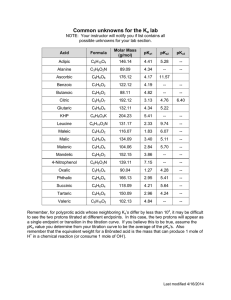
Introduction to HPLC Introduction to LC and the HPLC ChemStation Course H5929A This 4 day laboratory and lecture course is designed to introduce users to the practical aspects of performing routine analyses. What is HPLC From Wikipedia, the free encyclopedia High-performance liquid chromatography (HPLC; formerly referred to as high-pressure liquid chromatography), is a technique in analytical chemistry used to separate the components in a mixture, to identify each component, and to quantify each component. It relies on pumps to pass a pressurized liquid solvent containing the sample mixture through a column filled with a solid adsorbent material. Each component in the sample interacts slightly differently with the adsorbent material, causing different flow rates for the different components and leading to the separation of the components as they flow out the column. Comparison of HPLC and GC Mass Analyzer The spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical structures of molecules, such as peptides and other chemical compounds. Octopole Ion Pulser DC Quad Rough Pump Turbo 1 Turbo 1 Turbo 1 Turbo 2 Reversed-Phase Chromatography Why RPC? • Mostly Water as mobile phase • Gradients • pH selective • Hydrophobic stationary phases work for most organic analytes. 29 Sample Preparation A typical requirement of the pharmaceutical industry is the quality control of active ingredients ( ) in drugs. It is very important to identify drug impurities ( ) that may occur during synthesis or by decomposition of the active ingredient. Quality control ensures patient safety. Page 35 Step 1: Sample made soluble Page 36 Step 2: Separation Column Stationary Phase Compound separation occurs in a separation column. The column consists of a metal tube filled with silica gel. The silica gel is considered the stationary phase. Page 37 Step 2: Separation Column The dissolved compounds are injected onto the separation column. Stationary Phase Page 38 Useful Solvents for Reversed-Phase Chromatography Water Methanol Acetonitrile Isopropanol Dioxane Tetrahydrofuran Elution Strength Solvent Miscibility Name Acetic Acid Acetone Acetonitrile Benzene Butyl Alcohol Carbon Tetrachloride Immiscible Miscible Chloroform Cyclohexane Cyclopentane Dichloroethane Dichloromethane Dimethylformamide Dimethyl Sulfoxide Dioxan Ethylacetate Ethyl Alcohol Di-Ethylether Heptane Hexane Methyl Alcohol Methylethyl Ketone I-Octane Pentane I-Propyl Alcohol Di-Propylether Tetrachloroethane Tetrahydrofuran Toluene Trichloroethane Water Xylene 2-Propanol is an excellent intermediate solvent Structures and pKa values Benzoic Acid pKa 4.19 Phenylacetic Acid pKa 4.3 Vanillin pKa 7.38 Phenol pKa 10 Sorbic Acid pKa 4.8 Structure graphics: Wikipedia pH Blending C and D (C+D=10%) meter pH 9.00 8.00 7.81 7.00 Measured pH 6.00 5.00 4.00 3.00 3.07 3.66 5.34 5.06 4.85 Acetate pK 4.7 4.42 4.63 4.24 4.00 5.78 meter pH 2.00 1.00 0.00 0.0 0.2 0.4 0.6 0.8 1.0 1.2 Fraction of 7.8 buffer vs. 3.07 Where C = 1M HOAc, D=~1M HOAc pH ~7.8 (NH4OH) and the final molarity is 0.1M The pKa of the acetate ion is approximately 4.7, while the formate pKa is 3.7 Selectivity vs. pH AmmOAc 0.1M 1 Initial test pH 3.07 10% C 1 Vanillin 2 Phenol 3 Benzoic 4 Sorbic 5 Phenylacetic 3,4 2 5 Selectivity vs. pH AmmOAc 0.1M 3 1 5 4 2 Mid-pH test pH 4.63 5% C, 5% D 1 Vanillin 2 Phenol 3 Benzoic 4 Sorbic 5 Phenylacetic Selectivity vs. pH AmmOAc 0.1M 3 4 5 1 Optimized pH 4.85 4% C, 6% D 2 1 Vanillin 2 Phenol 3 Benzoic 4 Sorbic 5 Phenylacetic Quick Evaluation of Different pH on Selectivity 7 pH 2.7 0.1% formic acid/acn 4 6 1 3 2 5 0 2 4 6 pH 4.8 NH4AC/acn 1 0 2 pH 7 NaPO4/acn 4 5 4 2 3 8 6 7 min 2 1 3 4 0 min 6 2 7 8 4 6 6 5 8 Group/Presentation Title Agilent Restricted Month ##, 200X