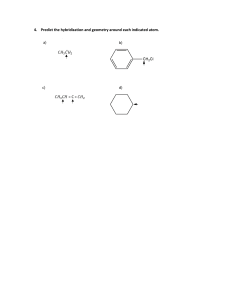
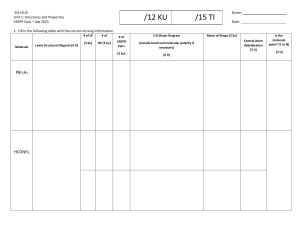
Chemical bonding -2Molecular geometry and hybridization of Atomic Orbital 10.1 Molecular Geometry P. 411 - 420 Molecular - Molecular Geometry is the three-dimensional arrangement of atoms in a molecule. Geometry - Any molecule should be made up of two or more atoms. - Molecule Example’s: CCl4, HCl, N2, …. - Shape may be determined by VSEPR. VSEPR Planar triangular Trigonal bipyramidal Tetrahedral Octahedral Valence Shell Electron Pair Repulsion Essential Question How is VSEPR Theory used to determine molecular geometry? Valence Shell Electron Pair Repulsion Theory What is VSEPR Theory? Predicts molecular geometry (shape) Only works for: Covalent compounds & polyatomic ions Compounds with central atom Steps to Determine Shape 1. Draw Lewis Dot Structure 2. Count # of lone pairs on central atom 3. Count # of bonded atoms on central atom 4. Determine shape using the VSEPR Shape Chart The shape of a molecule can be categorized into 5 general shapes. 5 Types of Shapes 1. Linear 2. Trigonal Planar 3. Tetrahedral 4. Trigonal Bipyramidal 5. Octahedral The geometrical shape is the arrangement of the atoms around a central atom based on the number of atoms, and the amount of lone pairs, which contribute to the overall shape. AB2: BeCl2 Cl—Be–Cl Bonding pairs repel each other, they must be at opposite ends (To be as far as possible). The predicted angle is to be 180 so the molecule is linear. AB2 Molecules Example: HgCl2, BeH2 and CO2. AB3: BF3 AB3 Molecules Boron trifluoride has three bonding pairs repelling each other as far as they can. The geometry is Trigonal Planer, because all four atoms are in the same plane, and the three end atoms form an equilateral triangle. Examples: BH2, SO3, NO3. B