Measurement & Data Processing: Uncertainties & Errors
advertisement

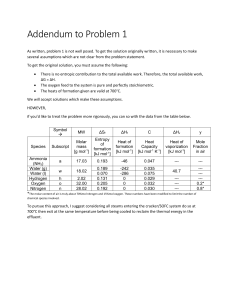
Topic 11 - Measurement and Data Processing 11.1 Uncertainties and errors in measurement and results Four groups have done a calorimetric experiment to determine the enthalpy change of a reaction in aqueous solution, for which the literature value is 38.73 kJ mol-1. The values found by the groups (in kJ mol-1) were: A 35.1 ± 0.3 C 33.2 ± 0.1 B 36.5 ± 0.5 D 34.7 ± 0.2 1. Which result is the most precise? 2. Which result is the most accurate? 3. Do you think the major problem is A B C D 4. C D Using a more precise thermometer. Not assuming that the specific heat of the solution equalled that of water. Using pipettes rather than measuring cylinders. Improving the insulation of the calorimeter. Which of the following is the best way to report the mean result of the group? A B C D 6. Which result has the lowest absolute uncertainty? A B C D 8. A B C D 9. 34.875 ± 0.100 kJ mol-1. 34.9 ± 0.5 kJ mol-1. 34.9 ± 0.8 kJ mol-1. 35 ± 2 kJ mol-1. Repeating an experiment a number of times will lead to a decrease in: A B C D the random error. the systematic error. both the random and the systematic error. neither the random and the systematic error. 34.875 ± 0.017 749 ± 5 0.0004 ± 0.0001 87500 ± 200 Which result has the lowest percentage uncertainty? 34.875 ± 0.017 749 ± 5 0.0004 ± 0.0001 87500 ± 200 If x = 749 ± 5 and y = 34.8 ± 0.7, which one of the following will have the greatest percentage uncertainty? A B C D If you were to repeat the experiment which of the following is most likely to be the improvement that would most improve the result? A B 5. Random error Systematic error Both are equally important It is not possible to tell from the data given 7. x+y x-y x/y x.√y 10. Give the result and the absolute uncertainty, to the correct precision, of the following calculations: a) 20.1 ± 0.1 + 2.75 ± 0.05 b) 115.4 ± 0.2 × 8.137 ± 0.001 c) 0.572 ± 0.001 (17.6 0.2 - 114 ± 1) d) (1 - 0.276 ± 0.002)/0.024 ± 0.001 e) 52800 ± 100/ √(17.2 ± 0.2 - 2.37 ± 0.01)