr2O7(aq) → Ag2Cr2O7(s) + KNO3(aq)
This equation has the general form of an exchange reaction:
Thus precipitation reactions are a subclass of exchange reactions that
occur between ionic compounds when one of the products is insoluble.
Because both components of each compound change partners, such reactions
are sometimes called double-displacement reactions. Two important uses of
precipitation reactions are to isolate metals that have been extracted
from their ores and to recover precious metals for recycling.
Whether or not such a reaction occurs can be determined by using the
solubility rules for common ionic solids. You will determine whether a
double-displacement reaction occurs when aqueous solutions of the four
compounds listed in Table 2 are mixed. You will be assigned an unknown
solution containing one of the four compounds listed in Table 2. Based on
your observations, you will identify the unknown inorganic compound.
Materials: 0.5 M NaOH, 0.5 M CuSO4, 0.5 M Pb(NO3)2, 0.5 M BaCl2, distilled
water, testtubes, droppers or pipette.
Procedure:
1. Develop and carry out a procedure for determining whether double
displacement reactions occur when solutions of the following four compounds are mixed. There are 6 possible reaction combinations. Use only ten
drops of each solution for each reaction. Note: if no dropper is available,
obtain 3 mL of each solution.
Record the colors of the solutions listed
below on the Data Sheet.
a. 0.5 M NaOH
b. 0.5 M CuSO4
c. 0.5 M Pb(NO3)2
d. 0.5 M BaCl2
2. Carefully observe the mixtures in each test tube for any evidence of
reaction. Record your observations in the table on
the Data Sheet. The table in the Data Sheet will
assist
you
with
determining
the
6
possible
combinations.
3. After recording your observations, empty the contents of the test tubes
into the Waste Container.
4. Thoroughly clean and dry all test tubes.
5. Write the balanced net ionic chemical equations for all reactions that
occur in Steps 1-2.
6. Obtain an unknown inorganic compound solution from your instructor.
Record the unknown number and color of the solution on the Data Sheet.
7. In five separate test tubes, mix four drops of the unknown inorganic
compound with each of the test solutions from Step 1.
8. Carefully observe the mixtures in each test tube for any evidence of
reaction. Record your observations in the table on the Data Sheet.
9. After recording your observations, pour the contents of the test tubes
into the Waste Container.
10. Thoroughly clean and dry all test tubes.
11. From your observations of the reactions of the known and the unknown
inorganic compounds, identify the unknown inorganic compound. The unknown
is one of the compounds listed in Step 1.
Table of Observations/ Data Sheets:
1. Record the color of the known inorganic compound solutions:
0.5 M NaOH
_______________
0.5 M CuSO4
________________
0.5 M Pb(NO3)2
________________
0.5 M BaCl2
________________
1-2. In the top (white space) of each box in the table below, record your
observations (e.g., white precipitate, solution turns green, etc.). In the
bottom (gray space) of each box, write the formula for the sub- stance
(e.g., precipitates or complex ions) formed when the ions are mixed. If
no reaction occurs, write NR in the boxes.
BaCl2
NaOH
Pb(NO3)2
CuSO4
CuSO4
Pb(NO3)2
5. Write balanced net ionic chemical equations for all reactions that
occurred in Steps 1-2.
6.
Unknown number of inorganic compound
Color
of
unknown
inorganic
________________________
compound
solution
________________________
7-8. Observations for the unknown metal ion.
NaOH
CuSO4
Pb(NO3)2
BaCl2
Unknown
inorganic
compound
11. Identity of the unknown inorganic compound. _______________________
 0
0