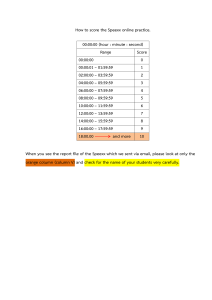
Practice Exercises for Process Synthesis & Simulation I A Karimi Department of Chemical & Biomolecular Engineering National University of Singapore Pointers & Guidelines on Aspen Hysys (or simply Hysys) (1) Do not follow instructions mechanically. THINK what you are doing and why. Fully read and understand each exercise and each sentence. (2) Do not receive/donate solutions from/to friends. (Remember No pain, No gain) (3) Do exercises in the stated sequence. Do not skip parts within an exercise. (4) Double-clicking an Aspen Hysys file always opens a new license. Hence, always open multiple Aspen Hysys files from one Hysys window to avoid using multiple licenses. (5) Save your work REPEATEDLY. You will need the files of all your exercises. Find your own H: drive on the network. (6) No exercise involves unsteady state operations. Do not use any module meant only for dynamic or unsteady state operations (e.g. Furnace, Tank, PID, etc.) (7) Numerical answers, where given, are obtained using the following choices. First, components are chosen from Hysys and not Aspen Properties. Second, fluid packages are chosen from either Hysys or COMThermo, and not Aspen Properties. (8) Numerical answers are ROUGH GUIDES only. Do not be concerned, if you cannot get them EXACTLY. Answers do vary with your data (e.g. temperatures, pressure drops), Hysys version, fluid package, numerical convergence, etc. (9) Before simulating any process, first develop a BFD on paper using our process synthesis algorithm, and assign appropriate Hysys module to each operation. (10) Draw the BFD in Hysys one module at a time in the sequence of material flow. Specify stream data and equipment parameters one module at a time. Converge each module before installing the next. Hysys Tutorials Page 1 of 24 Copyright © IA Karimi Exercise P1: A process receives two separate feeds. One is pure water at 100 kg/s, 1 atm, and 40 C. The other is 80:20 wt% ethanol:water at 150 kg/s, 2 atm, and 95 C. Their mixture is to be stored in a tank at 40 C (exactly) and 3 atm, but the tank has only one inlet. Use the process synthesis algorithm discussed in the class to develop three BFDs for this process using 1 atm, 2 atm, and 3 atm as three possible mixing pressures. Simulate the BFDs. Which process uses the fewest equipment items? Which process is likely to have the least capital cost (CAPEX)? Do not attempt to use Economic Analyzer in Hysys. It is NOT required. Just compare costs relatively based on simple assumptions. Which process uses the most heating energy (kW)? Which uses the most cooling energy (kW)? Which uses the most electrical energy (power kW)? Which utility (hot, cold, or power) in general costs the most? Which costs the least? Which process will have the lowest total utility cost? Which will have the highest total utility cost? Which cost (CAPEX or OPEX) will generally dominate in a chemical process? Which process will be the easiest to maintain? Which process would you recommend? Why? Hysys Tutorials Page 2 of 24 Copyright © IA Karimi Exercise P2: A shell and tube heat exchanger (H-100) cools aniline (120,000 lb/h, 1.5 atm, 275 F) to 200 F by using benzene (2 atm, 100 F). Aniline flows on the shell side with a pressure drop of 1 psi and benzene on the tube side with a pressure drop of 2 psi. Hysys has three modules for heating/cooling: Heater, Cooler, and Heat Exchanger. The first two do not allow two streams, hence each by itself cannot simulate the exchange of heat between two streams. In contrast, Heat Exchanger allows two streams (hot and cold), and simulates a shell and tube exchanger. Here, you will simulate heat exchange between two process streams using Heater and Cooler ONLY. Select PRSV as the fluid package (No COMThermo). (Hint: Take energy from Cooler and supply to Heater) What is the duty of H-100? (4.399e6 Btu/h) If the benzene flow is 100,000 lb/h, then what is its exit temperature? (204.1 F) If benzene exits at 200 F, then what is its flow rate? (104400 lb/h) Exercise P3: A fully sealed (no vent) tank (T-100) holds liquefied natural gas or LNG (97% methane, 2% ethane, and 1% propane by mol). At time zero, T-100 is at 1.2 bar, and 100% FULL with LNG (i.e. no vapor in T-100). Assume the pressure is 1.2 bar everywhere in T-100. LNG has no polar components, so COMThermo is not needed for a fluid package. Use Peng Robinson from Hysys to describe both phases. What is the name for the maximum temperature that T-100 can have at time zero? Which Hysys module can you use to find that temperature? What is that maximum temperature? Tmax = (–159.1 C) ____________. If T-100 is at Tmax at time zero, then what is the density of its LNG? (431.2 kg/m3) _________. T-100 has a pressure-indicator (PI-100) and a temperature-indicator (TI-100). After two days, PI-100 shows 1.5 bar and TI-100 shows –156 C. Why did T-100 temperature increase? Why did T-100 pressure increase? What percentage (wt%) of LNG evaporated during the two days? (32.17 wt%) ___________. Now, let us say T-100 has a vent with a relief valve, which keeps its pressure constant at 1.2 bar. In other words, T-100 is NOT sealed any more, and keeps losing some evaporated LNG continuously. It has 10 Mg of LNG at time zero with a temperature of Tmax. Assume that 10 MJ Hysys Tutorials Page 3 of 24 Copyright © IA Karimi of heat leaks into T-100 over two days at a constant rate. Assuming that the evaporated amount is negligible compared to the total LNG in T-100, we wish to compute the percentage (wt%) of LNG lost from T-100 in two days. For this, we need to imagine a continuous process, because T-100 is a batch system. Hence, consider the below continuous process. Convince yourself that 10 Mg of LNG receives 10 MJ of heat in one hour. Since the total evaporation of LNG depends only on the total amount of heat and not the duration of heating, the evaporation in the below process should match the evaporation from T-100. Find the percentage of LNG lost from T100. (0.1921 wt%) Exercise P4: Toluene and Hydrogen produce Benzene via the below gas-phase reactions. Toluene + H2 → Benzene + Methane (1) 2 Benzene (2) ↔ Diphenyl (or Biphenyl) + H2 A toluene feed (100% toluene, 100 lbmole/h) is available at 77 F and 14.7 psia. A hydrogen feed (95 mol% hydrogen and 5 mol% methane) is available at 100 F and 550 psia. The total molar flow of the hydrogen feed is automatically controlled to be five times that of the toluene feed. The reactor needs one single feed at 500 psia and 1150 F exactly. The following heuristic rules are useful in developing a process flow diagram (PFD). Rule 1: Avoid mixing a liquid stream with a gas stream, whenever possible. Rule 2: Prefer 1-phase flows over 2-phase flows. Rule 3: Never mix two streams at different pressures. Rule 4: Pumps cost much less than compressors in general. Rule 5: Pumping a liquid needs much less energy than compressing a gas. Rule 6: Evaporators normally produce only saturated vapors. Using the above rules, develop/draw on paper a BFD for the below feed preparation system. Hysys Tutorials Page 4 of 24 Copyright © IA Karimi Simulate your BFD. Use Peng Robinson from Hysys. Do not add the two reactions now, but include all the necessary components. You will add the reactions in Exercise P8. Does your BFD use compression? If yes, then how can you avoid it? Does your BFD mix a gas with a liquid? If yes, then how can you avoid it? Does your BFD have a 2-phase flow? If yes, then how can you avoid it? Exercise P5: A refinery produces a petroleum gas (PG) at 70 C and 200 kPa. Its composition (mol%) is 3.0% methane, 7.0% ethane, 60.0% propane, 15.0% n-butane, 12.0% i-butane, 1.0% n-pentane and 1.0% i-pentane, 1% water). The refinery liquefies this gas at a high pressure to get a liquid called Liquefied PG or LPG. This LPG product is sent to domestic and industrial users in thick and heavy LPG cylinders via non-refer (non-refrigerated) trucks. The pressure inside each cylinder must be high to keep its contents liquid, even when it is transported or stored outside. This exercise aims to develop a process to produce 95% full LPG cylinders at 40 C. Use Peng Robinson from Hysys. Is PG fully vapor or has some liquid? If we cool PG at 200 kPa, then at what temperature will we see the first liquid drop? (~ 17.9 C) Which component will dominate that liquid drop? If we pressurize PG, while maintaining the temperature at 70 C, at what pressure will it begin to liquefy? _________________ (1813 kPa) If we continue to pressurize as above, at what pressure will it become fully liquid? ________ (2880 kPa) Hysys Tutorials Page 5 of 24 Copyright © IA Karimi Now, let us see if we can liquefy PG by increasing its pressure to about 2880 kPa. So, let us use a compressor to pressurize PG from (200 kPa, 70 C) to 2880 kPa. Does the exit stream have any liquid? Why? What will you do to get LPG? A process produces LPG at 40 C. What minimum pressure must the LPG stream have? _____ (1800 kPa). Unlike the last question, do not use a compressor for this question. Develop and simulate a BFD for producing LPG at 40 C from PG for household use. Assume LPG pressure to be 400 kPa higher than the minimum from the previous question. Respect the following heuristics for the design and operation of compressors. Please minimize the total power consumption of the process. Use the following heuristic rules or guidelines in developing your process. Rule 1: The outlet temperature of a compressor is usually kept below 200 C. Rule 2: A single-stage compressor may not increase pressure by a ratio of more than 5. Rule 3: The feed to a compressor must not have any liquid. Rule 4: Compressing a colder feed saves energy. Rule 5: Cooling water at 30 C can cool a gas stream to 40 C or higher, but not lower. Rule 6: Pumping a liquid is far cheaper than compressing a gas. Let us now fill your LPG product (2200 kPa, 40 C) into a cylinder full with nitrogen at 1 atm. Assume that this nitrogen does not condense, as LPG flows in and the entering LPG also does not evaporate. Also, assume that the cylinder and its contents remain at 40 C all the time. Describe what happens as LPG fills the cylinder. When will LPG stop flowing into the cylinder? What is the fractional volume of nitrogen at this point as compared to its initial volume? What fraction of cylinder is liquid? _____ (~ 95.3 vol%) How will you change your proposed LPG plant to get LPG cylinders that are 95 vol% full with liquid? ___________ (The process should produce LPG at ~ 2026 kPa vs 2200 kPa) Hysys Tutorials Page 6 of 24 Copyright © IA Karimi Exercise P6: Consider a 50:50 (wt%) mixture of ethanol and water at 1 atm. Use the COMThermo package with van Laar and HysysPR. Find the temperature at which the mixture is 50 wt% vapor. Please strictly follow the procedure in the notes for using Adjust. Temperature for 50 wt% vapor = ________________________ (84.44 C). Exercise P7: Consider Benzene (C6H6) + 3H2 Cyclohexane (C6H12) at 23 atm with a stoichiometric gas feed. Use Peng Robinson from Hysys. Use partial pressure in atm as the basis and Gibbs free energy as the source of equilibrium constant. Find the temperature at which 97.5% of C6H6 converts. Please strictly follow the procedure in the notes for using Adjust. (~ 256 C) State how the equilibrium conversion will change, if some C6H12 product is recycled back to the reactor? Verify your answer by adding a separate feed stream of pure C6H12 to the reactor and gradually increasing its flow to see the effect on the conversion. Do not recycle the product back to the reactor in your simulation. Exercise P8: Conversion and Equilibrium are simpler reactor blocks. CSTR (Continuous Stirred Tank Reactor) and PFR (Plug Flow Reactor) are more detailed and realistic, but need more data. PFR simulates a tubular reactor, which is common in the industry. Take the single feed from Exercise P4 to produce benzene in a PFR via the following gas-phase catalytic reactions. Toluene + H2 → Benzene + Methane (1) 2 Benzene (2) ↔ Diphenyl + H2 The reaction rates (r1 and r2) in lbmol/min-ft3 are: r1 3.6858 107 exp( r2 5.987 104 exp( 50906 kcal/kgmol ) pT p1/H 2 RT 50906 kcal/kgmol 2 50906 kcal/kgmol ) pB 2.553105 exp( ) pD pH RT RT where, T is temperature (K), pT, pH, pB, and pD are the partial pressures (psia) of toluene, hydrogen, benzene, and diphenyl respectively. Classify the above reactions as reversible or irreversible. What are the units of reaction rates? Hysys Tutorials Page 7 of 24 Copyright © IA Karimi What does the first term in 𝑟 represent? What does the second term represent? What component property is used as the basis in the above rate expressions? What is its unit? What is the order of reaction (1) with respect to Toluene? Hydrogen? Remember that the order of a reaction is NOT the same as the stoichiometric coefficient. Which parts of 𝑟 and 𝑟 are Arrhenius rate constants (𝑘 and 𝑘 )? What are the pre-exponential factors for the two reactions? What are the activation energies for the two reactions? Use Peng Robinson. Add reactions (1) and (2) in Hysys using the kinetic model as RM. Use the correct units and note that E is positive. Specify the right phase for each reaction. The reactor has the following design and operating data: 1. Feed temperature is 1150 °F in order to achieve good reaction rates. 2. Feed pressure is 500 psia. (Can pressure increase across the reactor?) 3. Product temperature should not exceed 1310 °F to avoid coking. 4. The molar flow of hydrogen feed is at least five times that of the toluene fee. (Why should you use a higher ratio? What are its effects?) 5. The reactor uses 250 tubes with 4.53 cm internal diameter and 2.8 mm wall thickness. Specify 4.53 cm as tube diameter in Hysys. 6. The void fraction of catalyst is 0.4. Assume the Hysys-default data for the catalyst. As the first step, make reactor adiabatic and specify pressure drop as zero. Use Adjust to find the tube length (𝐿) to achieve 70% conversion of toluene via the primary reaction. Please strictly follow the procedure in the notes for using Adjust. (~ 4.5 m) What is the product temperature? (~ 1303 F) Keep the reactor adiabatic, but remove the user-specified pressure drop of zero and use Ergun equation for all subsequent parts. Hysys Tutorials Page 8 of 24 Copyright © IA Karimi What is the pressure drop on the tube side? (~ 161.1 kPa) For the above pressure drop, what tube length will give 70% conversion? (~ 4.66 m) What is the product temperature? (~ 1303 F) Explain why the pressure drop affects the required tube length. Remove Adjust and set L = 4.5 m. Study and describe the effect of feed temperature on the reactor conversion. Exercise P9: A furnace operates at 100 kPa. It uses a producer gas (2 atm, 50 C) and ambient air (1 atm, 30 C). The producer gas is 2% CH4, 5% CO2, 14% H2, 20% CO and 59% N2 by mol. The air is 75.59% N2, 20.28% O2, 0.90% Ar, 3.19% H2O, and 0.04% CO2 by mol. Use Peng Robinson as the FP. What is the primary use of a furnace or fired heater in the chemical industry? What does it do to achieve this objective? What type of process is combustion (separation, absorption, mixing, heating, reaction, etc.)? Which components are combustible in the producer gas? What reaction conversions will you target for these components in the furnace? Hysys has a Fired Heater, but it is only for dynamic simulation. Thus, we need another module. Select the simplest module that can simulate combustion. What operation is required before the producer gas can enter the furnace? What about the air? Which modules in Hysys can simulate those operations? For any furnace, one must decide the amount of air to use for combustion. It is normally indicated as an air-to-fuel ratio (call it 𝐴𝐹𝑅): Hysys Tutorials Page 9 of 24 Copyright © IA Karimi Air-to-fuel Ratio = 𝐴𝐹𝑅 When the airflow is just sufficient (no more, no less) to burn a fuel fully (100% conversions for the combustion reactions), the corresponding 𝐴𝐹𝑅 is called the minimum air-to-fuel ratio or 𝐴𝐹𝑅 . For a given fuel flow, as we increase air flow from zero to 𝐴𝐹𝑅 and beyond, the flow rates of various components in the gas exiting the furnace (called flue or stack gas) change. Plot qualitatively by hand (do not use Hysys) how the exit flow of each component will vary, as and beyond. From your plots, answer the following. 𝐴𝐹𝑅 changes from zero to 𝐴𝐹𝑅 Which components will have zero exit flow rates for 0 ponents. Which components will have zero exit flows for 𝐴𝐹𝑅 Which components will have zero flows at 𝐴𝐹𝑅 𝐴𝐹𝑅 𝐴𝐹𝑅 𝐴𝐹𝑅 𝐴𝐹𝑅 ? Call them O-com- ? Call them F-components. ? Let FF = total molar flow of F-components and FO = total molar flow of O-components. What is the value of FF–FO at 𝐴𝐹𝑅 𝐴𝐹𝑅 ? What is the sign of FF–FO (+ve or –ve) for 𝐴𝐹𝑅 𝐴𝐹𝑅 ? For 𝐴𝐹𝑅 𝐴𝐹𝑅 ? Assume the producer gas flow is 1 kgmol/h. Use Adjust and for the producer gas. (~ 1.037). Spreadsheet to find an accurate 𝐴𝐹𝑅 Save this work as P9-1.hsc file. A furnace normally uses 𝐴𝐹𝑅 as follows. %𝐸𝐴 𝐴𝐹𝑅 . Why? The extra air is called % excess air and defined 100 𝐴𝐹𝑅 𝐴𝐹𝑅 /𝐴𝐹𝑅 Make a copy of P9-1.hsc file. Call it P9-2.hsc. Remove Adjust, and set producer gas flow to 100 kgmol/h, and delete the molar flow value in the air stream. Assume %𝐸𝐴 25 or 𝐴𝐹𝑅 1.25 ∗ 𝐴𝐹𝑅 . Use Spreadsheet to compute and set the molar flow of air using 𝐴𝐹𝑅 and the producer gas flow. At what temperature does the flue gas leave the furnace? (~ 1435 C). What is its moisture content? (~ 6.43 wt%). How will the flue gas temperature change, if you remove some heat from the furnace? Normally, a process stream removes some heat from the furnace, before the flue gas exits. Assume a process stream removes 1.0e8 Btu/h from your furnace. Does Hysys give an error? What does it say? Explain what happened. Hint: What happens to the flue gas temperature, as you remove more and more heat from the furnace? How will you correct the error? Save this work as P9-2.hsc file. Hysys Tutorials Page 10 of 24 Copyright © IA Karimi Exercise P10: Copy P9-2.hsc as P9-3.hsc. Open P9-3.hsc file. Assume you want the furnace to supply 1e8 Btu/h of heat to a process stream. However, at the same time, you want the flue gas temperature to be 700 C. How much producer gas does the furnace need? Use the procedure for Adjust from the notes. (~ 1777 kgmol/h). The flue gas normally has some SOx and NOx. If it cools substantially, then its moisture can liquefy and dissolve these gases to corrode pipes. Add a Cooler to cool the flue gas and estimate the temperature at which such corrosion could. (~ 42-43 C) One way to reduce fuel use in a furnace is to preheat air. Use a Heater to heat the air entering the furnace to 200 C. How much producer gas do you save? (~ 10%) In the previous two questions, you added heat to the air, and removed heat from the flue gas. Is it possible to use some heat from the flue gas to preheat the air to 200 C? That would save us heating and cooling energy! For this, take the energy stream from the cooler and connect to the heater. At what temperature does the flue gas leave the Cooler? (~ 613.6 C) If the Heater and the Cooler were combined (Do not attempt to combine) inside a Shell & Tube Heat Exchanger, then what would be the minimum temperature approach for this exchanger? The minimum temperature approach for a 2-stream exchanger is defined as follows. If the hot fluid cools from T1 to T2 and the cold fluid heats from t1 to t2, then minimum temperature approach is the minimum of (T1–t2) and (T2–t1). If we preheat air too much, then the flue gas may cool and condense partially. This may cause pipes to corrode. Therefore, we should not cool the flue gas below 150 C. Moreover, we want a minimum temperature approach of at least 100 C for the exchanger. Therefore, preheat the air gradually beyond 200 C. Stop when the flue gas cools to 150 C or the minimum temperature approach reaches 100 C, whichever is earlier. How much fuel can you reduce by air preheating? (~ 27.8 %) What is the temperature of the preheated air? (600 C) At what temperature does the flue gas leave the Cooler? (~ 392 C) Hysys Tutorials Page 11 of 24 Copyright © IA Karimi Exercise P11: Consider an ammonia reactor (N2+3H22NH3) operating at 250 C and 2 MPa with a stoichiometric feed and 20% conversion wrt N2. Is the reactor product liquid or gas? Which components does it have? Which is the most volatile component? Which is the least volatile component? Use a Cooler after the reactor to cool the product to say 0 C. Is there any liquid in the cooled product? If you continue to cool further, which component will liquefy first? At what temperature does the product begin to liquefy? Cool the product to –40 C. Is there any liquid in the cooled product? What percentage (wt%) of that liquid is ammonia? Hysys defines three types of flows for a stream: Molar Flow = Total molar flow of the stream Master Comp Molar Flow = Total molar flow of a component in the stream Phase Comp Molar Flow = Molar flow of a component in a particular phase (vapor, liquid, or aqueous) of the stream. Use Spreadsheet to compute liquid ammonia recovery at –40 C by using the following: Ammonia Recovery (%) = 100 ∗ (~ 66%) What should be the temperature of the cooler to achieve 80% recovery for ammonia liquid? (~ –47.5 C) __________________. Hysys Tutorials Page 12 of 24 Copyright © IA Karimi Exercise P12: A distillation column with a total condenser and a partial reboiler operates at 1.5 atm. Its feed (1.5 atm, 80 C, 100 kgmol/h) is a 60:40 mol% mixture of benzene:toluene. 98% of the benzene is recovered in the top product, and 95% of toluene in the bottom product. Are benzene and toluene polar? Are they similar in nature? Do you expect them to form ideal solutions? What is Raoult’s law? What is its expression? Which property does it need? Is it valid for ideal solutions? Which fluid package can help you use Raoult’s law for the benzene-toluene system? Condenser Overhead Product Distillation Column Reboiler Bottom Product Component Splitter Use Component Splitter to simulate the ENTIRE distillation column including the reboiler and condenser. What are the temperatures of the condenser and reboiler? (93.25 C and 124.1 C) What are the product purities? (96.71 mol% benzene and 96.94 mol% toluene) Distillation Column in Hysys simulates a distillation column. It cannot design the column. It cannot tell you how many stages or what reflux ratio you need directly. For approximate design, Hysys Tutorials Page 13 of 24 Copyright © IA Karimi Hysys offers a Shortcut Distillation Column. This is based on the Fenske-Underwood-Gilliland (FUG) method for multicomponent distillation. Please refer your CN3123 notes or textbook to refresh your memory. Its primary objective is to compute the minimum number of stages (𝑁 ) and minimum reflux ratio (𝑅 ) for a given feed and its separation. The FUG method requires you to select a Light Key (LK) and Heavy Key (HK), and specify the desired compositions of these components in the top and bottom products. Using 𝑁 , one can specify 𝑅/𝑅 to estimate the number of theoretical stages (𝑁) in a real column. For the above product purities of benzene and toluene, find 𝑅 using Shortcut Distillation Column. (~ 0.972) and 𝑁 (~ 7.625) For 𝑅⁄𝑅 1.4, how many theoretical stages (𝑁) do you need for this column? (~ 15.964). What is the optimum feed stage location? (~ 7.171). Now, use 𝑁 16 and feed at stage 7 to simulate the real column using Distillation Column in Hysys. Find 𝑅 for 98% recovery of benzene in the top product and 95% recovery of toluene in the bottom product. Compare your results such as product flows, temperatures, purities, and component recoveries with those from Component Splitter. Also, compare 𝑅 with that predicted by Shortcut Distillation Column. (R=1.134 vs R=1.3608). Please find the main assumptions behind the FUG method from your CN3132 notes. Exercise P13: A fermenter in an acetone-butanol-ethanol (ABE) process produces a liquid stream at 1.1 atm and 40 C. The stream has 90.3 wt% water, 3.0% acetone, 0.6% ethanol, 6.0% 1-butanol, and 0.1% CO2. It is heated to 70 C and then distilled in a beer column at 1.1 atm with 20 stages and a total condenser. The feed enters at stage 8 from the top. Use COMThermo with NRTL and PRSV. Compare the boiling points of acetone, ethanol, water, and 1-butanol. If we want to recover most of water in the bottom product, then where (top product or bottom product) do you expect 1-butanol to go? If 𝑅 2.0 and the distillate flow is 15% of the feed by weight, then what are the purities of the top and bottom products and the recoveries of various components? Set mass as the basis on “Specs” page. Use Column Recovery as a spec. Use “Reset”, if you have convergence problems. (The top product has 40 wt% 1-butanol, 35.33 wt% water, 20 wt% acetone, 4 wt% ethanol, and 0.67 wt% CO2. The recoveries of ABE and CO2 are 100% in the top product, and the recovery of water is 94.13% in the bottom product.) Hysys Tutorials Page 14 of 24 Copyright © IA Karimi Propose a possible reason why 1-butanol may go to the distillate in spite of its higher boiling point. Which component is most likely to go in the bottoms? Study how 𝑅 and 𝑁 affect various component recoveries. Study how the feed stage and feed temperature affect component recoveries. What is the condenser temperature? Is the temperature rather high or low? Why? Can you use cooling water at 30 C in this condenser? Why? Is using total condenser a good idea? What will you change to be able to use cooling water at 30 C? Simulate the column again with the change you propose in the last part. If you need an additional spec, then specify 60 C as the condenser temperature. What are the recoveries of ABE in the distillate product and water in the bottom product? (Recoveries in the distillate are 98.4% for acetone, 99.6% for ethanol, and 99.9% for 1-butanol. Water recovery in the bottom product reduces slightly to 94.0%) Hysys Tutorials Page 15 of 24 Copyright © IA Karimi Exercise P14: A continuous fermenter (CSTR) operates at 40 C and 1.2 atm. It has two product streams: one liquid (L) from the bottom and one vapor (V) from the top. These two streams TOGETHER contain 930 kg/h water, 70 kg/h ethanol, and 70 kg/h CO2. Assuming the two product streams are in vapor-liquid equilibrium, what are the component flows in L? (928.1 kg/h water, 68.1 kg/h ethanol, and 1.3 kg/h CO2). You can use the Separator module in Hysys to obtain the L and V streams from the combined product stream from the reactor. High purity ethanol is the desired product from this reactor. Since ethanol and water form a minimum boiling azeotrope at 78.2 C with a composition of 95.63 wt% ethanol and 4.37 wt% water, let us distill the liquid stream from the reactor to get >90 wt% pure ethanol with >99% recovery. Which type (total, partial, full reflux, total reflux) of condenser will you use? Why? How many specs will you need? If the condenser must use cooling water at 30 C, then what condenser temperatures are possible? What will you use as column pressure (𝑃)? Why? Design (Find 𝑃, 𝑁, 𝑅, etc.) and simulate a distillation column to achieve the desired process objectives. Use the COMThermo option with NRTL for liquid, and SRK for vapor. What are 𝑁, 𝑅, ethanol recovery, reboiler duty, and condenser duty for your column? Vary feed stage manually to find the best. How did you define best? Hysys Tutorials Page 16 of 24 Copyright © IA Karimi Exercise P15: Consider the below PFD for Cyclohexane manufacture from the lecture notes. Simulate this process using the following data. All compositions are in mol% and stream names are in parentheses. (a) Benzene Feed: 100 kgmol/h, 100% Benzene, 100 F, 15 psia. (b) Makeup Hydrogen: 97.5% H2, 2.0% CH4, 0.5% N2, 120 F, 340 psia. (c) Feed Preheater: Inlet pressure = 340 psia, ΔP = 5 psia, Outlet temperature = 570 F. (d) Reactor: ΔP = 15 psia; 99.85% conversion for C6H6; C6H6:H2 = 1:5 mol/mol in feed; product at 430 F. (e) Product Cooler: ΔP = 5 psia, outlet temperature = 120 F. (f) Flash: ΔP = 0 psia, adiabatic. (g) Purge 8% of gas from Flash. Recycle 30% of liquid from Flash. (h) Stabilizer: Pressure = 20 psia, full reflux condenser, partial reboiler. Use only if needed. (i) Cyclohexane Product: 99.90 wt% C6H12. (j) BFW (Boiler Feed Water) inlet temperature = 50 C. Identify the Hysys modules most appropriate for modeling the above process. Use HysysPR. Use Recycle to break loops. How will you set the flow of Makeup Hydrogen to achieve C6H6:H2 = 1:5 mol/mol in the reactor feed for any given flow of Benzene Feed? What are the energy needs (heating, cooling, power) of various equipment in this process? Hysys Tutorials Page 17 of 24 Copyright © IA Karimi What is the overall yield of this process? (Percent of benzene converted to cyclohezxane) What is the Makeup Hydrogen to Benzene Feed ratio (mol/mol)? What will you do with the Light Ends? What are the losses (%) of H2 and C6H6 from the process? How much MP steam (Saturated at 17 bar) can you generate in Reactor? How can you conserve some energy in this process? Modify your PFD to simulate such a process. Hysys Tutorials Page 18 of 24 Copyright © IA Karimi OPTIONAL but RECOMMENDED EXERCISES Exercise P16: A distillation column has 15 stages, total condenser and partial reboiler. It operates at 1 atm with no pressure drop. Its feed is a saturated liquid (25 mol% toluene, 75 mol% phenol, 1 atm, 100 kgmol/h). Use COMThermo with van Laar and PRSV. If you have trouble converging the distillation column, it may help to reset and run again. A) For a distillate rate of 26 kgmol/h, study the effects of feed stage location and R on product purities and recoveries by running the following scenarios. 1. Feed at stage 10 from top and R = 0.60 2. Feed at stage 10 from top and R = 0.55 3. Feed at stage 10 from top and R = 0.65 4. Feed at stage 08 from top and R = 0.60 (1: Purities are 96.12 mol% toluene and 99.99 mol% phenol. Recoveries are 99.96% for toluene and 98.65% for phenol) B) For R = 0.6 and feed at stage 10, use toluene purity of 98 mol% min as the Spec instead of distillate rate. What is the distillate flow rate now? (25.49 kgmol/h) What are the product purities and recoveries? (Purities are 98.00 mol% toluene and 99.98 mol% phenol. Recoveries are 99.93 % for toluene and 99.32% for phenol) Examine the temperature and flow (liquid and vapor) profiles on the column trays. Is the constant molal overflow assumption valid? D) What is the minimum number of stages required for 98 mol% distillate and bottom products? Use Short-cut Distillation column. What is the optimum feed stage? Hysys Tutorials Page 19 of 24 Copyright © IA Karimi Exercise P17: Ethylene oxide (C2H4O) is partially soluble in water and is produced via the following gas phase reactions at 20 atm and 400 C. 2C2H4 + O2 = 2C2H4O (Conversion 30% with respect to C2H4) C2H4 + 3O2 = 2CO2 + 2H2O (Conversion 10% with respect to C2H4) C2H4 is available at 2 atm and 50 C, while O2 is available at 2 atm and 30 C. They enter the process in the mole ratio of C2H4:O2 = 1:3. The process uses a preheater before the reactor to heat the reactor feed using the reactor products as much as possible. Use UNIQUAC for liquid and HysysPR for vapor. It is desired to remove C2H4O from the gas stream using water. What operation or unit can achieve that? How will the temperatures of water and gas stream affect the performance of this unit? Will higher temperature be better for water? Will it be better for gas? How will you achieve the desired temperature? How much water is required per mol of C2H4O? What is the purity of crude ethylene oxide obtained from this unit? Is it a one-phase or two-phase stream? The gas leaving the above unit is scrubbed with a suitable solvent to remove 99% of CO2. Model this scrubbing as a Component Splitter. The scrubbed gas is recycled back to the reactor. The crude C2H4O stream is purified further using stripping and distillation to get 99% pure C2H4O product. Make appropriate assumptions and develop a Hysys model to simulate this process. Hysys Tutorials Page 20 of 24 Copyright © IA Karimi Exercise P18: Develop a process to manufacture 5000 tonne/day of 99 wt% pure methanol (30 C, 1.5 atm) using reformer gas and simulate it using Hysys. A reformer unit supplies the feed gas (H2 78%, CO 6%, CO2 15%, CH4 1% by volume) at 50 psia and 70 C. You must decide what unit operations the process must have, what should be their operating conditions, etc. You need not design the unit operations in detail. Use PRSV for vapor phase and UNIQUAC for liquid phase. Methanol synthesis is carried out in gas phase at 300 C and 100 atm in a fixed bed reactor with a suitable catalyst. Two reactions take place in the reactor: 1. CO + 2H2 = CH3OH and 2. CO2 + H2 = CO + H2O. Per pass conversion of CO by reaction 1 is 25% and that of CO2 by reaction 2 is 35%. Since the conversions are low, a recycle is used in the process with a purge:recycle = 1:9. Exercise P19: Reconsider Exercise P5. An LPG cylinder has an internal volume of 44.5 L. Its contents weigh 19 kg. If it is at 30 C, then what is its internal pressure? What will happen to its pressure, if it is heated to 70 C? Hysys Tutorials Page 21 of 24 Copyright © IA Karimi Exercise P20: A distillation column has 20 ideal stages or trays. Its feed (100,000 kg/h, 90 C, 4 bar) with an overall composition of 78 wt% methanol, 21 wt% methyl formate, and 1 wt% carbon monoxide enters the column via a valve at stage 10. Textbook heuristics recommend a pressure drop of 2-5 psia across reboilers and condensers, and 0.1-0.2 psia for a tray. Use COMThermo with Redlich-Kwong and UNIQUAC as fluid packages. If the top product exits the condenser at 3 bar, then what is the reboiler pressure? __________ (~ 382.7 kPa for 0.1 psi/tray and 5 psi each for the reboiler and condenser) What type of condenser will you use in Hysys? Why? How many specs does the column need? Let us assume that the column must produce 95 wt% pure methyl formate at 3 bar and 15 C as the top product with a recovery of 90 %. The condenser will use chilled water at 5 C. Simulate this column to find the required reflux ratio _________ (~ 2.380) and top product rate ________ (~345.8 kgmol/h). Note these numbers for a later question. If heating via LP steam costs 0.10 $/kWh and cooling via the chilled water costs 0.30 $/kWh, what is the total utility cost for this column? ___________ ($4202 /h). Can the top product exit the condenser at 15 C, if you use a 30 C cooling water in the condenser? Why? For the 30 C cooling water in the condenser, what is the exit temperature of the top product? Estimate the total utility cost _______ (~ 1057 $/h) for the column, if the 30 C cooling water costs 0.02 $/kWh. Reset your column to use the 5 C chilled water instead of the 30 C cooling water. Review your separation textbook to define tray and column efficiencies. When and where did you ask Hysys to use ideal trays? Hysys Tutorials Page 22 of 24 Copyright © IA Karimi View Parameters Efficiencies to see the tray efficiencies assumed by Hysys. What tray efficiency did Hysys use? Set each efficiency as 0.80. Rerun the column. How does the lower efficiency impact the column operation? Please refer “Predict distillation tray efficiency,” (Chemical Engineering Progress, pp. 24-30, 2018.) by M Duss and R Taylor for various methods for estimating tray efficiency. O’Connell defined “column” (vs tray) efficiency and proposed the following correlation. 𝜂 0.503 𝜇 𝛼 . Estimate the O’Connell’s efficiency for your column using results from Performance Plots. __________ (~ 0.51). How many real trays should your column have? _________ (~ 40). Set 40 trays and 51% tray efficiency for your column, and change column pressures. Estimate the total utility cost again. __________ ($4,259 /h) Please explain why the cost increased or decreased from your previous ideal estimate. Are “column” and “tray” efficiencies the same? Do they have the same definition? Is it okay to use O’Connell’s efficiency as tray efficiency? To answer the above questions, replace the purity and recovery specs with the reflux ratio and top product rate you obtained earlier. With 51% tray efficiency, does your column yield the desired product purity and recovery? Why? Hysys Tutorials Page 23 of 24 Copyright © IA Karimi Change tray efficiency manually to achieve your desired purity and recovery targets. What is the tray efficiency? __________ (53 %) Is it higher or lower than the O’Connell’s column efficiency of 51%? What do you conclude? What is the utility cost for this column? ________ ($4,164 /h). Hysys offers a utility (called Tray Utility) that allows the user to design/size each tray, and assess its hydraulic performance. This utility can be activated under “Internals” tab, only after the column has converged fully (GREEN). Hysys allows a column to have multiple sections with different diameters. Tray utility works in two modes: Interactive Sizing and Rating. Interactive Sizing mode fixes tray / column dimensions automatically based on certain default or user-specified parameters. It also assesses the tray performance. Rating mode allows us to assess tray performance for fixed or given tray/column designs. Use the Interactive Sizing mode to study the performance of your column with 40 trays and 53% efficiency. Define two column sections: Section 1 above the feed stage, and Section 2 below and including the feed stage. Under View Geometry, select 1-pass trays and 2.5 m diameter for Section 1, and 2-pass trays and 2.0 m diameter for Section 2. View the maximum jet flooding level for each section under Internals View Results Summary. ________ (~ 63 %) for Section 1 and ________ (~ 89 %) for Section 2. Use Rating Pressure Drops to view the pressure drop computed by Hysys for each tray. Are pressure drop the same for all trays? Why? Hysys allows you to export these pressure drops back to your column simulation. Why should you do that? Check View Messages and View Hydraulic Plots to make sure the tray utility has no error or warning. Export the tray pressure drops back to your column simulation. Do the pressure drops meet the heuristic recommendations from the textbook? Use F1, see Hysys Help, or read Chapter 6 of Distillation Design by Henry Z Kister to learn about the methods for tray sizing and performance. Hysys Tutorials Page 24 of 24 Copyright © IA Karimi